Healthcare Workers’ Hand Microbiome May Mediate Carriage of Hospital Pathogens
Abstract
:1. Introduction
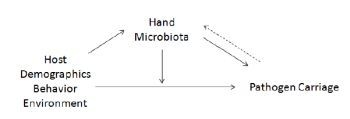
2. Results and Discussion
2.1. Survey Results
| Pathogen (targeted gene) | Collection Visit | Ɏ * Mean copies/uL | * Positive (%, n = 34) |
|---|---|---|---|
| Staphylococcus aureus (nuc) | 1 | 951.3 | 41.2 |
| 2 | 6,623.1 | 41.2 | |
| 3 | 351.4 | 52.9 | |
| Enterococcus spp. (16S) | 1 | 1,702.9 | 52.9 |
| 2 | 2,877.1 | 70.6 | |
| 3 | 1,823.6 | 61.8 | |
| Candida albicans (18S) | 1 | 663.8 | 8.8 |
| 2 | 336.3 | 5.9 | |
| 3 | 142.7 | 2.9 | |
| MRSA (mecA|orfX) | 1 | 173.5 | 2.9 |
| 2 | 1,405.8 | 5.9 | |
| 3 | 3,763.5 | 2.9 |
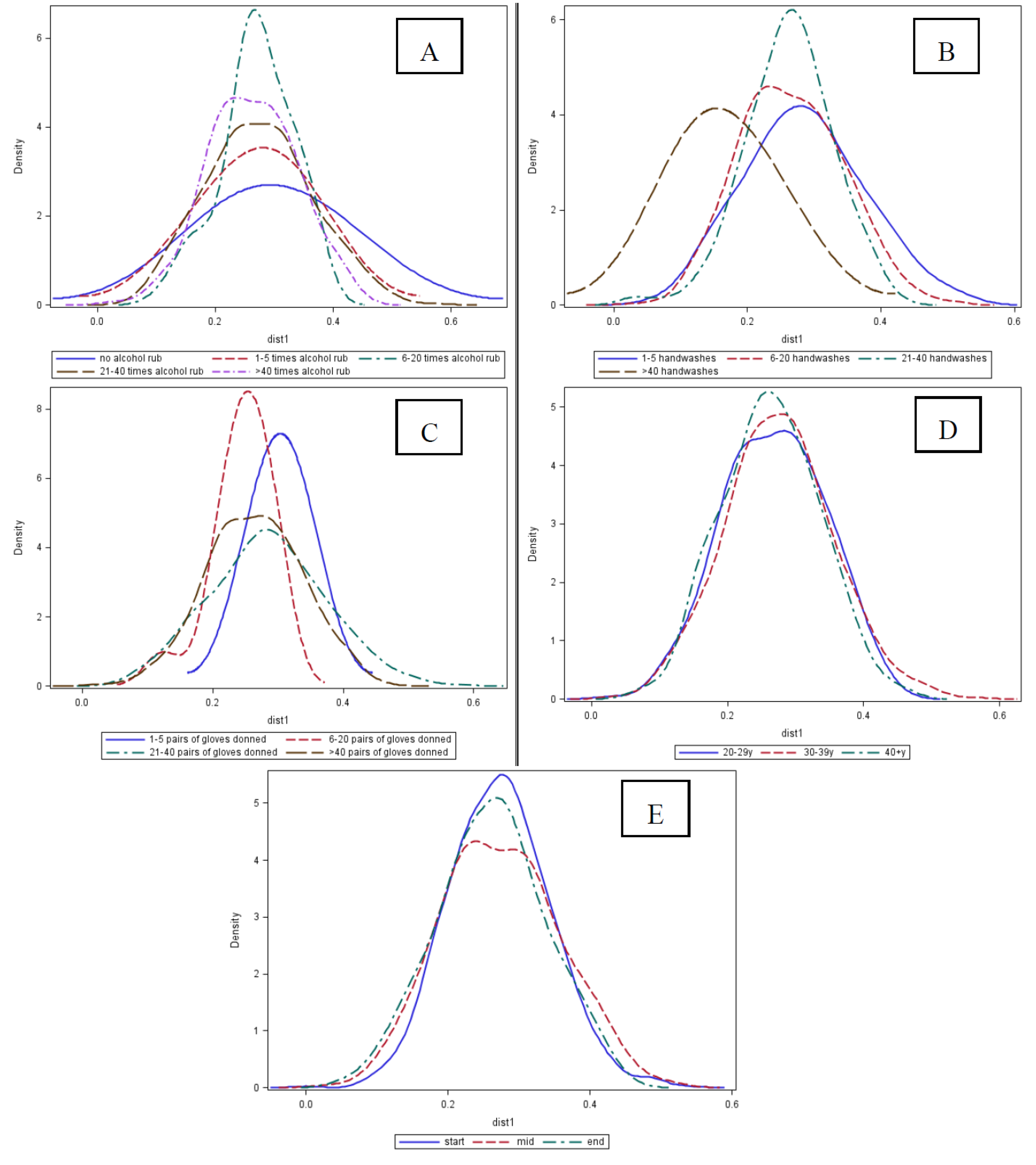
2.3. Association between Risk Factors for Pathogen Carriage and HCW Hand Microbiota
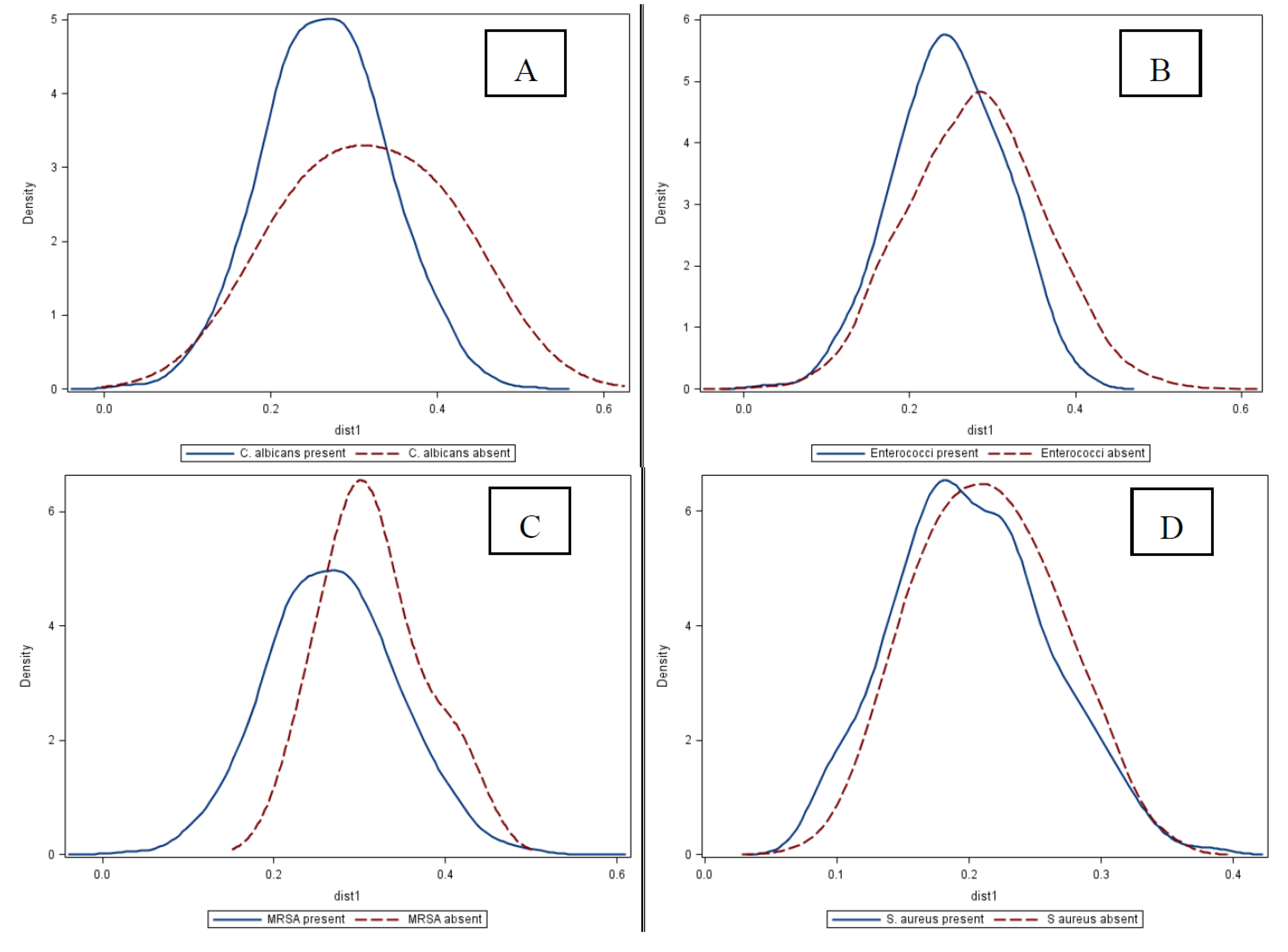
2.4. Association between HCW Hand Microbiota and Pathogen Carriage
2.5. Discussion
3. Experimental Section
3.1. Study Population
3.2. Survey Instruments for Acquiring Potential Risk Factors for Pathogen Carriage
3.3. Sample Collection
3.4. DNA Extraction, Purification and Amplification
3.5. Real-time Quantitative PCR for Pathogen Carriage Detection
| Target | Primers (5' → 3') | Reference | |
|---|---|---|---|
| Staphylococcus aureus | Nuc | GCGATTGATGGTGATACGGTT AGCCAAGCCTTGACGAACTAAAGC | [24] |
| MRSA | mecA|orfX | TATGATATGCTTCTCC AACGTTTAGGCCCATACACCA | [25] |
| Enterococcus spp. | 16S | CCCTTATTGTTAGTTGCCATCATT ACTCGTTGTACTTCCCATTGT | [26] |
| Candida albicans | 18S | GGATCGCTTTGACAATGG GCGGGTAGTCCTACCTGATTT | [27] |
3.6. Statistics
4. Conclusions
Supplementary Files
Acknowledgments
Conflicts of Interest
References
- Cho, I.; Blaser, M. The human microbiome: At the interface of health and disease. Nature 2012, 13, 260–270. [Google Scholar]
- Kinross, J.; Darzi, A.; Nicholson, J. Gut microbiome-host interactions in health and disease. Genome Med. 2011, 3, 14. [Google Scholar] [CrossRef]
