3.1. No-Choice Bioassay with Fresh and Aged Baits
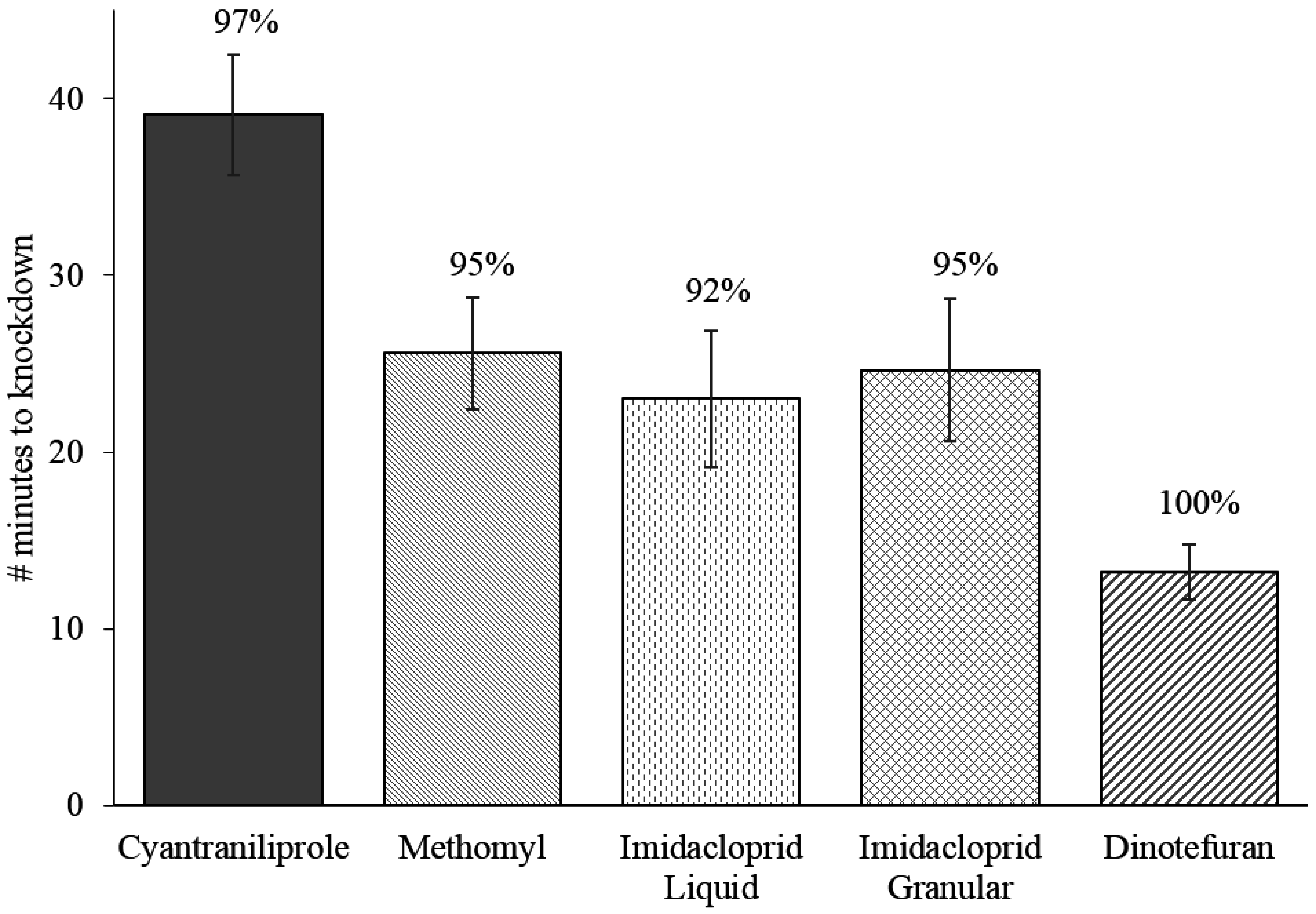
The average time to knockdown for flies in minutes was 39.1 ± 3.4 (mean ± SEM) for cyantraniliprole bait, 25.6 ± 3.2 for methomyl bait, 23.0 ± 3.9 for imidacloprid liquid bait, 24.7 ± 4.0 for imidacloprid granular bait, and 13.2 ± 1.5 for dinotefuran bait (
Figure 2). Flies exposed to cyantraniliprole bait had a significantly slower time to knockdown compared to flies exposed to all other baits. Imidacloprid liquid bait had a significantly slower time to knockdown than dinotefuran bait, but was not significantly different from imidacloprid granular and methomyl baits (
F = 12.4;
df = 4,276;
p ≤ 0.0001).
