An Overview of Pest Species of Bactrocera Fruit Flies (Diptera: Tephritidae) and the Integration of Biopesticides with Other Biological Approaches for Their Management with a Focus on the Pacific Region
Abstract
:1. Introduction
2. Overview of Pest Bactrocera Species
| Species | Hosts | Distribution |
|---|---|---|
| CATEGORY A pests | ||
| B. carambolae Drew & Hancock | Polyphagous fruit pest | Vietnam to Indonesia. Introduced into South America. |
| B. correcta (Bezzi) | Polyphagous fruit pest | Pakistan to Vietnam. |
| B. cucurbitae (Coquillett) | Primarily Cucurbitaceae (fruit & flower) | Tropical Asia (widespread). Introduced into Africa and Oceania. |
| B. dorsalis (Hendel) | Polyphagous fruit pest | Tropical Asia (widespread). Introduced into Africa and Oceania. |
| B. latifrons (Hendel) | Mainly Solanaceae | Pakistan to Taiwan; south to Sulawesi. Introduced into Hawaii and Africa. |
| B. neohumeralis (Hardy) | Polyphagous fruit pest | Australia, New Guinea. |
| B. oleae (Gmelin) | Olive | Africa. Introduced into southern Europe, the Middle East and California. |
| B. tryoni (Froggatt) | Polyphagous fruit pest | Australia. Introduced in Oceania. |
| B. zonata (Saunders) | Polyphagous fruit pest | India to Vietnam. |
| CATEGORY B pests | ||
| B. aquilonis (May) | Polyphagous fruit pest | Australia. May be conspecific with B. tryoni. |
| B. caryeae (Kapoor) | Oligophagous fruit pest | Southern India. |
| B. cucumis (French) | Cucurbitaceae (fruit) | Australia. |
| B. curvipennis (Froggatt) | Polyphagous fruit pest | New Caledonia. |
| B. facialis (Coquillett) | Polyphagous fruit pest | Tonga. |
| B. frauenfeldi (Schiner) | Polyphagous fruit pest | Australia, Micronesia (except Marianas), New Guinea, Solomon Islands. |
| B. jarvisi (Tryon) | Polyphagous fruit pest | Australia. |
| B. kandiensis Drew & Hancock | Oligophagous fruit pest | Sri Lanka. |
| B. kirki (Froggatt) | Polyphagous fruit pest | French Polynesia, Fiji (Rotuma), Niue, Samoa (American & Western), Tonga. |
| B. kraussi (Hardy) | Polyphagous fruit pest | Australia. |
| B. melanotus (Coquillett) | Polyphagous fruit pest | Cook Islands. |
| B. minax (Enderlein) | Citrus | Bhutan, China, Nepal. |
| B. musae (Tryon) | Banana | Australia, New Guinea. |
| B. occipitalis (Bezzi) | Oligophagous fruit pest | Kalimantan, Philippines. |
| B. passiflorae (Froggatt) | Polyphagous fruit pest | Fiji, Wallis & Futuna, Niue. |
| B. psidii (Froggatt) | Polyphagous fruit pest | New Caledonia. |
| B. tau (Walker) | Cucurbitaceae (fruit) | Pakistan to Philippines; south to Sumatra & Sulawesi. |
| B. trilineola Drew | Polyphagous fruit pest | Vanuatu. |
| B. tsuneonis (Miyake) | Citrus | China, Japan. |
| B. xanthodes (Broun) | Polyphagous fruit pest | Cook Islands, Fiji, French Polynesia (Austral group), Niue, Samoa (American & Western), Tonga, Wallis & Futuna. |
| CATEGORY C pests | ||
| B. albistrigata (deMeijere) | Oligophagous fruit pest | Indonesia, Malaysia |
| B. atrisetosa (Perkins) | Cucurbitaceae (fruit) | New Guinea. |
| B. bryoniae (Tryon) | Banana, chili pepper | Australia, New Guinea. |
| B. caudata (Fabricius) | Cucurbitaceae (flowers) | India to Taiwan; south to Indonesia (Lesser Sundas). |
