Using a Geospatial Model to Relate Fluvial Geomorphology to Macroinvertebrate Habitat in a Prairie River—Part 2: Matching Family-Level Indices to Geomorphological Response Units (GRUs)
Abstract
:1. Introduction
2. Methods
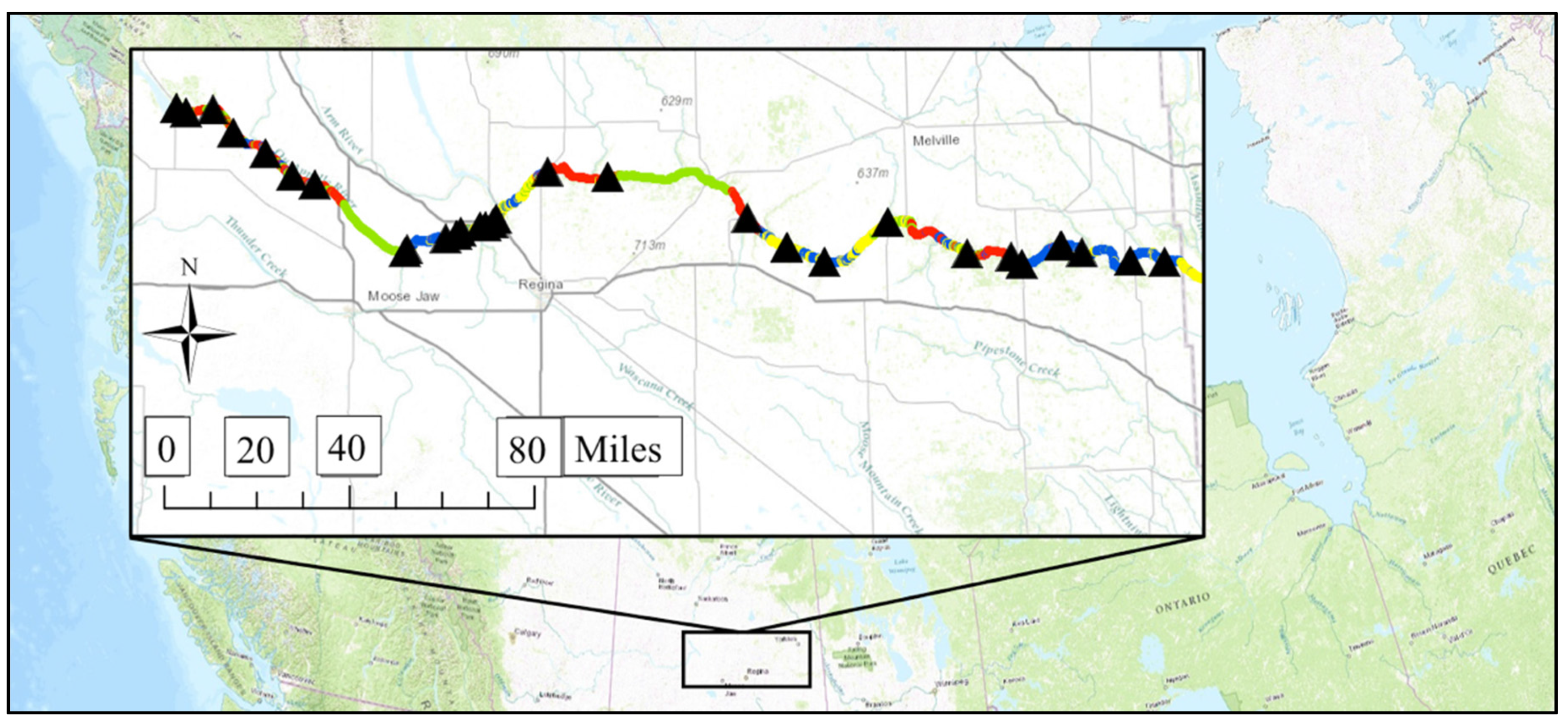
2.1. Study River
2.2. Creating the Geospatial Model
2.3. Macroinvertebrate Sampling
2.4. EPT/C Ratio
2.5. Shannon Diversity Index
2.6. Family Biotic Index
2.7. Comparing GRUs to Macroinvertebrate Data
3. Results and Discussion
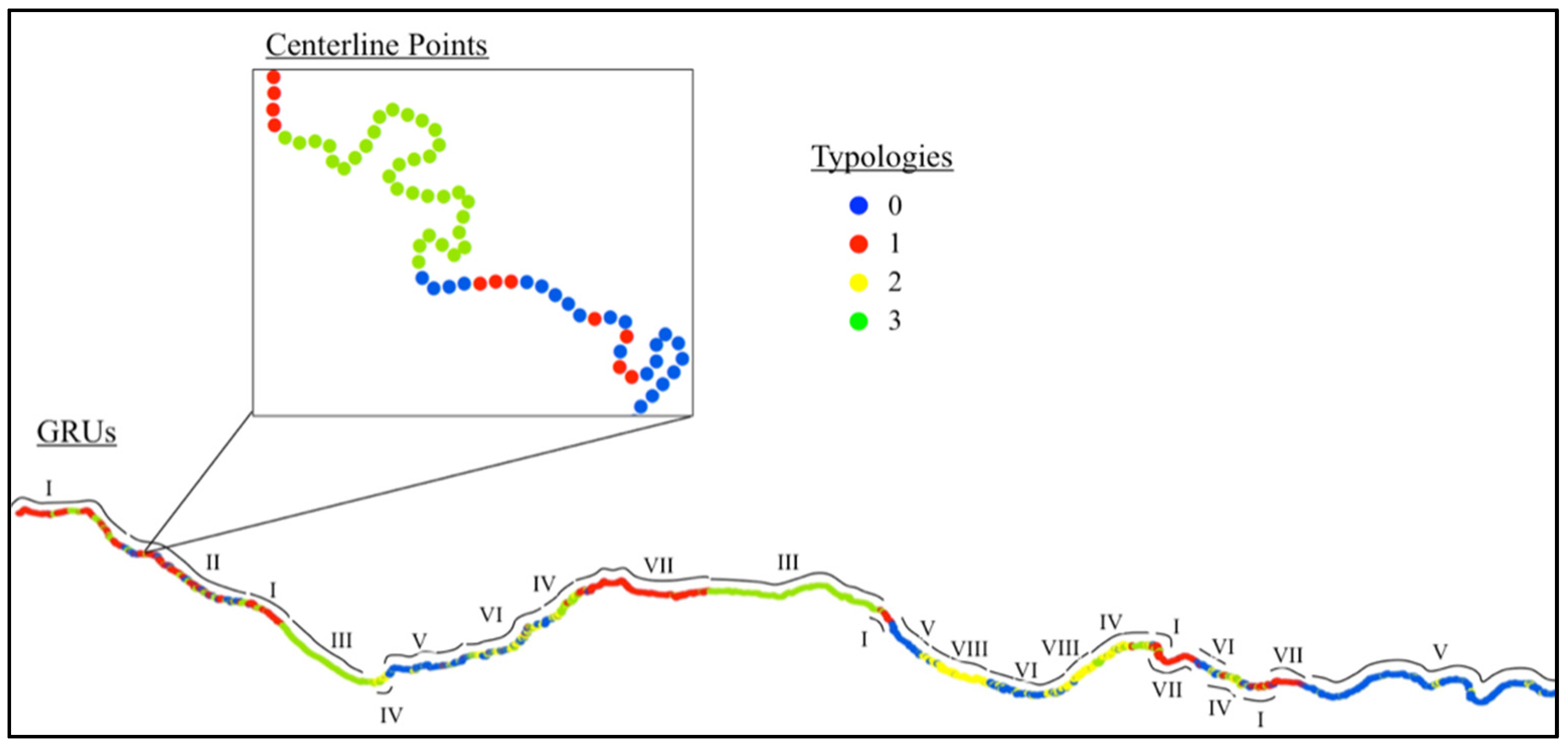
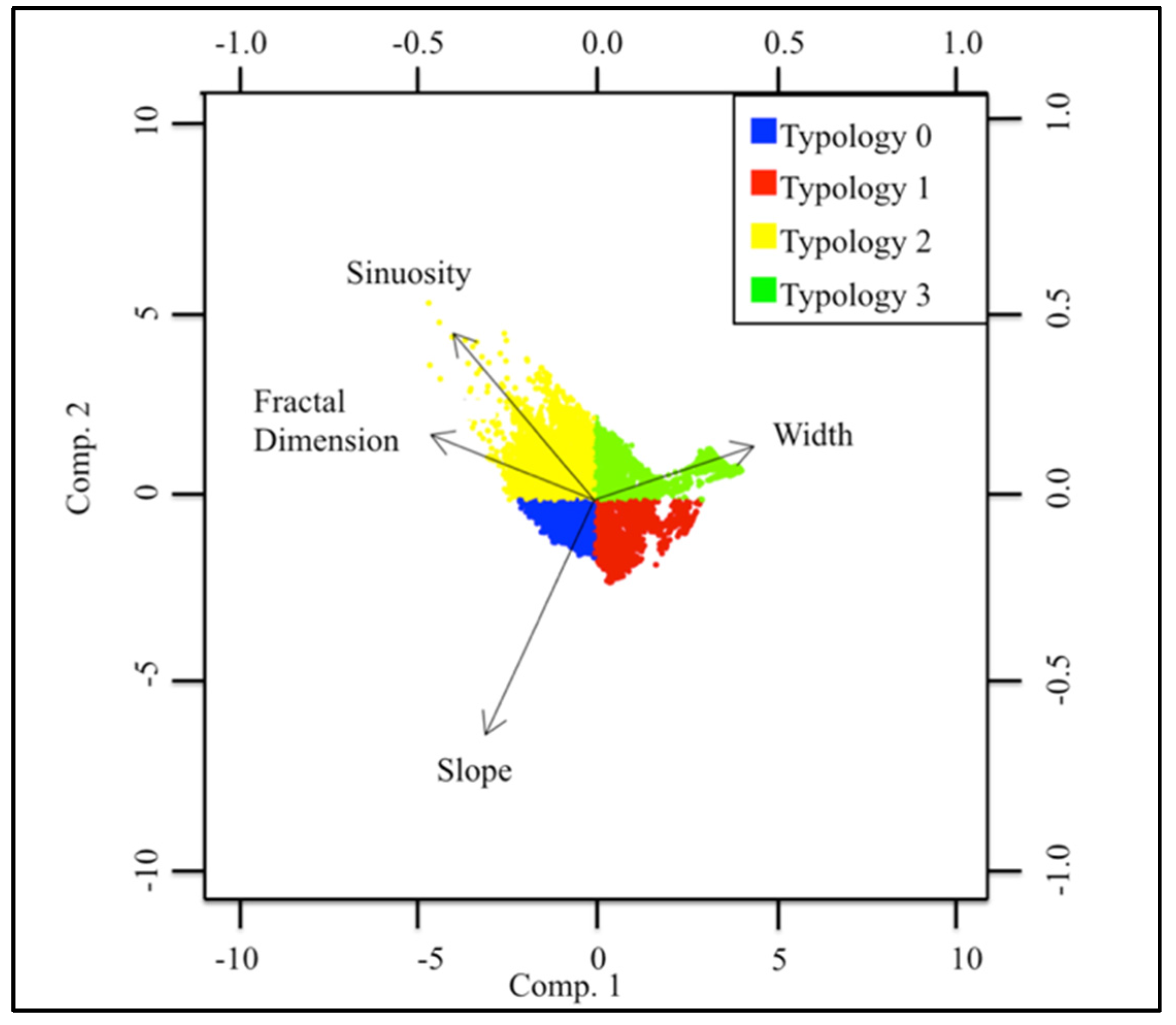
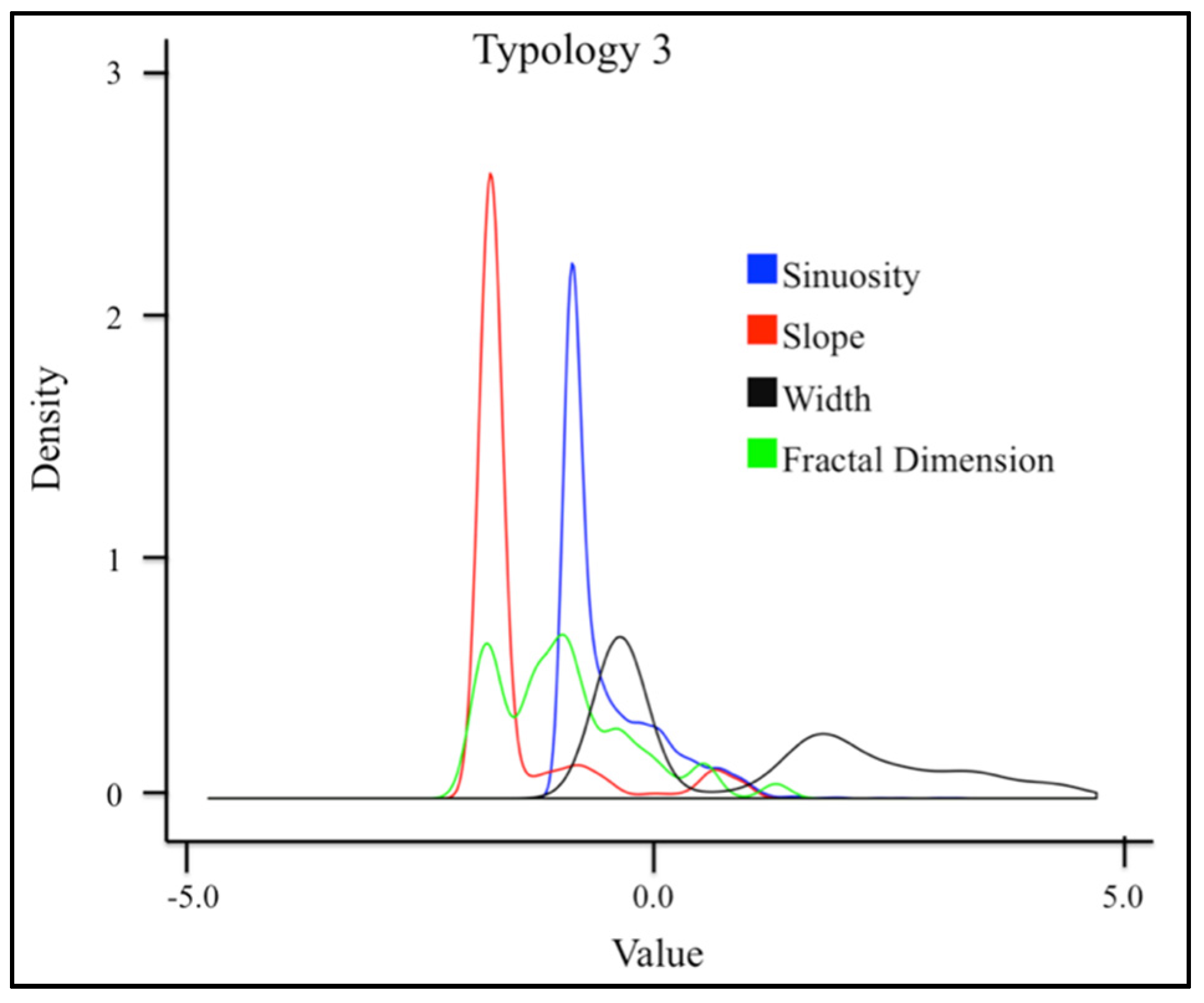
3.1. Geospatial Factors
3.2. Overall Macroinvertebrate Indices Results
3.2.1. EPT/C Ratio
3.2.2. Family Biotic Index
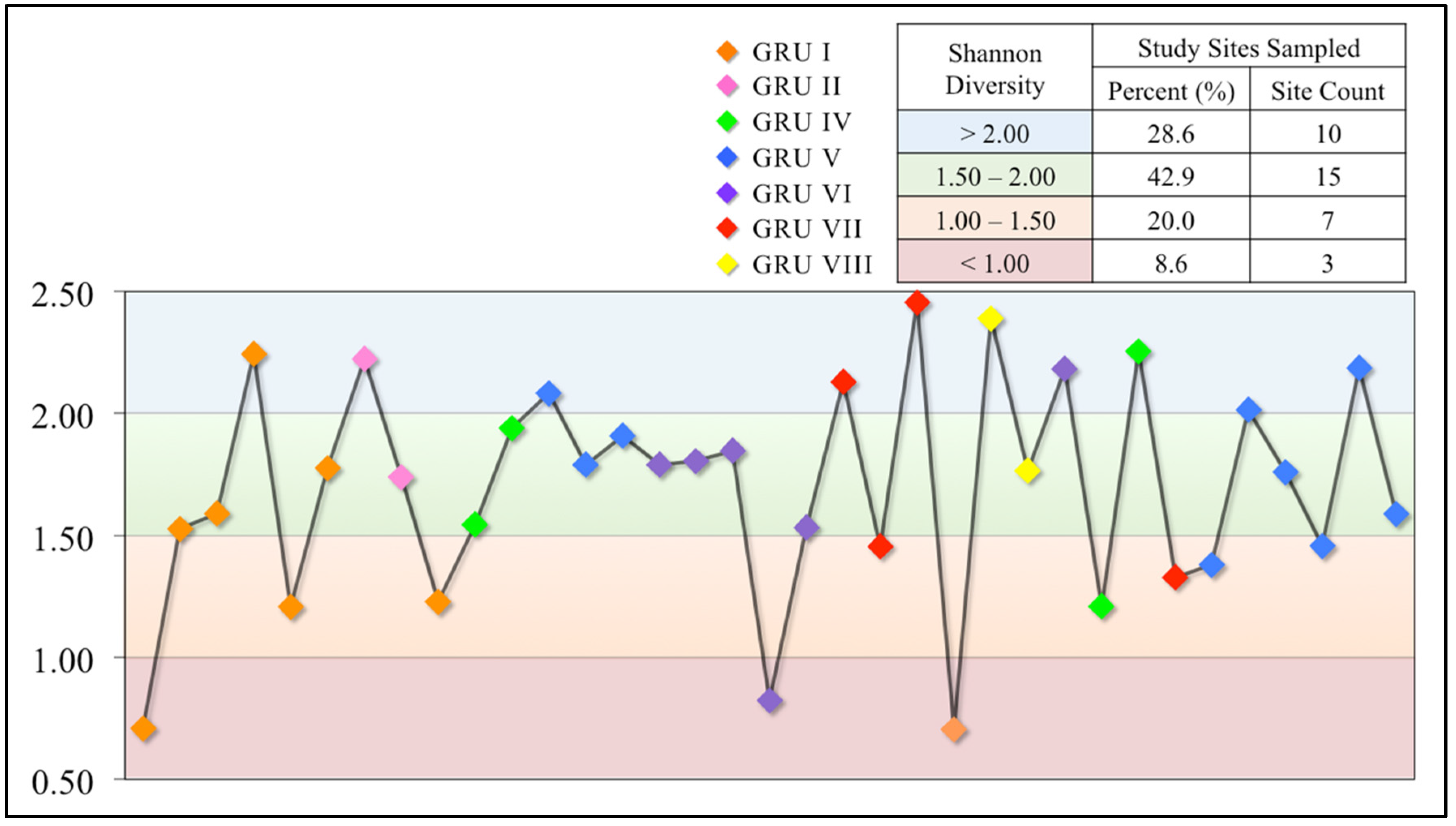
3.2.3. Shannon Diversity Index
3.3. Comparing GRUs to Macroinvertebrate Data
3.3.1. EPT Families
3.3.2. Dipteran Families
3.3.3. Lepidopteran Families
3.3.4. Amphipoda Families
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
| Qu’Appelle River Macroinvertebrate Taxa List | |||||||
|---|---|---|---|---|---|---|---|
| Order | Family | Functional Feeding Groups | Total | Order | Family | Functional Feeding Groups | Total |
| Acari | Acari | Predators | 15 | Gastropoda | Ancylidae | Scrapers | 14 |
