Microgravity-Induced Fluid Shift and Ophthalmic Changes
Abstract
:1. Introduction
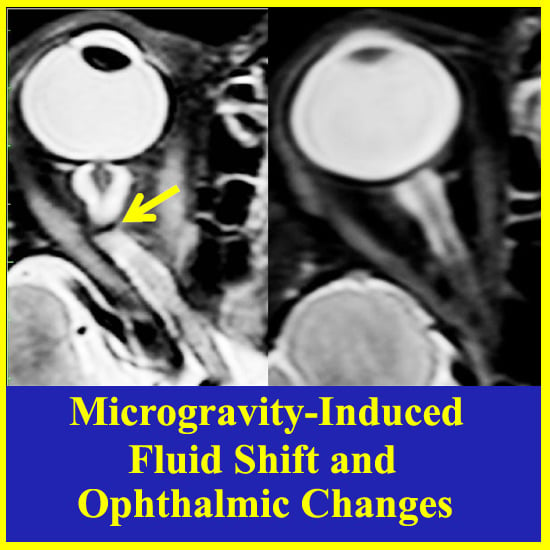
2. The Visual Impairment and Intracranial Pressure (VIIP) Syndrome
| CPG Class | Definition | No. of Affected Crewmembers |
|---|---|---|
| Non Cases |
| 2 |
| 1 | (repeat Optical Coherence Tomography (OCT) and visual acuity in 6 weeks)
| 2 |
| 2 | (repeat OCT, cycloplegic refraction, fundus exam and threshold visual field every four to six weeks x six months, repeat Magnetic Resonance Imaging (MRI) in six months)
| 8 |
| 3 | (repeat OCT, cycloplegic refraction, fundus exam and threshold visual field every four to six weeks x six months, repeat MRI in six months)
| 1 |
| 4 | (institute treatment protocol as per CPG)
| 4 |
| Unclassified |
| 19 |
| Ophthalmic Condition | Total Affected |
|---|---|
| Optic nerve sheath distension | 6/7 (86%) |
| Nerve fiber layer thickening | 6/7 (86%) |
| Optic disc edema | 5/7 (71%) |
| Posterior globe flattening | 5/7 (71%) |
| Hyperopic shift in one or both eyes by >+0.50 diopters | 5/7 (71%) |
| Choroidal folds | 4/7 (57%) |
| Elevated postflight CSF pressure (indicative of increased ICP) | 4/7 (57%) |
| Cotton wool spots | 3/7 (43%) |
| Decreased intraocular pressure (IOP) postflight | 3/7 (43%) |
| Tortuous optic nerve | 2/7 (29%) |
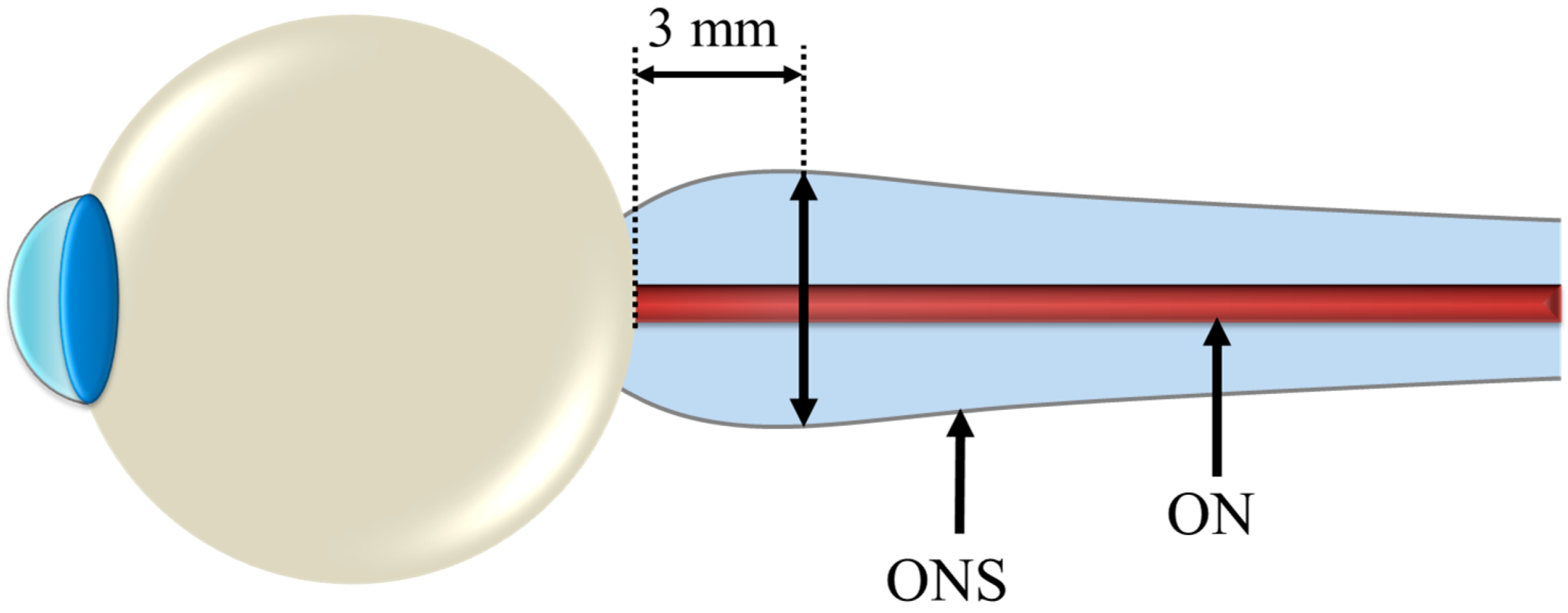






3. Fluid Redistribution in Microgravity
3.1. Reduced Gravity Environments
3.2. Simple Physical Systems
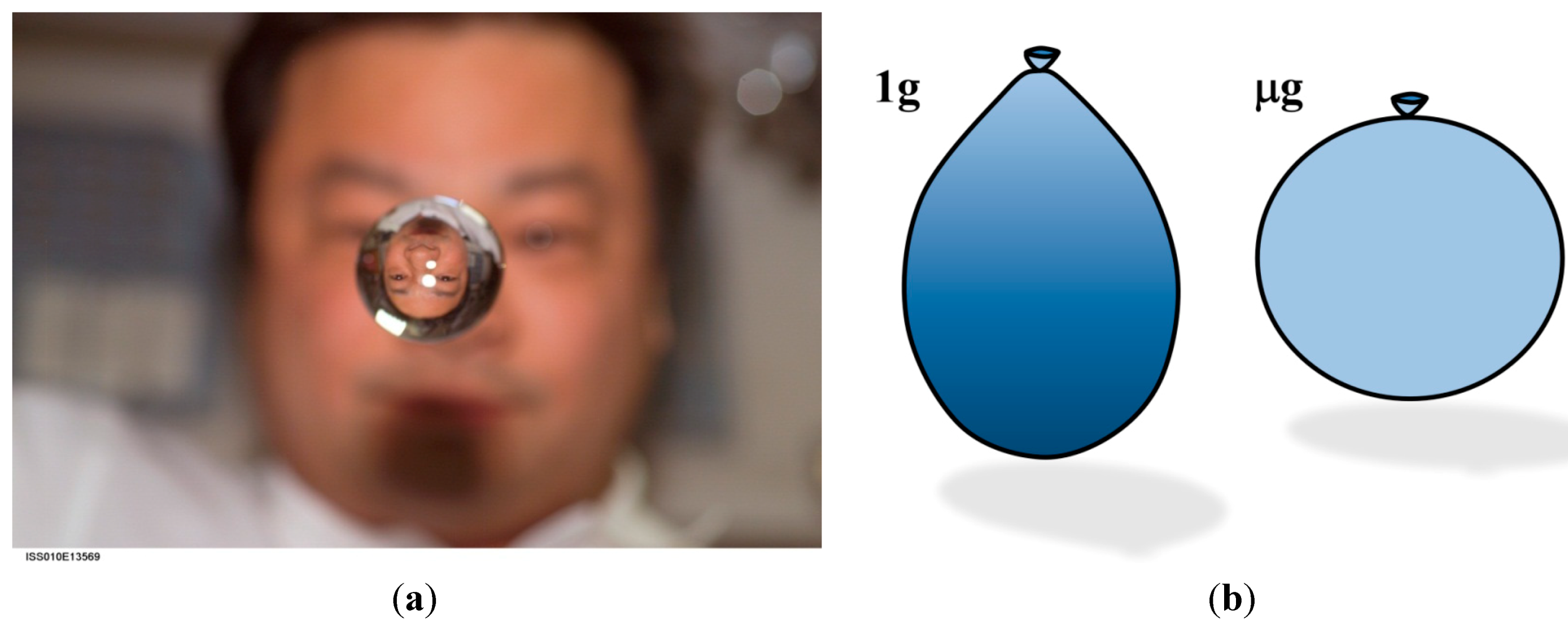
3.3. Human Beings in Space and the Cephalic Fluid Shift
3.3.1. Hydrostatic and Biomechanical Effects on Humans in Spaceflight and Simulated Spaceflight

- The height of the fluid column,
- The initial fluid volume,
- The volume, saturation potential and material properties of fat, muscle and other porous tissues,
- The bones, cartilage and other tissues that may limit volumetric expansion,
- The water storage capacity and compliance of vascular and extravascular systems,
- The presence of semipermeable membranes that separate fluid volumes, such as the blood/brain barrier, and
- The distribution of all these components along the height of the body.
