Phylogenetic Analysis of Fusarium solani Associated with the Asian Longhorned Beetle, Anoplophora glabripennis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Rearing Colony Derived A. glabripennis on Different Host Trees
2.2. Collection of Larval A. glabripennis and Fungal Cultures from Introduced Wild Populations
2.3. Anoplophora Glabripennis Larval Gut Dissection and DNA Extraction
2.4. Culture Independent Fungal Community Analysis
2.5. Aerobic Culturing of Gut Fungus on Restrictive Media
2.6. Multi-Locus Sequencing from Cultured Fungal DNA Extraction
| Tree Replicate Number | |||||
|---|---|---|---|---|---|
| Host tree | 1 | 2 | 3 | Total | |
| PSU-derived | Sugar Maple | 7 | 6 | 3 | 16 |
| NYC-derived | Sycamore Maple | 1 | - | - | 1 |
| MA-derived | Silver Maple | 4 | 1 | - | 5 |
| Total | 12 | 7 | 3 | 22 | |
2.7. Sequence Editing, Alignment and Operational Taxonomic Unit (OTU) Analysis of TEF1-α
2.8. Single- and Multi-locus Phylogenetic Analysis
3. Results and Discussion
3.1. Culture Independent Fungal Community Analysis
| Culture Independent | Host tree | OTU Identification Number | ||
|---|---|---|---|---|
| 1 | 2 | Total | ||
| PSU-derived | Sugar Maple | 39 | 39 | |
| Pin Oak | 27 | 6 | 33 | |
| Callery Pear | 46 | 46 | ||
| Artificial Diet | 32 | 3 | 35 | |
| NYC-derived | Silver Maple (tree 1) | 30 | 30 | |