| Amphipoda | Amphipoda | Collector Gatherers | 14269.91 | Gastropoda | Scrapers | 1 | |
| Gammaridae | Collector Gatherers | 2046.05 | Hydrobiidae | Scrapers | 165 | ||
| Dogielinotidae | Collector Gatherers | 44289.81 | Lymnaeidae | Scrapers | 140 | ||
| Total | 60605.76 | Physidae | Scrapers | 3825.57 | |||
| Coleoptera | Dytiscidae | Predators | 106 | Planorbidae | Scrapers | 506 | |
| Elmidae | Collector Gatherers | 122.82 | Valvatidae | Scrapers | 643 | ||
| Gyrinidae | Predators | 70 | Total | 5294.57 | |||
| Haliplidae | Shredders | 43 | Hemiptera | Corixidae | Herbivores | 3021.56 | |
| Hydraenidae | Scrapers | 4 | Predators | 586.02 | |||
| Hydrophilidae | Collector Gatherers | 10 | Notonectidae | Predators | 14 | ||
| Herbivores | 1 | Total | 3621.58 | ||||
| Predators | 1 | Hydrachnidia | Hydrachnidia | Predators | 53 | ||
| Shredders | 1 | Lepidoptera | Crambidae | Herbivores | 546 | ||
| Total | 358.82 | Lepidoptera | Herbivores | 10 | |||
| Collembola | Collembola | Collector Gatherers | 1 | Noctuidae | Herbivores | 264 | |
| Decopoda | Cambaridae | Omnivores | 166 | Total | 820 | ||
| Diptera | Athericidae | Predators | 2 | Malacostraca | Cambaridae | Omnivores | 257 |