Figure 2.
Mean (±SEM) time to knockdown of flies after ingestion of different fly baits. Four replications of 15 flies were allowed to feed on the baits in a no-choice bioassay for a total of
n = 60 for each fly bait. Note: Percent knockdown of flies (numbers above bars) used in time to knockdown experiment are shown in
Figure 3 as week 0.
Figure 2.
Mean (±SEM) time to knockdown of flies after ingestion of different fly baits. Four replications of 15 flies were allowed to feed on the baits in a no-choice bioassay for a total of
n = 60 for each fly bait. Note: Percent knockdown of flies (numbers above bars) used in time to knockdown experiment are shown in
Figure 3 as week 0.
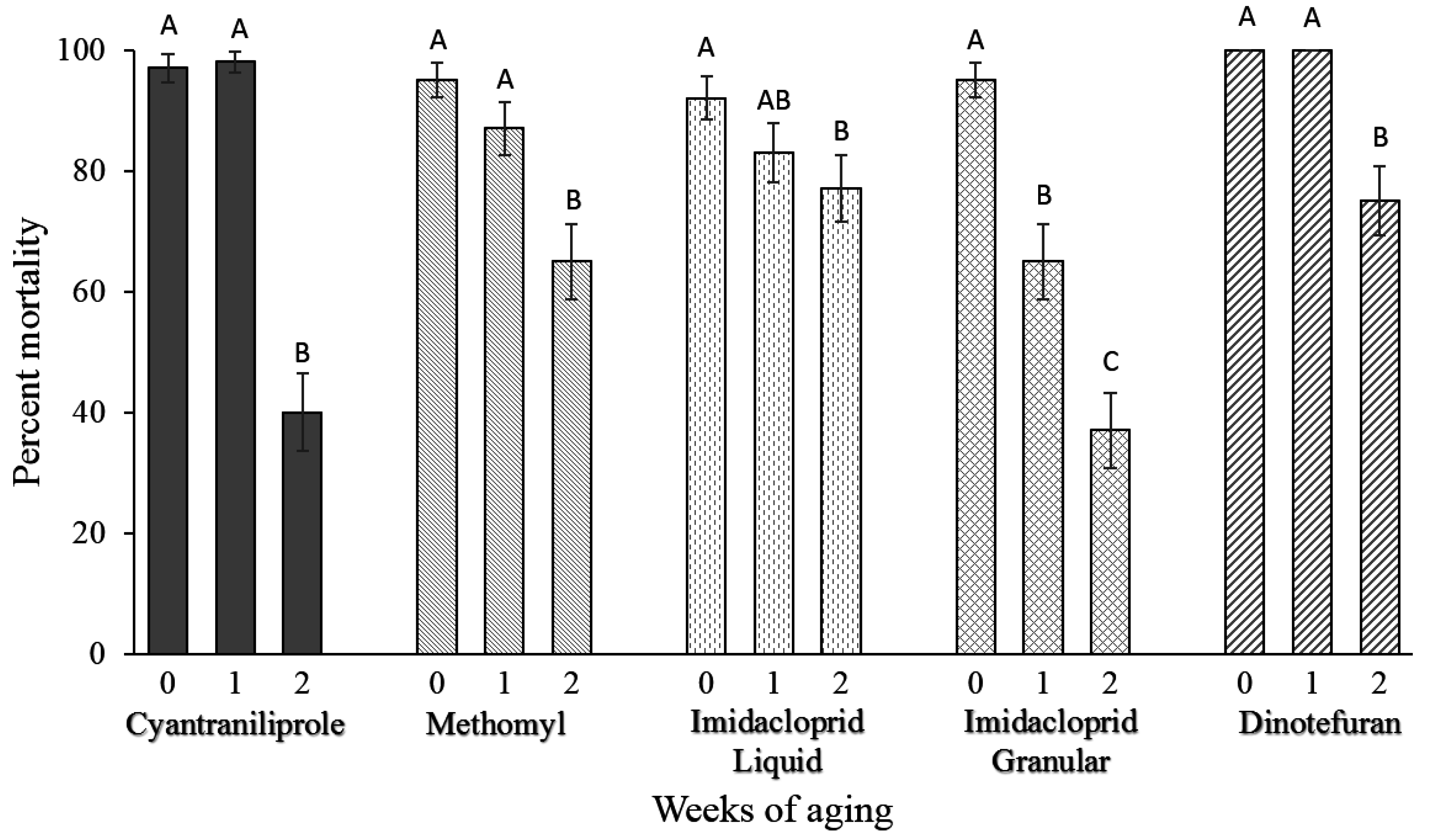
Over the 2-week aging period, degradation of the baits was observed leading to a significant decrease in the percent knockdown of flies killed by different baits (
F = 21.4,
df = 2,885,
p ≤ 0.0001) (
Figure 3). Significant degradation of baits was not observed between week 0 and week 1, but was observed from week 1 to week 2 for cyantraniliprole (
F = 62.5;
df = 2177,
p ≤ 0.0001), methomyl (
F = 10.9;
df = 2177;
p ≤ 0.0001), and dinotefuran baits (
F = 19.7;
df = 2177;
p ≤ 0.0001). Imidacloprid liquid bait did not show a significant degradation from week 0 to week 1 or from week 1 to week 2, but there was a significant difference from week 0 to week 2 (
F = 2.5;
df = 2177;
p ≤ 0.0001). Strongest quality degradation was observed with imidacloprid granular bait, with significant decrease in the efficacy between weeks 0 and 1, and between weeks 1 and 2 (
F = 25.0;
df = 2177;
p ≤ 0.0001).
Figure 3.