| Silver Maple (tree 2) | 32 | 32 | ||
| Sycamore Maple | 33 | 33 | ||
| Horse-Chestnut | 29 | 29 | ||
| Total | 268 | 9 | 277 | |
3.2. Culture-Dependent Fungal Analysis
| Cultured | Host tree | OTU Identification Number | ||
|---|---|---|---|---|
| 1 | 2 | Total | ||
| PSU | Sugar Maple | 9 | 8 | 17 |
| NYC | Horse chestnut | 1 | 0 | 1 |
| MA | Silver Maple | 0 | 6 | 6 |
| Total | 10 | 14 | 24 | |
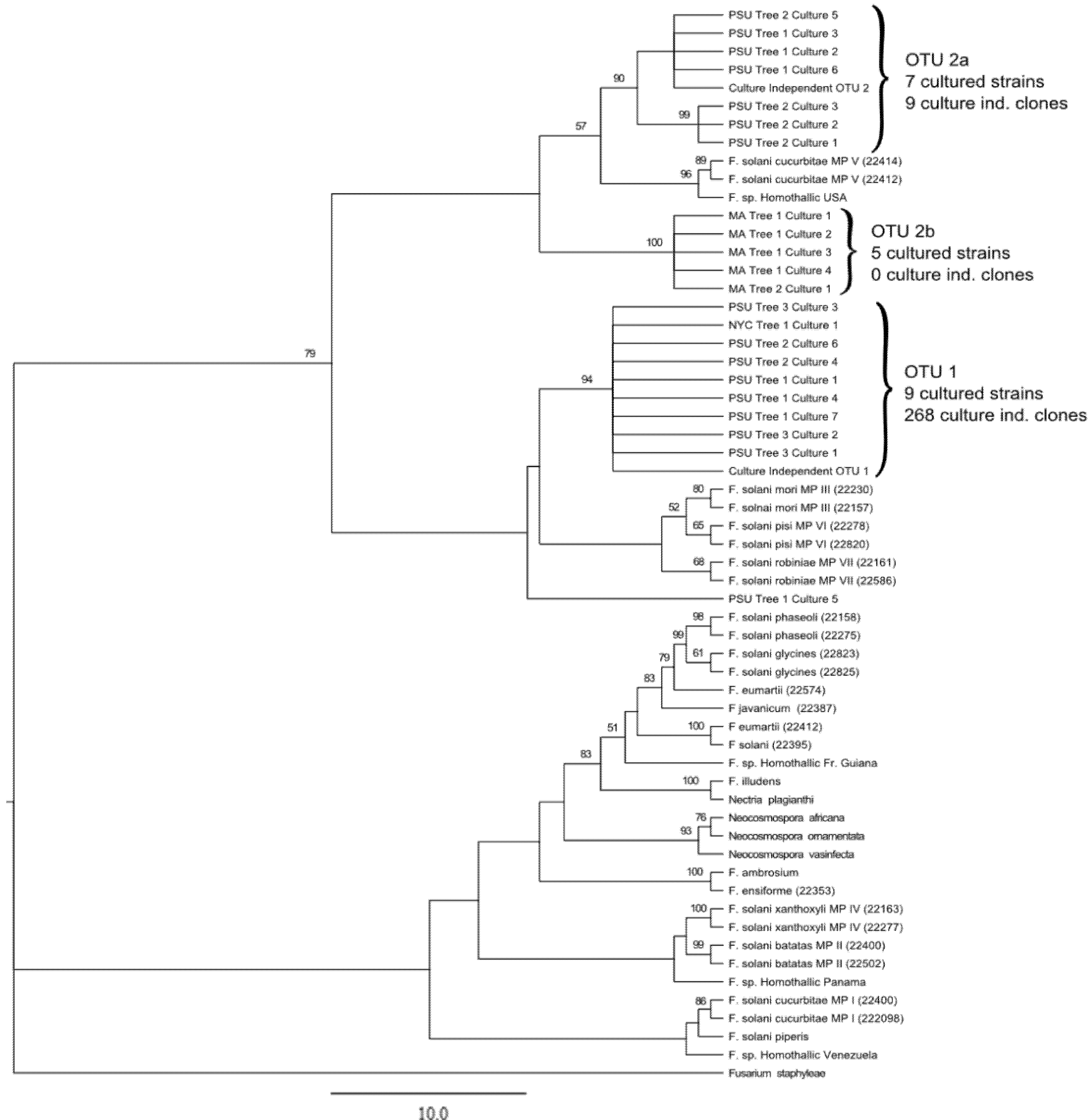
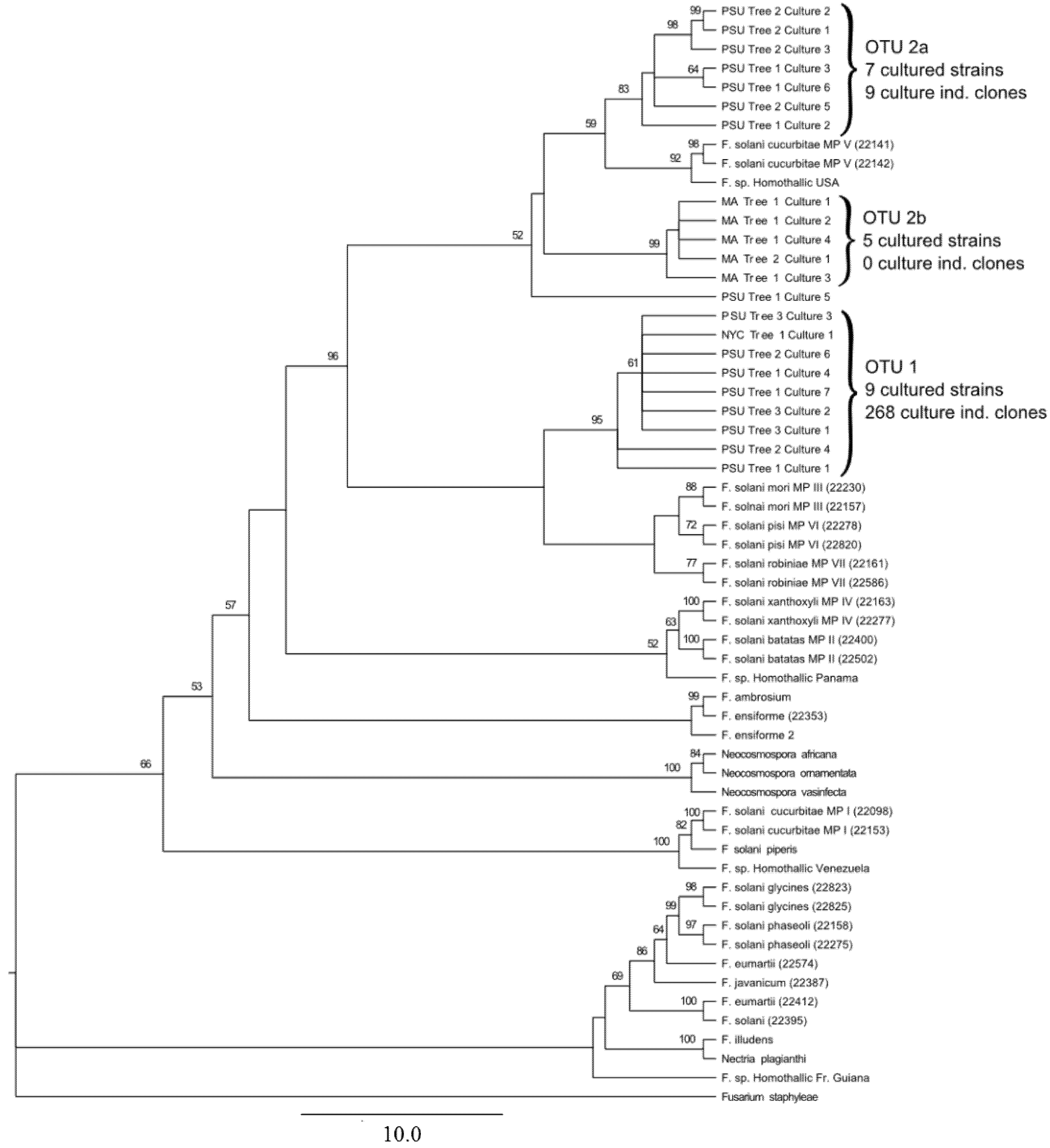
3.3. Discussion
4. Conclusions
Acknowledgments
References
- Whitney, A.N.; Keena, M.A. Effects of Host Wood Moisture on the Lifecycle Development of the ASIAN Longhorned Beetle; McManus, K.A.G., Kurt, W., Eds.; Department of Agriculture, Forest Service, Northern Research Station: Annapolis, MD, USA, 2010; p. 169. [Google Scholar]
- Moser, W.K.; Barnard, E.L.; Billings, R.F.; Crocker, S.J.; Dix, M.E.; Gray, A.N.; Ice, G.G.; Kim, M.S.; Reid, R.; Rodman, S.U.; McWilliams, W.H. Impacts of Nonnative Invasive Species on US Forests and Recommendations for Policy and Management. J. For. 2009, 107, 320–327. [Google Scholar]
- Haack, R. New York's battle with the Asian long-horned beetle. J. For. 1997, 95, 11–15. [Google Scholar]
- Kukor, J.J.; Martin, M.M. Cellulose digestion in Monochamus marmorator Kby (Coleoptera, Cerambycidae)—Role of acquired fungal enzymes. J. Chem. Ecol. 1986, 12, 1057–1070. [Google Scholar] [CrossRef]