| B. decipiens (Drew) | Cucurbitaceae (fruit) | New Guinea. |
| B. depressa (Shiraki) | Cucurbitaceae (fruit) | Japan, South Korea, Taiwan. |
| B. distincta (Malloch) | Sapotaceae | Fiji, Samoa (American & Western), Tonga, Wallis Is. |
| B. diversa (Coquillett) | Cucurbitaceae (flowers) | Pakistan to Vietnam. |
| B. halfordiae (Tryon) | Oligophagous fruit pest | Australia. |
| B. melas (Perkins & May) | Polyphagous fruit pest | Australia. May be conspecific with B. tryoni. |
| B. moluccensis (Perkins) | Inocarpus fagifer | Java to New Guinea, Solomon Islands. |
| B. obliqua (Malloch) | Guava, Syzygium | New Guinea. |
| B. passiflorae (sp. nr.) | Oligophagous fruit pest | Fiji, Tokelau, Tonga (Niuas Group), Tuvalu. |
| B. pyrifoliae Drew & Hancock | Guava, peach, pear | Thailand, Vietnam. (Member of B. dorsalis complex). |
| B. scutellaris (Bezzi) | Cucurbitaceae (flowers) | India to Vietnam; south to peninsular Malaysia. |
| B. scutellata (Hendel) | Cucurbitaceae (flowers) | Bhutan to Vietnam; north to Taiwan & Japan (Ryukus). |
| B. strigifinis (Walker) | Cucurbitaceae (flowers) | Australia, New Guinea. |
| B. triangularis (Drew) | Cucurbitaceae (flowers) | New Guinea. |
| B. trivialis (Drew) | Oligophagous fruit pest | New Guinea. (Member of B. dorsalis complex). |
| B. tuberculata (Bezzi) | Oligophagous fruit pest | Bangladesh to Vietnam. |
| B. umbrosa (Fabricius) | Breadfruit, jackfruit | Widespread from southern Thailand through New Guinea to New Caledonia. |
| CATEGORY D pests | ||
| B. arecae (Fabricius) | Betel nut | Malaysia (Peninsular), Singapore, Thailand. |
| B. atramentata (Hering) | Pometia pinnata | New Guinea. |
| B. bancroftii (Tryon) | Mulberry | Australia. |
| B. expandens (Walker) | Mangosteen | Australia, Indonesia (Moluccas), New Guinea. |
| B. hastigerina (Hardy) | Spondias | New Guinea, Solomon Islands. |
| B. hochii (Zia) | Luffa cylindrica (fruit) | Bangladesh to Vietnam; south to Sumatra. |
| B. lineata (Perkins) | Pometia pinnata | New Guinea. |
| B. mesomelas (Bezzi) | Guava | Africa. |
| B. mucronis (Drew) | Guava, sweetsop | New Caledonia. |
| B. munda (Bezzi) | Squash (fruit) | Philippines, Taiwan. |
| B. murrayi (Perkins) | Mango, Surinam cherry | Australia, New Guinea. |
| B. mutabilis (May) | Guava, kumquat | Australia. |
| B. nigrofemoralis White & Tsuruta | Pomelo, mamey sapote | Indian subcontinent, including Sri Lanka. |
| B. nigrotibialis (Perkins) | Guava, rose-apple | India to Vietnam; south to Indonesia (Lesser Sundas). |
| B. ochroma Drew & Romig | Mango | Indonesia. |
| B. perfusca (Aubertin) | Mango, rose-apple | French Polynesia (Marquesas only). |
| B. pruniae Drew & Romig | Peach | Vietnam. |
| B. quadrisetosa (Bezzi) | Pometia pinnata | Solomon Islands, Vanuatu. |
| B. speculifera (Walker) | Breadfruit | New Guinea. |
| B. tapervitta Mahmood | Luffa cylindrica (fruit) | Philippines. |
| B. trichosanthes Drew & Romig | Bittergourd (fruit) | Indonesia (Java), Malaysia (Peninsular & East), Thailand. |
| B. trimaculata (Hardy & Adachi) | Bittergourd (fruit) | Philippines. |
| B. versicolor (Bezzi) | Sapodilla | India, Sri Lanka. |
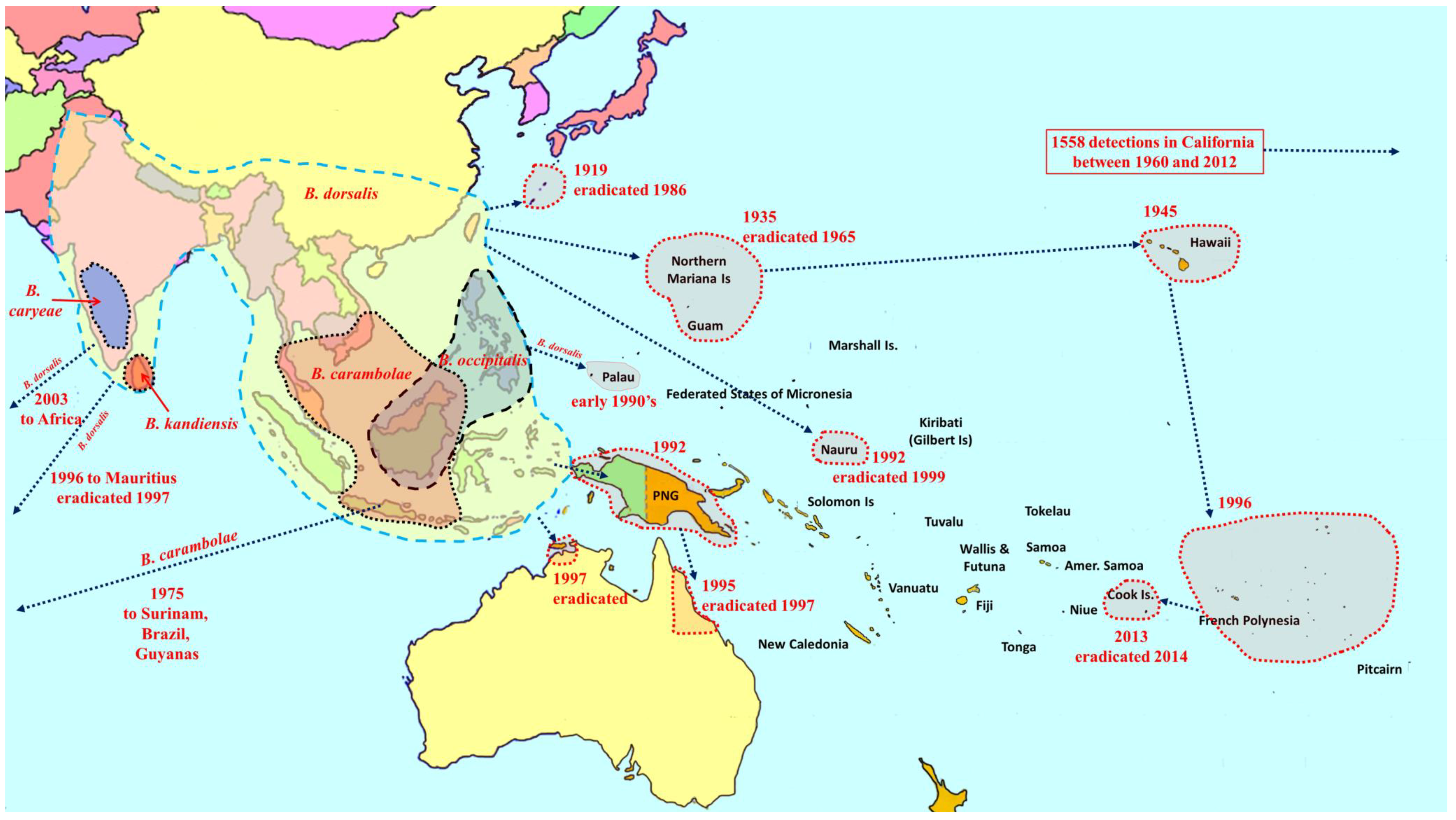