| Ceratopogonidae | Predators | 167.04 | Nematoda | Nematoda | Predators | 15 | |
| Chironomidae | Collector Gatherers | 7747.06 | Odonata | Coenagrionidae | Predators | 165 | |
| Predators | 442.26 | Gomphidae | Predators | 16 | |||
| Diptera | Collector Gatherers | 18 | Lestidae | Predators | 16 | ||
| Empididae | Predators | 39 | Total | 197 | |||
| Leptoceridae | Shredders | 22 | Oligochaeta | Oligochaeta | Detritivores | 1514.70 | |
| Simuliidae | Filterers | 19958 | Ostracoda | Ostracoda | Filterers | 8 | |
| Stratiomyidae | Collector Gatherers | 1 | Pelecypoda | Pisidiidae | Filterers | 691.10 | |
| Tabanidae | Predators | 12 | Pharyngobdellida | Erpobdellidae | Predators | 24 | |
| Tipulidae | Predators | 10 | Plecoptera | Perlidae | Predators | 5 | |
| Total | 28418.35 | Plecoptera | Shredders | 1 | |||
| Pteronarcyidae | Shredders | 10 | |||||
| Total | 16 | ||||||
| Ephemeroptera | Baetidae | Scrapers | 3088.10 | Rhynchobdellida | Glossiphoniidae | Predators | 20 |
| Caenidae | Collector Gatherers Scrapers | 491 | |||||
| Filterers | 279 | ||||||
| Ephemeridae | Collector Gatherers | 2666.82 | Trichoptera | Brachycentridae | Filterers | 238 | |
| Ephemeridae/polymitarcyidae | Collector Gatherers | 891 | Hydropsychidae | Filterers | 2748.02 | ||