- Petrof, E.; Gloor, G.; Vanner, S.; Weese, S.; Carter, D.; Daigneault, M.; Brown, E.; Schroeter, K.; Allen-Vercoe, E. Stool substitute transplant therapy for the eradication of Clostridium difficile infection: “RePOOPulating” the gut. Microbiome 2013. [Google Scholar] [CrossRef]
- McBride, M.; Duncan, W.; Knox, J. The environment and the microbial ecology of human skin. Appl. Environ. Microbiol. 1977, 33, 603–608. [Google Scholar]
- Selwyn, S. Natural antibiosis among skin bacteria as a primary defence against infection. Brit. J. Dermatol. 1975, 93, 487–493. [Google Scholar] [CrossRef]
- Fredricks, D. Microbial Ecology of Human Skin in Health and Disease. J. Investig. Dermatol. Symp. Proc. 2001, 6, 167–169. [Google Scholar] [CrossRef]
- Van Belkum, A.; Melles, D.; Nouwen, J.; van Leeuwen, W.; van Wamel, W.; Vos, M.; Wertheim, H.; Verbrugh, H. Co-evolutionary aspects of human colonization and infection by Staphylococcus aureus. Infect. Genet. Evol. 2009, 9, 32–47. [Google Scholar] [CrossRef]
- Horn, P.; Schouenborg, P.; Brandslund, I. Quantification of Staphylococcus aureus and Staphylococcus epidermidis on the hands of health-care workers using a real-time polymerase chain reaction method. Scand. J. Clin. Lab. Invest. 2007, 67, 165–177. [Google Scholar] [CrossRef]
- Hayden, M. Insights into the epidemiology and control of infection with vancomycin-resistant enterococci. Clin. Infect. Dis. 2001, 31, 1058–1065. [Google Scholar] [CrossRef]
- Creamer, E.; Dorrian, S.; Dolan, A.; Sherlock, O.; Fitzgerald-Hughes, D.; Thomas, T.; Walsh, J.; Shore, A.; Sullivan, D.; Kinnevey, P.; et al. When are the hands of healthcare workers positive for methicillin-resistant Staphylococcus aureus? J. Hosp. Infect. 2010, 75, 107–111. [Google Scholar] [CrossRef]
