Individual Oral Therapy with Immediate Release and Effervescent Formulations Delivered by the Solid Dosage Pen
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effervescent Formulation
2.1.1. Formulation Development of Effervescent Formulation
| Effervescent formulation | |
|---|---|
| Metoprolol tartrate | 18 |
| Mannitol | 38 |
| Hydrogen carbonate | 38 |
| Tartaric acid | 6 |

2.1.2. Dosing of Effervescent Dosage Forms


| Dosage units (DU) | Nominal content of a single dose (mg) | Content (%)(MW ± SD) | Acceptance value(AV) |
|---|---|---|---|
| 3 | 1.5 | 101.8 ± 4.5 | 11.18 |
| 5 | 2.5 | 101.4 ± 2.9 | 6.98 |
| 10 | 5 | 99.8 ± 3.1 | 7.49 |
2.1.3. Characterization of cut Effervescent Doses
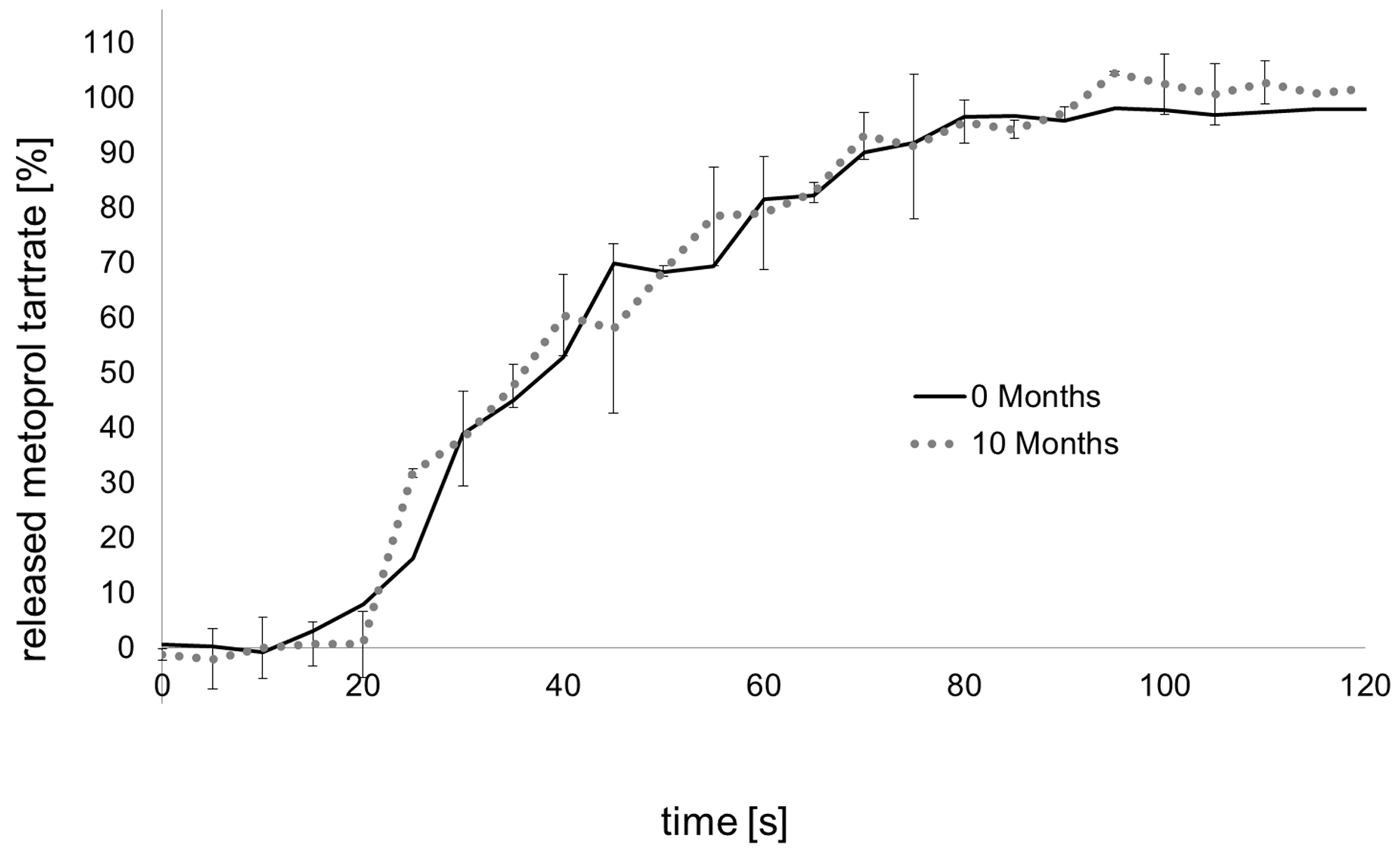
2.2. Immediate Release Melt-Extrudates
2.2.1. Formulation Development of Immediate Release Melt-Extrudates
| Batch acronyms | LuCarv | LumiCarv | LuMet | LumiMet |
|---|---|---|---|---|
| Carvedilol | 5 | 5 | ||
| Metoprolol tartrate | 20 | 20 | ||
| Mannitol | 45 | 45 | 34 | 34 |
| Poloxamer 188 | 50 | 46 | ||
| Poloxamer 188 micro | 50 | 46 |