- Kukor, J.; Cowan, D.; Martin, M. The role of ingested fungal enzymes in cellulose digestion in the larvae of cerambycid beetles. Physiol. Zool. 1988, 61, 364–371. [Google Scholar]
- Morewood, W.D.; Hoover, K.; Neiner, P.R.; Sellmer, J.C. Complete development of Anoplophora glabripennis (Coleoptera: Cerambycidae) in northern red oak trees. Can. Entomologist 2005, 137, 376–379. [Google Scholar] [CrossRef]
- Morewood, W.D.; Neiner, P.R.; McNeil, J.R.; Sellmer, J.C.; Hoover, K. Oviposition preference and larval performance of Anoplophora glabripennis (Coleoptera: Cerambycidae) in four eastern North American hardwood tree species. Environ. Entomology 2003, 32, 1028–1034. [Google Scholar] [CrossRef]
- Morewood, W.D.; Neiner, P.R.; Sellmer, J.C.; Hoover, K. Behavior of adult Anoplophora glabripennis on different tree species under greenhouse conditions. J. Insect Behav. 2004, 17, 215–226. [Google Scholar] [CrossRef]
- Sellmer, J.C.; Morewood, W.D.; Neiner, P.; Hoover, K. Evaluating Asian Longhorned Beetle Adult Preference and Larval Performance Among Commonly Planted Landscape Trees. In Processeding of 13th Metropolitan Tree Improvement Alliance and Landscape Plant Development Center Conference, Lisle, IL, USA, 16–19 June 2004.
