Lettuce (Lactuca sativa L. var. Sucrine) Growth Performance in Complemented Aquaponic Solution Outperforms Hydroponics
Abstract
:1. Introduction
2. Materials and Methods
2.1. Nutrient Solution Formulation and Control
2.2. Lettuce Growth and Leaf Nutrient Content
3. Results
3.1. Shoot and Root Fresh Weight
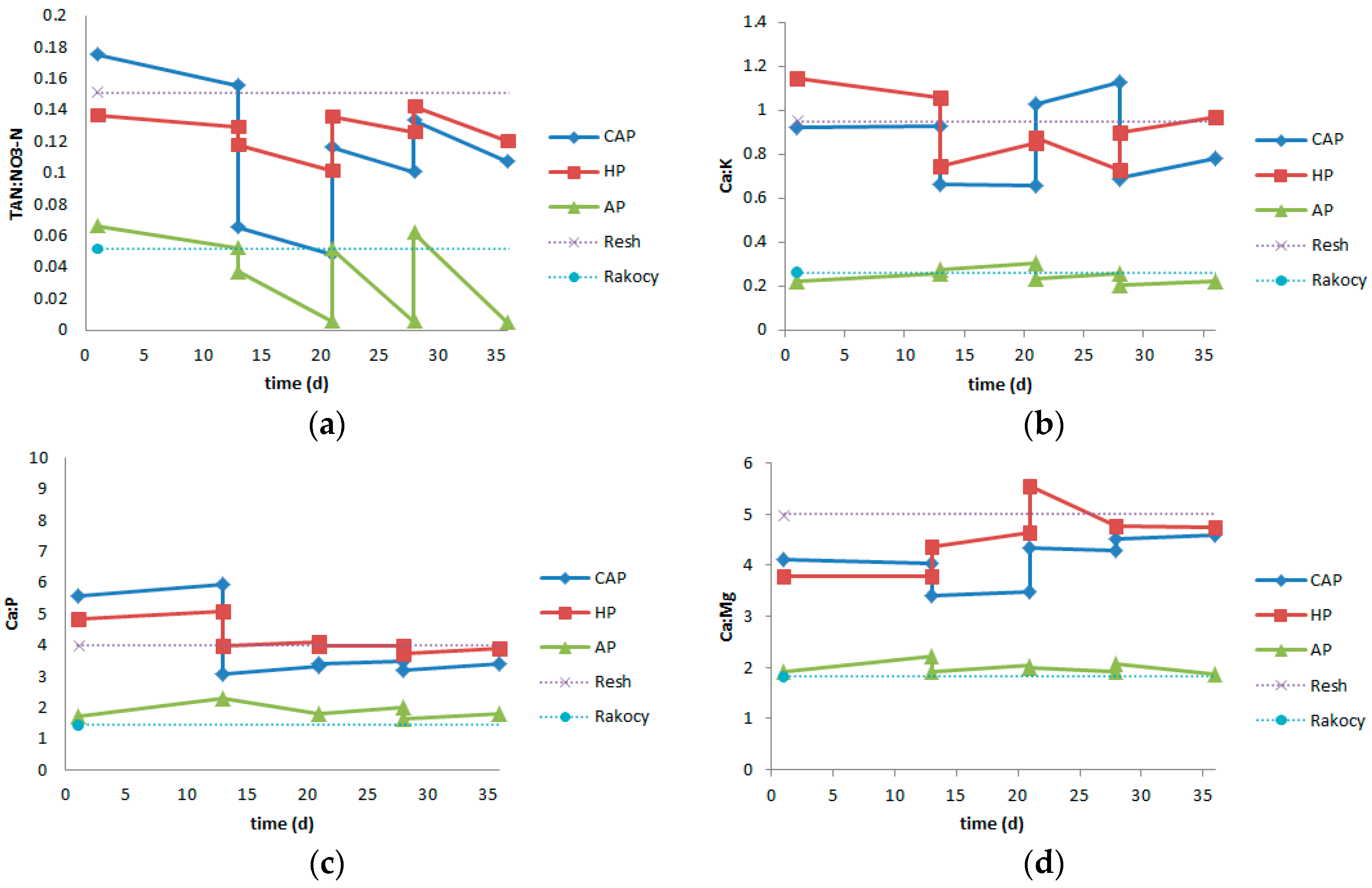
3.2. Nutrient Solutions
3.3. Lettuce Leaf Nutrient Content
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Goddek, S.; Delaide, B.; Mankasingh, U.; Ragnarsdottir, K.; Jijakli, H.; Thorarinsdottir, R. Challenges of Sustainable and Commercial Aquaponics. Sustainability 2015, 7, 4199–4224. [Google Scholar] [CrossRef]
- Endut, A.; Jusoh, A.; Ali, N.; Nik, W.B.W.; Hassan, A. A study on the optimal hydraulic loading rate and plant ratios in recirculation aquaponic system. Bioresour. Technol. 2010, 101, 1511–1517. [Google Scholar] [CrossRef] [PubMed]
- Graber, A.; Junge, R. Aquaponic Systems: Nutrient recycling from fish wastewater by vegetable production. Desalination 2009, 246, 147–156. [Google Scholar] [CrossRef]
- Goddek, S.; Espinal, C.; Delaide, B.; Jijakli, H.M.; Schmautz, Z.; Wuertz, S.; Keesman, K.J. Navigating towards Decoupled Aquaponic Systems: A System Dynamics Design Approach. Water 2016, 8, 303. [Google Scholar] [CrossRef]
- Kloas, W.; Groß, R.; Baganz, D.; Graupner, J.; Monsees, H.; Schmidt, U.; Staaks, G.; Suhl, J.; Tschirner, M.; Wittstock, B.; et al. A new concept for aquaponic systems to improve sustainability, increase productivity, and reduce environmental impacts. Aquac. Environ. Interact. 2015, 7, 179–192. [Google Scholar] [CrossRef]
- Resh, H.M. Hydroponic food Production: A Definitive Guidebook for the Advanced Home Gardener and the Commercial Hydroponic Grower; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- Sonneveld, C.; Voogt, W. Nutrient Solutions for Soilless Cultures. In Plant Nutrition of Greenhouse Crops; Springer: Houten, The Netherlands, 2009; pp. 257–275. [Google Scholar]
- Jones, B.J. Hydroponics—A Practical Guide for the Soilless Grower, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Savvas, D.; Passam, H.C.; Olympios, C.; Nasi, E.; Moustaka, E.; Mantzos, N.; Barouchas, P. Effects of ammonium nitrogen on lettuce grown on pumice in a closed hydroponic system. HortScience 2006, 41, 1667–1673. [Google Scholar]
- Sonneveld, C. Composition of nutrient solutions. In Hydroponic Production of Vegetables and Ornamentals; Embryo Publisher: Athens, Greece, 2002; pp. 179–210. [Google Scholar]
