Cancer Genetic Counselor Information Needs for Risk Communication: A Qualitative Evaluation of Interview Transcripts
Abstract
:1. Introduction
2. Methods
2.1. Setting
2.2. The Interview
3. Results
3.1. Sample Characteristics
| Characteristics | Genetic counselors (N = 8) N (%) |
|---|---|
| Gender | |
| Female | 8 (100.0) |
| Male | 0 |
| Geographic location | |
| New York, NY | 2 (25.0) |
| Salt Lake City, Utah | 3 (37.5) |
| Philadelphia, PA | 3 (37.5) |
| Education | |
| Master’s degree (M.S.), genetic counseling | 8 (100.0) |
3.2. Summary of Findings
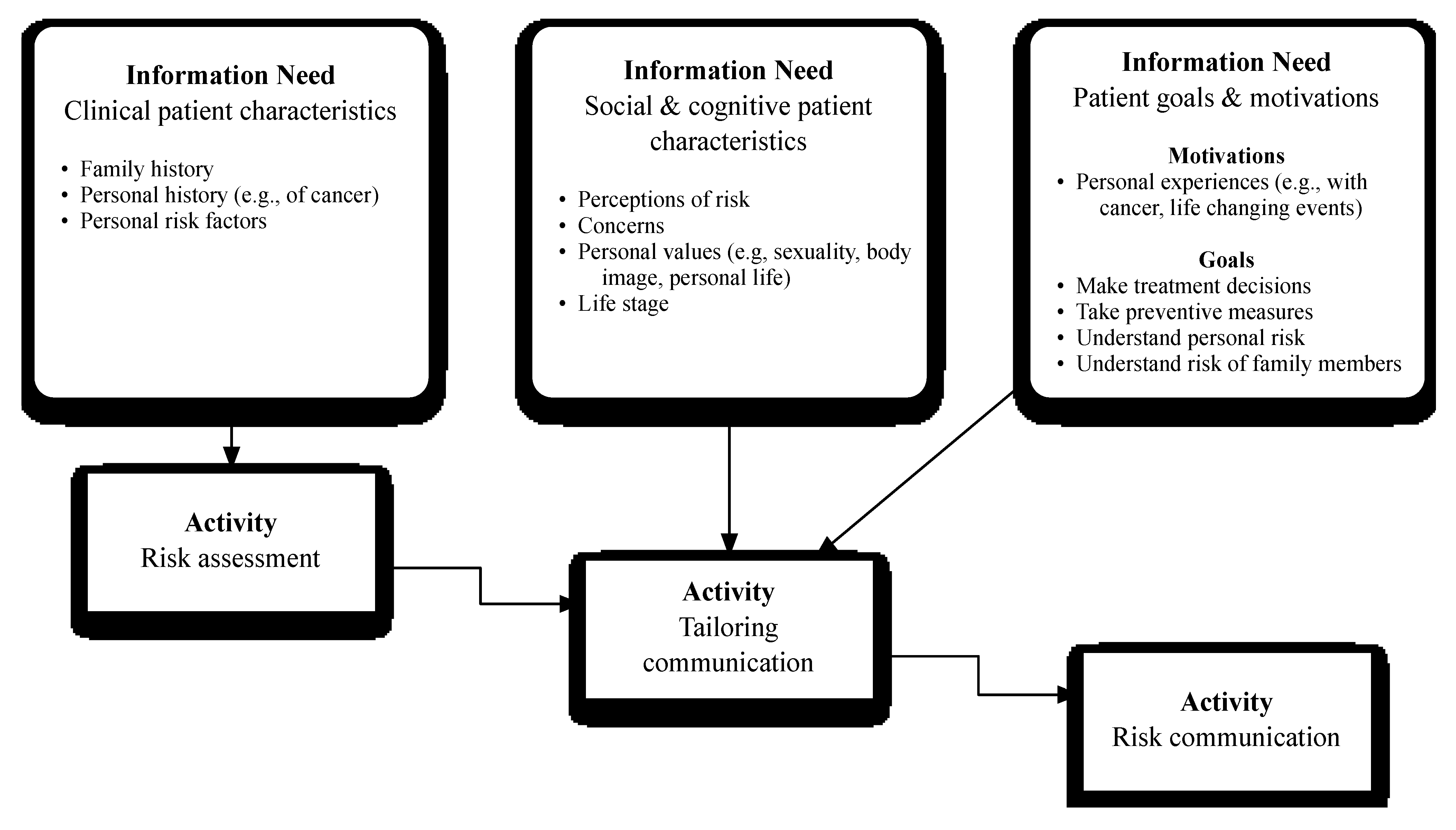
3.2.1. Theme 1: Clinical Patient Characteristics
“We send out what we call our health history questionnaire, and that’s many, many questions on your health and things that you’ve been exposed to. Now, it’s extremely comprehensive, because we’ve put together all of our risk assessment programs, so it has to do with tobacco exposure and alcohol exposure and a lot of different medication exposures, radiation exposures […]”Subject 3
“On the medical history questionnaire, we ask about cancer diagnosis, as well as personal screening, when they started getting mammograms, if they’ve had breast MRIs in the past, how many breast biopsies they’ve had, enough information for us to do a Gail Model Risk if they haven’t had breast cancer themselves. …Then, with the family history questionnaire, if they return that ahead of time, our schedulers put that into Progeny, so we already have a full pedigree drawn out when we go into the session and we’re just kind of asking for updates or any changes if they learned more information about family members in the meantime.”Subject 4
“So, we basically have most of the family history information when the patient comes in to see us; so based on that information, we can use various different models that we have to assess the likelihood that somebody may actually carry a mutation in either BRCA 1 or 2.”Subject 7
“We will get family history information, and we can plug that into Progeny and BRCAPro. So, we can get those kinds of [risk assessment] numbers before somebody actually walks in the door.”Subject 8
3.2.2. Theme 2: Social and Cognitive Patient Characteristics
“[…] for one, it was her body image, and to the other one, it was her fear of getting a new breast cancer. So, even though numerically the risk of getting a new breast cancer for both was probably 25%, the way they put that together was totally different.”Subject 3
“I try and get a sense from them if they’ve thought ahead of time, you know, if they did come back positive, what management would they be comfortable with. Would they go more towards screening or would they go more towards surgery. And for all the patients, regardless of what their response is, I do present surgery as an option, but I definitely emphasize in all cases that it’s an option and that it’s not a strict recommendation to get it. If they are very much saying they wouldn’t consider surgery, I wouldn’t go into as many details about different surgical options or different cosmetic outcomes or whatnot.”Subject 4
“I think it’s important to figure out what the patient initially perceives their risk to be. Whether they indicate that numerically or, you know, with words like high or low - so, you know where they’re coming from [and] so that you can present your risk information in the appropriate context. I think that’s figuring out how they’re perceiving things. If they’re overestimating, underestimating, maybe estimating correctly, but not necessarily assigning the right numerical value to, it helps you know what you need to present to them to hopefully get them to a point of more accurate understanding.”Subject 6