- Lee, S.J.; Kim, S.R.; Yoon, H.J.; Kim, I.; Lee, K.S.; Je, Y.H.; Lee, S.M.; Seo, S.J.; Sohn, H.D.; Jin, B.R. cDNA cloning, expression, and enzymatic activity of a cellulase from the mulberry longicorn beetle, Apriona germari. Comp. Biochem. Physiol. B Comp. Biochem. 2004, 139, 107–116. [Google Scholar] [CrossRef]
- Lee, S.J.; Lee, K.S.; Kim, S.R.; Gui, Z.Z.; Kim, Y.S.; Yoon, H.J.; Kim, I.; Kang, P.D.; Sohn, H.D.; Jin, B.R. A novel cellulase gene from the mulberry longicorn beetle, Apriona germari: Gene structure, expression, and enzymatic activity. Comp. Biochem. Physiol. B Comp. Biochem. 2005, 140, 551–560. [Google Scholar]
- Sugimura, M.; Watanabe, H.; Lo, N.; Saito, H. Purification, characterization, cDNA cloning and nucleotide sequencing of a cellulase from the yellow-spotted longicorn beetle, Psacothea hilaris. Eur. J. Biochem. 2003, 270, 3455–3460. [Google Scholar] [CrossRef]
- Holtzapple, M.; Cognata, M.; Shu, Y.; Hendrickson, C. Inhibition of Trichoderma reesei cellulase by sugars and solvents. Biotechnol. Bioeng. 1990, 36, 275–287. [Google Scholar] [CrossRef]
- Geib, S.M.; Filley, T.R.; Hatcher, P.G.; Hoover, K.; Carlson, J.E.; Jimenez-Gasco Mdel, M.; Nakagawa-Izumi, A.; Sleighter, R.L.; Tien, M. Lignin degradation in wood-feeding insects. Proc. Natl. Acad. Sci. USA 2008, 105, 12932–12937. [Google Scholar]
- Dillon, R.J.; Dillon, V.M. The gut bacteria of insects: Nonpathogenic interactions. Annu. Rev. Entomol. 2004, 49, 71–92. [Google Scholar] [CrossRef]
- Schloss, P.D.; Delalibera, I.; Handelsman, J.; Raffa, K.F. Bacteria associated with the guts of two wood-boring beetles: Anoplophora glabripennis and Saperda vestita (Cerambycidae). Environ. Entomol. 2006, 35, 625–629. [Google Scholar] [CrossRef]
- Delalibera, I.; Handelsman, J.; Raffa, K.F. Contrasts in cellulolytic activities of gut microorganisms between the wood borer, Saperda vestita (Coleoptera: Cerambycidae), and the bark beetles, Ips pini and Dendroctonus frontalis (Coleoptera: Curculionidae). Environ. Entomol. 2005, 34, 541–547. [Google Scholar] [CrossRef]
- Geib, S.M.; Jimenez-Gasco, M.D.M.; Carlson, J. E.; Tien, M.; Hoover, K. Effect of Host Tree Species on Cellulase Activity and Bacterial Community Composition in the Gut of Larval Asian Longhorned Beetle. Environ. Entomol. 2009, 38, 686–699. [Google Scholar] [CrossRef]
- Geib, S.M.; Jimenez-Gasco Mdel, M.; Carlson, J.E.; Tien, M.; Jabbour, R.; Hoover, K. Microbial community profiling to investigate transmission of bacteria between life stages of the wood-boring beetle, Anoplophora glabripennis. Microb. Ecol. 2009, 58, 199–211. [Google Scholar] [CrossRef]
- Suh, S.O.; Marshall, C.J.; McHugh, J.V.; Blackwell, M. Wood ingestion by passalid beetles in the presence of xylose-fermenting gut yeasts. Mol. Ecol. 2003, 12, 3137–3145. [Google Scholar] [CrossRef]
- Suh, S.O.; McHugh, J.V.; Pollock, D.D.; Blackwell, M. The beetle gut: A hyperdiverse source of novel yeasts. Mycol. Res. 2005, 109, 261–265. [Google Scholar] [CrossRef]