3.3.2. Experimental Data on the Impact of Cephalic Fluid Shift

4. The Ocular Environment in Spaceflight
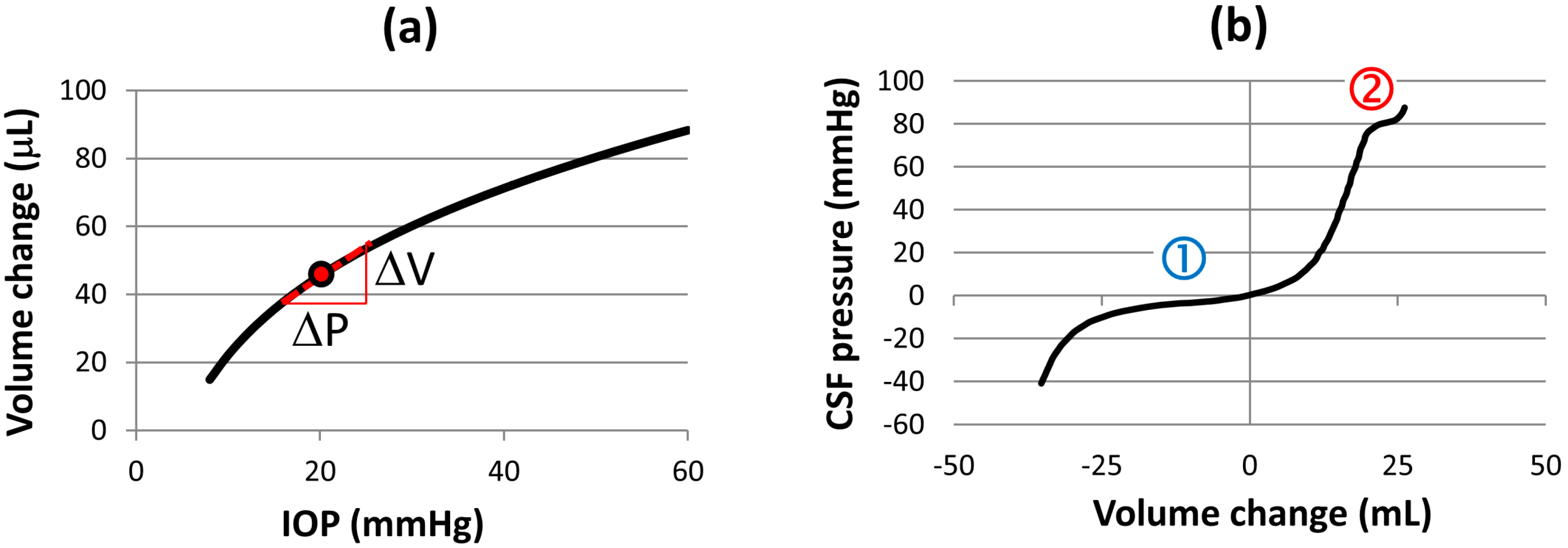
4.1. Intracranial Pressure Dynamics, Ocular Blood Flow and Biomechanics
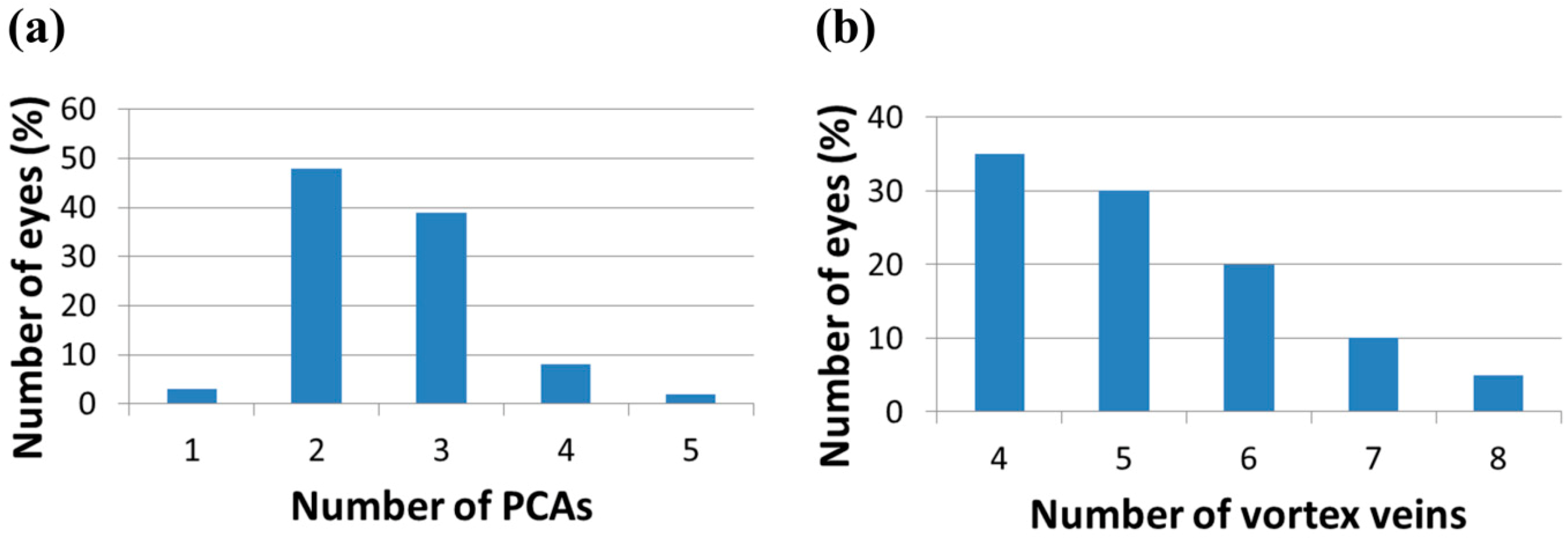
4.2. The Spacecraft Environment and Other Potential Factors in the Development of VIIP
- Low humidity and high CO2 concentrations, with periods of exposure to low total pressure and 100% O2 during extravehicular activity,
- Increased radiation levels with the potential of exposure to high energy ‘cosmic ray’ radiation,
- A high salt diet resulting from a change in taste acuity, and
- A disrupted circadian rhythm.
5. Concluding Remarks
Nomenclature and Abbreviations
| A | Mean cross-sectional area inside the blood vessel |
| COP | Colloid Osmotic Pressure |
| CPG | Clinical Practice Guidelines |
| CRA | Central Retinal Artery |
| CSF | Cerebrospinal fluid |
| CWS | Cotton wool spots |
| d | Diameter |
| GCR | Galactic Cosmic Rays |
| g | Gravitational or net acceleration |
| ge | Gravitational acceleration at sea level on earth |
| h | Axis along which gravity acts |
| ĥ | Local axis aligned with the height of the body |
| Hb | Total height of the body |
| Ht | Total height of the tubular balloon |
| HDT | Head-Down Tilt |
| ICP | Intracranial Pressure |
| IIH | Idiopathic Intracranial Hypertension |
| IOP | Intraocular Pressure |
| ISS | International Space Station |
| m | Mass |
| MCA | Middle Cerebral Artery |
| MR | Magnetic resonance (imaging) |
| OCT | Optical coherence tomography |
| OA | Ophthalmic artery |
| OD | Oculus dexter (right eye) |
| ON | Optic nerve |
| OND | Optic nerve disc |
| ONS | Optic nerve sheath |
| ONSD | Optic nerve sheath diameter |
| OS | Oculus sinister (left eye) |
| OV | Ophthalmic vein |
| pcsf | Cerebrospinal fluid pressure |
| PV | Plasma Volume |
| Q | Mean volumetric flow rate |
| RBC | Red blood cell |
| SPE | Solar particle event |
| SRB | Solid Rocket Boosters |
| SSME | Space Shuttle Main Engines |
| V | Mean velocity |
| VIIP | Visual Impairment and Intracranial Pressure |
| W | Weight |
| Δph | Hydrostatic pressure gradient |
| ρ | Density |
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- This cephalad shifting of fluid from the extremities toward the head is often referred to as the “cephalad fluid shift” in the spaceflight community.
- LeBlanc, A.; Schneider, V.; Shackelford, L.; West, S.; Oganov, V.; Bakulin, A.; Voronin, L. Bone mineral and lean tissue loss after long duration space flight. J. Musculoskelet. Neuronal Interact. 2000, 1, 157–160. [Google Scholar] [PubMed]
- LeBlanc, A.; Lin, C.; Shackelford, L.; Sinitsyn, V.; Evans, H.; Belichenko, O.; Schenkman, B.; Kozlovskaya, I.; Oganov, V.; Bakulin, A.; et al. Muscle volume, MRI relaxation times (T2), and body composition after spaceflight. J. Appl. Physiol. 2000, 89, 2158–2164. [Google Scholar]
- Blaber, A.P.; Goswami, N.; Bondar, R.L.; Kassam, M.S. Impairment of cerebral blood flow regulation in astronauts with orthostatic intolerance after flight. Stroke 2011, 42, 1844–1850. [Google Scholar] [CrossRef] [PubMed]
- Clement, G.; Ngo-Anh, J.T. Space physiology II: Adaptation of the central nervous system to space flight--past, current, and future studies. Eur. J. Appl. Physiol. 2013, 113, 1655–1672. [Google Scholar] [CrossRef]
- Pietrzyk, R.A.; Jones, J.A.; Sams, C.F.; Whitson, P.A. Renal stone formation among astronauts. Aviat. Space Environ. Med. 2007, 78, A9–A13. [Google Scholar] [PubMed]
- Alexander, D.J.; Gibson, C.R.; Hamilton, D.R.; Lee, S.M.C.; Mader, T.H.; Otto, C.; Oubre, C.M.; Pass, A.F.; Platts, S.; Scott, J.M.; et al. Evidence Report: Risk of Spaceflight-Induced Intracranial Hypertension and Vision Alterations; NASA: Washington, DC, USA, 2012. Available online: http://humanresearchroadmap.nasa.gov/evidence/reports/VIIP.pdf (accessed on 4 November 2014).