- Khan, Z.; Chandy, R.; Metwali, K. Candida albicans strain carriage in patients and nursing staff of an intensive care unit: A study of morphotypes and resistotypes. Mycoses 2003, 46, 479–486. [Google Scholar] [CrossRef]
- Boyce, J.; Pittet, D. Healthcare Infection Control Practices Advisory Committee; HICPAC/SHEA/APIC/IDSA Hand Hygiene Task Force. Guideline for hand hygiene in health-care settings. Recommendations of the Healthcare Infection Control Practices Advisory Committee and the HICPAC/SHEA/APIC/IDSA Hand Hygiene Task Force. Society for Healthcare Epidemiology of America/Association for Professionals in Infection Control/Infectious Diseases Society of America. MMWR Recomm. Rep. 2002, 51, 1–45. [Google Scholar]
- The WHO Guidelines on Hand Hygiene in Health Care. 2009. Available online: http://whqlibdoc.who.int/publications/2009/9789241597906_eng.pdf (accessed on 9 October 2013).
- Rosenthal, M.; Goldberg, D.; Aiello, A.; Larson, E.; Foxman, B. Skin microbiota: Microbial community structure and its potential association with health and disease. Infect. Genet. Evol. 2011, 11, 839–848. [Google Scholar] [CrossRef]
- Larson, E.; Hughes, A.; Pyrek, J.; Sparks, S.; Cagatay, E.; Bartkus, J. Changes in bacterial flora associated with skin damage on hands of health care personnel. Am. J. Infect. Control 1988, 26, 513–521. [Google Scholar]
- Larson, E.; Butz, A.; Gullette, D.; Laughon, B. Alcohol for surgical scrubbing? Infect. Control Hosp. Epidemiol. 1990, 11, 139–143. [Google Scholar]
- Larson, E.; Friedman, C.; Cohran, J.; Treston-Aurand, J.; Green, S. Prevalence and correlates of skin damage on the hands of nurses. Heart Lung 1997, 26, 404–412. [Google Scholar] [CrossRef]
- Berg, M. Evaluation of a questionnaire used in dermatological epidemiology. Discrepancy between self-reported symptoms and objective signs. Acta Derm. Venereol. Suppl. (Stockh.) 1991, 156, 13–17. [Google Scholar]