Percentage (±SEM) of flies killed by the fly baits over a two-week aging period. SEM bars with different letters for the same treatment represent a significant difference (T-test α = 0.05). Four replications of 15 flies were allowed to feed on the baits in a no-choice bioassay for a total of n = 60 for each fly bait during each week. Note: In control treatment, one fly died in one of the four replicates (1.7% control mortality).
Figure 3.
Percentage (±SEM) of flies killed by the fly baits over a two-week aging period. SEM bars with different letters for the same treatment represent a significant difference (T-test α = 0.05). Four replications of 15 flies were allowed to feed on the baits in a no-choice bioassay for a total of n = 60 for each fly bait during each week. Note: In control treatment, one fly died in one of the four replicates (1.7% control mortality).
Cyantraniliprole was slower in killing the flies due to its mode of action. Cyantraniliprole is a diamide, which acts on the ryanodine receptor in the muscle cells of flies. Consequently, the muscle cells continuously release calcium causing a slow paralysis [
14]. This is a slow mode of action compared to carbamates and neonicotinoids. Carbamates are acetyl cholinesterase inhibitors [
15] and neonicotinoids are nicotinic agonists that interact with the nicotinic acetylcholine receptor [
16]. These different modes of actions account for the significant difference in the time to knockdown between cyantraniliprole and the other baits. It is important to note that flies did not recover from effects of active ingredients during the 2 h study period, but were discarded after the 2 h experimental time and could have recovered after being discarded.