- Convertino, V.A.; Rickards, C.A. Human Models of Space Physiology. In Sourcebook of Models for Biomedical Research; Conn, P.M., Ed.; Humana Press: New York, NY, USA, 2008; pp. 457–464. [Google Scholar]
- Alessandri, N.; Petrassi, M.; Tufano, F.; Giudici, A.D.; de Angelis, S.; Urciuoli, F.; Alessandri, C.; de Angelis, C.; Tomao, E. Functional changes cardiovascular: Normobaric activity and microgravity in young healthy human subjects. Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 310–315. [Google Scholar] [PubMed]
- Thornton, W.E.; Hoffler, G.W.; Rummel, J.A. Anthropometric changes and fluid shifts. In Biomedical Results from Skylab; Johnston, R.S., Dietlein, L.F., Eds.; NASA: Washington, DC, USA, 1987; pp. 330–338. [Google Scholar]
- Johnston, S.L.; Campbell, M.R.; Scheuring, R.; Feiveson, A.H. Risk of herniated nucleus pulposus among US astronauts. Aviat. Space Environ. Med. 2010, 81, 566–574. [Google Scholar] [CrossRef] [PubMed]
- Blaber, A.P.; Zuj, K.A.; Goswami, N. Cerebrovascular autoregulation: Lessons learned from spaceflight research. Eur. J. Appl. Physiol. 2013, 113, 1909–1917. [Google Scholar] [CrossRef] [PubMed]
- Prisk, G.K. Invited review: Microgravity and the lung. J. Appl. Physiol. 2000, 89, 385–396. [Google Scholar] [PubMed]
- Taibbi, G.; Cromwell, R.L.; Kapoor, K.G.; Godley, B.F.; Vizzeri, G. The effect of microgravity on ocular structures and visual function: A review. Surv. Ophthalmol. 2013, 58, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Kramer, L.A.; Sargsyan, A.E.; Hasan, K.M.; Polk, J.D.; Hamilton, D.R. Orbital and intracranial effects of microgravity: Findings at 3-T MR imaging. Radiology 2012, 263, 819–827. [Google Scholar] [CrossRef] [PubMed]
- Mader, T.H.; Gibson, C.R.; Pass, A.F.; Kramer, L.A.; Lee, A.G.; Fogarty, J.; Tarver, W.J.; Dervay, J.P.; Hamilton, D.R.; Sargsyan, A.; et al. Optic disc edema, globe glattening, choroidal folds, and hyperopic shifts observed in astronauts after long-duration space flight. Ophthalmology 2011, 118, 2058–2069. [Google Scholar] [CrossRef] [PubMed]
- Duntley, S.Q.; Austin, R.W.; Taylor, J.L. Experiments S-8/D-13, Visual Acuity and Astronaut Visibility. In Gemini Midprogram Conference; NASA: Washington, DC, USA, 1966. [Google Scholar]
- Nicogossian, A.E.; Huntoon, C.; Pool, S.L. Space Physiology and Medicine; Lea & Febiger: Philadelphia, PA, USA; London, UK, 1989. [Google Scholar]
- Mader, T.H. Intraocular pressure in microgravity. J. Clin. Pharmacol. 1991, 31, 947–950. [Google Scholar] [CrossRef] [PubMed]
- Mader, T.H.; Gibson, C.R.; Caputo, M.; Hunter, N.; Taylor, G.; Charles, J.; Meehan, R.T. Intraocular pressure and retinal vascular changes during transient exposure to microgravity. Am. J. Ophthalmol. 1993, 115, 347–350. [Google Scholar] [CrossRef] [PubMed]
- Chiquet, C.; Custaud, M.A.; Le Traon, A.P.; Millet, C.; Gharib, C.; Denis, P. Changes in intraocular pressure during prolonged (7-day) head-down tilt bedrest. J. Glaucoma 2003, 12, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Kergoat, H.; Lovasik, J.V. Seven-degree head-down tilt reduces choroidal pulsatile ocular blood flow. Aviat. Space Environ. Med. 2005, 76, 930–934. [Google Scholar] [PubMed]
- Longo, A.; Geiser, M.H.; Riva, C.E. Posture changes and subfoveal choroidal blood flow. Invest. Ophthalmol. Vis. Sci. 2004, 45, 546–551. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Li, L.; Cao, R.D.; Tao, Y.; Guo, Q.; Geng, J.; Li, Y.Z.; Zhang, Z.M. Intraocular pressure and ocular perfusion pressure in myopes during 21 min head-down rest. Aviat. Space Environ. Med. 2010, 81, 418–422. [Google Scholar] [CrossRef] [PubMed]
- Tarver, W.J.; Otto, C. NASA’s Spaceflight Visual Impairment Intracranial Pressure (VIIP) Risk: Clinical Correlations and Pathophysiology. In Aerospace Medicine Grand Rounds; NASA: Washington, DC, USA, 2012. [Google Scholar]
- Bershad, E.M. Intracranial Pressure in Microgravity: The Shifting of Water. In Papilledema Summit; Baylor College of Medicine: Houston, TX, USA, 2011. [Google Scholar]
- Roach, R. Analogs: Intracranial Pressure at High Altitude. In Papilledema Summit; Baylor College of Medicine: Houston, TX, USA, 2011. [Google Scholar]
- Jonas, J.B.; Nangia, V.; Matin, A.; Sinha, A.; Kulkarni, M.; Bhojwani, K. Intraocular pressure and associated factors: The Central India Eye and Medical Study. J. Glaucoma 2011, 20, 405–409. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P.; Lee, A.J.; Wang, J.J.; Rochtchina, E. Intraocular pressure over the clinical range of blood pressure: Blue Mountains Eye Study findings. Am. J. Ophthalmol. 2005, 140, 131–132. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Wong, T.Y.; Mitchell, P.; Friedman, D.S.; He, M.; Aung, T. Distribution of ocular perfusion pressure and its relationship with open-angle glaucoma: The Singapore Malay Eye Study. Invest. Ophthalmol. Vis. Sci. 2010, 51, 3399–404. [Google Scholar] [CrossRef]
- Geeraerts, T.; Newcombe, V.; Coles, J.; Abate, M.; Perkes, I.; Hutchinson, P.J.; Outtrim, J.; Chatfield, D.; Menon, D. Use of T2-weighted magnetic resonance imaging of the optic nerve sheath to detect raised intracranial pressure. Crit. Care 2008, 12, 1–7. [Google Scholar] [CrossRef]
- Brodsky, M.C.; Vaphiades, M. Magnetic resonance imaging in pseudotumor cerebri. Ophthalmology 1998, 105, 1686–1693. [Google Scholar] [CrossRef] [PubMed]
- Degnan, A.J.; Levy, L.M. Pseudotumor cerebri: Brief review of clinical syndrome and imaging findings. Am. J. Neuroradiol. 2011, 32, 1986–1993. [Google Scholar] [CrossRef] [PubMed]
- Arora, A.; Upreti, L.; Kapoor, A.; Gupta, R.; Sharma, P.; Puri, S.K. Pseudotumor Cerebri: MR Imaging Findings; Department of Radiodiagnosis, G.B. Pant Hospital and associated Maulana Azad Medical College: New Delhi, India, 2010. [Google Scholar]
- George, U.; Bansal, G.; Pandian, J. Magnetic resonance “flip-flop” in idiopathic intracranial hypertension. J. Neurosci. Rural Pract. 2011, 2, 84–86. [Google Scholar]
- Wall, M. Idiopathic intracranial hypertension. Neurol. Clin. 2010, 28, 593–617. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Lai, T.F.; Leibovitch, I.; Franzco, D.S. Persistent posterior globe flattening after orbital cavernous haemangioma excision. Clin. Exp. Ophthalmol. 2005, 33, 424–425. [Google Scholar] [CrossRef]
- Bullock, J.D.; Egbert, P.R. The origin of choroidal folds: A clinical, histopathological and experimental study. Doc. Ophthalmol. 1974, 37, 261–293. [Google Scholar] [CrossRef]
- Jaworski, A.; Wolffsohn, J.S.; Napper, G.A. Aetiology and management of choroidal folds. Clin. Exp. Optom. 1999, 82, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Kashiwada, S.; Ferrucci, S.; Peschke, K.; Grimes, A.L. Idiopathic choroidal folds. Clin. Refract. Optom. 2004, 15, 298–302. [Google Scholar]
- Yuh, W.T.; Zhu, M.; Taoka, T.; Quets, J.P.; Maley, J.E.; Muhonen, M.G.; Schuster, M.E.; Kardon, R.H. MR imaging of pituitary morphology in idiopathic intracranial hypertension. J. Magn. Reson. Imaging 2000, 12, 808–813. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, D. The mystery of cotton-wool spots - a review of recent and historical descriptions. Eur. J. Med. Res. 2008, 13, 231–266. [Google Scholar] [PubMed]
- Thornton, W.E. Anthropometric changes in weightlessness. Anthropometric Source Book; Technical Report RP 1024, N79-11735. NASA: Washington, DC, USA, 1978; pp. I-1–I-102. Available online: https://archive.org/stream/nasa_techdoc_19790003563/19790003563#page/ (accessed on 4 November 2014).