“[If] I’m getting the sense that they’re not as savvy [with numbers] as some of the patients, I really try to make it less number-based and just try to help them understand the overall concept that, you know, something increases versus decreases risks. Then, I have some patients that are just really savvy and have really great questions, and then, I’ll get into much more detail with them. So, I’ll kind of let the patient give me the cues to help me tailor.”Subject 5
“I think people need to hear the information in several different ways, so increased risks, you know, decreased risks, no risk or population risk of breast cancer, that can mean very different things to people.”Subject 2
“I had a patient last week that said ‘I don’t think numbers mean anything to me. Numbers can lie, they don’t mean anything.’ So, for that person, I really tried to tailor that session to not be just about let’s look at this bar graph and let’s talk about risk, but let’s talk about what you think […], in genetic counseling we’re told to avoid being subjective, like [using] subjective terms, like low, medium and high. But, for somebody who can’t really grasp or doesn’t like numbers, you have to really categorize these risks for them.”Subject 9
“[…] I had a patient tell me that she’s had cancer and there’s no way that she’s ever going to be cut, period, and she told me how she was in labor for three days, and they wanted to do a C-section and there was no way she was going to let them give her any kind of scar on her body. So, from there, I learned how important her body image was to her. [After hearing that] I’m really not going to spend that much time talking about risk reducing surgeries, such as prophylactic mastectomy. […] We could focus on something else.”Subject 9
3.2.3. Theme 3: Patient Goals and Motivations for the Genetic Counseling Session
“[…] my patients come in for several different reasons. I’ll see patients who are newly diagnosed with cancer, and they’re trying to make treatment decisions, so they want to know how likely it is that their cancer is hereditary and how aggressive they should be in their treatment decisions. That’s one avenue of patients. I have another avenue of patients who have never been diagnosed with cancer, but have a strong family history of cancer, and they want to find out how likely it is that the cancers in their family are hereditary cancers. And, what they can do, what steps they can take to protect themselves against getting these cancers. And then, I have other people who come in because they’ve been diagnosed with cancer in the past and are now ready to deal with whether or not the cancer is hereditary. And maybe, they want to provide helpful information for other family members or children, siblings.”Subject 3
“I don’t think when people are thinking about their risk, they’re thinking about them in numbers the way we are, so as geneticists, we’re thinking about your risk as 15 percent versus 10 percent, but for the average woman, they might just be feeling like, ‘Yeah, my mom and sister got breast cancer; it’s probably going to happen to me too.’”Subject 6