A previous study presented results that contrast those presented in this article [
3]. In that no-choice study, the UCR lab strain of flies took anywhere from 1 to 3 days to achieve 95%–100% mortality. In our no-choice lab study, 95%–100% knockdown was observed on fresh baits within an hour. This contrast in data could be due to a number of factors including fly strain and different bioassays. The Murillo study released 50 flies into 1 L test arena while in the presented study, 15 flies were released into a 1 L mason jar. This may have elicited feeding in a shorter period of time due to lower competition for food. Flies in the present study were also allowed to acclimate in the test jar for an hour before bait was placed while the Murillo study placed flies directly in the arena with baits.
Significant degradation of baits was observed even when the baits were allowed to age in a covered outdoor environment. Baits were exposed to natural temperature and humidity levels and absorbed water readily. The hydrophilic nature of the baits may have contributed to either a degradation of the active ingredient, the attractive agent, or both. Quick degradation of baits, even in protected conditions, demonstrates the need for repeated reapplication of baits for maximum efficacy.
3.2. Choice Bioassay
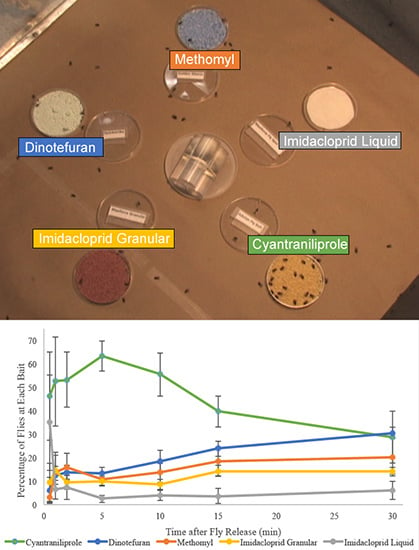
Significant differences were observed in the number of flies attracted to different baits. At the 0.5 (
F = 1.2;
df = 4, 12;
p = 0.37) and 1-min (
F = 1.4;
df = 4, 12;
p = 0.29) readings after initiation of bait exposure, no significant difference was observed in the attractance of baits, but starting after the 2-min reading, cyantraniliprole bait attracted significantly more flies than all other baits (
F = 3.9;
df = 4, 12;
p = 0.03). The greatest differentiation in bait attractance occurred at 5-min when approximately 63% of the responding flies were on the cyantraniliprole bait and only 2.5% of flies were on imidacloprid liquid bait, which attracted significantly fewer flies than all other products (
F = 24.0;
df = 4, 12;
p ≤ 0.0001). At the 15-min reading, cyantraniliprole bait continued to be more attractive than imidacloprid granular, methomyl, and imidacloprid liquid baits (
F = 5.0;
df = 4, 12;
p = 0.013), but after the 30-min reading, there was no significant difference in the number of flies attracted to the different baits (
F = 1.8;
df = 4, 12;
p = 0.19). On average, cyantraniliprole was the most preferred bait throughout the replications (
F = 18.1;
df = 4,127;
p ≤ 0.0001) (
Figure 4).
Figure 4.
Percentage (±SEM) of responding flies on each fly bait at different time intervals over a 30-min period. Four replications of approximately 150 flies were performed (n = 4).
Figure 4.
Percentage (±SEM) of responding flies on each fly bait at different time intervals over a 30-min period. Four replications of approximately 150 flies were performed (n = 4).
When given the choice of a variety of commercially available baits, the flies preferred cyantraniliprole. Bait preference has been evaluated in field scenarios, but this experiment shows clearly how the baits compare in terms of attractance over a short time. In situations where the user prefers a more attractive bait over a bait with a quick time to mortality, cyantraniliprole seems an ideal choice. Flies were observed spending a longer period of time on cyantraniliprole than the other baits. This may be due to the combination of a slow mode of action and the presence of food attractant in this bait. The other baits used in this study have a relatively quick mode of action and the feeding time was shorter.
