Abstract
Scutellariae radix (S. radix) is one of the most important crude drugs used in Kampo medicines (KMs). A part of its pharmaceutical properties is due to flavone glycoside, baicalin (BI). A technique named eastern blotting was developed for the specific and easy identification of BI in the extracts of crude drugs and KMs using anti-BI monoclonal antibody (MAb). BI separated by silica gel thin-layer chromatography (TLC) transferred to a polyethersulfone (PES) membrane was treated with a NaIO4 solution and reacted with bovine serum albumin (BSA) preparing BI-BSA conjugate on the PES membrane. Anti-BI MAb was bound and then antibody labeled with peroxidase directed against anti-BI MAb. Finally, a substrate was added and then BI was detected. As little as 1 μg of BI was still detected on the PES membrane under immunostaining method. Various samples of S. radix and KMs which contain S. radix were qualitatively analyzed, and BI was visually detected by eastern blotting technique. Furthermore, this method was applied for the immunohistochemical study to investigate the distribution of BI in the fresh root of Scutellaria baicalensis using immunoblotting by transferred from fresh root to the PES membrane.
1. Introduction
Scutellaria baicalensis Georgi (Labiatae) is a well-known herb and widely found in East Asian from the north of China to east of Siberia. Scutellariae radix (S. radix), the dried root of S. baicalensis is one of the most important crude drugs used widely in Kampo medicines (KMs) and traditional Chinese medicines (TCMs) for the treatment of fever, inflammation, hepatitis and hypertension [1]. S. radix contains more than 30 kinds of flavonoids, such as baicalin (BI), baicalein (BE), wogonin and wogonin glucuronide (Figure 1). The pharmacological efficacy of S. radix is due to BI and BE and they show anti-allergic [2], anti-inflammation [3], anti-viral effects on diseases such as human immunodeficiency virus (HIV) [4,5], anti-cancer [6] and free-radical scavenging effect [7]. The defense mechanism of S. baicalensis including oxidative burst against plant disease and insect attack was investigated and showed BI must be the most important substance of its mechanism [8,9].
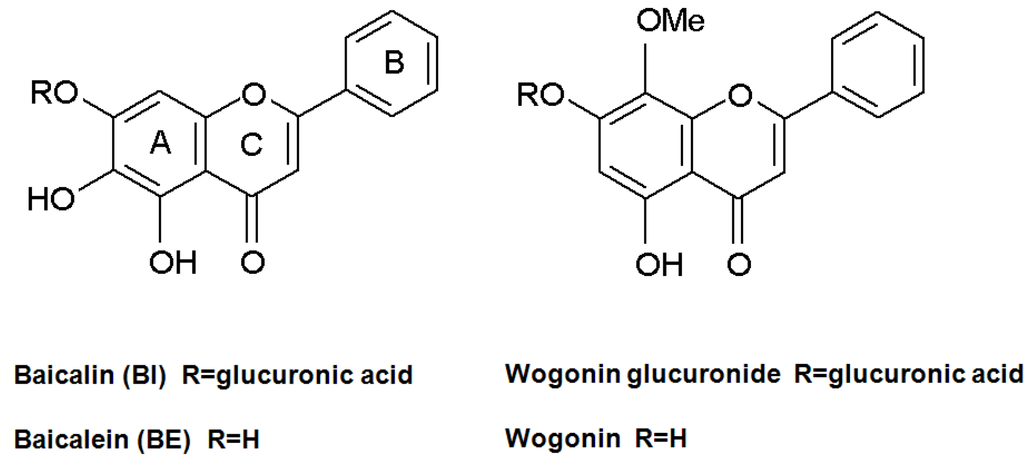
Figure 1.
Structures of BI and its related compounds.
Several methods have been investigated for qualitative and quantitative analysis of BI in S. radix and KMs. Thin-layer chromatography (TLC) and gas chromatography with mass spectrometry (GC-MS) were applied to qualitative analysis of BI [10]. The quantitative analysis of BI using high-performance liquid chromatography (HPLC) with ultraviolet detection has been adopted as a quality control for S. radix and the concentration of BI in S. radix used clinically should be ≥10% (w/w) as authorized by the Japanese Pharmacopoeia. HPLC with electrochemical detection showed a high sensitivity in the pharmacokinetic study of BI [11,12]. Furthermore, micellar electrokinetic capillary electrophoresis was established to separate, detection and identify BI and other flavonoids in S. radix [13].