| Ephemeroptera | Scrapers | 353.27 | Hydroptilidae | Herbivores | 770 | ||
| Heptageniidae | Collector Gatherers | 437 | Leptoceridae | Collector Gatherers | 54 | ||
| Scrapers | 1189.82 | Herbivores | 16 | ||||
| Isonychiidae | Filterers | 4 | Shredders | 92.02 | |||
| Leptohyphidae | Collector Gatherers | 79 | Phryganeidae | Shredders | 31.02 | ||
| Leptophlebiidae | Collector Gatherers | 52.85 | Polycentropodidae | Filterers | 8 | ||
| Scrapers | 2 | Predators | 20 | ||||
| Polymitarcyidae | Collector Gatherers | 82 | Trichoptera | Shredders | 6 | ||
| Total | 9615.86 | Total | 3983.05 | ||||
| Grand Total | 115695.80 | ||||||
References
- D’Ambrosio, J.L.; Williams, L.R.; Witter, J.D.; Ward, A. Effects of Geomorphology, Habitat, and Spatial Location on Fish Assemblages in a Watershed in Ohio, USA. Environ. Monit. Assess. 2009, 148, 325–341. [Google Scholar] [CrossRef] [PubMed]
- Allan, J.D.; Castillo, M.M. Stream Ecology; Springer Science & Business Media: Dordrecht, The Netherlands, 2007. [Google Scholar]
- Rodríguez-Iturbe, I.; Valdés, J.B. The Geomorphologic Structure of Hydrologic Response. Water Resour. Res. 1979, 15, 1409–1420. [Google Scholar] [CrossRef]
