Pyrazines Attract Catocheilus Thynnine Wasps
Abstract
:1. Introduction
2. Experimental Section
2.1. GC-MS/EAD
| Group | Species |  |  |  |  |  |  |
|---|---|---|---|---|---|---|---|
| SEXUALLY DECEPTIVE ORCHIDS | Drakaea livida | x | x | x | x | x | |
| Drakaea thynniphila | x | ||||||
| Drakaea micrantha | x | ||||||
| Drakaea confluens | x | ||||||
| Caladenia barbarossa [17] | x | x | |||||
| THYNNINE WASPS | Zaspilothynnus nigripes | x | |||||
| Zaspilothynnus gilesi | x* | ||||||
| Zaspilothynnus seductor | x* | ||||||
| Zaspilothynnus rugicollis | x* | ||||||
| Macrothynnus insignis | x* |
2.2. Chemical Survey
2.3. Field Bioassays
3. Results and Discussion
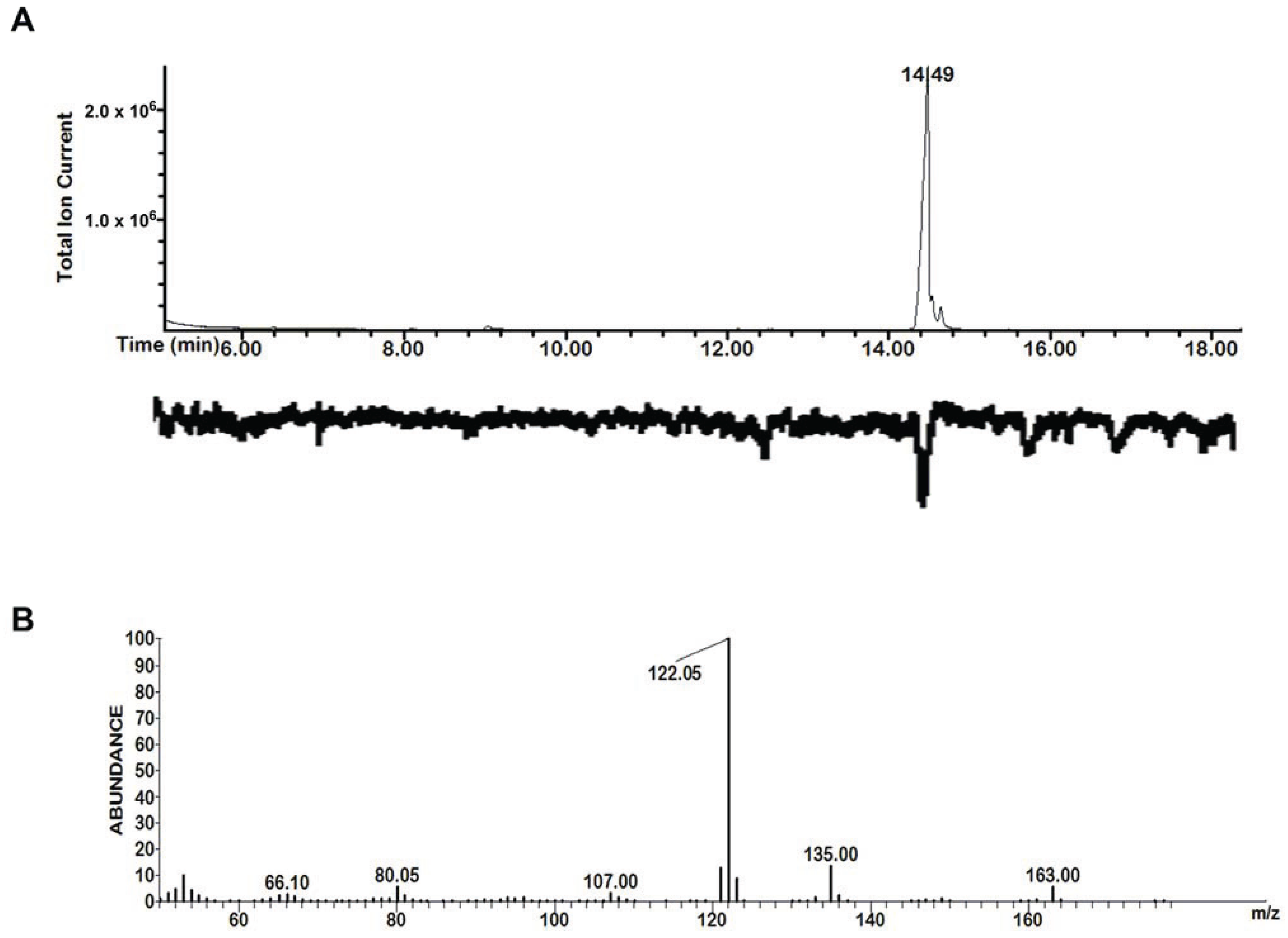
3.1. GC-MS/EAD
3.2. Chemical Survey
3.3. Field Bioassays

3.4. Evolutionary Implications
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cozzolino, S.; Widmer, A. Orchid diversity: An evolutionary consequence of deception? Trends Ecol. Evol. 2005, 20, 487–494. [Google Scholar] [CrossRef]
- Schiestl, F.P.; Ayasse, M.; Paulus, H.F.; Lofstedt, C.; Hansson, B.S.; Ibarra, F.; Francke, W. Orchid pollination by sexual swindle. Nature 1999, 399, 421–421. [Google Scholar] [CrossRef]
- Peakall, R. Responses of male Zaspilothynnus trilobatus wasps to females and the sexually deceptive orchid it pollinates. Funct. Ecol. 1990, 4, 159–167. [Google Scholar] [CrossRef]
- Stoutamire, W.P. Pseudocopulation in Australian orchids. Am. Orchid Soc. Bull. 1975, 44, 226–233. [Google Scholar]
- Peakall, R.; Ebert, D.; Poldy, J.; Barrow, R.A.; Francke, W.; Bower, C.C.; Schiestl, F.P. Pollinator specificity, floral odour chemistry and the phylogeny of Australian sexually deceptive Chiloglottis orchids: Implications for pollinator-driven speciation. New Phytol. 2010, 188, 437–450. [Google Scholar] [CrossRef]
- Gaskett, A.C. Orchid pollination by sexual deception: Pollinator perspectives. Biol. Rev. 2011, 86, 33–75. [Google Scholar] [CrossRef]
- Griffiths, K.E.; Trueman, J.W.H.; Brown, G.R.; Peakall, R. Molecular genetic analysis and ecological evidence reveals multiple cryptic species among thynnine wasp pollinators of sexually deceptive orchids. Mol. Phylogenet. Evol. 2011, 59, 195–205. [Google Scholar] [CrossRef]
- Mant, J.; Brändli, C.; Vereecken, N.J.; Schulz, C.M.; Francke, W.; Schiestl, F.P. Cuticular hydrocarbons as sex pheromone of the bee Colletes cunicularius and the key to its mimicry by the sexually deceptive orchid, Ophrys exaltata. J. Chem. Ecol. 2005, 31, 1765–1787. [Google Scholar] [CrossRef]