Immunoassay systems using monoclonal antibodies (MAbs) against bioactive compounds of low molecular weight have become important tools for the qualitative and quantitative analyses of active constituents from natural medicines and natural products [14]. The production and characterization of anti-BI MAb have already been described [15]. Fortunately the anti-BI MAb has the cross-reactivity only with both BI and BE which is an aglycon part of BI (100% and 51.4%, respectively) because BI and its metabolite of BE functioned as the major pharmacological activities of S. radix. Therefore an enzyme-linked immunosorbent assay (ELISA) system was developed for measurement of BI in crude drugs and KMs and BE in biological fluid. We also succeeded to construct a one-step immunochromatographic assay system for the rapid qualitative detection of BI in large numbers of samples [16]. In the investigation of staining technique for several low molecular weight compounds such as natural glycosides and phospholipids, the first successful immunostaining of natural glycosides on polyvinylidene difluoride (PVDF) membrane was reported on solasodine glycosides by our group following to the thin-layer chromatography blotting procedure of phospholipids [17,18]. Recently we suggested a new name, eastern blotting for glycyrrhizin as a new immunostaining technique [19]. However, an immunostaining technique of BI on the membrane such as PVDF, nylon, nitrocellurose or polyethersulfone (PES) membranes has not been reported.
Therefore, as an extension of anti-BI MAb application, the present paper describes the development of eastern blotting technique for the specific analysis of BI in S. radix and KMs. Furthermore, immunohistochemical study to investigate the distribution of BI in the fresh root of S. baicalensis was also demonstrated using immunoblotting by transferred from fresh root to the PES membrane.
2. Results and Discussion
2.1. Dot Blot Assay of BI
In the present study, anti-BI MAb was applied to the immunostaining technique for the visual detection of BI on the membrane. First, the specificity of anti-BI MAb was tested in a dot blot assay using BI and other glycosides such as flavonoids and terpenoids. From the staining data as shown in Figure 2, anti-BI MAb specifically recognized BI when 10 μg was present on the membrane. This result agrees with cross-reactivity data of anti-BI MAb against various glycosides in competitive ELISA [15]. In the methodology of immunostaining of BI, BI molecule was separated into two functional parts, the epitope part (mainly aglycone) and the sugar part. The sugar part of BI is oxidized to give aldehyde groups which react with amino groups of lysine or arginine or both on the protein that can bind strongly to the adsorbent membrane. The aglycone part of BI was bound by anti-BI MAb as shown diagrammatically in Figure 3. From this, it becomes evident that anti-BI MAb interacts with the A ring including the hydroxyl groups at C-5 and C-6 positions of BI. Figure 4 showed the sensitivity of anti-BI MAb and detection of BI under dot blot assay conditions. As little as 0.5 μg of BI was still detected by dot blot assay. These characteristics of anti-BI MAb might be an important advantage for the specific analysis of BI which is a major pharmacologically active component of S. radix using on-membrane assisted immunostaining in a plant extract. Furthermore, the results of dot blot assay suggest that the PES membrane is suitable for immunodetection of BI.
2.2. Eastern Blotting of BI
Direct detection of small molecular compounds on a TLC plate by immunostaining is impossible, because they are easily washed out by buffer solution without fixing. We designed the system that BI was blotted onto a PES membrane from the developed TLC plate for immunostaining. After BI was transferred to the PES membrane, it was treated with NaIO4 solution, followed by the addition of BSA. This reaction enhanced the fixation of BI via BI-BSA conjugate preparation on the PES membrane. When the blotted PES membrane was incubated in the absence of BSA, it was essentially free of immunostaining (data not shown). A comparison of the detection limit of BI was made between the eastern blotting technique and the TLC plate stained with FeCl3 (Figure 5). It became apparent that the eastern blotting technique was almost same sensitivity compared to FeCl3 staining. The eastern blotting technique could detect 1 μg of BI on the PES membrane. In regard to blotting, two points should be taken into consideration. First, BI on the TLC plate should be easily dissolvable in the blotting solution. Second, diffusion should be avoided so that the spots of BI on the TLC plate are not destroyed during the transferring manipulation. It is difficult to estimate the blotting efficiency, because the percentage of BI that can be blotted onto the PES membrane is unknown.

Figure 2.
Dot blot analysis of BI and its specificity against various glycosides.