- Simion, F.; Rhein, L.; Morrison, B.; Scala, D.; Salko, K.; Kligman, A. Self perceived sensory responses to soap and synthetic detergent bars correlate with clinical signs of irritation. J. Am. Acad. Dermatol. 1993, 22, 288–293. [Google Scholar]
- Smit, H.; Coenraads, P.; Lavrijsen, A.; Nater, J. Evaluation of a self administered questionnaire on hand dermatitis. Contact Dermatitis 1992, 26, 11–16. [Google Scholar] [CrossRef]
- Farage, M.; Maibach, H.; Andersen, K.; Lachapelle, J.; Kern, P.; Ryan, C.; Ely, J.; Kanti, A. Historical perspective on the use of visual grading scales in evaluating skin irritation and sensitization. Contact Dermatitis 2011, 65, 65–75. [Google Scholar] [CrossRef]
- Hummelen, R.; Fernandes, A.; Macklaim, J.; Dickson, R.; Changalucha, J.; Gloor, G.; Reid, G. Deep sequencing of the vaginal microbiota of women with HIV. PLoS One 2010, 5, e12078. [Google Scholar]
- Hidron, A.; Edwards, J.; Patel, J.; Horan, T.; Sievert, D.; Pollock, D.; Fridkin, S. Antimicrobial-resistant pathogens associated with healthcare-associated infections: Annual summary of data reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2006–2007. Infect. Control Hosp. Epidemiol. 2008, 29, 996–1011. [Google Scholar] [CrossRef]
- Brakstad, O.; Aasbakk, K.; Maeland, J. Detection of Staphylococcus aureus by polymerase chain reaction amplification of the nuc gene. J. Clin. Microbiol. 1992, 30, 1654–1660. [Google Scholar]
- Cuny, C.; Witte, W. PCR for the identification of methicillin-resistant Staphylococcus aureus (MRSA) strains using a single primer pair specific for SCCmec elements and the neighbouring chromosome-borne orfX. Clin. Microbiol. Infect. 2005, 11, 834–837. [Google Scholar] [CrossRef]
- Rinttilä, T.; Kassinen, A.; Malinen, E.; Krogius, L.; Palva, A. Development of an extensive set of 16S rDNA-targeted primers for quantification of pathogenic and indigenous bacteria in faecal samples by real-time PCR. J. Appl. Microbiol. 2004, 97, 1166–1177. [Google Scholar] [CrossRef]