- Moore, T.P.; Thornton, W.E. Space shuttle inflight and postflight fluid shifts measured by leg volume changes. Aviat. Space Environ. Med. 1987, 58, A91–A96. [Google Scholar] [PubMed]
- Lathers, C.M.; Charles, J.B.; Elton, K.F.; Holt, T.A.; Mukai, C.; Bennett, B.S.; Bungo, M.W. Acute hemodynamic responses to weightlessness in humans. J. Clin. Pharmacol. 1989, 29, 615–627. [Google Scholar] [CrossRef] [PubMed]
- Mukai, C.N.; Lathers, C.M.; Charles, J.B.; Bennett, B.S.; Igarashi, M.; Patel, S. Acute hemodynamic responses to weightlessness during parabolic flight. J. Clin. Pharmacol. 1991, 31, 993–1000. [Google Scholar] [CrossRef] [PubMed]
- Kirsch, K.A.; Baartz, F.J.; Gunga, H.C.; Rocker, L.; Wicke, H.J.; Bunsch, B. Fluid shifts into and out of superficial tissues under microgravity and terrestrial conditions. Clin. Investig. 1993, 71, 687–689. [Google Scholar] [PubMed]
- Nelson, E.S. An Examination of Anticipated G-Jitter on Space Station and its Effects on Material Processes; Technical Report NASA TM 103775; NASA: Washington, DC, USA, 1994. Available online: http://ntrs.nasa.gov/archive/nasa/casi.ntrs.nasa.gov/19950006290.pdf (accessed on 4 November 2014).
- Jules, K. Low-gravity orbiting research laboratory environment potential impact on space biology research. In Proceedings of the 57th International Astronautical Congress, Valencia, Spain, 2–6 October 2006.
- Sharp, L.M.; Dietrich, D.L.; Motil, B.J. Microgravity fluids and combustion research at NASA Glenn Research Center. J. Aerosp. Eng. 2013, 26, 439–450. [Google Scholar] [CrossRef]
- Herault, S.; Tobal, N.; Norman, H.; Roumy, J.; Denise, P.; Arbeille, P. Effect of human head flexion on the control of peripheral blood flow in microgravity and in 1 g. Eur. J. Appl. Physiol. 2002, 87, 296–303. [Google Scholar]
- Norsk, P.; Damgaard, M.; Petersen, L.; Gybel, M.; Pump, B.; Gabrielsen, A.; Christensen, N.J. Vasorelaxation in space. Hypertension 2006, 47, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Pletser, V. Short duration microgravity experiments in physical and life sciences during parabolic flights: The first 30 ESA campaigns. Acta Astronaut. 2004, 55, 829–854. [Google Scholar] [CrossRef] [PubMed]
- Schlegel, T.T.; Benavides, E.W.; Barker, D.C.; Brown, T.E.; Harm, D.L.; DeSilva, S.J.; Low, P.A. Cardiovascular and valsalva responses during parabolic flight. J. Appl. Physiol. 1998, 85, 1957–1965. [Google Scholar]
- Verheyden, B.; Beckers, F.; Aubert, A.E. Spectral characteristics of heart rate fluctuations during parabolic flight. Eur. J. Appl. Physiol. 2005, 95, 557–568. [Google Scholar] [CrossRef]
- Fung, Y.C. The Meaning of the Constitutive Equation. In Biomechanics: Mechanical Properties of Living Tissues; Springer: New York, NY, USA, 1993; pp. 23–65. [Google Scholar]
- Pritchard, P.J. Fluid Statics. In Fox and McDonald’s Introduction to Fluid Mechanics; Wiley: New York, NY, USA, 2011; pp. 55–95. [Google Scholar]
- Fung, Y.C. The Flow Properties of Blood. In Biomechanics: Mechanical Properties of Living Tissues; Springer: New York, NY, USA, 1993; pp. 66–105. [Google Scholar]
- Fung, Y.C. Bioviscoelastic Solids. In Biomechanics: Mechanical Properties of Living Tissues; Springer: New York, NY, USA, 1993; pp. 242–314. [Google Scholar]
- Caiani, E.G.; Sugeng, L.; Weinert, L.; Capderou, A.; Lang, R.M.; Vaida, P. Objective evaluation of changes in left ventricular and atrial volumes during parabolic flight using real-time three-dimensional echocardiography. J. Appl. Physiol. 2006, 101, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Caiani, E.G.; Weinert, L.; Lang, R.M.; Vaida, P. The role of echocardiography in the assessment of cardiac function in weightlessness—Our experience during parabolic flights. Respir. Physiol. Neurobiol. 2009, 169, S6–S9. [Google Scholar] [CrossRef] [PubMed]
- Carey, V.P. Interfacial Tension. In Liquid-Vapor Phase-Change Phenomena; Hemisphere Publishing Corporation: Managua, Nicaragua, 1992; pp. 25–56. [Google Scholar]
- Subramanian, R.S.; Balasubramanian, R. The Role of Gravity and Interfacial Tension in the Motion of Bubbles and Drops. In The Motion of Drops and Bubbles in Reduced Gravity; Cambridge University Press: Cambridge, MA, USA, 2001; pp. 3–46. [Google Scholar]
- Weislogel, M.M.; Jenson, R.; Chen, Y.; Collicott, S.H.; Klatte, J.; Dreyer, M. The capillary flow experiments aboard the International Space Station: Status. Acta Astronaut. 2009, 65, 861–869. [Google Scholar] [CrossRef]
- Water balloons [sic] in zero gravity. Available online: http://www.youtube.com/watch?v=r7fEHYkGxd0 (accessed on 23 October 2014).
- Picture from National Aeronautics and Astronautics Association. Available online: http://www.nasa.gov/images/content/112515main_exp10_image_009hires.jpg (accessed on 23 October 2014).
- McGowan, C. Patients’ confidentiality. Crit. Care Nurse 2012, 32, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Foldager, N.; Andersen, T.A.E.; Jessen, F.B.; Ellegaard, P.; Stadeager, C.; Videbaek, R.; Norsk, P. Central venous pressure in humans during microgravity. J. Appl. Physiol. 1996, 81, 408–412. [Google Scholar] [PubMed]
- Ansari, R.; Manuel, F.K.; Geiser, M.; Moret, F.; Messer, R.K.; King, J.F.; Suh, K.I. Measurement of Choroidal Blood Flow in Zero Gravity. In Proceedings of the Opthalmic Technologies XII, San Jose, CA, USA, 25 January 2002.
- Meck, J.V.; Dreyer, S.A.; Warren, L.E. Long-duration head-down bed rest: Project overview, vital signs, and fluid balance. Aviat. Space Environ. Med. 2009, 80, A1–A8. [Google Scholar] [CrossRef] [PubMed]
- Regnard, J.; Heer, M.; Drummer, C.; Norsk, P. Validity of microgravity simulation models on earth. Am. J. Kidney Dis. 2001, 38, 668–674. [Google Scholar] [CrossRef] [PubMed]
- Stein, T.P. The relationship between dietary intake, exercise, energy balance and the space craft environment. Pflugers Archiv.-Eur. J. Physiol. 2000, 441, R21–R31. [Google Scholar] [CrossRef]
- Hinghofer-Szalkay, H. Gravity, the hydrostatic indifference concept and the cardiovascular system. Eur. J. Appl. Physiol. 2011, 111, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Stevens, S.A.; Lakin, W.D. Local compliance effects on the global pressure-volume relationship in models of intracranial pressure dynamics. Math. Comput. Model. Dyn. Syst. 2000, 6, 445–465. [Google Scholar] [CrossRef]
- Silver, D.M.; Geyer, O. Pressure-volume relation for the living human eye. Curr. Eye Res. 2000, 20, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Tain, R.W.; Bagci, A.M.; Lam, B.L.; Sklar, E.M.; Ertl-Wagner, B.; Alperin, N. Determination of cranio-spinal canal compliance distribution by MRI: Methodology and early application in idiopathic intracranial hypertension. J. Magn. Reson. Imaging 2011, 34, 1397–1404. [Google Scholar] [CrossRef] [PubMed]
- Tain, R.W.; Alperin, N. Noninvasive intracranial compliance from MRI-based measurements of transcranial blood and CSF flows: Indirect versus direct approach. IEEE Trans. Biomed. Eng. 2009, 56, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Holzapfel, G.A.; Eberlein, R.; Wriggers, P.; Weizsacker, H.W. Large strain analysis of soft biological membranes: Formulation and finite element analysis. Comput. Methods Appl. Mech. Eng. 1996, 132, 45–61. [Google Scholar] [CrossRef]
- Kaviany, M. Flow in Porous Media. In Handbook of Fluid Dynamics; Johnson, R.W., Ed.; CRC Press: Boca Raton, FL, USA, 1998. [Google Scholar]
- Fung, Y.C. Basic Transport Equations according to Thermodynamics, Molecular Diffusion, Mechanisms in Membranes and Multiphasic Structure. In Biomechanics: Motion, Flow, Stress and Growth; Springer: New York, NY, USA, 1990; pp. 225–308. [Google Scholar]
- Balzani, D.; Brands, D.; Klawonn, A.; Rheinbach, O.; Schroder, J. On the mechanical modeling of anisotropic biological tissue and iterative parallel solution strategies. Arch. Appl. Mech. 2010, 80, 479–488. [Google Scholar] [CrossRef]