“[…] if they’ve had a lot of neighbors that grew up getting cancer, they could be really worried about the environmental effects. And, some people do live in areas where there are certain higher incidences of cancer, but oftentimes, they can consider any cancer to be greatly inflated with that. Where, at least with some of the studies, more cancers can be prone to [environment] than others. And, some of the cancer in the family may not be related to the environmental effects as much or just be very difficult to prove. So, [someone] who may be a good genetic testing candidate may still be focused on the environmental effects.”Subject 4
4. Discussion
Limitations and Implications for Future Research
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Hood, L.; Flores, M. A personal view on systems medicine and the emergence of proactive P4 medicine: Predictive, preventive, personalized and participatory. N. Biotechnol. 2012, 29, 613–624. [Google Scholar] [CrossRef]
- Escher, M.; Sappino, A.P. Primary care physicians’ knowledge and attitudes towards genetic testing for breast-ovarian cancer predisposition. Ann. Oncol. 2000, 11, 1131–1135. [Google Scholar] [CrossRef]
- Freedman, A.N.; Wideroff, L.; Olson, L.; Davis, W.; Klabunde, C.; Srinath, K.P.; Reeve, B.B.; Croyle, R.T.; Ballard-Barbash, R. US physicians’ attitudes toward genetic testing for cancer susceptibility. Am. J. Med. Genet. Part A 2003, 120A, 63–71. [Google Scholar] [CrossRef]
- Marzuillo, C.; de Vito, C.; Boccia, S.; D’ÄôAddario, M.; D’ÄôAndrea, E.; Santini, P.; Boccia, A.; Villari, P. Knowledge, attitudes and behavior of physicians regarding predictive genetic tests for breast and colorectal cancer. Prev. Med. 2013. [Google Scholar] [CrossRef] [Green Version]
- Bellcross, C.A.; Kolor, K.; Goddard, K.A.; Coates, R.J.; Reyes, M.; Khoury, M.J. Awareness and utilization of BRCA1/2 testing among U.S. primary care physicians. Am. J. Prev. Med. 2011, 40, 61–66. [Google Scholar] [CrossRef]
- Klitzman, R.; Chung, W.; Marder, K.; Shanmugham, A.; Chin, L.J.; Stark, M.; Leu, C.S.; Appelbaum, P.S. Attitudes and practices among internists concerning genetic testing. J. Genet. Couns 2013, 22, 90–100. [Google Scholar] [CrossRef]
- Wideroff, L.; Vadaparampil, S.T.; Greene, M.H.; Taplin, S.; Olson, L.; Freedman, A.N. Hereditary breast/ovarian and colorectal cancer genetics knowledge in a national sample of US physicians. J. Med. Genet. 2005, 42, 749–755. [Google Scholar] [CrossRef]
- Veach, P.M.; Bartels, D.M.; Leroy, B.S. Coming full circle: A reciprocal-engagement model of genetic counseling practice. J. Genet. Couns. 2007, 16, 713–728. [Google Scholar] [CrossRef]
- Vos, J.; Stiggelbout, A.M.; Oosterwijk, J.; Gomez-Garcia, E.; Menko, F.; Collee, J.M.; van Asperen, C.J.; Tibben, A. A counselee-oriented perspective on risk communication in genetic counseling: Explaining the inaccuracy of the counselees’ risk perception shortly after BRCA1/2 test result disclosure. Genet. Med. 2011, 13, 800–811. [Google Scholar] [CrossRef]
- Miller, S.J.; Hope, T.; Talbot, D.C. The development of a structured rating schedule (the BAS) to assess skills in breaking bad news. Br. J. Cancer 1999, 80, 792–800. [Google Scholar] [CrossRef]
- Charles, C.; Gafni, A.; Whelan, T. Shared decision-making in the medical encounter: What does it mean? (or it takes at least two to tango). Soc. Sci. Med. 1997, 44, 681–692. [Google Scholar] [CrossRef]
- Dy, S.M.; Purnell, T.S. Key concepts relevant to quality of complex and shared decision-making in health care: A literature review. Soc. Sci. Med. 2012, 74, 582–587. [Google Scholar] [CrossRef]