Methomyl, imidacloprid liquid, imidacloprid granular, and dinotefuran baits all incorporate the sex pheromone, (
Z)-9 tricosene into their respective bait formulations. The cyantraniliprole bait, on the other hand, incorporates a food matrix as the attractant and no sex pheromone. The difference in attractants may be one of the reasons for the cyantraniliprole bait’s success in controlling the flies and demonstrates that a fly bait does not need to contain the sex pheromone, (
Z)-9-tricosene to be attractive to house flies. A previous study shows the addition of (
Z)-9 tricosene to water did not significantly increase the number of flies attracted [
17].
After 30-min, greater than 75% of the flies were dead or immobile, and most flies had fed on at least one of the baits and were being physically affected by the insecticides. An active ingredient with minimum immediate effect on the flies may be important to guarantee the consumption of sufficient bait to cause higher levels of mortality. Therefore, slow-acting baits can still be effective if they are preferred while flies are feeding.
3.3. Field Attractiveness Trial
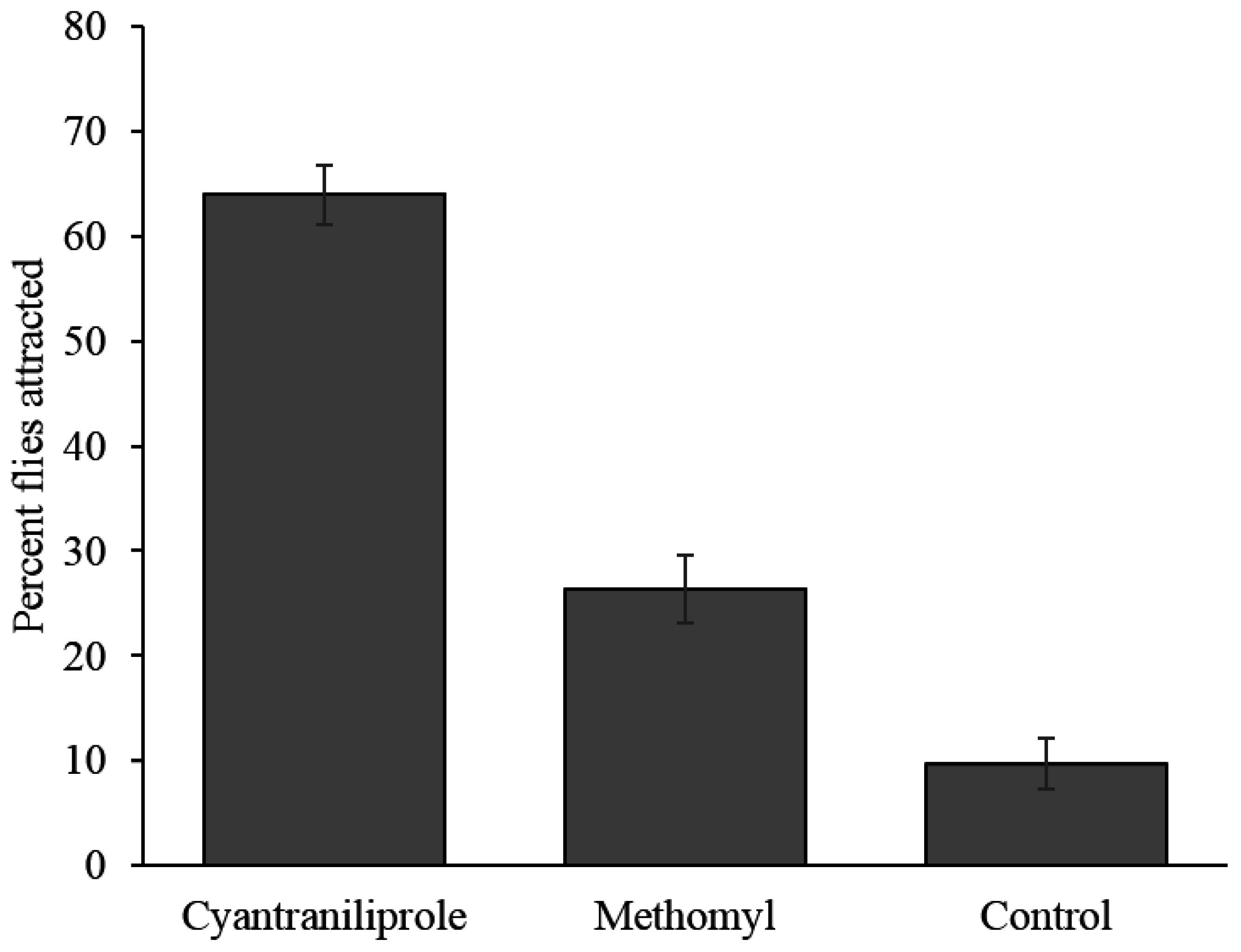
Flies did not distribute themselves randomly among the treatments in the field and had a significantly higher proportion of them preferring cyantraniliprole bait (64.0%) over methomyl bait (26.3%) and the control (9.7%) (
F = 95.1;
df = 2, 24;
p ≤ 0.0001) (
Figure 5). The number of flies attracted to methomyl bait was not significantly different from the control (
p = 0.10).
Figure 5.