- Dollar, E.S.J.; James, C.S.; Rogers, K.H.; Thoms, M.C. A Framework for Interdisciplinary Understanding of Rivers as Ecosystems. Geomorphology 2007, 89, 147–162. [Google Scholar] [CrossRef]
- Chakona, A.; Phiri, C.; Day, J. Potential for Trichoptera Communities as Biological Indicators of Morphological Degradation in Riverine Systems. Hydrobiologia 2008, 621, 155–167. [Google Scholar] [CrossRef]
- O’Laughlin, K. The Streamkeeper’s Field Guide: Watershed Inventory and Stream Monitoring Methods; Adopt-a-Stream Foundation: Washington, DC, USA, 1996. [Google Scholar]
- Dufrêne, M.; Legendre, P. Species Assemblages and Indicator Species: The Need for a Flexible Asymmetrical Approach. Ecol. Monogr. 1997, 67, 345–366. [Google Scholar] [CrossRef]
- Kubosova, K.; Brabec, K.; Jarkovsky, J.; Syrovatka, V. Selection of Indicative Taxa for River Habitats: A Case Study on Benthic Macroinvertebrates Using Indicator Species Analysis and the Random Forest Methods. Hydrobiologia 2010, 651, 101–114. [Google Scholar] [CrossRef]
- Bovee, K.D.; Lamb, B.L.; Bartholow, J.M.; Stalnaker, C.B.; Taylor, J.; Henriksen, J. Stream Habitat Analysis Using the Instream Flow Incremental Methodology; U.S. Geological Survey, Biological Resources Division Information and Technology Report: Fort Collins, CO, USA, 1998.