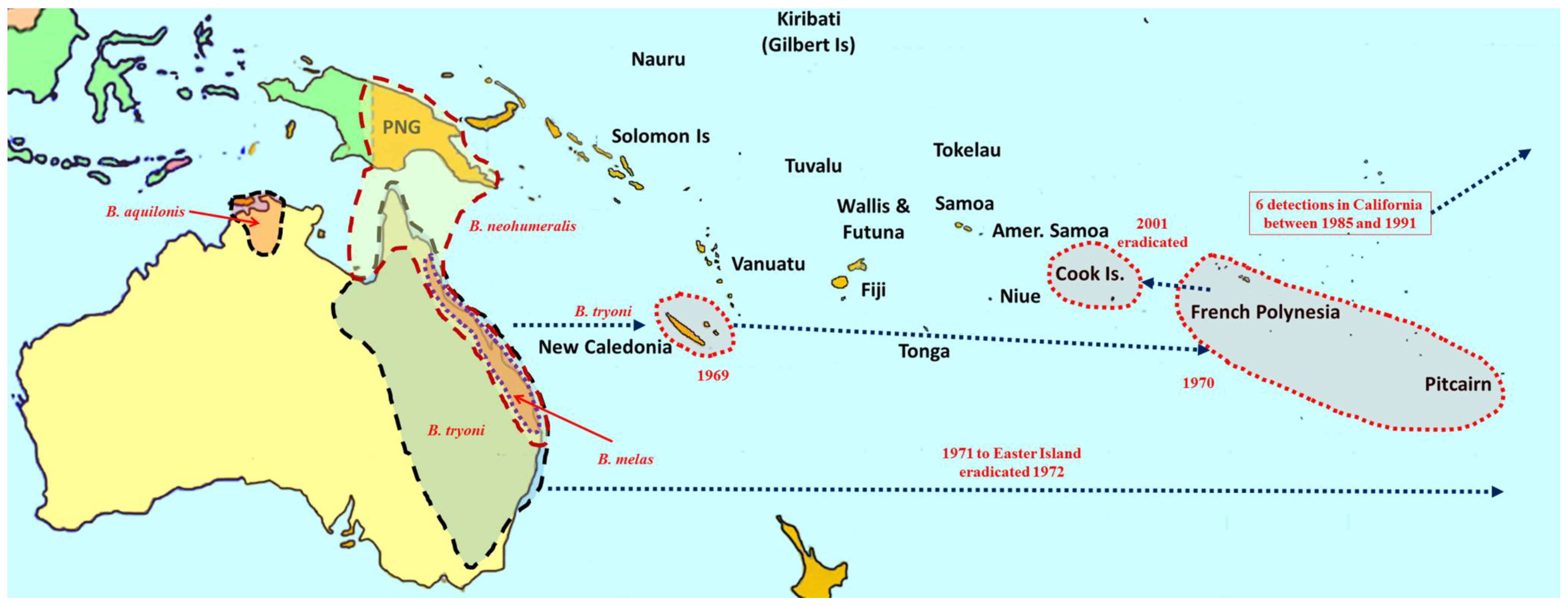
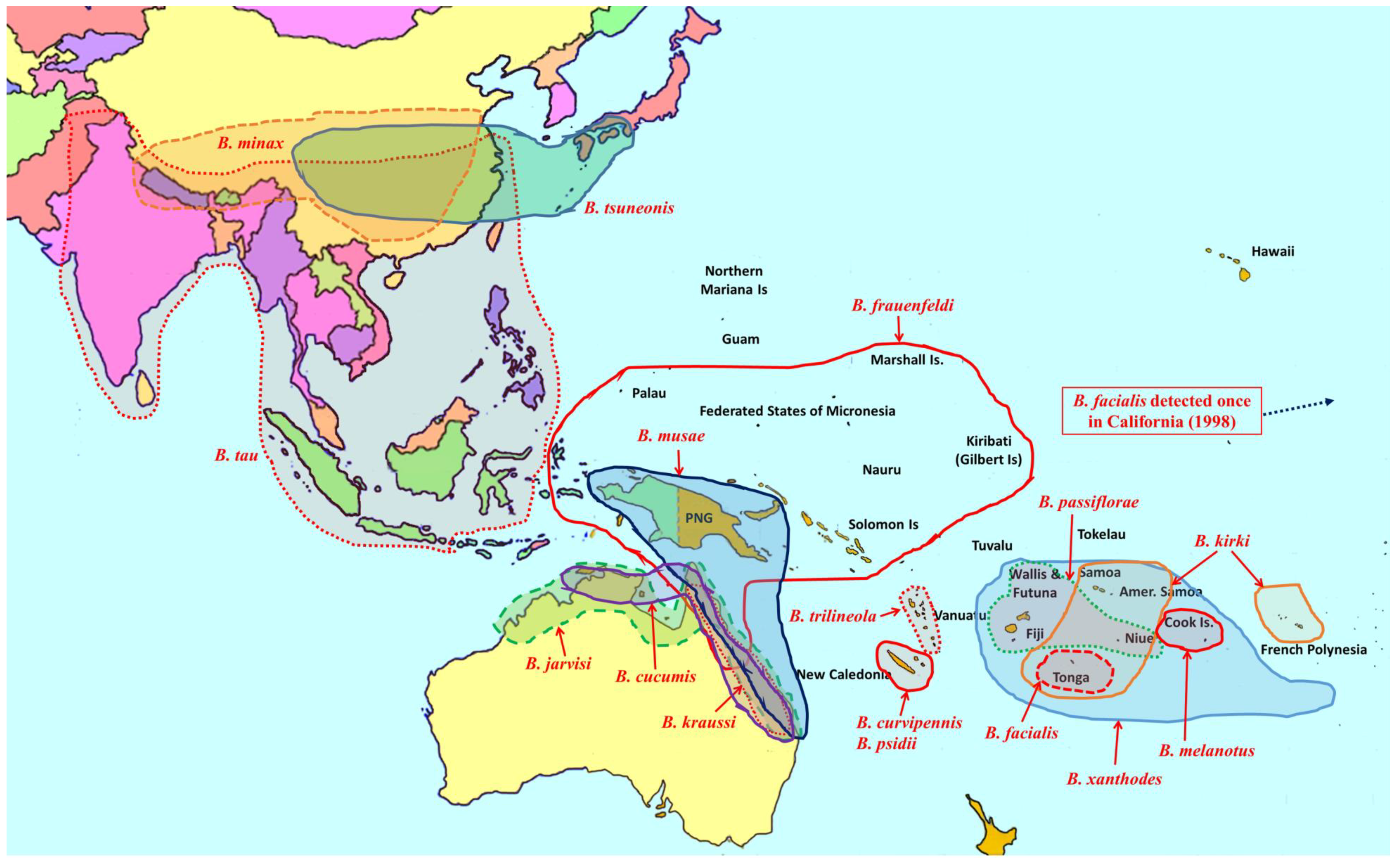
3. Area-Wide Control Technologies
3.1. Insecticide Cover Sprays, Protein Bait Sprays, and Soil Drenches
3.2. Male Annihilation
3.3. Sterile Insect Releases
3.4. Releases of Natural Enemies
3.5. Cultural Controls
3.5.1. Sanitation
3.5.2. Fruit Bagging
3.5.3. Augmentorium
3.5.4. Soil Disturbance
4. Examples of Successful Fruit Fly IPM Systems against Bactrocera spp.
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- White, I.M.; Elson-Harris, M.M. Fruit Flies of Economic Significance: Their Identification and Bionomics; CABI International: Wallingford, UK, 1992. [Google Scholar]
- Vargas, R.I.; Mau, R.F.L.; Jang, E.B.; Faust, R.M.; Wong, L. The Hawaii fruit fly areawide pest management programme. In Areawide Pest Management: Theory and Implementation; Koul, O., Cuperus, G., Eds.; CABI Books: London, UK, 2008; pp. 300–325. [Google Scholar]
- Kogan, M.; Bajwa, W.I. Integrated pest management: A global reality? Ann. Soc. Entomol. Brasil 1999, 28, 1–25. [Google Scholar] [CrossRef]
- Pedigo, L.P.; Rice, M. Entomology and Pest Management, 6th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2008. [Google Scholar]
- Dacine Fruit Flies of the Asia-Pacific. Available online: http://www.herbarium.hawaii.edu/fruitfly/ (accessed on 10 January 2015).
- Drew, R.; Tsuruta, K.; White, I. A new species of pest fruit fly (Diptera: Tephritidae: Dacinae) from Sri Lanka and Africa. Afr. Entomol. 2005, 13, 149–154. [Google Scholar]
- Rousse, P.; Harris, E.J.; Quilici, S. Fopius arisanus, an egg-pupal parasitoid of Tephritidae. Overview. Biocontrol News Inform. 2005, 26, 59N–69N. [Google Scholar]
- Allwood, A.J.; Chinajariyawong, A.; Drew, R.A.I.; Hamacek, E.L.; Hancock, D.L.; Hengsawad, C.; Jipanin, J.C.; Jirasurat, M.; Kong Krong, C.; Kritsaeneepaiboon, S.; et al. Host plant records for fruit flies (Diptera: Tephritidae) in Southeast Asia. Raffles Bull. Zool. 1999, 47, 1–92. [Google Scholar]