- Fung, Y.C. Mechanical Properties and Active Remodeling of Blood Vessels. In Biomechanics: Mechanical Properties of Living Tissue; Springer: New York, NY, USA, 1993; pp. 321–391. [Google Scholar]
- Iskovitz, I.; Kassemi, M.; Thomas, J.D. Impact of weightlessness on cardiac shape and left ventricular stress/strain distributions. J. Biomech. Eng. 2013, 135, 121008. [Google Scholar] [CrossRef] [PubMed]
- Fung, Y.C. Stress, Strain and Stability of Organs. In Biomechanics: Motion, Flow, Stress and Growth; Springer: New York, NY, USA, 1990; pp. 382–451. [Google Scholar]
- Lakin, W.D.; Stevens, S.A.; Penar, P.L. Modeling intracranial pressures in microgravity: The influence of the blood-brain barrier. Aviat. Space Environ. Med. 2007, 78, 932–936. [Google Scholar] [CrossRef] [PubMed]
- Lakin, W.D.; Stevens, S.A.; Tranmer, B.I.; Penar, P.L. A whole-body mathematical model for intracranial pressure dynamics. J. Math. Biol. 2003, 46, 347–383. [Google Scholar] [CrossRef] [PubMed]
- Linninger, A.A.; Sweetman, B.; Penn, R. Normal and hydrocephalic brain dynamics: The role of reduced cerebrospinal fluid reabsorption in ventricular enlargement. Ann. Biomed. Eng. 2009, 37, 1434–1447. [Google Scholar] [CrossRef] [PubMed]
- Stevens, S.A.; Lakin, W.D.; Penar, P.L. Modeling steady-state intracranial pressures in supine, head-down tilt and microgravity conditions. Aviat. Space Environ. Med. 2005, 76, 329–338. [Google Scholar] [PubMed]
- Stevens, S.A.; Stimpson, J.; Lakin, W.D.; Thakore, N.J.; Penar, P.L. A model for idiopathic intracranial hypertension and associated pathological ICP wave-forms. IEEE Trans. Biomed. Eng. 2008, 55, 388–398. [Google Scholar] [CrossRef] [PubMed]
- Ursino, M.; Lodi, C.A. A simple mathematical model of the interaction between intracranial pressure and cerebral hemodynamics. J. Appl. Physiol. 1997, 82, 1256–1269. [Google Scholar] [PubMed]
- Ursino, M.; Minassian, A.T.; Lodi, C.A.; Beydon, L. Cerebral hemodynamics during arterial and CO2 pressure changes: In vivo prediction by a mathematical model. Am. J. Physiol.-Heart Circ. Physiol. 2000, 279, H2439–H2455. [Google Scholar] [PubMed]
- Vacaitis, N.M.; Sweetman, B.; Linninger, A.A. A Computational Model of Cerebral Vasculature, Brain Tissue and Cerebrospinal Fluid. In 21st European Symposium on Computer-Aided Process Engineering; Pistikopoulos, E.N., Georgiadis, M.C., Kokossis, A.C., Eds.; Elsevier: Amsterdam, The Netherlands, 2011. [Google Scholar]
- National Aeronautics and Space Administration (NASA). Evidence Book: Risk of Orthostatic Intolerance during Re-Exposure to Gravity; NASA, Lyndon B. Johnson Space Center: Houston, TX, USA, 2008. [Google Scholar]
- Summers, R.L.; Platts, S.; Myers, J.G.; Coleman, T.G. Theoretical analysis of the mechanisms of a gender differentiation in the propensity for orthostatic intolerance after spaceflight. Theor. Biol. Med. Model. 2010, 7. [Google Scholar] [CrossRef]
- Christensen, N.J.; Heer, M.; Ivanova, K.; Norsk, P. Sympathetic nervous activity decreases during head down bed rest but not during microgravity. Microgravity Sci. Technol. 2007, 19, 95–97. [Google Scholar] [CrossRef]
- Arbeille, P.; Provost, R.; Vincent, N.; Aubert, A. Adaptation of the main peripheral artery and vein to long term confinement (Mars 500). PLoS One 2014, 9, e83063. [Google Scholar]
- Grytz, R.; Meschke, G. A computational remodeling approach to predict the physiological architecture of the collagen fibril network in corneo-scleral shells. Biomech. Model. Mechanobiol. 2010, 9, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Gleason, R.L.; Humphrey, J.D. A mixture model of arterial growth and remodeling in hypertension: Altered muscle tone and tissue turnover. J. Vasc. Res. 2004, 41, 352–363. [Google Scholar] [CrossRef] [PubMed]
- Nickla, D.L.; Wallman, J. The multifunctional choroid. Progr. Retin. Eye Res. 2010, 29, 144–168. [Google Scholar] [CrossRef]
- Arbeille, P.; Fomina, G.; Roumy, J.; Alferova, I.; Tobal, N.; Herault, S. Adaptation of the left heart, cerebral and femoral arteries, and jugular and femoral veins during short- and long-term head-down tilt and spaceflights. Eur. J. Appl. Physiol. 2001, 86, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.; Kuipers, A.; Mukai, C.; Thirsk, R. Acclimation during space flight: Effects on human physiology. Can. Med. Assoc. J. 2009, 180, 1317–1323. [Google Scholar] [CrossRef]
- White, R.J.; Blomqvist, C.G. Central venous pressure and cardiac function during spaceflight. J. Appl. Physiol. 1998, 85, 738–746. [Google Scholar] [PubMed]
- Sayson, J.V.; Hargens, A.R. Pathophysiology of low back pain during exposure to microgravity. Aviat. Space Environ. Med. 2008, 79, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Convertino, V.A. Clinical aspects of the control of plasma volume at microgravity and during return to one gravity. Med. Sci. Sports Exerc. 1996, 28, S45–S52. [Google Scholar] [CrossRef] [PubMed]
- Convertino, V.A.; Engelke, K.A.; Ludwig, D.A.; Doerr, D.F. Restoration of plasma volume after 16 days of head-down tilt induced by a single bout of maximal exercise. Am. J. Physiol.—Regul. Integr. Comp. Physiol. 1996, 270, R3–R10. [Google Scholar]
- Leach, C.S.; Rambaut, P.C. Endocrine responses in long-duration manned space flight. Acta Astronaut. 1975, 2, 115–127. [Google Scholar] [CrossRef] [PubMed]
- Leach, C.S. Fluid control mechanisms in weightlessness. Aviat. Space Environ. Med. 1987, 58, A74–A79. [Google Scholar]
- Leach, C.S.; Alfrey, C.P.; Suki, W.N.; Leonard, J.I.; Rambaut, P.C.; Inners, L.D.; Smith, S.M.; Lane, H.W.; Krauhs, J.M. Regulation of body fluid compartments during short-term spaceflight. J. Appl. Physiol. 1996, 81, 105–116. [Google Scholar] [PubMed]
- Drummer, C.; Heer, M.; Dressendorfer, R.A.; Strasburger, C.J.; Gerzer, R. Reduced natriuresis during weightlessness. Clin. Investig. 1993, 71, 678–686. [Google Scholar] [PubMed]
- Branch, J.D.; Pate, R.R.; Bodary, P.F.; Convertino, V.A. Red cell volume and erythropoietin responses during exposure to simulated microgravity. Aviat. Space Environ. Med. 1998, 69, 347–351. [Google Scholar] [PubMed]
- Maillet, A.; Fagette, S.; Allevard, A.M.; Traon, A.P.; Guell, A.; Gharib, C.; Gauquelin, G. Cardiovascular and hormonal response during a 4-week head-down tilt with and without exercise and LBNP countermeasures. J. Gravit. Physiol. 1996, 3, 37–48. [Google Scholar] [PubMed]
- Hsieh, S.T.; Ballard, R.E.; Murthy, G.; Hargens, A.R.; Convertino, V.A. Plasma colloid osmotic pressure increases in humans during simulated microgravity. Aviat. Space Environ. Med. 1998, 69, 23–26. [Google Scholar] [PubMed]
- Stein, T.P. Nutrition in the space station era. Nutr. Res. Rev. 2001, 14, 87–118. [Google Scholar] [CrossRef] [PubMed]
- Drummer, C.; Gerzer, R.; Baisch, F.; Heer, M. Body fluid regulation in μ-gravity differs from that on Earth: An overview. Pflugers Archiv 2000, 441, R66–R72. [Google Scholar] [CrossRef] [PubMed]
- Young, K.; Rajulu, S. The Effects of Microgravity on Seated Height (Spinal Elongation). In NASA Human Research Program Investigators’ Workshop; NASA: Houston, TX, USA, 2012. Available online: http://ntrs.nasa.gov/archive/nasa/casi.ntrs.nasa.gov/20110023150.pdf (accessed on 4 November 2014). [Google Scholar]
- Ledsome, J.R. Spinal Changes in Microgravity. In Second International Microgravity Laboratory (IML-2) Final Report; Snyder, R.S., Ed.; NASA Marshall Space Flight Center: Huntsville, AL, USA, 1987; pp. 182–185. [Google Scholar]
- Hargens, A.; Chiang, S.B.; Sayson, J.V.; Parazynski, S.E.; Chang, D.G.; Garfin, S.R.; Lotz, J.C. Mechanism of Post-Flight Herniation of Intervertebral Discs. In NASA Human Research Program Investigators’ Workshop; NASA: Houston, TX, USA, 2012. [Google Scholar]
- Thornton, I.L.; Dupps, W.J.; Roy, A.S.; Krueger, R.R. Biomechanical effects of intraocular pressure elevation on optic nerve/lamina cribrosa before and after peripapillary scleral collagen cross-linking. Investig. Ophthalmol. Vis. Sci. 2009, 50, 1227–1233. [Google Scholar] [CrossRef]