2.2.2. Dosing of Immediate Release Melt-Extrudates

2.2.3. Characterization of Cut Immediate Release Melt-Extrudate Doses

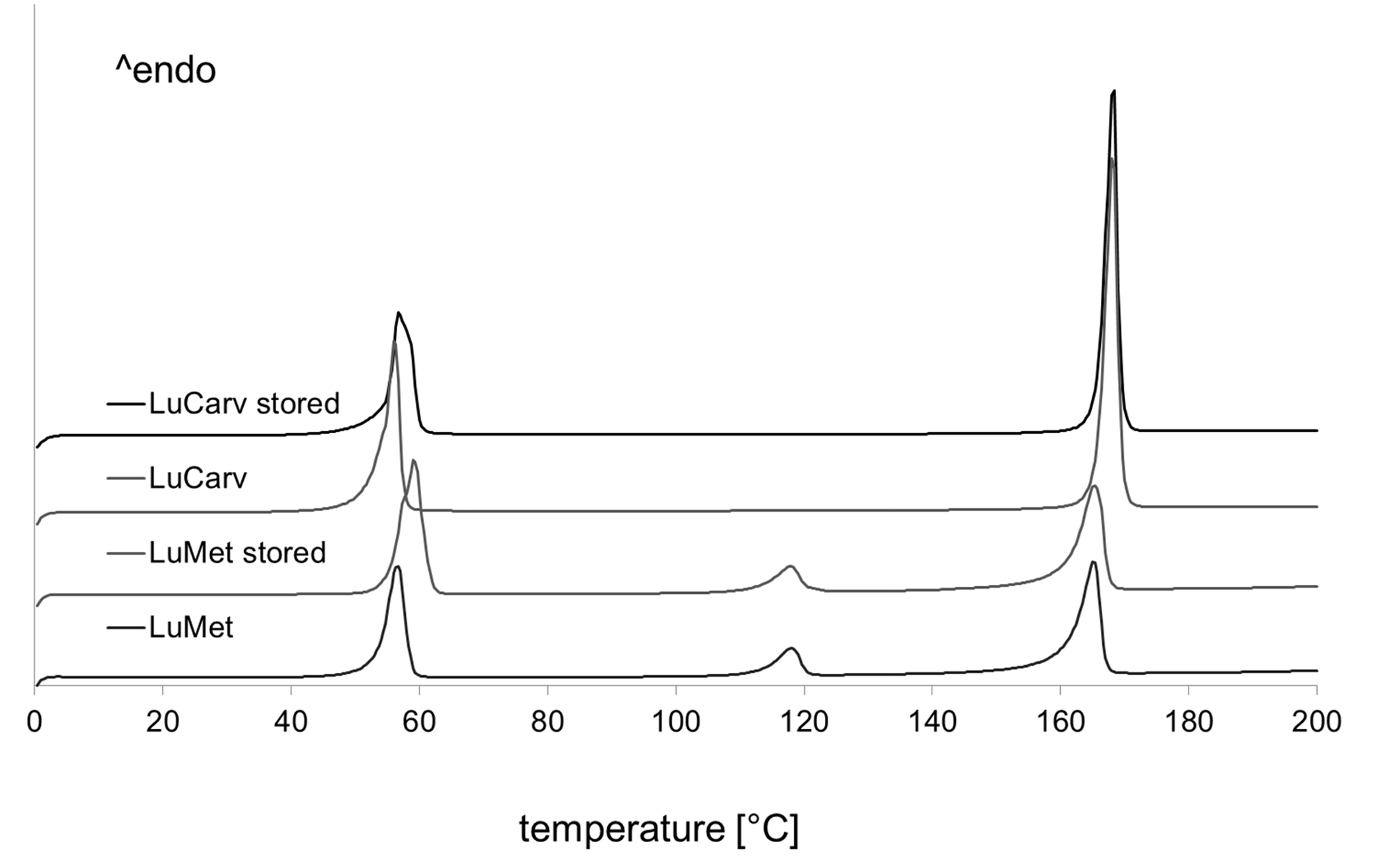

3. Experimental
3.1. Materials
3.2. Methods
3.2.1. Extrusion
3.2.2. Drug Release

3.2.3. Uniformity of Content of Single Doses
3.2.4. AV-Value
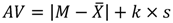
3.2.5. Differential Scanning Calorimetry (DSC)
3.2.6. Determination of Cutting-Forces
4. Conclusions
Acknowledgments
Conflict of Interest
References and Notes
- Stehle, S.; Kirchheiner, J.; Lazar, A.; Fuhr, U. Pharmacogenetics of oral anticoagulants-A basis for dose individualization. Clin. Pharmacokinet. 2008, 47, 565–594. [Google Scholar] [CrossRef]
- Cholerton, S.; Daly, A.K.; Idle, J.R. The role of individual human cytochromes-P450 in drug-metabolism and clinical-response. Trends Pharmacol. Sci. 1992, 13, 434–439. [Google Scholar] [CrossRef]
- Ingelman-Sundberg, M.; Sim, S.C.; Gomez, A.; Rodriguez-Antona, C. Influence of cytochrome P450 polymorphisms on drug therapies: Pharmacogenetic, pharmacoepigenetic and clinical aspects. Pharmacol. Ther. 2007, 116, 496–526. [Google Scholar] [CrossRef]
- Zhou, S.F.; Di, Y.M.; Chan, E.; Du, Y.M.; Chow, V.D.W.; Xue, C.C.L.; Lai, X.S.; Wang, J.C.; Li, C.G.; Tian, M.; et al. Clinical Pharmacogenetics and Potential Application in Personalized Medicine. Curr. Drug Metab. 2008, 9, 738–784. [Google Scholar] [CrossRef]
- Kearns, G.L.; Abdel-Rahman, S.M.; Alander, S.W.; Blowey, D.L.; Leeder, J.S.; Kauffman, R.E. Developmental pharmacology-Drug disposition, action, and therapy in infants and children. N. Engl. J. Med. 2003, 349, 1157–1167. [Google Scholar] [CrossRef]
- Standing, J.F.; Tuleu, C. Paediatric formulations-Getting to the heart of the problem. Int. J. Pharm. 2005, 300, 56–66. [Google Scholar] [CrossRef]
- Breitkreutz, J.; Boos, J. Paediatric and geriatric drug delivery. Expert Opin. Drug Deliv. 2007, 4, 37–45. [Google Scholar] [CrossRef]
- Stegemann, S.; Ecker, F.; Maio, M.; Kraahs, P.; Wohlfart, R.; Breitkreutz, J.; Zimmer, A.; Bar-Shalom, D.; Hettrich, P.; Broegmann, B. Geriatric drug therapy: Neglecting the inevitable majority. Ageing Res. Rev. 2010, 9, 384–398. [Google Scholar] [CrossRef]