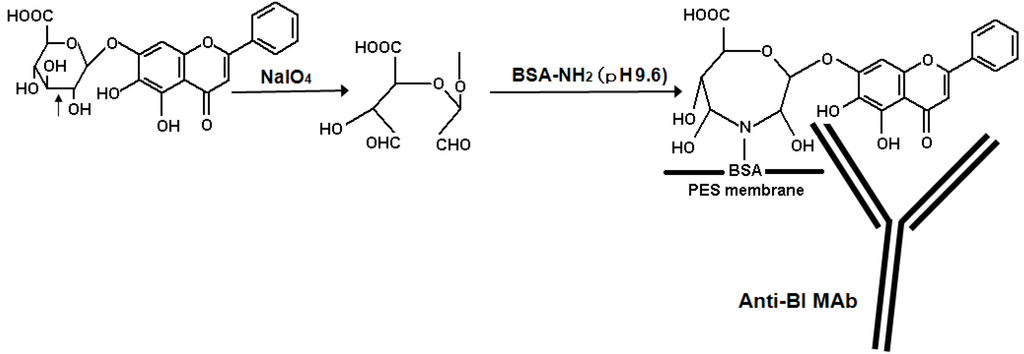
Figure 3.
Schematic diagram illustrating the immunostaining of BI onto polyethersulfone (PES) membrane and the detection using an anti-BI MAb.
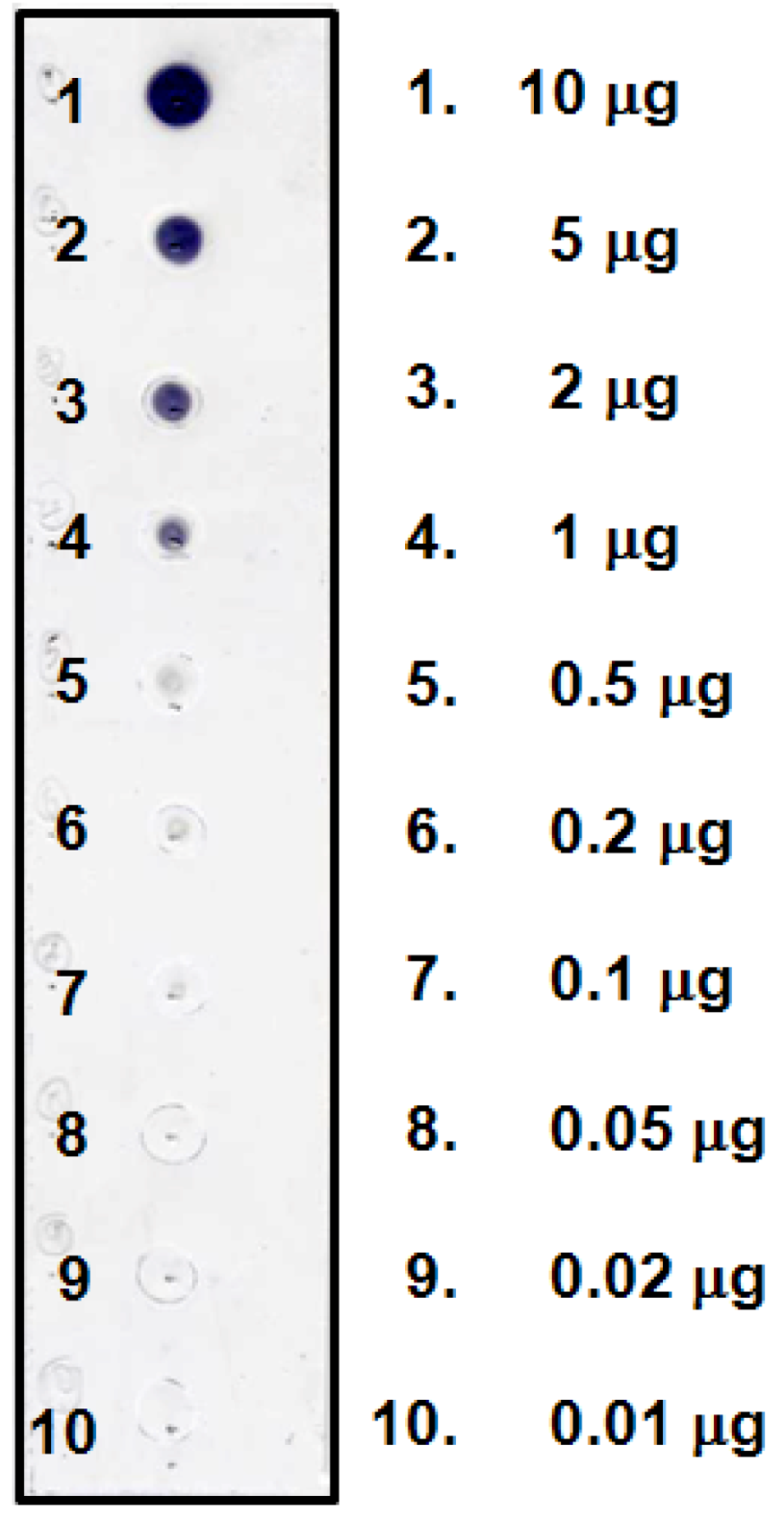
Figure 4.