- Thornton, W.E.; Moore, T.P.; Pool, S.L. Fluid shifts in weightlessness. Aviat. Space Environ. Med. 1987, 58, A86–A90. [Google Scholar] [PubMed]
- Hoffler, G.W.; Wolthuis, R.A.; Johnson, R.L. Apollo space crew cardiovascular evaluations. Aerosp. Med. 1974, 45, 807–823. [Google Scholar] [PubMed]
- Thornton, W.E.; Hedge, V.; Coleman, E.; Uri, J.J.; Moore, T.P. Changes in leg volume during microgravity simulation. Aviat. Space Environ. Med. 1992, 63, 789–794. [Google Scholar] [PubMed]
- Smith, S.M.; Krauhs, J.M.; Leach, C.S. Regulation of body fluid volume and electrolyte concentrations in spaceflight. Adv. Space Biol. Med. 1997, 6, 123–165. [Google Scholar] [PubMed]
- Parazynski, S.E.; Hargens, A.R.; Tucker, B.; Aratow, M.; Styf, J.; Crenshaw, A. Transcapillary fluid shifts in tissues of the head and neck during and after simulated microgravity. J. Appl. Physiol. 1991, 71, 2469–2475. [Google Scholar]
- Leach, C.S.; Johnson, P.C. Influence of spaceflight on erythrokinetics in man. Science 1984, 225, 216–218. [Google Scholar] [CrossRef] [PubMed]
- Alfrey, C.P.; Udden, M.M.; Leach-Huntoon, C.; Driscoll, T.; Pickett, M.H. Control of red blood cell mass in spaceflight. J. Appl. Physiol. 1996, 81, 98–104. [Google Scholar]
- Udden, M.M.; Driscoll, T.B.; Pickett, M.H.; Leach-Huntoon, C.S.; Alfrey, C.P. Decreased production of red blood cells in human subjects exposed to microgravity. J. Lab. Clin. Med. 1995, 125, 442–449. [Google Scholar] [PubMed]
- Leach, C.S.; Johnson, P.C.; Cintron, N.M. The endocrine system in space flight. Acta Astronaut. 1988, 17, 161–166. [Google Scholar] [CrossRef] [PubMed]
- De Santo, N.G.; Cirillo, M.; Kirsch, K.A.; Correale, G.; Drummer, C.; Frassl, W.; Perna, A.F.; di Stazio, E.; Bellini, L.; Gunga, H.C. Anemia and erythropoietin in space flights. Semin. Nephrol. 2005, 25, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Kerstman, E.L.; Scheuring, R.A.; Barnes, M.G.; DeKorse, T.B.; Saile, L.G. Space adaptation to back pain: A retrospective study. Aviat. Space Environ. Med. 2012, 83, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, K.J.; Watenpaugh, D.E.; Murthy, G.; Convertino, V.A.; Hargens, A.R. Back pain during 6 degrees head-down tilt approximates that during actual microgravity. Aviat. Space Environ. Med. 1995, 66, 256–259. [Google Scholar] [PubMed]
- Styf, J.R.; Ballard, R.E.; Fechner, K.; Watenpaugh, D.E.; Kahan, N.J.; Hargens, A.R. Height increase, neuromuscular function, and back pain during 6 degrees head-down tilt with traction. Aviat. Space Environ. Med. 1997, 68, 24–29. [Google Scholar] [PubMed]
- Caprihan, A.; Sanders, J.A.; Cheng, H.A.; Loeppky, J.A. Effect of head-down tilt on brain water distribution. Eur. J. Appl. Physiol. Occup. Physiol. 1999, 79, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Taibbi, G.; Cromwell, R.L.; Zanello, S.B.; Yarbrough, P.O.; Vizzeri, G. Evaluation of ocular outcomes in two 14-day bed rest studies. Available online: http://abstracts.iovs.org/cgi/content/short/53/6/4903 (accessed on 24 October 2014).
- Zamir, M.; Norton, K.; Fleischhauer, A.; Frances, M.F.; Goswami, R.; Usselman, C.W.; Nolan, R.P.; Shoemaker, J.K. Dynamic responsiveness of the vascular bed as a regulatory mechanism in vasomotor control. J. Gen. Physiol. 2009, 134, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Jonas, J.B.; Wang, N.; Wang, Y.X.; You, Q.S.; Xie, X.; Yang, D.; Xu, L. Body height, estimated cerebrospinal fluid pressure and open-angle glaucoma. The Beijing Eye Study 2011. PLoS One 2014, 9, e86678. [Google Scholar] [CrossRef] [PubMed]
- Grytz, R.; Meschke, G. Constitutive modeling of crimped collagen fibrils in soft tissues. J. Mech. Behav. Biomed. Mater. 2009, 2, 522–533. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.F.; Hargens, A.R. Intraocular/Intracranial pressure mismatch hypothesis for visual impairment syndrome in space. Aviat. Space Environ. Med. 2014, 85, 78–80. [Google Scholar] [CrossRef] [PubMed]
- Berdahl, J.P.; Yu, D.Y.; Morgan, W.H. The translaminar pressure gradient in sustained zero gravity, idiopathic intracranial hypertension, and glaucoma. Med. Hypotheses 2012, 79, 719–724. [Google Scholar] [CrossRef] [PubMed]
- Jonas, J.B. Trans-lamina cribrosa pressure difference. Arch. Ophthalmol. 2007, 125, 431–431. [Google Scholar] [CrossRef] [PubMed]
- Marek, B.; Harris, A.; Kanakamedala, P.; Lee, E.; Amireskandari, A.; Carichino, L.; Guidoboni, G.; Tobe, L.A.; Siesky, B. Cerebrospinal fluid pressure and glaucoma: Regulation of trans-lamina cribrosa pressure. Br. J. Ophthalmol. 2014, 98, 721–725. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, P.J. Introduction to differential analysis of fluid motion. In Fox and McDonald's Introduction to Fluid Mechanics; Wiley: New York, NY, USA, 2011; pp. 171–234. [Google Scholar]
- Hargens, A.R. Recent bed rest results and countermeasure development at NASA. Acta Physiol. Scand. Suppl. 1994, 616, 103–114. [Google Scholar] [PubMed]
- Alperin, N.; Lam, B.L.; Tain, R.W.; Ranganathan, S.; Letzing, M.; Bloom, M.; Alexander, B.; Aroucha, P.R.; Sklar, E. Evidence for altered spinal canal compliance and cerebral venous drainage in untreated idiopathic intracranial hypertension. Acta Neurochir. Suppl. 2012, 114, 201–205. [Google Scholar] [PubMed]
- Thakore, N.J.; Stevens, S.A.; Lakin, W.D. A mathematical model of idiopathic intracranial hypertension. Headache 2005, 45, 801–802. [Google Scholar]
- Vein, A.A.; Koppen, H.; Haan, J.; Terwindt, G.M.; Ferrari, M.D. Space headache: A new secondary headache. Cephalalgia 2009, 29, 683–686. [Google Scholar] [PubMed]
- Oosterhout, W.V.; Terwindt, G.; Vein, A.; Ferrari, M. Space headache on Earth: Head-down-tilted bed rest studies simulating outer-space microgravity. Cephalalgia 2014. [Google Scholar] [CrossRef]
- Kawai, Y.; Doi, M.; Setogawa, A.; Shimoyama, R.; Ueda, K.; Asai, Y.; Tatebayashi, K. Effect of microgravity on cerebral hemodynamics. Yonago Acta Med. 2003, 46, 1–8. [Google Scholar]
- Aoi, M.; Gremaud, P.; Tran, H.T.; Novak, V.; Olufsen, M.S. Modeling cerebral blood flow and regulation. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2009, 2009, 5470–5473. [Google Scholar] [PubMed]
- Doepp, F.; Schreiber, S.J.; von Munster, T.; Rademacher, J.; Klingebiel, R.; Valdueza, J.M. How does the blood leave the brain? A systematic ultrasound analysis of cerebral venous drainage patterns. Neuroradiology 2004, 46, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Gisolf, J.; van Lieshout, J.J.; van Heusden, K.; Pott, F.; Stok, W.J.; Karemaker, J.M. Human cerebral venous outflow pathway depends on posture and central venous pressure. J. Physiol. 2004, 560, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Monti, L.; Menci, E.; Ulivelli, M.; Cerase, A.; Bartalini, S.; Piu, P.; Marotti, N.; Leonini, S.; Galluzzi, P.; Romano, D.G.; et al. Quantitative ColourDopplerSonography evaluation of cerebral venous outflow: A comparative study between patients with multiple sclerosis and controls. PLoS One 2011, 6, e25012. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.Y.; Chang, F.C.; Chao, A.C.; Chung, C.P.; Hu, H.H. Internal jugular venous abnormalities in transient monocular blindness. BMC Neurol. 2013, 13. [Google Scholar] [CrossRef] [PubMed]
- Darge, K.; Brandis, U.; Zieger, B.; Rohrschneider, W.; Wunsch, R.; Troeger, J. Internal jugular venous valves in children: High-resolution US findings. Eur. Radiol. 2001, 11, 655–658. [Google Scholar] [CrossRef] [PubMed]
- Diaconu, C.I.; Staugaitis, S.M.; Fox, R.J.; Rae-Grant, A.; Schwanger, C.; McBride, J.M. A technical approach to dissecting and assessing cadaveric veins pertinent to chronic cerebrospinal venous insufficiency in multiple sclerosis. Neurol Res. 2012, 34, 810–818. [Google Scholar]
- Lepori, D.; Capasso, P.; Fournier, D.; Genton, C.Y.; Schnyder, P. High-resolution ultrasound evaluation of internal jugular venous valves. Eur. Radiol. 1999, 9, 1222–1226. [Google Scholar] [CrossRef] [PubMed]