- Srinivasan, U.; Ponnaluri, S.; Villareal, L.; Gillespie, B.; Wen, A.; Miles, A.; Bucholz, B.; Marrs, C.; Iyer, R.; Misra, D.; et al. Gram stains: A resource for retrospective analysis of bacterial pathogens in clinical studies. PLoS One 2012, 7, e42898. [Google Scholar] [CrossRef]
- West, B.; Welch, K.; Galecki, A. Linear Mixed Models: A Practical Guide Using Statistical Software, 1st ed.; Chapman Hall/CRC Press: Boca Raton, FL, USA, 2006; pp. 41–43. [Google Scholar]
- Rosenthal, M.; Aiello, A.; Chenoweth, C.; Goldberg, D.; Larson, E.; Gloor, G.; Foxman, B. Impact of technical sources of variation on the hand microbiome dynamics of healthcare workers. PLoS ONE. submitted.
- Kamada, N.; Seo, S.; Chen, G.; Nunez, G. Role of the gut microbiota in immunity and inflammatory disease. Nature Immunol. 2013, 13, 321–335. [Google Scholar]
- Tlaskalová-Hogenová, H.; Štepánková, R.; Hudcovic, T.; Tucková, L.; Cukrowska, B.; Lodinová-Žádnıková, R.; Kozáková, H.; Rossmanna, P.; Bártová, J.; Sokol, D.; et al. Commensal bacteria (normal microflora), mucosal immunity and chronic inflammatory and autoimmune diseases. Immunol. Lett. 2004, 93, 97–108. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rosenthal, M.; Aiello, A.; Larson, E.; Chenoweth, C.; Foxman, B. Healthcare Workers’ Hand Microbiome May Mediate Carriage of Hospital Pathogens. Pathogens 2014, 3, 1-13. https://doi.org/10.3390/pathogens3010001
Rosenthal M, Aiello A, Larson E, Chenoweth C, Foxman B. Healthcare Workers’ Hand Microbiome May Mediate Carriage of Hospital Pathogens. Pathogens. 2014; 3(1):1-13. https://doi.org/10.3390/pathogens3010001
Chicago/Turabian StyleRosenthal, Mariana, Allison Aiello, Elaine Larson, Carol Chenoweth, and Betsy Foxman. 2014. "Healthcare Workers’ Hand Microbiome May Mediate Carriage of Hospital Pathogens" Pathogens 3, no. 1: 1-13. https://doi.org/10.3390/pathogens3010001