- Suh, S.O.; Nguyen, N.H.; Blackwell, M. Yeasts isolated from plant-associated beetles and other insects: Seven novel Candida species near Candida albicans. FEMS Yeast Res. 2008, 8, 88–102. [Google Scholar] [CrossRef]
- Suh, S.-O.; Blackwell, M. The beetle gut as a habitat for new species of yeasts. In Insect Fungal Associations: Ecology and Evolution; Vega, F., Blackwell, M., Eds.; Oxford University: New York, NY, USA, 2005. [Google Scholar]
- Zhang, N.; Suh, S.O.; Blackwell, M. Microorganisms in the gut of beetles: Evidence from molecular cloning. J. Invertebr. Pathol. 2003, 84, 226–233. [Google Scholar] [CrossRef]
- Jones, K.; Dowd, P.; Blackwell, M. Polyphyletic origins of yeast-like endocytobionts from anobiid and cerambycid beetles. Mycol. Res. 1999, 103, 542–546. [Google Scholar] [CrossRef]
- Suh, S.-O.; Blackwell, M. Three new beetle-associated yeast species in the Pichia guilliermondii clade. FEMS Yeast Res. 2004, 5, 87–95. [Google Scholar] [CrossRef]
- Chararas, C.; Eberhard, R.; Courtois, J.E.; Petek, F. Purification of three cellulases from the zylophageous larvae of Ergates faber (Coleoptera: Cerambycidae). Insect Biochem. 1983, 13, 213–218. [Google Scholar] [CrossRef]
- Dowd, P. Symbiont-mediated detoxification in insect herbivores. In Microbial Mediation of Plant-Herbivore Interactions; Barbosa, P., Krischik, V., Jones, C., Eds.; John Wiley & Sons: New York, NY, USA, 1991; pp. 411–440. [Google Scholar]
- Jones, C.G. Microorganisms as mediators of plant resource exploitation by insect herbivores. In A New Ecology: Novel Approaches to Interactive Systems; Price, P.W., Slobodchikoff, C.N., Gaud, W.S., Northern Arizona, U., Eds.; Wiley: New York, NY, USA, 1984; pp. 53–99. [Google Scholar]
- Vega, F.E.; Dowd, P.F. The role of yeasts as insect endosymbionts. In Insect-Fungal Associations: Ecology and Evolution; Vega, F.E., Blackwell, M., Eds.; Oxford University Press: New York, NY, USA, 2005; pp. 211–243. [Google Scholar]
- Nardon, P.; Grenier, A. Endosymbiosis in Coleoptera: Biological, biochemical, and genetic aspects. In Insect Endocytobiosis: Morphology, Physiology, Genetics, Evolution; Schwemmler, W., Gassner, G., Eds.; CRC Press: Boca Raton, FL, USA, 1989; pp. 175–216. [Google Scholar]
- Berkov, A.; Feinstein, J.; Small, J.; Nkamany, M. Yeasts isolated from neotropical wood-boring beetles in SE Peru. Biotropica 2007, 39, 530–538. [Google Scholar] [CrossRef]
- Teetorbarsch, G.H.; Roberts, D.W. EntomogenousFusarium Species. Mycopathologia 1983, 84, 3–16. [Google Scholar] [CrossRef]
- Morales-Ramos; Rojas, M.; Sittertz-Bhatkar, H.; Saldana, G. Symbiotic relationshiop between Hypothenemus hampei (Coleoptera: Scolytidae) and Fusarium solani (Moniliales: Tuberculariaceae). Ann. Entomol. Soc. Am. 2000, 93, 541–547. [Google Scholar] [CrossRef]
- Morewood, W.D.; Hoover, K.; Neiner, P.R.; McNeil, J.R.; Sellmer, J.C. Host tree resistance against the polyphagous wood-boring beetle Anoplophora glabripennis. Entomologia Experimentalis et Applicata 2004, 110, 79–86. [Google Scholar] [CrossRef]