- Allwood, A.J.; Leblanc, L.; Tora Vueti, E.; Bull, R. Fruit Fly Control Methods for Pacific Island Countries and Territories. Available online: http://bem.bime.ntu.edu.tw/clchuang/PAL40 (accessed on 10 January 2015).
- Allwood, A.J.; Vueti, E.T.; Leblanc, L.; Bull, R. Eradication of introduced Bactrocera species (Diptera: Tephritidae) in Nauru using male annihilation and protein bait application techniques. In Turning the Tide: The Eradication of Invasive Species; Proceedings of the International Conference on Eradication Of Island Invasives, Auckland, New Zealand, 19–23 February 2001; Veitch, C.R., Clout, M.N., Eds.; IUCN Publications Services Unit: Cambridge, UK, 2002; pp. 19–25. [Google Scholar]
- Mwatawala, M.W.; DeMeyer, M.; Makundi, R.H.; Maerere, A.P. Host range and distribution of fruit-infesting pestiferous fruit flies (Diptera, Tephritidae) in selected areas of Central Tanzania. Bull. Entomol. Res. 2009, 99, 629–641. [Google Scholar] [CrossRef] [PubMed]
- Vargas, R.I.; Mau, R.F.L.; Stark, J.D.; Piñero, J.C. Evaluation of methyl eugenol and cue-lure traps with solid lure and insecticide dispensers for monitoring and male annihilation in Hawaii. J. Econ. Entomol. 2010, 103, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Drew, R.A.I. The tropical fruit flies (Diptera: Tephritidae: Dacinae) of the Australasian and Oceanian regions. Mem. Queensl. Mus. 1989, 26, 1–521. [Google Scholar]
- Drew, R.A.I.; Hancock, D.L. The Bactrocera dorsalis complex of fruit flies (Diptera: Tephritidae: Dacinae) in Asia. Bull. Entol. Res. 1994, 84 (Suppl. S2), 1–68. [Google Scholar] [CrossRef]
- White, I.M. Taxonomy of the Dacina (Diptera: Tephritidae) of Africa and the Middle East; Entomological Society of Southern Africa: Hatfield, South Africa, 2006. [Google Scholar]
- Drew, R.A.I.; Romig, M.C. Tropical Fruit Flies of South-East Asia; CAB International: Wallingford, UK, 2013. [Google Scholar]
- Hancock, D.L.; Hamacek, E.L.; Lloyd, A.C.; Elson-Harris, M.M. The Distribution and Host Plants of Fruit Flies (Diptera: Tephritidae) in Australia; Queensland Department of Primary Industries: Brisbane, Queensland, Australia, 2000; pp. 1–75.