- Stökl, J.; Twele, R.; Erdmann, D.H.; Francke, W.; Ayasse, M. Comparison of the flower scent of the sexually deceptive orchid Ophrys iricolor and the female sex pheromone of its pollinator Andrena morio. Chemoecology 2007, 17, 231–233. [Google Scholar] [CrossRef]
- Ayasse, M.; Stökl, J.; Francke, W. Chemical ecology and pollinator-driven speciation in sexually deceptive orchids. Phytochemistry 2011, 72, 1667–1677. [Google Scholar] [CrossRef]
- Schiestl, F.P.; Peakall, R.; Mant, J.G.; Ibarra, F.; Schulz, C.; Franke, S.; Francke, W. The chemistry of sexual deception in an orchid-wasp pollination system. Science 2003, 302, 437–438. [Google Scholar] [CrossRef]
- Franke, S.; Ibarra, F.; Schulz, C.M.; Twele, R.; Poldy, J.; Barrow, R.A.; Peakall, R.; Schiestl, F.P.; Francke, W. The discovery of 2,5-dialkylcyclohexan-1,3-diones as a new class of natural products. Proc. Natl. Acad. Sci. USA 2009, 106, 8877–8882. [Google Scholar] [CrossRef]
- Bohman, B.; Jeffares, L.; Flematti, G.; Byrne, L.T.; Skelton, B.W.; Phillips, R.D.; Dixon, K.W.; Peakall, R.; Barrow, R.A. Discovery of tetrasubstituted pyrazines as semiochemicals in a sexually deceptive orchid. J. Nat. Prod. 2012, 75, 1589–1594. [Google Scholar] [CrossRef]
- Bohman, B.; Jeffares, L.; Flematti, G.; Phillips, R.D.; Dixon, K.W.; Peakall, R.; Barrow, R.A. The discovery of 2-hydroxymethyl-3-(3-methylbutyl)-5-methylpyrazine: A semiochemical in orchid pollination. Org. Lett. 2012, 14, 2576–2578. [Google Scholar]
- Bohman, B.; Phillips, R.D.; Menz, M.H.M.; Berntsson, B.W.; Flematti, G.R.; Barrow, R.A.; Dixon, K.W.; Peakall, R. Discovery of pyrazines as pollinator sex pheromones and orchid semiochemicals: Implications for the evolution of sexual deception. New Phytologist 2014. [Google Scholar] [CrossRef]
- Phillips, R.D.; Peakall, R.; Hutchinson, M.F.; Linde, C.C.; Xu, T.; Dixon, K.W.; Hopper, S.D. Specialized ecological interactions and plant species rarity: The role of pollinators and mycorrhizal fungi across multiple spatial scales. Biol. Conservat. 2014, 169, 285–295. [Google Scholar] [CrossRef]
- Bohman, B.; Phillips, R.D.; Flematti, G.; Peakall, R.; Barrow, R.A. Sharing of pyrazine semiochemicals between genera of sexually deceptive orchids. Nat. Prod. Comm. 2013, 8, 701–702. [Google Scholar]
- Syntech. Electroantennography, a Practical Introduction; Syntech: Kirchzarten, Germany, 2004. [Google Scholar]
- Syntech. GcEad/2011. Available online: http://sourceforge.net/projects/gcead/files/gcead/v1.2/ (accessed on 16 June 2014).