Dot blot analysis of BI and its detection limit.
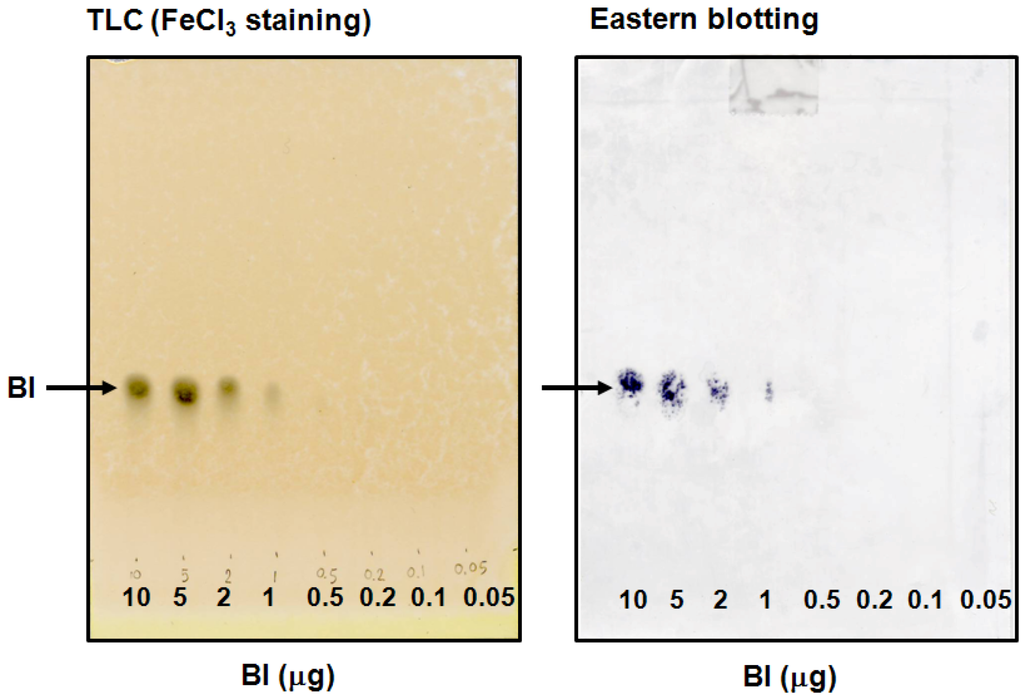
Figure 5.
Eastern blotting of BI and its detection limit.
2.3. Qualitative and Quantitative Analysis of BI Using Eastern Blotting Technique and Competitive ELISA in Crude Extracts of Various S. radix Samples and KMs
Various S. radix samples and KMs were qualitatively analyzed by eastern blotting technique (Figure 6), and the amount of BI was quantitatively determined by competitive ELISA using anti-BI MAb (Table 1). Lane 6 is shakuyakukanzoto extract which does not contain S. radix and no band was indicated by immunostaining. On the other hand, the stained band in shosaikoto and saikokaryukotsuboreito extracts (Figure 6, lanes 7 and 8), which contain S. radix, and various extracts of S. radix samples clearly appeared (Figure 6, lanes 2–5). Furthermore, the concentration of BI was quantitatively analyzed by competitive ELISA using anti-BI MAb (Table 1). Collectively, this established eastern blotting technique is more useful than the TLC analysis for the specific detection of BI in samples containing various crude drugs such as KMs with various colored components (e.g., flavonoids, anthraquinones and alkaloids) and impurities.