- Leblanc, L.; Tora Vueti, E.; Drew, R.A.I.; Allwood, A.J. Host plant records for fruit flies (Diptera: Tephritidae: Dacini) in the Pacific Islands. Proc. Hawaiian Entomol. Soc. 2012, 44, 11–53. [Google Scholar]
- Leblanc, L.; Vueti, E.T.; Allwood, A.J. Host plant records for fruit flies (Diptera: Tephritidae: Dacini) in the Pacific Islands: 2. Infestation statistics on economic hosts. Proc. Hawaiian Entomol. Soc. 2013, 45, 83–117. [Google Scholar]
- Clarke, A.R.; Armstrong, K.F.; Carmichael, A.E.; Milne, J.R.; Raghu, S.; Roderick, G.K.; Yeates, D.K. Invasive phytophagous pests arising through a recent tropical evolutionary radiation: The Bactrocera dorsalis complex of fruit flies. Annu. Rev. Entomol. 2005, 50, 293–319. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schutze, M.K.; Aketarawong, N.; Amornsak, W.; Armstrong, K.; Augustinos, A.A.; Barr, N.; Bo, W.; Bourtzis, K.; Boykin, L.M.; Cáceres, C.; et al. Synonymization of key pest species within the Bactrocera dorsalis species complex (Diptera: 3 Tephritidae): Taxonomic changes based on 20 years of integrative morphological, molecular, 4 cytogenetic, behavioral, and chemoecological data. Syst. Entomol. 2014. [Google Scholar] [CrossRef]
- Papadopoulos, N.T.; Plant, R.E.; Carey, J.R. From trickle to flood: The large-scale, cryptic invasion of California by tropical fruit flies. Proc. R. Soc. Series B. 2013, 280. [Google Scholar] [CrossRef]
- Suckling, D.M.; Kean, J.M.; Stringer, L.D.; Caceres-Barrios, C.; Hendrichs, J.; Reyes-Flores, J. Eradication of tephritid fruit fly pest populations: Outcomes and prospects. Pest Manag. Sci. 2014. [Google Scholar] [CrossRef]
- Clarke, A.R.; Powell, K.S.; Weldon, C.W.; Taylor, P.W. The ecology of Bactrocera tryoni (Diptera: Tephritidae): What do we know to assist pest management? Ann. Appl. Biol. 2011, 158, 26–54. [Google Scholar] [CrossRef] [Green Version]
- Roessler, Y. Insecticidal bait and cover spray. In Fruit Flies, Their Biology, Natural Enemies and Control; Robinson, A.S., Hooper, G., Eds.; Elsevier: Amsterdam, the Netherlands, 1989; Volume 3A, pp. 329–335. [Google Scholar]
- Prokopy, R.J.; Papaj, D.R.; Hendrichs, J.; Wong, T.T.Y. Behavioral responses of Ceratitis capitata flies to bait spray droplets and natural food. Entomol. Exp. Appl. 1992, 64, 247–257. [Google Scholar] [CrossRef]
- Peck, S.L.; McQuate, G.T. Field tests of environmentally friendly malathion replacements to suppress wild Mediterranean fruit fly (Diptera: Tephritidae) populations. J. Econ. Entomol. 2000, 93, 280–289. [Google Scholar] [CrossRef] [PubMed]
- Prokopy, R.J.; Miller, N.W.; Piñero, J.C.; Barry, J.D.; Tran, L.C.; Oride, L.; Vargas, R.I. Effectiveness of GF-120 fruit fly bait spray applied to border area plants for control of Melon flies (Diptera: Tephritidae). J. Econ. Entomol. 2003, 96, 1485–1493. [Google Scholar] [CrossRef] [PubMed]
- Piñero, J.C.; Mau, R.F.L.; McQuate, G.T.; Vargas, R.I. Novel bait stations for attract-and-kill of pestiferous fruit flies. Entomol. Exp. Appl. 2009, 133, 208–216. [Google Scholar] [CrossRef]
- Stark, J.D.; Vargas, R. An Evaluation of alternative insecticides to diazinon for control of Tephritid fruit flies (Diptera: Tephritidae) in Soil. J. Econ. Entomol. 2009, 102, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Stark, J.D.; Vargas, R.I.; Souder, S.; Fox, A.J.; Smith, T.R.; Mackey, B. A Comparison of the Bioinsecticide, Spinosad, the Semi-synthetic Insecticide, Spinetoram and Synthetic Insecticides as Soil Drenches for Control of Tephritid Fruit Flies. Biopestic. Int. 2013, 9, 120–126. [Google Scholar]
- Stark, J.D.; Vargas, R.I.; Souder, S.K.; Fox, A.J.; Smith, T.R.; Leblanc, L.; Mackey, B. Simulated field applications of insecticide soil drenches for control of tephritid fruit flies. Biopestic. Int. 2014, 10, 136–142. [Google Scholar]
- Steiner, L.F.; Mitchell, W.C.; Harris, E.J.; Kozuma, T.T.; Fujimoto, M.S. Oriental fruit fly eradication by male annihilation. J. Econ. Entomol. 1965, 58, 961–964. [Google Scholar] [CrossRef]
- Koyama, J.; Tadashi, T.; Kenji, T. Eradication of the oriental fruit fly (Diptera: Tephritidae) from Okinawa Islands by male annihilation method. J. Econ. Entomol. 1984, 77, 468–472. [Google Scholar]
- Vargas, R.I.; Stark, J.D.; Kido, M.H.; Ketter, H.M.; Whitehand, L.C. Methyl eugenol and cue-lure traps for suppression of male oriental fruit flies and melon flies (Diptera: Tephritidae) in Hawaii: Effects of lure mixtures and weathering. J. Econ. Entomol. 2000, 93, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Vargas, R.I.; Leblanc, L.; Piñero, J.C.; Hoffman, K.M. Male annihilation, past, present, and future. In Trapping Tephritid Fruit Flies. Lures, Area-Wide Programs, and Trade Implications; Shelly, T., Epsky, N., Jang, E.B., Reyes-Flores, J., Vargas, R., Eds.; Springer: Berlin, Germany, 2014; pp. 493–511. [Google Scholar]
- Steiner, L.F.; Hart, W.G.; Harris, E.J.; Cunningham, R.T.; Ohinata, K.; Kamakahi, D.C. Eradication of the oriental fruit fly from the Mariana Islands by the methods of male annihilation and sterile insect release. J. Econ. Entomol. 1970, 63, 131–135. [Google Scholar] [CrossRef]
- Koyama, J. Eradication of the melon fly, Bactrocera cucurbitae by the sterile insect technique in Japan. In Proceedings of IAEA Training Course on the Use of Sterile Insect and Related Techniques for the Area-Wide Management of Insect Pests, Gainesville, FL, USA, 8 May–19 June 1996.