- Lindenschmidt, K.E.; Long, J. A GIS Approach to Define the Hydro-Geomorphological Regime for Instream Flow Requirements Using Geomorphic Response Units (GRU). River Syst. 2013, 20, 261–275. [Google Scholar] [CrossRef]
- Carr, M.K.; Lacho, C.; Pollock, M.; Watkinson, D.A.; Lindenschmidt, K.-E. Development of geomorphic typologies for identifying Lake Sturgeon (Acipenser fulvescens) habitat in the Saskatchewan River System. River Syst. 2015, 21, 215–227. [Google Scholar] [CrossRef]
- Carr, M.K.; Watkinson, D.A.; Svendsen, J.C.; Enders, E.C.; Long, J.M.; Lindenschmidt, K.-E. Geospatial modeling of the Birch River: Distribution of Carmine Shiner (Notropis percobromus) in Geomorphic Response Units (GRU). Int. Rev. Hydrobiol. 2015, 100, 129–140. [Google Scholar] [CrossRef]
- Carr, M.K.; Watkinson, D.A.; Lindenschmidt, K.-E. Identifying links between Fluvial Geomorphic Response Units (FGRU) and fish species in the Assiniboine River, Manitoba. Ecohydrology 2016. [Google Scholar] [CrossRef]
- Dodds, W.K.; Whiles, M.R. Freshwater Ecology: Concepts and Environmental Applications of Limnology; Academic Press: San Diego, CA, USA, 2010. [Google Scholar]
- Merritt, R.W.; Cummins, K.W.; Hunt, K. An Introduction to the Aquatic Insects of North America; Kendall/Hunt Publishing Company: Dubuque, IA, USA, 1996. [Google Scholar]
- Ernst, A.G.; Warren, D.R.; Baldigo, B.P. Natural–Channel–Design Restorations That Changed Geomorphology Have Little Effect on Macroinvertebrate Communities in Headwater Streams. Restor. Ecol. 2012, 20, 532–540. [Google Scholar] [CrossRef]
- Meissner, A.G.N.; Carr, M.K.; Phillips, I.D.; Lindenschmidt, K-E. Using a Geospatial Model to Relate Fluvial Geomorphology to Macroinvertebrate Habitat in a Prairie River—Part 1: Genus-Level Relationships with Geomorphic Typologies. Water 2016, 8, 42. [Google Scholar] [CrossRef]
- Mollard, J.D. Morphological Study of the Upper Qu’Appelle River; Friends of Wascana Marsh: Regina, SK, Canada, 2004. [Google Scholar]
- Clifton Associates Ltd. Upper Qu’Appelle Water Supply Project: Economic Impact and Sensitivity Analysis; Report to Water Security Agency; Clifton Associates Ltd.: Regina, SK, Canada, 2012. [Google Scholar]
- Pomeroy, J.; de Boer, D.; Martz, L. Hydrology and Water Resources of Saskatchewan: Centre for Hydrology Report #1; University of Saskatchewan: SK, Canada, 2005. [Google Scholar]
- Shen, X.H.; Zou, L.J.; Zhang, G.F.; Su, N.; Wu, W.Y.; Yang, S.F. Fractal Characteristics of the Main Channel of Yellow River and Its Relation to Regional Tectonic Evolution. Geomorphology 2011, 127, 64–70. [Google Scholar] [CrossRef]
- Güneralp, İ.; Abad, J.D.; Zolezzi, G.; Hooke, J. Advances and Challenges in Meandering Channels Research. Geomorphology 2012, 163–164, 1–9. [Google Scholar] [CrossRef]
- Ahnert, F. Einführung in Die Geomorphologie; Verlag Eugen Ulmer: Stuttgart, Germany, 2015. [Google Scholar]
- Lüderitz, V.; Kunz, C.; Wüstemann, O.; Remy, D.; Feuerstein, B. Typisierung und Bewertung für die leitbildorientierte Sanierung von Altwässern. In Flussaltwässer: Ökologie und Sanierung; Lüderitz, V., Langheinrich, U., Kunz, C., Eds.; Vieweg + Teubner: Wiesbaden, Germany, 2009; pp. 91–168. [Google Scholar]
- Zumbroich, T.; Müller, A. Das Verfahren der Gewässerstrukturkartierung. In Strukturgüte von Fließgewässern: Grundlagen und Kartierung, 1st ed.; Zumbroich, T., Müller, A., Günther, F., Eds.; Springer-Verlag Berlin Heidelberg: Berlin, Germany, 1999; pp. 97–121. [Google Scholar]
- Schumm, S.A. The Fluvial System; John Wiley & Sons: New York, NY, USA, 1977; p. 338. [Google Scholar]
- Schuller, D.J.; Rao, A.R.; Jeong, G.D. Fractal Characteristics of Dense Stream Networks. J. Hydrol. 2001, 243, 1–16. [Google Scholar] [CrossRef]
- MathSoft Inc. Mathcad v.15; MathSoft Inc.: Cambridge, MA, USA, 2012. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2012; Available online: https://www.r-project.org/ (accessed on 10 September 2015).