- Peakall, R.; Smouse, P.E. GENALEX 6: Genetic analysis in excel. Population genetic software for teaching and research. Mol. Ecol. Notes 2006, 6, 288–295. [Google Scholar] [CrossRef]
- Peakall, R.; Smouse, P.E. GenAlEx 6.5: Genetic analysis in excel. Population genetic software for teaching and research-an update. Bioinformatics 2012, 28, 2537–2539. [Google Scholar] [CrossRef]
- Jürgens, A.; Dötterl, S.; Meve, U. The chemical nature of fetid floral odours in stapeliads (Apocynaceae-Asclepiadoideae-Ceropegieae). New Phytologist 2006, 172, 452–468. [Google Scholar] [CrossRef]
- Citron, C.A.; Rabe, P.; Dickschat, J.S. The scent of bacteria: Headspace analysis for the discovery of natural products. J. Nat. Prod. 2012, 75, 1765–1776. [Google Scholar] [CrossRef]
- Dickschat, J.S.; Reichenbach, H.; Wagner-Döbler, I.; Schulz, S. Novel pyrazines from the myxobacterium Chondromyces crocatus and marine bacteria. Euro. J. Org. Chem. 2005, 2005, 4141–4153. [Google Scholar]
- Dickschat, J.S.; Wagner-Döbler, I.; Schulz, S. The chafer pheromone buibuilactone and ant pyrazines are also produced by marine bacteria. J. Chem. Ecol. 2005, 31, 925–947. [Google Scholar] [CrossRef]
- Dossey, A.T.; Gottardo, M.; Whitaker, J.M.; Roush, W.R.; Edison, A.S. Alkyldimethylpyrazines in the defensive spray of Phyllium westwoodii: A first for order phasmatodea. J. Chem. Ecol. 2009, 35, 861–870. [Google Scholar] [CrossRef]
- Brophy, J.J.; Cavill, G.W.K.; Duke, R.K. Volatile constituents in a methylene chloride extract of a ponerine ant, Rhytidoponera aciculata (Smith). Insect Biochem. 1983, 13, 503–505. [Google Scholar] [CrossRef]
- Cavill, G.W.K.; Houghton, E. Some pyrazine derivatives from the argentine ant, Iridomyrmex humilis. Aust. J. Chem. 1974, 27, 879–889. [Google Scholar] [CrossRef]
- Fales, H.M. Structure and synthesis of tetrasubstituted pyrazines in ants in the genus Mesoponera. Tetrahedron 1988, 44, 5045–5050. [Google Scholar] [CrossRef]
- Jones, T.H.; Flournoy, R.C.; Torres, J.A.; Snelling, R.R.; Spande, T.F.; Garraffo, H.M. 3-methyl-4-phenylpyrrole from the ants Anochetus kempfi and Anochetus mayri. J. Nat. Prod. 1999, 62, 1343–1345. [Google Scholar] [CrossRef]
- Morgan, E.D.; Do Nascimento, R.R.; Keegans, S.J.; Billen, J. Comparative study of mandibular gland secretions of workers of ponerine ants. J. Chem. Ecol. 1999, 25, 1395–1409. [Google Scholar] [CrossRef]
- Ayasse, M.; Schiestl, F.P.; Paulus, H.F.; Ibarra, F.; Francke, W. Pollinator attraction in a sexually deceptive orchid by means of unconventional chemicals. Proc. Roy. Soc. Lond. B Biol. Sci. 2003, 270, 517–522. [Google Scholar] [CrossRef]
- Stökl, J.; Paulus, H.; Dafni, A.; Schulz, C.; Francke, W.; Ayasse, M. Pollinator attracting odour signals in sexually deceptive orchids of the Ophrys fusca group. Plant Systemat. Evol. 2005, 254, 105–120. [Google Scholar] [CrossRef]
- Vereecken, N.J.; Schiestl, F.P. The evolution of imperfect floral mimicry. Proc. Natl. Acad. Sci. USA 2008, 105, 7484–7488. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bohman, B.; Peakall, R. Pyrazines Attract Catocheilus Thynnine Wasps. Insects 2014, 5, 474-487. https://doi.org/10.3390/insects5020474
Bohman B, Peakall R. Pyrazines Attract Catocheilus Thynnine Wasps. Insects. 2014; 5(2):474-487. https://doi.org/10.3390/insects5020474
Chicago/Turabian StyleBohman, Bjorn, and Rod Peakall. 2014. "Pyrazines Attract Catocheilus Thynnine Wasps" Insects 5, no. 2: 474-487. https://doi.org/10.3390/insects5020474
APA StyleBohman, B., & Peakall, R. (2014). Pyrazines Attract Catocheilus Thynnine Wasps. Insects, 5(2), 474-487. https://doi.org/10.3390/insects5020474