- McInnis, D.; Leblanc, L.; Mau, R. Melon fly (Diptera: Tephritidae) genetic sexing: All-male sterile fly releases in Hawaii. Proc. Hawaiian Entomol. Soc. 2007, 39, 105–110. [Google Scholar]
- Vargas, R.I.; Leblanc, L.; Putoa, R.; Eitam, A. Impact of introduction of Bactrocera dorsalis (Diptera: Tephritidae) and classical biological control releases of Fopius arisanus (Hymenoptera: Braconidae) on economically important fruit flies in French Polynesia. J. Econ. Entomol. 2007, 100, 670–679. [Google Scholar] [CrossRef] [PubMed]
- Vargas, R.I.; Leblanc, L.; Harris, E.J.; Manoukis, N.C. Regional suppression of Bactrocera fruit flies (Diptera: Tephritidae) in the Pacific through biological control and prospects for future introductions into other areas of the world. Insects 2012, 3, 727–742. [Google Scholar] [CrossRef]
- Allwood, A.J. Control strategies for fruit flies (Diptera: Tephritidae) in the South Pacific. In Management of Fruit Flies in the Pacific. A Regional Symposium, Nadi, Fiji 28–31 October 1996; Allwood, A.J., Drew, R.A.I., Eds.; Australian Centre for International Agricultural Research: Canberra, Australia, 1997; pp. 171–178. [Google Scholar]
- Klungness, L.M.; Jang, E.B.; Mau, R.F.L.; Vargas, R.I.; Sugano, J.S.; Fujitani, E. New sanitation techniques for controlling tephritid fruit flies (Diptera: Tephritidae) in Hawaii. J. Appl. Sci. Environ. Mgt. 2005, 9, 4–14. [Google Scholar]
- Steiner, L.F.; Rohwer, G.G.; Ayers, E.L.; Christenson, L.D. The role of attractants in the recent Mediterranean fruit fly eradication program in Florida. J. Econ. Entomol. 1961, 54, 30–35. [Google Scholar] [CrossRef]
- Steiner, L.F. Methyl eugenol as an attractant for oriental fruit fly. J. Econ. Entomol. 1952, 45, 241–248. [Google Scholar] [CrossRef]
- Vargas, R.I.; Miller, N.W.; Prokopy, R.J. Attraction and feeding responses of Mediterranean fruit fly and a natural enemy to protein baits laced with two novel toxins, Phloxine B and Spinosad. Entomol. Exp. Appl. 2002, 102, 273–282. [Google Scholar] [CrossRef]
- Canale, A.; Benelli, G.; Conti, B.; Lenzi, G.; Flamini, G.; Francini, A.; Cioni, P.L. Ingestion toxicity of three Lamiaceae essential oils incorporated in protein baits against the olive fruit fly, Bactrocera oleae (Rossi) (Diptera Tephritidae). Nat. Prod. Res. 2013, 27, 2091–2099. [Google Scholar] [CrossRef] [PubMed]
- Stark, J.D.; Vargas, R.I.; Miller, N.W. Toxicity of spinosad in protein bait to three economically important tephritid fruit fly species (Diptera: Tephritidae) and their parasitoids Hymenoptera: Braconidae). J. Econ. Entomol. 2004, 97, 911–915. [Google Scholar] [CrossRef] [PubMed]
- Moreno, D.S.; Mangan, R.L. Responses of the Mexican fruit fly (Diptera: Tephritidae) to twohydrolyzed proteins and incorporation of phloxine B to kill adults. In Light Activated Pest Control; Heitz, J.R., Downum, K.R., Eds.; ACS Symposium Series: Washington, DC, USA, 1995; pp. 257–279. [Google Scholar]
- DowElanco. Spinosad Technical Guide; DowElanco: Indianapolis, IN, USA, 1994. [Google Scholar]
- Nishida, T.; Bess, H.A.; Ota, A. Comparative effectiveness of malathion and malathion-yeast hydrolysate bait sprays for control of the melon fly. J. Econ. Entomol. 1957, 50, 680–684. [Google Scholar] [CrossRef]
- Piñero, J.C.; Mau, R.F.L.; Vargas, R.I. Managing oriental fruit fly (Diptera: Tephritidae) with spinosad-based protein bait sprays and sanitation in papaya orchards in Hawaii. J. Econ. Entomol. 2009, 102, 1123–1132. [Google Scholar] [CrossRef] [PubMed]
- Ekesi, S.; Dimbi, S.; Maniania, N.K. The role of entomopathogenic fungi in the integrated management of tephritid fruit flies (Diptera: Tephritidae) with emphasis on species occurring in Africa. In Use of Entomopathogenic Fungi in Biological Pest Management; Ekesi, S., Maniania, N.K., Eds.; Research SignPost: Kerala, India, 2007; pp. 239–274. [Google Scholar]
- Steiner, L.F.; Lee, R.K.S. Large area tests of a male-annihilation method for oriental fruit fly control. J. Econ. Entomol. 1955, 48, 311–317. [Google Scholar] [CrossRef]
- Bateman, M.A.; Insungza, V.; Arreta, P. The eradication of Queensland fruit fly from Easter Island. FAO Plant Prot. Bull. 1973, 21, 114. [Google Scholar]
- Vargas, R.I.; Stark, J.D.; Hertlein, M.; Mafra-Neto, A.; Coler, R.; Piñero, J.C. Evaluation of SPLAT with spinosad and methyl eugenol or cue-lure for “attract-and-kill” of oriental and melon fruit flies (Diptera: Tephritidae) in Hawaii. J. Econ. Entomol. 2008, 101, 750–768. [Google Scholar] [CrossRef] [PubMed]