- Legendre, P.; Legendre, L. Numerical Ecology, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Jolliffe, I.T. Discarding Variables in a Principal Component Analysis. I: Artificial Data. J. R. Stat. Soc. C (Appl. Stat.) 1972, 21, 160–173. [Google Scholar] [CrossRef]
- Jolliffe, I.T. Principal Component Analysis, 2nd ed.; Springer: New York, NY, USA, 2002; p. 488. [Google Scholar]
- Dosdall, L.M.; Lehmkuhl, D.M. Stoneflies (Plecoptera) of Saskatchewan. Quaest. Entomol. 1979, 15, 3–116. [Google Scholar]
- Clifford, H.F. Aquatic Invertebrates of Alberta; The University of Alberta Press: Edmonton, AB, Canada, 1991. [Google Scholar]
- Webb, J.M. The Mayflies of Saskatchewan. Master’s Thesis, University of Saskatchewan, Saskatoon, SK, Canada, 2002. [Google Scholar]
- Berg, M.B.; Hellenthal, R.A. The Role of Chironomidae in Energy Flow of a Lotic Ecosystem. Netherland J. Aquat. Ecol. 1992, 26, 471–476. [Google Scholar] [CrossRef]
- Karaus, U.; Larsen, S.; Guillong, H.; Tockner, K. The Contribution of Lateral Aquatic Habitats to Insect Diversity along River Corridors in the Alps. Landsc. Ecol. 2013, 28, 1755–1767. [Google Scholar] [CrossRef]
- Mandaville, S.M. Benthic Macroinvertebrates in Taxa Tolerance Values, Metrics, and Protocols; Project H–1; Soil and Water Conservation Society of Metro Halifax: Halifax, NS, Canada, 2002. [Google Scholar]
- Hilsenhoff, W.L. Rapid Field Assessment of Organic Pollution with a Family-Level Biotic Index. J. North Am. Benthol. Soc. 1988, 7, 65–68. [Google Scholar] [CrossRef]
- Strand, R.M.; Merritt, R.W. Effects of Episodic Sedimentation on the Net- Spinning Caddisflies Hydropsyche betteni and Ceratopsyche Sparna (Trichoptera: Hydropsychidae). Environ. Pollut. 1998, 98, 129–134. [Google Scholar] [CrossRef]
- Jones, J.I.; Murphy, J.F.; Collins, A.L.; Sear, D.A.; Naden, P.S.; Armitage, P.D. The Impact of Fine Sediment on Macroinvertebrates. River. Res. Appl. 2012, 28, 1055–1071. [Google Scholar] [CrossRef]
- Krieger, K.A.; Bur, M.T.; Ciborowski, J.J.H.; Barton, D.R.; Schloesser, D.W. Distribution and Abundance of Burrowing Mayflies (Hexagenia Spp.) in Lake Erie 1997–2005. J. Great Lakes Res. 2007, 33, 20–33. [Google Scholar] [CrossRef]
- Eymann, M. Flow Patterns Around Cocoons and Pupae of Black Flies of the Genus Simulium (Diptera: Simuliidae). Hydrobiologia 1991, 215, 223–229. [Google Scholar] [CrossRef]
- Eymann, M.; Friend, W.G. Behaviors of Larvae of the Black Flies Simulium vittatum and S. decorum (Diptera: Simuliidae) Associated with Establishing and Maintaining Dispersion Patterns on Natural and Artificial Substrates. J. Insect. Behav. 1988, 1, 169–186. [Google Scholar] [CrossRef]
- Stoops, C.A.; Adler, P.H.; McCreadie, J.W. Ecology of Aquatic Lepidoptera (Crambidae: Nymphulinae). Hydrobiologia 1998, 379, 33–40. [Google Scholar] [CrossRef]