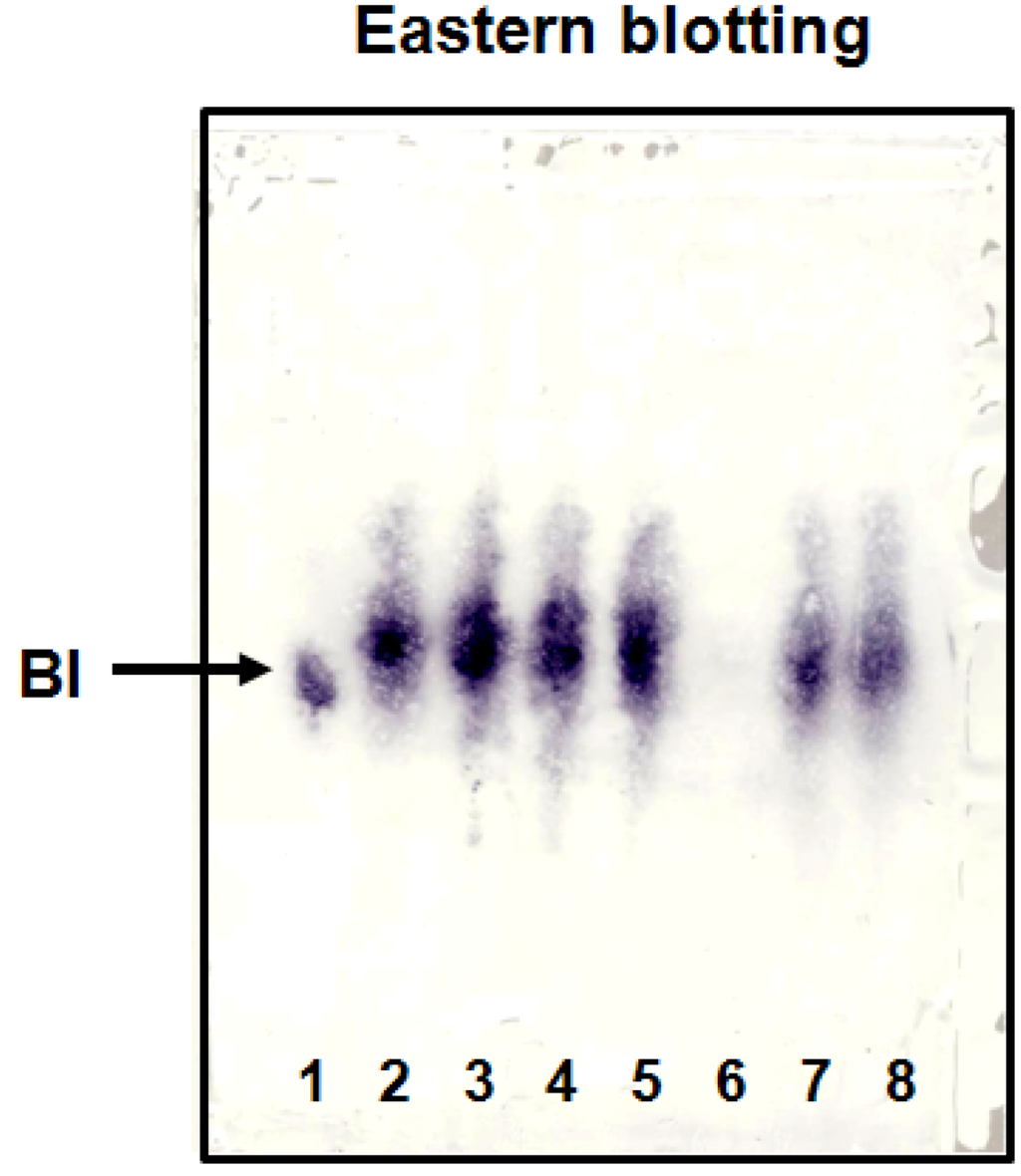
Figure 6.
Eastern blotting of BI in S. radixes and KMs. 1. Baicalin standard, 2. S. radix main root, 3. S. radix A, 4. S. radix B, 5. S. radix C, 6. Shakuyakukanzoto, 7. Shosaikoto, 8. Saikokaryukotsuboreito

Table 1.
The concentration of BI in S. radixes and KMs determined by ELISA.
| Sample name | BI (μg/mg dry wt.) |
|---|---|
| Scutellariae radix main root | 74.2 ± 7.8 |
| S. radix A | 126.6 ± 17.3 |
| S. radix B | 177.8 ± 6.1 |
| S. radix C | 80.4 ± 10.0 |
| Shakuyakukanzoto | N.D. |
| Shosaikoto | 29.0 ± 4.7 |
| Saikokaryukotsuboreito | 9.2 ± 1.1 |
Data were the means of triplicate assays. ND= not detectable.