- Nicolaides, A.N.; Morovic, S.; Menegatti, E.; Viselner, G.; Zamboni, P. Screening for chronic cerebrospinal venous insufficiency (CCSVI) using ultrasound: Recommendations for a protocol. Funct. Neurol 2011, 26, 229–248. [Google Scholar] [PubMed]
- Schreiber, S.J.; Doepp, F.; Klingebiel, R.; Valdueza, J.M. Internal jugular vein valve incompetence and intracranial venous anatomy in transient global amnesia. J. Neurol Neurosurg. Psychiatry 2005, 76, 509–513. [Google Scholar] [CrossRef] [PubMed]
- Harmon, J.V., Jr.; Edwards, W.D. Venous valves in subclavian and internal jugular veins. Frequency, position, and structure in 100 autopsy cases. Am. J. Cardiovasc. Pathol. 1987, 1, 51–54. [Google Scholar] [PubMed]
- Caggiati, A.; Phillips, M.; Lametschwandtner, A.; Allegra, C. Valves in small veins and venules. Eur. J. Vasc Endovasc. Surg. 2006, 32, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Phillips, M.N.; Jones, G.T.; van Rij, A.M.; Zhang, M. Micro-venous valves in the superficial veins of the human lower limb. Clin. Anat. 2004, 17, 55–60. [Google Scholar] [PubMed]
- Iliff, J.; Wang, M.; Liao, Y.; Plogg, B.; Peng, W.; Gunderson, G.; Benveniste, H.; Vates, G.; Deane, R.; Goldman, S.; et al. A paravascular pathway facilitates CSF flow through the brain parenchyma and clearance of interstitial solutes, including amyloid β. Sci. Transl. Med. 2012, 4, 1–11. [Google Scholar]
- Johnston, M.; Zakharov, A.; Papaiconomou, C.; Salmasi, G.; Armstrong, D. Evidence of connections between cerebrospinal fluid and nasal lymphatic vessels in humans, non-human primates and other mammalian species. Cerebrospinal Fluid Res. 2004, 1. [Google Scholar] [CrossRef] [Green Version]
- Schmidt, C.; Wiener, E.; Hoffmann, J.; Klingebiel, R.; Schmidt, F.; Hofmann, T.; Harms, L.; Kunte, H. Structural olfactory nerve changes in patients suffering from idiopathic intracranial hypertension. PLoS One 2012, 7, e35221. [Google Scholar] [CrossRef] [PubMed]
- Weller, R.O.; Djuanda, E.; Yow, H.Y.; Carare, R.O. Lymphatic drainage of the brain and the pathophysiology of neurological disease. Acta Neuropathol. 2009, 117, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Margaris, K.N.; Black, R.A. Modelling the lymphatic system: Challenges and opportunities. J. R. Soc. Interface 2012, 9, 601–612. [Google Scholar] [CrossRef] [PubMed]
- Carlson, K.H.; McLaren, J.W.; Topper, J.E.; Brubaker, R.F. Effect of body position on intraocular pressure and aqueous flow. Invest. Ophthalmol. Vis. Sci. 1987, 28, 1346–1352. [Google Scholar] [PubMed]
- Selvadurai, D.; Hodge, D.; Sit, A.J. Aqueous humor outflow facility by tonography does not change with body position. Invest. Ophthalmol. Vis. Sci. 2010, 51, 1453–1457. [Google Scholar] [CrossRef] [PubMed]
- Prata, T.S.; de Moraes, C.G.; Kanadani, F.N.; Ritch, R.; Paranhos, A., Jr. Posture-induced intraocular pressure changes: Considerations regarding body position in glaucoma patients. Surv. Ophthalmol. 2010, 55, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Malihi, M.; Sit, A.J. Effect of head and body position on intraocular pressure. Ophthalmology 2012, 119, 987–991. [Google Scholar] [PubMed]
- Friberg, T.R.; Weinreb, R.N. Ocular manifestations of gravity inversion. JAMA 1985, 253, 1755–1757. [Google Scholar] [CrossRef] [PubMed]
- Weinreb, R.N.; Cook, J.; Friberg, T.R. Effect of inverted body position on intraocular pressure. Am. J. Ophthalmol. 1984, 98, 784–787. [Google Scholar] [CrossRef] [PubMed]
- Silver, D.M.; Farrell, R.A. Validity of pulsatile ocular blood flow measurements. Surv. Ophthalmol. 1994, 38 (Suppl), S72–S80. [Google Scholar]
- Dastiridou, A.I.; Ginis, H.S.; de Brouwere, D.; Tsilimbaris, M.K.; Pallikaris, I.G. Ocular rigidity, ocular pulse amplitude, and pulsatile ocular blood flow: The effect of intraocular pressure. Investig. Ophthalmol. Vis. Sci. 2009, 50, 5718–5722. [Google Scholar] [CrossRef]
- Mader, T.H.; Taylor, G.R.; Hunter, N.; Caputo, M.; Meehan, R.T. Intraocular pressure, retinal vascular, and visual acuity changes during 48 hours of 10 degree head-down tilt. Aviat. Space Environ. Med. 1990, 61, 810–813. [Google Scholar] [PubMed]
- Shinojima, A.; Iwasaki, K.; Aoki, K.; Ogawa, Y.; Yanagida, R.; Yuzawa, M. Subfoveal choroidal thickness and foveal retinal thickness during head-down tilt. Aviat. Space Environ. Med. 2012, 83, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Kiel, J.W. Choroidal myogenic autoregulation and intraocular pressure. Exp. Eye Res. 1994, 58, 529–543. [Google Scholar] [CrossRef]
- Baer, R.M.; Hill, D.W. Retinal vessel responses to passive tilting. Eye 1990, 4, 751–756. [Google Scholar] [PubMed]
- Frey, M.A.B.; Mader, T.H.; Bagian, J.P.; Charles, J.B.; Meehan, R.T. Cerebral blood velocity and other cardiovascular responses to 2 days of head-down tilt. J. Appl. Physiol. 1993, 74, 319–325. [Google Scholar] [PubMed]
- Zuj, K.A.; Arbeille, P.; Shoemaker, J.K.; Blaber, A.P.; Greaves, D.K.; Xu, D.; Hughson, R.L. Impaired cerebrovascular autoregulation and reduced CO2 reactivity after long duration spaceflight. Am. J. Physiol. Heart Circ. Physiol. 2012, 302, H2592–H2598. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, K.I.; Levine, B.D.; Zhang, R.; Zuckerman, J.H.; Pawelczyk, J.A.; Diedrich, A.; Ertl, A.C.; Cox, J.F.; Cooke, W.H.; Giller, C.A.; et al. Human cerebral autoregulation before, during and after spaceflight. J. Physiol.-Lond. 2007, 579, 799–810. [Google Scholar] [CrossRef] [PubMed]
- Verheyden, B.; Liu, J.; Beckers, F.; Aubert, A.E. Operational point of neural cardiovascular regulation in humans up to 6 months in space. J. Appl. Physiol. 2010, 108, 646–654. [Google Scholar] [PubMed]
- Cox, J.F.; Tahvanainen, K.U.O.; Kuusela, T.A.; Levine, B.D.; Cooke, W.H.; Mano, T.; Iwase, S.; Saito, M.; Sugiyama, Y.; Ertl, A.C.; et al. Influence of microgravity on astronauts’ sympathetic and vagal responses to Valsalva’s manoeuvre. J. Physiol.-Lon. 2002, 538, 309–320. [Google Scholar] [CrossRef]
- Zhang, L.F. Vascular adaptation to microgravity: What have we learned? J. Appl. Physiol. 2001, 91, 2415–2430. [Google Scholar]
- Baevsky, R.M.; Bennett, B.S.; Bungo, M.W.; Charles, J.B.; Goldberger, A.L.; Nikulina, G.A. Adaptive responses of the cardiovascular system to prolonged spaceflight conditions: Assessment with Holter monitoring. J. Cardiovasc. Diagn. Proced. 1997, 14, 53–57. [Google Scholar] [PubMed]
- Cirovic, S.; Kim, M. A one-dimensional model of the spinal cerebrospinal-fluid compartment. J. Biomech. Eng. 2012, 134. [Google Scholar] [CrossRef]
- Feke, G.T.; Tagawa, H.; Deupree, D.M.; Goger, D.G.; Sebag, J.; Weiter, J.J. Blood flow in the normal human retina. Invest. Ophthalmol. Vis. Sci. 1989, 30, 58–65. [Google Scholar] [PubMed]
- Werkmeister, R.M.; Dragostinoff, N.; Palkovits, S.; Told, R.; Boltz, A.; Leitgeb, R.A.; Groschl, M.; Garhofer, G.; Schmetterer, L. Measurement of absolute blood flow velocity and blood flow in the human retina by dual-beam bidirectional Doppler fourier-domain optical coherence tomography. Invest. Ophthalmol. Vis. Sci. 2012, 53, 6062–6071. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Lawford, P.; Hose, R. Review of zero-D and 1-D models of blood flow in the cardiovascular system. BioMed. Eng. Online 2011, 10. [Google Scholar] [CrossRef]
- Wakeland, W.; Goldstein, B. A review of physiological simulation models of intracranial pressure dynamics. Comput. Biol. Med. 2008, 38, 1024–1041. [Google Scholar] [CrossRef] [PubMed]
- Ethier, C.R.; Simmons, C.A. Introductory Biomechanics: From Cells to Organisms; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Lorenzi, M.; Feke, G.T.; Cagliero, E.; Pitler, L.; Schaumberg, D.A.; Berisha, F.; Nathan, D.M.; McMeel, J.W. Retinal haemodynamics in individuals with well-controlled type 1 diabetes. Diabetologia 2008, 51, 361–364. [Google Scholar] [CrossRef] [PubMed]