- Wening, K.; Breitkreutz, J. Oral drug delivery in personalized medicine: Unmet needs and novel approaches. Int. J. Pharm. 2011, 404, 1–9. [Google Scholar] [CrossRef]
- Stoltenberg, I.; Winzenburg, G.; Breitkreutz, J. Solid oral dosage forms for children- Formulations, excipients and acceptance issues. J. Appl. Ther. Res. 2010, 7, 141–146. [Google Scholar]
- Walsh, J.; Bickmann, D.; Breitkreutz, J.; Chariot-Goulet, M. Delivery devices for the administration of paediatric formulations: Overview of current practice, challenges and recent developments. Int. J. Pharm. 2011, 415, 221–231. [Google Scholar] [CrossRef]
- Breitkreutz, J.; Wessel, T.; Boos, J. Dosage Forms for Peroral Drug Administration to Children. Paediatr. Perinat. Drug Ther. 1999, 3, 25–33. [Google Scholar]
- van Santen, E.; Barends, D.M.; Frijlink, H.W. Breaking of scored tablets: A review. Eur. J. Pharm. Biopharm. 2002, 53, 139–145. [Google Scholar] [CrossRef]
- Schoemakers, J.; Grummel, A. Dosing Stick Containing Rod-Shaped Tablets. WO 02/102296 A102291 2002. [Google Scholar]
- Wening, K.; Breitkreutz, J. Novel delivery device for monolithical solid oral dosage forms for personalized medicine. Int. J. Pharm. 2010, 395, 174–181. [Google Scholar] [CrossRef]
- Frobel, A.K. Untersuchung der Pharmakokinetik von Bisoprolol bei Kindern in einer klinischen Studie und mit physiologiebasierter Simulation als Konsequenz aus einem systematischen Cochrane-Review und als Beitrag zur Optimierung zukünftiger Studien (in German). Ph.D. Thesis, University of Duesseldorf, Duesseldorf, Germany, 2010. [Google Scholar]
- Laer, S.; Mir, T.S.; Behn, F.; Eiselt, M.; Scholz, H.; Venzke, A.; Meibohm, B.; Weil, J. Carvedilol therapy in pediatric patients with congestive heart failure: A study investigating clinical and pharmacokinetic parameters. Am. Heart J. 2002, 143, 916–922. [Google Scholar] [CrossRef]
- Hermes, M.; Barnscheid, L.; Garsuch, V.; Schoettler, P.; Dominguez-Hirschi, J.; Breitkreutz, J. Arzneimittelversorgung von Kindern: Ergebnisse einer Studie zur praktischen Auswirkung der EU-Verordnung 1901/2006 "Better Medicines for children" seit ihrem Inkrafttreten im Januar 2007 (in German); [Drug prescription for children: Results of a study on the practical effect of the EU regulation No. 1901/2006 "Better medicines for children" since its coming into effect in January 2007]. Pharm. Ind. 2010, 72, 1334–1344. [Google Scholar]