2.4. Validation of Eastern Blotting Technique to Identify the Distribution of BI in Fresh Root of S. Baicalensis
As another application of eastern blotting technique, the immunostaining of BI was investigated to identify the distribution of BI in fresh root of S. baicalensis. A piece of PES membrane was placed onto a glass microfiber filter sheet. A sliced fresh root of S. baicalensis was treated with methanol to inactivate endogenous peroxidase. Then the sliced root was placed onto the PES membrane, and pressed evenly for 10 min. The blotted PES membrane was immunostained using a procedure similar to that described in dot blot assay. Figure 7 illustrates the immunohistochemical localization of BI in fresh S. baicalensis root. The cortex (part B) contained a higher concentration of BI than the xylem (part A). In order to confirm this result, these tissues were analyzed individually by competitive ELISA. The concentrations of BI, as determined by ELISA, were 33.7 ± 3.3 and 72.2 ± 6.8 μg/mg dry weight for xylem and cortex, respectively.
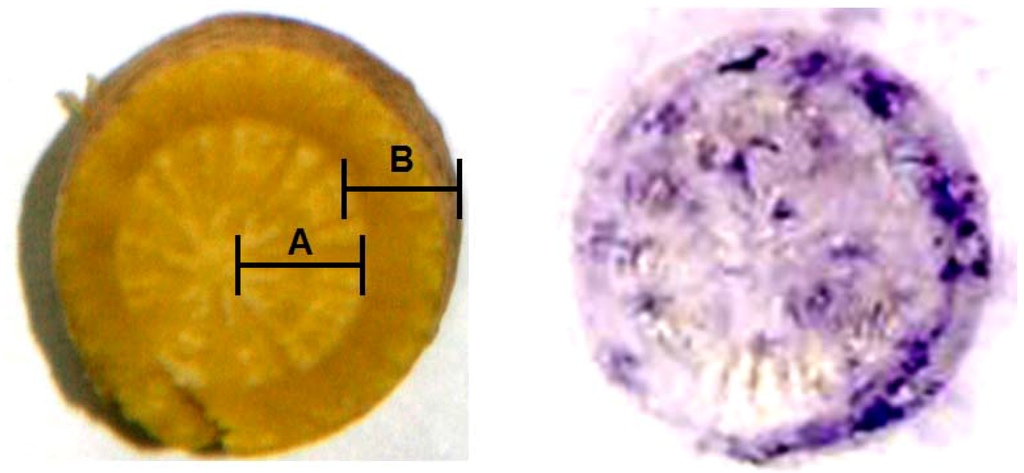
Figure 7.
Immunohistochemical analysis of BI in the rootof S. baicalensis.
3. Experimental
3.1. Chemicals and Immunochemicals
BI, BE and other flavonoids were purchased from Wako Pure Chemicals (Osaka, Japan). Bovine serum albumin (BSA) and peroxidase-labeled anti-mouse IgG (whole molecule) were purchased from Sigma (Steinheim, Germany). MustangTM E positive-charged PES membrane was purchased from Pall Corporation (East Hills, NY, USA). Glass microfiber filter sheets (GF/A) were purchased from Whatman International Ltd. (Maidstone, England). All other chemicals were standard commercial products of analytical-reagent grade.
3.2. Plant Materials and Extraction
Samples of various S. radixes were purchased from Tochimototenkaido Corporation (Osaka, Japan) and collected from the botanical garden of Nagasaki International University. The commercial KMs were procured from Tsumura & Co. (Tokyo, Japan).
Dried powders of various S. radixs (100 mg) and KMs (100 mg) were extracted five times with 1.2 mL methanol containing 0.1% NH4OH with sonication and combined into the tube. Then, the solvent of the extract was evaporated using N2 gas evaporation system and freeze-dryer. After freeze-drying, the residue was dissolved in 1.0 mL methanol containing 0.1% NH4OH, filtered using a Cosmonice filter W (0.45 μm filter unit, Nacalai Tesque, Kyoto, Japan) and used for the experiments of eastern blotting and ELISA.
3.3. MAb against BI and Its Development of ELISA System for BI
Immunization, hybridization, purification and characterization of MAb against BI and its development of ELISA system for determination of BI have been established and described previously [15].