Percent (±SEM) of flies attracted to the different treatments in field study. Nine replications were done (n = 9).
Figure 5.
Percent (±SEM) of flies attracted to the different treatments in field study. Nine replications were done (n = 9).
Previous results [
3] show similar results with cyantraniliprole and spinosad (not evaluated in this study) being the most attractive baits in field studies. These authors also presented evidence that suggested resistance to methomyl, the active ingredient in Golden Malrin
®, by flies used in the study. The populations between the two study areas are different, but methomyl has been used commercially for decades. Consequently, behavioral or physiological resistance may have been a factor that contributed to cyantraniliprole bait being more attractive than methomyl bait in the field. Behavioral resistance to a bait would decrease the attractiveness of that bait to flies. This could happen through an increased sensitivity to certain aspects of the bait which would cause the flies to avoid consuming the bait [
18].
3.4. Field Efficacy
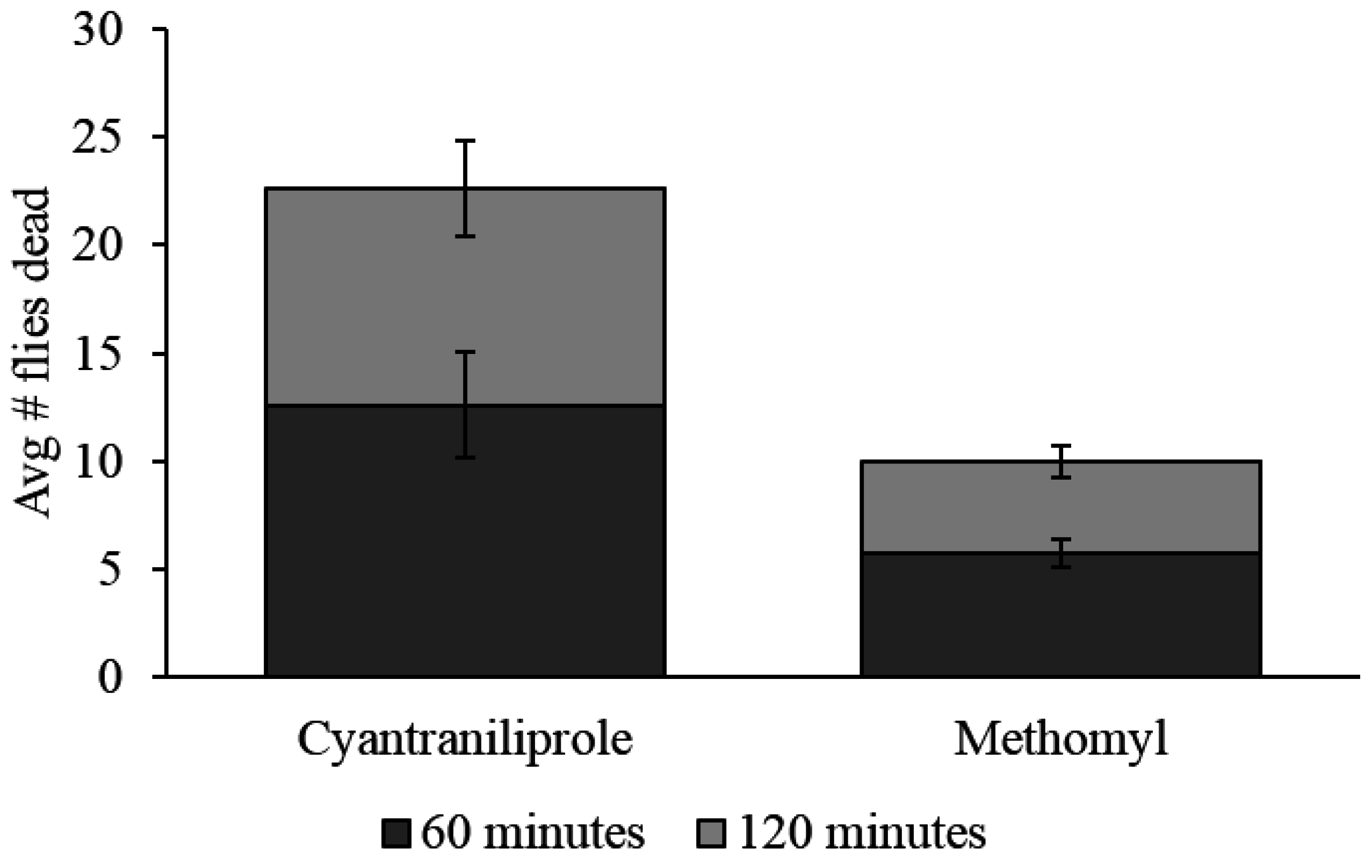
There was a significant difference between all baits when evaluated in the field. Cyantraniliprole bait caused a significantly higher mortality of flies than methomyl bait, which caused a significantly higher fly mortality than the control (
F = 37.8;
df = 2, 10;
p ≤ 0.0001). On average, after 60 min, 12.6 dead flies were found in the sampling area for cyantraniliprole compared to only 5.8 for methomyl. After 120 min, 22.6 flies were found in the sampling area for cyantraniliprole compared with 10 for methomyl (
Figure 6). Mortality was not observed in the control sampling areas.
Figure 6.
Average number of dead flies (±SEM) found in the sampling areas for each treatment after 60-min and 120-min. Three replications were done for a total of n = 3.
Figure 6.
Average number of dead flies (±SEM) found in the sampling areas for each treatment after 60-min and 120-min. Three replications were done for a total of n = 3.
During the field-testing of the baits, many dead flies were observed outside the sampling area for both cyantraniliprole and methomyl. However, numbers of cadavers outside the sampling area were higher for the cyantraniliprole than the methomyl treatment. Due to the slower mode of action for cyantraniliprole, flies exposed to this pesticide were likely able to disperse greater distances than flies affected by the methomyl bait. This behavior may have affected the number of flies observed in the sampling area. This is in contrast with flies affected by methomyl, which die quickly and, therefore, could not fly away from the established sampling area. Flies that have developed methomyl resistance would have been able to disperse from the test arena, therefore the low number of dead flies observed in methomyl testing arenas could be an indication of resistance in this field population. Despite the possibility of resistance and the limited number of active ingredients tested, the cyantraniliprole-containing bait was more effective than the methomyl-containing bait in this field trial.