- European Medicines Agency (EMA). Formulations of Choice for the Paediatric Population: Reflection Paper. London, UK, 2006. [Google Scholar]
- Lindberg, N.O.; Myrenas, M.; Tufvesson, C.; Olbjer, L. Extrusion of an effervescent granulation with a twin screw extruder, Baker Perkins MPF 50D-Determination of mean residence time. Drug Dev. Ind. Pharm. 1988, 14, 649–655. [Google Scholar] [CrossRef]
- Kircher, W. Arzneiformen richtig anwenden,ergonomische Aspekte (in German). [Correct Use of Dosage Forms,Ergonomic Aspects]; Deutscher Apothekerverlag Stuttgart: Stuttgart, Germany, 2007; pp. 17–29. [Google Scholar]
- Garsuch, V.; Breitkreutz, J. Novel analytical methods for the characterization of oral wafers. Eur. J. Pharm. Biopharm. 2009, 73, 195–201. [Google Scholar] [CrossRef]
- Dubey, R.; Jamalapuram, S. Development of intravenously injectable solution of carvedilol using a combination of pH adjustment and micellization. Eur. J. Parenter. Pharm. Sci. 2010, 15, 47–52. [Google Scholar]
- Thommes, M.; Baert, L.; Rosier, J. 800 mg Darunavir tablets prepared by hot melt extrusion. Pharm. Dev. Technol. 2011, 16, 645–650. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wening, K.; Laukamp, E.J.; Thommes, M.; Breitkreutz, J. Individual Oral Therapy with Immediate Release and Effervescent Formulations Delivered by the Solid Dosage Pen. J. Pers. Med. 2012, 2, 217-231. https://doi.org/10.3390/jpm2040217
Wening K, Laukamp EJ, Thommes M, Breitkreutz J. Individual Oral Therapy with Immediate Release and Effervescent Formulations Delivered by the Solid Dosage Pen. Journal of Personalized Medicine. 2012; 2(4):217-231. https://doi.org/10.3390/jpm2040217
Chicago/Turabian StyleWening, Klaus, Eva Julia Laukamp, Markus Thommes, and Jörg Breitkreutz. 2012. "Individual Oral Therapy with Immediate Release and Effervescent Formulations Delivered by the Solid Dosage Pen" Journal of Personalized Medicine 2, no. 4: 217-231. https://doi.org/10.3390/jpm2040217