3.4. Dot Blot Assay of BI
BI and various glycosides were dissolved in methanol (1 mg/mL) and aliquots (10 μg) were dotted onto a positive-charged PES membrane. All following steps were done at room temperature. After air drying, the PES membrane was dipped into water containing NaIO4 (10 mg/mL) and kept for 30 min. After washing with water, 50 mM carbonate buffer solution (pH 9.6) containing BSA (1%) was added, and shaken for 3 h. After washing the PES membrane with phosphate buffer saline (PBS), the membrane was treated with PBS containing 5% skim-milk for 2 h to reduce nonspecific adsorption. The PES membrane was immersed in anti-BI MAb and shaken for 3 h. After washing the PES membrane twice with PBS containing 0.05% Tween 20 (T-PBS), a peroxidase-labeled goat anti-mouse IgG (whole molecule) in T-PBS was added and shaken for 1 h. The PES membrane was washed twice with T-PBS and once with PBS and exposed to freshly prepared 1 mg/mL 4-chloro-1-naphthol-0.03% H2O2 in PBS for 15 min. The reaction was stopped by washing with PBS, and the immunostained PES membrane was allowed to dry.
3.5. Thin-Layer Chromatography and FeCl3 Staining
BI (10 μg) and appropriate other sample extracts were applied to a TLC plate (Silica gel 60, Merck Ltd., Darmstadt, Germany) and developed with 1-butanol-water-acetic acid (4:2:1, by volume). After drying the developed TLC plate, it was sprayed by FeCl3 solution for the detection of BI.
3.6. Eastern Blotting of BI
BI (10 μg) and appropriate other sample extracts were applied to a TLC plate and developed with 1-buranol-water-acetic acid (4:2:1, by volume). The developed TLC plate was dried and then sprayed with a blotting solution mixture of isopropanol-methanol-water (1:4:20, by volume). It was placed on a stainless steel plate and then covered with a PES membranes sheet. After covering with a glass microfiber filter sheet, the whole assembly was pressed evenly for 70 s with a 120 °C hot plate. The PES membrane was separated from the TLC plate and dried. The blotted PES membrane was treated for the immunostaining of BI as described in dot blot assay.
3.7. Immunohistochemical Localization of BI in Fresh Root of S. baicalensis
Fresh root of S. baicalensis was collected from the botanical garden of Nagasaki International University at the summer season. To identify the distribution of BI in the root, a sliced fresh root of S. baicalensis (thickness 2–3 mm) was placed on the PES membrane, and they were pressed together evenly 10 min for transfer all of the contents from the section to the PES membrane. Then, the blotted PES membrane was stained using the same procedure described in the dot blot assay.
4. Conclusions
In conclusion, the eastern blotting technique enabled visualizing a low molecular compound on a PES membrane using the immunostaining method. With the aid of the specificity of anti-BI MAb, the eastern blotting technique may prove to be useful for the identification of BI in a background containing a large amount of impurities, such as crude drugs and KMs. Moreover, this method can be used to investigate the distribution of BI in fresh S. baicalensis root using immunohistochemical localization.
Acknowledgments
This research was supported in part by Grant-in-Aid (No. 21790030 for O. M.) from the Ministry of Education, Culture, Sports, Science and Technology of Japan and the research grant from Takeda Science Foundation.
References
- Kawashima, K.; Nomura, A.; Makino, T.; Saito, K.; Kano, Y. Pharmacological properties of traditional medicine (XXIX): Effect of Hange-shashin-to and the combinations of its herbal constituents on rat experimental colitis. Biol. Pharm. Bull. 2006, 29, 1599–1603. [Google Scholar]
- Koda, A.; Watanabe, S.; Yanagihara, Y.; Nagai, H.; Sakamoto, K. A comparative study of the anti-allergic effects of disodium baicalein 6-phosphate (BPS) and disodium cromoglycate (DSCG). Jpn. J. Pharmacol. 1977, 27, 31–38. [Google Scholar] [CrossRef]