- Vargas, R.I.; Piñero, J.C.; Mau, R.F.L.; Stark, J.D.; Hertlein, M.; Mafra-Neto, A.; Coler, R.; Getchell, A. Attraction and mortality of oriental fruit flies (Diptera: Tephritidae) to SPLAT-MAT-methyl eugenol with spinosad. Entomol. Exp. Appl. 2009, 131, 286–293. [Google Scholar] [CrossRef]
- Cunningham, R.T.; Suda, D.Y. Male annihilation of the oriental fruit fly, Dacus dorsalis Hendel (Diptera: Tephritidae): A new thickener and extender for methyl eugenol formulations. J. Econ. Entomol. 1985, 75, 503–504. [Google Scholar] [CrossRef]
- Vargas, R.I.; Souder, S.K.; Hoffman, K.; Mercogliano, J.; Smith, T.R.; Hammond, J.H.; Davis, B.J.; Brodie, M.; Dripps, J.E. Attraction and mortality of Bactrocera dorsalis to STATICTM Spinosad ME weathered under operational conditions in California and Florida: A reduced-risk male annihilation treatment. J. Econ. Entomol. 2014, 107, 1362–1369. [Google Scholar] [CrossRef] [PubMed]
- Vargas, R.I.; Souder, S.K.; Borges, R.; Mafra-Neto, A.; Mackey, B.; Chou, M.Y.; Spafford, H. Effectiveness of a sprayable male annihilation treatment with a biopesticide against fruit flies (Dipera: Tephritidae) attacking tropical fruits. Biopest. Int. 2014, 10, 1–10. [Google Scholar]
- Knipling, E.F. Possibilities of insect control or eradication through the use of sexually sterile males. J. Econ. Entomol. 1955, 48, 59–62. [Google Scholar] [CrossRef]
- Baumhover, A.H.; Graham, A.J.; Bitter, B.A.; Hopkins, D.E.; New, W.D.; Dudley, F.H.; Bushland, R.C. Screwworm control through release of sterilized flies. J. Econ. Entomol. 1955, 48, 462–466. [Google Scholar] [CrossRef]
- Knipling, E.F. The Basic Principles of Insect Population Suppression and Management; U.S. Department of Agriculture: Washington, DC, USA, 1979; pp. 1–659.
- Benelli, G.; Daane, K.M.; Canale, A.; Niu, C.-Y.; Messing, R.H.; Vargas, R.I. Sexual communication and related behaviours in Tephritidae: Current knowledge and potential applications for Integrated Pest Management. J. Pest Sci. 2014, 87, 385–405. [Google Scholar] [CrossRef]
- Benelli, G. Aggression in Tephritidae flies: Where, when, why? Future directions for research in integrated pest management. Insects 2015, 6, 38–53. [Google Scholar] [CrossRef]
- Steiner, L.F.; Harris, E.J.; Mitchell, W.C.; Fujimoto, M.S.; Christenson, L.D. Melon fly eradication by overflooding with sterile flies. J. Econ. Entomol. 1965, 58, 519–521. [Google Scholar] [CrossRef]
- Andreawartha, H.G.; Monro, J.; Richardson, N.L. The use of sterile males to control populations of Queensland fruit fly, Dacus tryoni (Frogg.) (Diptera: Tephritidae). II. Filed experiments in New South Wales. Aust. J. Zool. 1967, 15, 461–473. [Google Scholar] [CrossRef]
- Harris, E.J.; Cunningham, R.T.; Tanaka, N.; Ohinata, K.; Schroeder, W.J. Development of the sterile-insect technique on the Island of Lanai, Hawaii for suppression of the Mediterranean fruit fly. Proc. Hawaii. Entomol. Soc. 1986, 26, 77–88. [Google Scholar]
- Cunningham, R.T.; Routhier, W.; Harris, E.J.; Cunningham, G.; Johnson, L.; Edwards, W.; Rosander, R.; Vettel, W.G. Eradication of medfly by sterile-male release: A case study. Citograph 1980, 65, 63–69. [Google Scholar]
- McInnis, D.O.; Tam, S.; Lim, R.; Komatsu, J.; Albrecht, C. Development of a pupal-color based genetic sexing strain of the melon fly, Bactrocera cucurbitae (Diptera: Tephritidae). Ann. Entomol. Soc. Am. 2004, 97, 1026–1033. [Google Scholar] [CrossRef]
- Harris, E.J.; Bautista, R.C.; Vargas, R.I.; Jang, E.B.; Eitam, A.; Leblanc, L. Suppression of melon fly (Diptera: Tephritidae) populations with releases of Fopius arisanus and Psyttalia fletcheri (Hymenoptera: Braconidae) in North Shore Oahu, HI, USA. Biocontrol 2010, 55, 593–599. [Google Scholar] [CrossRef]
- Vargas, R.I.; Stark, J.D.; Uchida, G.; Purcell, M. Opiine parasitoids (Hymenoptera, Braconidae) of oriental fruit-fly (Diptera, Tephritidae) on Kauai Island, Hawaii: Islandwide relative abundance and parasitism rates in wild and orchard guava habitats. Environ. Entomol. 1993, 22, 246–253. [Google Scholar] [CrossRef]
- Ekesi, S.; Billah, M.K. A Field Guide to the Management of Economically Important Tephritid Fruit Flies in Africa; ICIPE Science Press: Nairobi, Kenya, 2007. [Google Scholar]
- Liquido, N.J. Fruit on the ground as a reservoir of resident melon fly (Diptera: Tephritidae) population in Papaya orchards. Environ. Entomol. 1991, 20, 620–625. [Google Scholar] [CrossRef]
- Liquido, N.J. Reduction of Oriental fruit fly (Diptera: Tephritidae) populations in papaya orchards by field sanitation. J. Agric. Entomol. 1993, 10, 163–170. [Google Scholar]