- Nagahara, M.; Tamaki, Y.; Tomidokoro, A.; Araie, M. In vivo measurement of blood velocity in human major retinal vessels using the laser speckle method. Investig. Ophthalmol. Vis. Sci. 2011, 52, 87–92. [Google Scholar] [CrossRef]
- Garhofer, G.; Werkmeister, R.; Dragostinoff, N.; Schmetterer, L. Retinal blood flow in healthy young subjects. Invest. Ophthalmol. Vis. Sci. 2012, 53, 698–703. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lu, A.; Gil-Flamer, J.; Tan, O.; Izatt, J.A.; Huang, D. Measurement of total blood flow in the normal human retina using Doppler Fourier-domain optical coherence tomography. Br. J. Ophthalmol. 2009, 93, 634–637. [Google Scholar] [CrossRef] [PubMed]
- Feke, G.T.; Pasquale, L.R. Retinal blood flow response to posture change in glaucoma patients compared with healthy subjects. Ophthalmology 2008, 115, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Ambarki, K.; Hallberg, P.; Johannesson, G.; Linden, C.; Zarrinkoob, L.; Wahlin, A.; Birgander, R.; Malm, J.; Eklund, A. Blood flow of ophthalmic artery in healthy individuals determined by phase-contrast magnetic resonance imaging. Invest. Ophthalmol. Vis. Sci. 2013, 54, 2738–2745. [Google Scholar] [CrossRef] [PubMed]
- Kiss, B.; Dallinger, S.; Polak, K.; Findl, O.; Eichler, H.G.; Schmetterer, L. Ocular hemodynamics during isometric exercise. Microvasc. Res. 2001, 61, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Pasquale, L.R.; Jonas, J.B.; Anderson, D. Ocular Blood Flow in Glaucoma. In The 6th Consensus Report of the World Glaucoma Association; Weinreb, R.N., Harris, A., Eds.; World Glaucoma Association: Amsterdam, The Netherlands, 2009. [Google Scholar]
- Yu, P.K.; Tan, P.E.; Cringle, S.J.; McAllister, I.L.; Yu, D.Y. Phenotypic heterogeneity in the endothelium of the human vortex vein system. Exp. Eye Res. 2013, 115, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Kahn, M.T. Color Doppler orbital hemodynamics in healthy individuals and patients with vascular disorders in Taiwan. Orbit 2008, 27, 25–30. [Google Scholar] [PubMed]
- Stalmans, I.; Vandewalle, E.; Anderson, D.R.; Costa, V.P.; Frenkel, R.E.; Garhofer, G.; Grunwald, J.; Gugleta, K.; Harris, A.; Hudson, C.; et al. Use of colour Doppler imaging in ocular blood flow research. Acta Ophthalmol. 2011, 89, e609–e630. [Google Scholar] [CrossRef] [PubMed]
- Hayreh, S.S. Posterior ciliary artery circulation in health and disease: The Weisenfeld lecture. Invest. Ophthalmol. Vis. Sci. 2004, 45, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Hayreh, S.S. Physiological anatomy of the choroidal vascular bed. Int. Ophthalmol. 1983, 6, 85–93. [Google Scholar]
- Kutoglu, T.; Yalcin, B.; Kocabiyik, N.; Ozan, H. Vortex veins: Anatomic investigations on human eyes. Clin. Anatomy 2005, 18, 269–273. [Google Scholar] [CrossRef]
- Rutnin, U. Fundus appearance in normal eyes. I. The choroid. Am. J. Ophthalmol. 1967, 64, 821–839. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Stringer, M.D. Ophthalmic and facial veins are not valveless. Clin. Exp. Ophthalmol. 2010, 38, 502–510. [Google Scholar] [CrossRef]
- Hom, M.M.; Bielory, L. The anatomical and functional relationship between allergic conjunctivitis and allergic rhinitis. Allergy Rhinol 2013, 4, e110–e119. [Google Scholar] [CrossRef]
- Guidoboni, G.; Harris, A.; Arciero, J.C.; Siesky, B.A.; Amireskandari, A.; Gerber, A.L.; Huck, A.H.; Kim, N.J.; Cassani, S.; Carichino, L. Mathematical modeling approaches in the study of glaucoma disparities among people of African and European descents. J. Coupled Syst. Multiscale Dyn. 2013, 1, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Guidoboni, G.; Harris, A.; Carichino, L.; Arieli, Y.; Siesky, B.A. Effect of intraocular pressure on the hemodynamics of the central retinal artery: A mathematical model. Math. Biosci. Eng. 2014, 11, 523–546. [Google Scholar] [CrossRef] [PubMed]
- Delaey, C.; van de Voorde, J. Regulatory mechanisms in the retinal and choroidal circulation. Ophthalmic. Res. 2000, 32, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Kur, J.; Newman, E.A.; Chan-Ling, T. Cellular and physiological mechanisms underlying blood flow regulation in the retina and choroid in health and disease. Prog. Retin. Eye Res. 2012, 31, 377–406. [Google Scholar] [CrossRef] [PubMed]
- Arciero, J.; Harris, A.; Siesky, B.; Amireskandari, A.; Gershuny, V.; Pickrell, A.; Guidoboni, G. Theoretical analysis of vascular regulatory mechanisms contributing to retinal blood flow autoregulation. Invest. Ophthalmol. Vis. Sci. 2013, 54, 5584–5593. [Google Scholar] [CrossRef] [PubMed]
- Morgan, A.J.; Hosking, S.L. Non-invasive vascular impedance measures demonstrate ocular vasoconstriction during isometric exercise. Br. J. Ophthalmol. 2007, 91, 385–390. [Google Scholar] [CrossRef]
- Flammer, J.; Mozaffarieh, M. Autoregulation, a balancing act between supply and demand. Can. J. Ophthalmol. 2008, 43, 317–321. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.L. Carbon Dioxide. In Spacecraft Maximum Allowable Concentrations for Selected Airborne Contaminants; Council, N.R., Ed.; National Academy Press: Washington, DC, USA, 1996; pp. 105–188. [Google Scholar]
- Wei, E.P.; Seelig, J.M.; Kontos, H.A. Comparative responses of cerebellar and cerebral arterioles to changes in PaCO2 in cats. Am. J. Physiol. 1984, 246, H386–H388. [Google Scholar] [PubMed]
- Zwart, S.R.; Gibson, C.R.; Mader, T.H.; Ericson, K.; Ploutz-Snyder, R.; Heer, M.; Smith, S.M. Vision changes after spaceflight are related to alterations in folate- and vitamin B-12-dependent one-carbon metabolism. J. Nutr. 2012, 142, 427–431. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Kloeris, V.L.; Heer, M. Nutritional Biochemistry of Space Flight; Nova Science Publishers: New York, NY, USA, 2009. [Google Scholar]
- Cucinotta, F.A.; Kim, M.H.; Chappell, L.J.; Huff, J.L. How safe is safe enough? Radiation risk for a human mission to Mars. PLoS One 2013, 8, e74988. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, A.R.; Weissman, D.; Sanzari, J.K.; Krigsfeld, G.S.; Wan, X.S.; Romero-Weaver, A.L.; Diffenderfer, E.S.; Lin, L.; Cengel, K. Acute effects of solar particle event radiation. J. Radiat. Res. 2014, 55 (Suppl 1), i66–i67. [Google Scholar] [CrossRef]
- Smith, S.M.; Heer, M.A.; Shackelford, L.C.; Sibonga, J.D.; Ploutz-Snyder, L.; Zwart, S.R. Benefits for bone from resistance exercise and nutrition in long-duration spaceflight: Evidence from biochemistry and densitometry. J. Bone Miner. Res. 2012, 27, 1896–1906. [Google Scholar] [CrossRef] [PubMed]
- Haykowsky, M.J.; Eves, N.D.; Warburton, D.E.R.; Findlay, M.J. Resistance exercise, the Valsalva Maneuver, and cerebrovascular transmural pressure. Med. Sci. Sports Exerc. 2003, 35, 65–68. [Google Scholar]
- Brimioulle, S.; Moraine, J.J.; Norrenberg, D.; Kahn, R.J. Effects of positioning and exercise on intracranial pressure in a neurosurgical intensive care unit. Phys. Ther. 1997, 77, 1682–1689. [Google Scholar] [PubMed]
- Kelly, T.H.; Hienz, R.D.; Zarcone, T.J.; Wurster, R.M.; Brady, J.V. Crewmember performance before, during, and after spaceflight. J. Exp. Anal. Behav. 2005, 84, 227–241. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Kang, H.; Xu, Q.; Chen, M.J.; Liao, Y.; Thiyagarajan, M.; O’Donnell, J.; Christensen, D.J.; Nicholson, C.; Iliff, J.J.; et al. Sleep drives metabolite clearance from the adult brain. Science 2013, 342, 373–377. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nelson, E.S.; Mulugeta, L.; Myers, J.G. Microgravity-Induced Fluid Shift and Ophthalmic Changes. Life 2014, 4, 621-665. https://doi.org/10.3390/life4040621
Nelson ES, Mulugeta L, Myers JG. Microgravity-Induced Fluid Shift and Ophthalmic Changes. Life. 2014; 4(4):621-665. https://doi.org/10.3390/life4040621
Chicago/Turabian StyleNelson, Emily S., Lealem Mulugeta, and Jerry G. Myers. 2014. "Microgravity-Induced Fluid Shift and Ophthalmic Changes" Life 4, no. 4: 621-665. https://doi.org/10.3390/life4040621