- Li, B.Q.; Fu, T.; Gong, W.H.; Dunlop, N.; Kung, H.F.; Yan, Y.D.; Kang, J.; Wang, J.M. The flavonoid baicalin exhibits anti-inflammatory activity by binding to chemokines. Immunopharmacology 2000, 49, 295–306. [Google Scholar] [CrossRef]
- Li, B.Q.; Fu, T.; Yan, Y.D.; Baylor, N.W.; Ruscetti, F.W.; Kung, H.F. Inhibition of HIV infection by baicalin-a flavonoid compound purified from Chinese herbal medicine. Cell Mol. Biol. Res. 1993, 39, 119–124. [Google Scholar]
- Wu, J.A.; Attele, A.S.; Zhang, L.; Yuan, C.S. Anti-HIV activity of medicinal herbs: usage and potential development. Am. J. Chin. Med. 2001, 29, 69–81. [Google Scholar] [CrossRef]
- Konoshima, T.; Kokumai, M.; Kozuka, M.; Iinuma, M.; Mizuno, M.; Tanaka, T.; Tokuda, H.; Nishino, H.; Iwashima, A. Studies on inhibitors of skin tumor promotion. XI. Inhibitory effects of flavonoids from Scutellaria baicalensis on Epstein-Barr virus activation and their anti-tumor-promoting activities. Chem. Pharm. Bull. 1992, 40, 531–533. [Google Scholar] [CrossRef]
- Shieh, D.E.; Liu, L.T.; Lin, C.C. Antioxidant and free radical scavenging effects of baicalein, baicalin and wogonin. Anticancer Res. 2000, 20, 2861–2865. [Google Scholar]
- Morimoto, S.; Tateishi, N.; Matsuda, T.; Tanaka, H.; Taura, F.; Furuya, N.; Matsuyama, N.; Shoyama, Y. Novel hydrogen peroxide metabolism in suspension cells of Scutellaria baicalensis Georgi. J. Biol. Chem. 1998, 273, 12606–12611. [Google Scholar]
- Morimoto, S.; Tateishi, N.; Inuyama, M.; Taura, F.; Tanaka, H.; Shoyama, Y. Identification and molecular characterization of novel peroxidase with structural protein-like properties. J. Biol. Chem. 1999, 274, 26192–26198. [Google Scholar]
- Lin, M.C.; Tsai, M.; Wen, K.C. Supercritical fluid extraction of flavonoids from Scutellariae radix. J. Chromatogr. A 1999, 830, 387–395. [Google Scholar] [CrossRef]
- Zhang, L.; Xing, D.; Ding, Y.; Wang, R.; Wang, X.; Du, L. A chromatographic method for baicalin quantification in rat thalamus. Biomed. Chromatogr. 2005, 19, 494–497. [Google Scholar] [CrossRef]
- Kotani, A.; Kojima, S.; Hakamata, H.; Kusu, F. HPLC with electrochemical detection to examine the pharmacokinetics of baicalin and baicalein in rat plasma after oral administration of a Kampo medicine. Anal. Biochem. 2006, 350, 99–104. [Google Scholar]
- Zhang, H.; Tian, K.; Tang, J.; Qi, S.; Chen, H.; Chen, X.; Hu, Z. Analysis of baicalein, baicalin and wogonin in Scutellariae radix and its preparation by microemulsion electrokinetic chromatography with 1-butyl-3-methylimizolium tetrafluoborate ionic liquid as additive. J. Chromatogr. A 2006, 1129, 304–307. [Google Scholar] [CrossRef]
- Weiler, E.W.; Kruger, H.; Zenk, M.H. Radioimmunoassay for the determination of the steroidal alkaloid solasodine and related compounds in living plants and herbarium specimens. Planta Med. 1980, 39, 112–124. [Google Scholar] [CrossRef]
- Kido, K.; Morinaga, O.; Shoyama, Y.; Tanaka, H. Quick analysis of baicalin in Scutellariae radix by enzyme-linked immunosorbent assay using a monoclonal antibody. Talanta 2008, 77, 346–350. [Google Scholar] [CrossRef]
- Paudel, M.K.; Putalun, W.; Sritularak, B.; Morinaga, O.; Shoyama, Y.; Tanaka, H.; Morimoto, S. Development of a combined technique using a rapid one-step immunochromatographic assay and indirect competitive ELISA for the rapid detection of baicalin. Anal. Chim. Acta. 2011, 701, 189–193. [Google Scholar] [CrossRef]
- Tanaka, H.; Putalun, W.; Tsuzaki, C.; Shoyama, Y. A simple determination of steroidal alkaloid glycosides by thin-layer chromatography immunostaining using monoclonal antibody against solamargine. FEBS Lett. 1997, 404, 279–282. [Google Scholar] [CrossRef]
- Taki, T.; Kasama, T.; Handa, S.; Ishikawa, D. A simple and quantitative purification of glycosphingolipids and phospholipids by thin-layer chromatography blotting. Anal. Biochem. 1994, 223, 232–238. [Google Scholar]
- Shan, S.; Tanaka, H.; Shoyama, Y. Enzyme-linked immunosorbent assay for glycyrrhizin using anti-glycyrrhizin monoclonal antibody and an eastern blotting technique for glucuronides of glycyrrhetic acid. Anal. Chem. 2001, 73, 5784–5790. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).