- Sarker, D.; Rahman, M.M.; Barman, J.C. Efficacy of different bagging materials for the control of mango fruit fly. Bangladesh J. Agric. Res. 2009, 34, 165–168. [Google Scholar] [CrossRef]
- Jang, E.B.; Klungness, L.M.; McQuate, G. Extension of the use of Augmentoria for Sanitation in a cropping system susceptible to the alien terphritid fruit flies (Diptera: Tephritidae) in Hawaii. J. Appl. Sci. Environ. Manag. 2007, 11, 239–248. [Google Scholar]
- Deguine, J.P.; Atiama-Nurbel, T.; Quilici, S. Net choice is key to the augmentorium technique of fruit fly sequestration and parasitoid release. Crop Prot. 2011, 30, 198–202. [Google Scholar] [CrossRef]
- Verghese, A.; Tandon, P.L.; Stonehouse, J.M. Economic evaluation of the integrated management of the oriental fruit fly Bactrocera dorsalis (Diptera: Tephritidae) in mango in India. Crop Prot. 2004, 23, 61–63. [Google Scholar] [CrossRef]
- Mau, R.F.L.; Vargas, R.I.; Jang, E.B.; Wong, L. Hawaii Area-Wide Fruit Fly Integrated Pest Management Program: A Model System; College of Tropical Agriculture and Human Resources: Honolulu, HI, USA, 2009. [Google Scholar]
- Verghese, A.; Sreedevi, K.; Nagaraju, D.K. Pre and post-harvest IPM for the mango fruit fly, Bactrocera dorsalis (Hendel). In Proceedings of the 7th International Symposium on Fruit Flies of Economic Importance, Salvador, Brazil, 10–15 September 2006; pp. 179–182.
- Singh, M.; Gupta, D.; Gupta, P.R. Population suppression of fruit flies (Bactrocera spp.) in mango (Mangifera indica) orchards. Ielendian J. Agric. Sci. 2013, 83, 1064–1068. [Google Scholar]
- Gogi, M.D.; Arif, M.J.; Arshad, M.; Khan, M.A.; Bashir, M.H.; Zia, K.; Abdin, Z.U. Impact of sowing Times, Plant-to-Plant Distances, Sowing Methods and Sanitation on Infestation of Melon Fruit Fly (Bactrocera cucurbitae) and Yield Components of Bitter Gourd (Momordica charantia). Int. J. Agric. Biol. 2014, 16, 521–528. [Google Scholar]
- Collier, T.R.; van Steenwyk, R.A. Prospects for integrated control of olive fruit fly are promising in California. Calif. Agric. 2003, 57, 28–31. [Google Scholar] [CrossRef]
- Yokoyama, V.Y. Olive fruit fly (Diptera: Tephritidae) in California: Invasion, distribution, and management implications. J. Pest Manag. In Press.
- Johnson, M.W.; Zalom, F.G.; van Steenwyk, R.; Vossen, P.; Devarenne, A.K.; Daane, K.M.; Krueger, B.; Connell, J.H.; Yokoyama, V.; Bisabri, B.; et al. Olive Fruit Fly Management Guidelines for 2006. UC Plant Prot. Q. 2006, 16, 1–9. [Google Scholar]
- Yokoyama, V.Y. Olive fruit fly adult response to attract-and-kill traps in greenhouse cage tests with weathered bait spray and in a commercial table olive orchard. J. Asia-Pac. Entomol. 2014, 17, 717–721. [Google Scholar] [CrossRef]
- Yokoyama, V.Y. Response of olive fruit fly, Bactrocera oleae, to an attract and kill trap in greenhouse cage tests. J. Insect Sci. 2014, 14, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Mangan, R.L. Priorities in formulation and activity of adulticidal insecticide bait sprays for fruit flies. In Trapping Tephritid Fruit Flies. Lures, Area-Wide Programs, and Trade Implications; Shelly, T., Epsky, N., Jang, E.B., Reyes-Flores, J., Vargas, R., Eds.; Springer: Berlin, Germany, 2014; pp. 423–456. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vargas, R.I.; Piñero, J.C.; Leblanc, L. An Overview of Pest Species of Bactrocera Fruit Flies (Diptera: Tephritidae) and the Integration of Biopesticides with Other Biological Approaches for Their Management with a Focus on the Pacific Region. Insects 2015, 6, 297-318. https://doi.org/10.3390/insects6020297
Vargas RI, Piñero JC, Leblanc L. An Overview of Pest Species of Bactrocera Fruit Flies (Diptera: Tephritidae) and the Integration of Biopesticides with Other Biological Approaches for Their Management with a Focus on the Pacific Region. Insects. 2015; 6(2):297-318. https://doi.org/10.3390/insects6020297
Chicago/Turabian StyleVargas, Roger I., Jaime C. Piñero, and Luc Leblanc. 2015. "An Overview of Pest Species of Bactrocera Fruit Flies (Diptera: Tephritidae) and the Integration of Biopesticides with Other Biological Approaches for Their Management with a Focus on the Pacific Region" Insects 6, no. 2: 297-318. https://doi.org/10.3390/insects6020297
APA StyleVargas, R. I., Piñero, J. C., & Leblanc, L. (2015). An Overview of Pest Species of Bactrocera Fruit Flies (Diptera: Tephritidae) and the Integration of Biopesticides with Other Biological Approaches for Their Management with a Focus on the Pacific Region. Insects, 6(2), 297-318. https://doi.org/10.3390/insects6020297