- Dubois, T.; Hajek, A.E.; Smith, S. Methods for rearing the Asian longhorned beetle (Coleoptera: Cerambycidae) on artificial diet. Ann. Entomol. Soc. Am. 2002, 95, 223–230. [Google Scholar] [CrossRef]
- Ludwig, S.W.; Lazarus, L.; McCullough, D.G.; Hoover, K.; Montero, S.; Sellmer, J.C. Methods to evaluate host tree suitability to the Asian longhorned beetle, Anoplophora glabripennis. J. Environ. Hortic. 2002, 20, 175–180. [Google Scholar]
- Keena, M. Pourable artificial diet for rearing Anoplophora glabripennis (Coleoptera: Cerambycidae) and methods to optimize larval survival and synchronize development. Ann. Entomol. Soc. Am. 2005, 98, 536–547. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- O'Donnell, K.; Cigelnik, E. Two divergent intragenomic rDNA ITS2 types within a monophyletic lineage of the fungus Fusarium are nonorthologous. Mol. Phylogenet. Evol. 1997, 7, 103–117. [Google Scholar] [CrossRef]
- O'Donnell, K.; Cigelnik, E.; Nirenberg, H.L. Molecular systematics and phylogeography of the Gibberella fujikuroi species complex. Mycologia 1998, 90, 465–493. [Google Scholar] [CrossRef]
- Geiser, D.M.; Jimenez-Gasco, M.D.; Kang, S.C.; Makalowska, I.; Veeraraghavan, N.; Ward, T.J.; Zhang, N.; Kuldau, G.A.; O'Donnell, K. FUSARIUM-ID v. 1.0: A DNA sequence database for identifying Fusarium. Eur. J. Plant Pathol. 2004, 110, 473–479. [Google Scholar] [CrossRef]
- O'Donnell, K.; Kistler, H.C.; Cigelnik, E.; Ploetz, R.C. Multiple evolutionary origins of the fungus causing Panama disease of banana: Concordant evidence from nuclear and mitochondrial gene genealogies. Proc. Natl. Acad. Sci. USA 1998, 95, 2044–2049. [Google Scholar]
- O'Donnell, K. Molecular phylogeny of the Nectria haematococca-Fusarium solani species complex. Mycologia 2000, 92, 919–938. [Google Scholar] [CrossRef]
- Tamura, K.; Dudley, J.; Nei, M.; Kumar, S. MEGA4: Molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol. Biol. Evol. 2007, 24, 1596–1599. [Google Scholar] [CrossRef]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J.; Sahl, J.W.; Stres, B.; Thallinger, G.G.; Van Horn, D.J.; Weber, C.F. Introducing mothur: Open-Source, Platform-Independent, Community-Supported Software for Describing and Comparing Microbial Communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar]
- Morell, V. TreeBASE: The Roots of Phylogeny. Science 1996, 273, 569–560. [Google Scholar] [CrossRef]
- Swofford, D. PAUP*: Phylogenetic analysis using parsimony, version 4.0b10. 2003.
- Posada, D. jModelTest: Phylogenetic Model Averaging. Mol. Biol. Evol. 2008, 25, 1253–1256. [Google Scholar] [CrossRef]
- Guindon, S.; Gascuel, O. A Simple, Fast, and Accurate Algorithm to Estimate Large Phylogenies by Maximum Likelihood. Syst. Biol. 2003, 52, 696–704. [Google Scholar] [CrossRef]
- Zwickl, D. Genetic algorithm approaches for the phylogenetic analysis of large biological sequence datasets under the maxmum likelihood criterion. PhD Dissertation; University of Texas: Austin, TX, USA, 2006. Available online: http://www.nescent.org/wg_garli (accessed on 31 January 2012).