- Watson, A.M.; Ormerod, S.J. The Distribution of Three Uncommon Freshwater Gastropods in the Drainage Ditches of British Grazing Marshes. Biol. Conserv. 2004, 118, 455–466. [Google Scholar] [CrossRef]
- Government of Canada Fisheries and Oceans. Manual for the Culture of Selected Freshwater Invertebrates; Lawrence, S.G., Ed.; Department of Fisheries and Oceans Freshwater Institute: Winnipeg, MB, Canada, 1981. [Google Scholar]


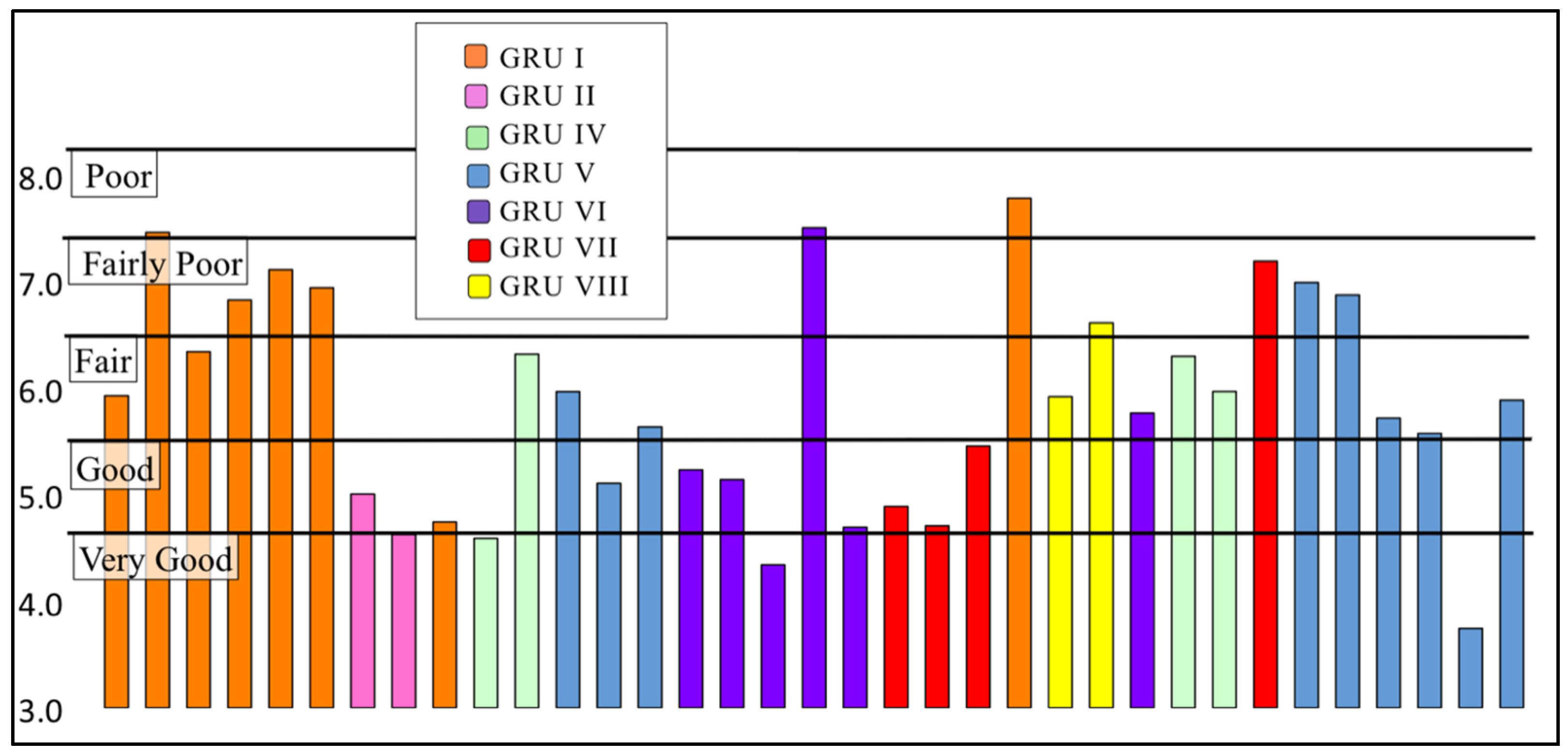

| Biotic Index Score | Water Quality | Degree of Organic Pollution | Percent of Sites |
|---|---|---|---|
| 0.00–3.50 | Excellent | No apparent organic pollution | 0% (0) |
| 3.51–4.50 | Very Good | Possible slight organic pollution | 6% (2) |
| 4.51–5.50 | Good | Some organic pollution | 31% (11) |
| 5.51–6.50 | Fair | Fairly significant organic pollution | 34% (12) |
| 6.51–7.50 | Fairly Poor | Significant organic pollution | 26% (9) |
| 7.51–8.50 | Poor | Very significant organic pollution | 3% (1) |
| 8.51–10.00 | Very Poor | Severe organic pollution | 0% (0) |
| Typology | Sinuosity | Slope | Fractal Dimension | Width |
|---|---|---|---|---|
| 0 | 0 | + | + | − |
| 1 | − | + | − | − |
| 2 | + | 0 | + | − |
| 3 | − | − | − | + |
| GRU | I | II | III | IV | V | VI | VII | VIII | |
|---|---|---|---|---|---|---|---|---|---|
| Typology Makeup | Most | 1 | 1, 0, 3 | 3 | 3, 2 | 0 | 0, 2 | 1 | 2 |
| Other | 3 | - | - | 1 | 1, 2, 3 | 3, 1 | 0, 2 | 0, 3 | |
| Macroinvertebrate Sample Sites | 8 | 2 | 0 | 4 | 9 | 6 | 4 | 2 | |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meissner, A.G.N.; Carr, M.K.; Phillips, I.D.; Lindenschmidt, K.-E. Using a Geospatial Model to Relate Fluvial Geomorphology to Macroinvertebrate Habitat in a Prairie River—Part 2: Matching Family-Level Indices to Geomorphological Response Units (GRUs). Water 2016, 8, 107. https://doi.org/10.3390/w8030107
Meissner AGN, Carr MK, Phillips ID, Lindenschmidt K-E. Using a Geospatial Model to Relate Fluvial Geomorphology to Macroinvertebrate Habitat in a Prairie River—Part 2: Matching Family-Level Indices to Geomorphological Response Units (GRUs). Water. 2016; 8(3):107. https://doi.org/10.3390/w8030107
Chicago/Turabian StyleMeissner, Anna Grace Nostbakken, Meghan Kathleen Carr, Iain David Phillips, and Karl-Erich Lindenschmidt. 2016. "Using a Geospatial Model to Relate Fluvial Geomorphology to Macroinvertebrate Habitat in a Prairie River—Part 2: Matching Family-Level Indices to Geomorphological Response Units (GRUs)" Water 8, no. 3: 107. https://doi.org/10.3390/w8030107
APA StyleMeissner, A. G. N., Carr, M. K., Phillips, I. D., & Lindenschmidt, K.-E. (2016). Using a Geospatial Model to Relate Fluvial Geomorphology to Macroinvertebrate Habitat in a Prairie River—Part 2: Matching Family-Level Indices to Geomorphological Response Units (GRUs). Water, 8(3), 107. https://doi.org/10.3390/w8030107