- Yen, P.Y.; Bakken, S. Review of health information technology usability study methodologies. J. Am. Med. Inform. Assoc. 2012, 19, 413–422. [Google Scholar] [CrossRef]
- Strauss, A.; Corbin, J. Basics of Qualitative Research: Grounded Theory Procedures and Technique; SAGE: Newbury Park, CA, USA, 1998. [Google Scholar]
- Gail, M.H.; Brinton, L.A.; Byar, D.P.; Corle, D.K.; Green, S.B.; Schairer, C.; Mulvihill, J.J. Projecting individualized probabilities of developing breast cancer for white females who are being examined annually. J Natl. Cancer Inst. 1989, 81, 1878–1886. [Google Scholar]
- Parmigiani, G.; Berry, D.; Aguilar, O. Determining carrier probabilities for breast cancer-susceptibility genes BRCA1 and BRCA2. Am. J. Hum. Genet. 1998, 62, 145–158. [Google Scholar] [CrossRef]
- Meiser, B.; Irle, J.; Lobb, E.; Barlow-Stewart, K. Assessment of the content and process of genetic counseling: A critical review of empirical studies. J. Genet. Couns. 2008, 17, 434–451. [Google Scholar] [CrossRef]
- Pieterse, A.H.; van Dulmen, A.M.; Ausems, M.G.; Beemer, F.A.; Bensing, J.M. Communication in cancer genetic counselling: Does it reflect counselees’ previsit needs and preferences? Br. J. Cancer 2005, 92, 1671–1678. [Google Scholar] [CrossRef]
- Bernhardt, B.A.; Biesecker, B.B.; Mastromarino, C.L. Goals, benefits, and outcomes of genetic counseling: Client and genetic counselor assessment. Am. J. Med. Genet. 2000, 94, 189–197. [Google Scholar] [CrossRef]
- Pieterse, A.; van Dulmen, S.; Ausems, M.; Schoemaker, A.; Beemer, F.; Bensing, J. QUOTE-gene(ca): Development of a counselee-centered instrument to measure needs and preferences in genetic counseling for hereditary cancer. Psychooncology 2005, 14, 361–375. [Google Scholar] [CrossRef]
- Albada, A.; van Dulmen, S.; Ausems, M.G.; Bensing, J.M. A pre-visit website with question prompt sheet for counselees facilitates communication in the first consultation for breast cancer genetic counseling: Findings from a randomized controlled trial. Genet. Med. 2012, 14, 535–542. [Google Scholar]
- Belkora, J.; Katapodi, M.; Moore, D.; Franklin, L.; Hopper, K.; Esserman, L. Evaluation of a visit preparation intervention implemented in two rural, underserved counties of Northern California. Patient Educ. Couns. 2006, 64, 350–359. [Google Scholar] [CrossRef]
- Sepucha, K.R.; Belkora, J.K.; Aviv, C.; Mutchnik, S.; Esserman, L.J. Improving the quality of decision making in breast cancer: Consultation planning template and consultation recording template. Oncol. Nurs. Forum 2003, 30, 99–106. [Google Scholar] [CrossRef]
|
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Overby, C.L.; Chung, W.K.; Hripcsak, G.; Kukafka, R. Cancer Genetic Counselor Information Needs for Risk Communication: A Qualitative Evaluation of Interview Transcripts. J. Pers. Med. 2013, 3, 238-250. https://doi.org/10.3390/jpm3030238
Overby CL, Chung WK, Hripcsak G, Kukafka R. Cancer Genetic Counselor Information Needs for Risk Communication: A Qualitative Evaluation of Interview Transcripts. Journal of Personalized Medicine. 2013; 3(3):238-250. https://doi.org/10.3390/jpm3030238
Chicago/Turabian StyleOverby, Casey Lynnette, Wendy K. Chung, George Hripcsak, and Rita Kukafka. 2013. "Cancer Genetic Counselor Information Needs for Risk Communication: A Qualitative Evaluation of Interview Transcripts" Journal of Personalized Medicine 3, no. 3: 238-250. https://doi.org/10.3390/jpm3030238