- Sukumaran, J.; Holder, M.T. DendroPy: A Python library for phylogenetic computing. Bioinformatics 2010, 26, 1569–1571. [Google Scholar] [CrossRef]
- Cheilas, T.; Stoupis, T.; Christakopoulos, P.; Katapodis, P.; Mamma, D.; Hatzinikolaou, D.G.; Kekos, D.; Macris, B.J. Hemicellulolytic activity of Fusarium oxysporum grown on sugar beet pulp: Production of extracellular arabinanase. Process Biochem. 2000, 35, 557–561. [Google Scholar] [CrossRef]
- Coleman, J.J.; Rounsley, S.D.; Rodriguez-Carres, M.; Kuo, A.; Wasmann, C.C.; Grimwood, J.; Schmutz, J.; Taga, M.; White, G.J.; Zhou, S.; Schwartz, D.C.; Freitag, M.; Ma, L.J.; Danchin, E.G.; Henrissat, B.; Coutinho, P.M.; Nelson, D.R.; Straney, D.; Napoli, C.A.; Barker, B.M.; Gribskov, M.; Rep, M.; Kroken, S.; Molnar, I.; Rensing, C.; Kennell, J.C.; Zamora, J.; Farman, M.L.; Selker, E.U.; Salamov, A.; Shapiro, H.; Pangilinan, J.; Lindquist, E.; Lamers, C.; Grigoriev, I.V.; Geiser, D.M.; Covert, S.F.; Temporini, E.; Vanetten, H.D. The genome of Nectria haematococca: Contribution of supernumerary chromosomes to gene expansion. PLoS Genet. 2009, 5, e1000618. [Google Scholar]
- Lozovaya, V.; Lygin, A.; Zernova, O.; Li, S.; Widholm, J. Lignin degradation by Fusarium solani f. sp. glycines. Plant Dis. 2006, 90, 77–82. [Google Scholar] [CrossRef]
- Manka, M. Cellulolytic activity of three Fusarium culmorum (W.G. Sm.) Sacc. isolates pathogenic towards wheat seedings. J. Phytophathol. 1988, 122, 113–117. [Google Scholar] [CrossRef]
- Panagiotou, G.; Kekos, D.; Macris, B.J.; Christakopoulos, P. Production of cellulolytic and xylanolytic enzymes by Fusarium oxysporum grown on corn stover in solid state fermentation. Ind. Crops Prod. 2003, 18, 37–45. [Google Scholar] [CrossRef]
- Sutherland, J.B.; Pometto, A.L.; Crawford, D.L. Lignocellulose Degradation by Fusarium Species. Can. J. Bot.-Rev. Canadienne De Botanique 1983, 61, 1194–1198. [Google Scholar] [CrossRef]
- Rodriguez, A.; Perestelo, F.; Carnicero, A.; Regalado, V.; Perez, R.; DelaFuente, G.; Falcon, M.A. Degradation of natural lignins and lignocellulosic substrates by soil-inhabiting fungi imperfecti. FEMS Microbiol. Ecol. 1996, 21, 213–219. [Google Scholar] [CrossRef]
- Falcon, M.A.; Rodriguez, A.; Carnicero, A.; Regalado, V.; Perestelo, F.; Milstein, O.; Delafuente, G. Isolation of Microorganisms with Lignin Transformation Potential from Soil of Tenerife Island. Soil. Biol. Biochem. 1995, 27, 121–126. [Google Scholar] [CrossRef]
- Teetor-Barsch, G.H.; Roberts, D.W. Entomogenous Fusarium species. Mycopathologia 1983, 84, 3–16. [Google Scholar] [CrossRef]
- Walter, S.; Nicholson, P.; Doohan, F.M. Action and reaction of host and pathogen during Fusarium head blight disease. New Phytol. 2010, 185, 54–66. [Google Scholar] [CrossRef]
- Dumestre, A.; Chone, T.; Portal, J.M.; Gerard, M.; Berthelin, J. Cyanide degradation under alkaline conditions by a strain of Fusarium solani isolated from contaminated soils. Appl. Environ. Microbiol. 1997, 63, 2729–2734. [Google Scholar]
- Zhang, N.; O'Donnell, K.; Sutton, D.A.; Nalim, F.A.; Summerbell, R.C.; Padhye, A.A.; Geiser, D.M. Members of the Fusarium solani Species Complex That Cause Infections in Both Humans and Plants Are Common in the Environment. J. Clin. Microbiol. 2006, 44, 2186–2190. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Geib, S.M.; Scully, E.D.; Jimenez-Gasco, M.d.M.; Carlson, J.E.; Tien, M.; Hoover, K. Phylogenetic Analysis of Fusarium solani Associated with the Asian Longhorned Beetle, Anoplophora glabripennis . Insects 2012, 3, 141-160. https://doi.org/10.3390/insects3010141
Geib SM, Scully ED, Jimenez-Gasco MdM, Carlson JE, Tien M, Hoover K. Phylogenetic Analysis of Fusarium solani Associated with the Asian Longhorned Beetle, Anoplophora glabripennis . Insects. 2012; 3(1):141-160. https://doi.org/10.3390/insects3010141
Chicago/Turabian StyleGeib, Scott M., Erin D. Scully, Maria del Mar Jimenez-Gasco, John E. Carlson, Ming Tien, and Kelli Hoover. 2012. "Phylogenetic Analysis of Fusarium solani Associated with the Asian Longhorned Beetle, Anoplophora glabripennis " Insects 3, no. 1: 141-160. https://doi.org/10.3390/insects3010141