- Polomski, R.F. South Carolina Master Gardener Training Manual. Master’s Thesis, Clemson University, Clemson, SC, USA, 2007. [Google Scholar]
- Lucas, R.E.; Davis, J.F. Relationships between pH values of organic soils and availabilities of 12 plant nutrients. Soil Sci. 1961, 92, 177–182. [Google Scholar] [CrossRef]
- Rakocy, J.E.; Shultz, R.C.; Bailey, D.S.; Thoman, E.S. Aquaponic production of tilapia and basil: Comparing a batch and staggered cropping system. Acta Hortic. 2004, 648, 63–69. [Google Scholar] [CrossRef]
- Sonneveld, C.; Voogt, W. Nutrient Management in Substrate Systems. In Plant Nutrition of Greenhouse Crops; Springer: Houten, The Netherlands, 2009; pp. 277–312. [Google Scholar]
- Licamele, J.D. Biomass Production and Nutrient Dynamics in an Aquaponics System. Ph.D. Thesis, The University of Arizona, Tucson, AZ, USA, 2009; p. 172. [Google Scholar]
- Pantanella, E.; Cardarelli, M.; Colla, G.; Rea, E.; Marcucci, A. Aquaponics vs. hydroponics: Production and quality of lettuce crop. Acta Hortic. 2012, 927, 887–893. [Google Scholar] [CrossRef]
- Setting Maximum Levels for Certain Contaminants in Foodstuffs; Commission Regulation (EC) No. 1881/2006; The Commission of the European Communities: Brussel, Belgium, December 2006; pp. 5–24.
- Olsen, C. The significance of concentration on the rate of ion absorption by higher plants in water culture. Physiol. Plant 1950, 3, 152–164. [Google Scholar] [CrossRef]
- Santos, B.M.; Dusky, J.A.; Stall, W.M.; Bewick, T.A.; Shilling, D.G.; Gilreath, J.P.; Santos, B.M.; Stall, W.M.; Bewick, T.A.; Shilling, D.G. Phosphorus absorption in lettuce, smooth pigweed (Amaranthus hybridus), and common purslane (Portulaca oleracea) mixtures. Weed Sci. 2004, 52, 389–394. [Google Scholar] [CrossRef]
- Clement, C.R.; Hopper, M.J.; Jones, L.H.P. The uptake of nitrate by Lolium perenne from flowing nutrient solution I. Effect of NO3− concentration. J. Exp. Bot. 1978, 29, 453–464. [Google Scholar] [CrossRef]
- Edwards, J.H.; Barber, S.A. Nitrogen uptake characteristics of corn roots at low N concentration as influenced by plant age. Agron. J. 1976, 68, 17–19. [Google Scholar] [CrossRef]
- Warncke, D.D.; Barber, S.A. Nitrate uptake effectiveness of four plant species. J. Environ. Qual. 1974, 3, 28–30. [Google Scholar] [CrossRef]
- Letey, J.; Jarrell, W.M.; Valoras, N. Nitrogen and water uptake patterns and growth of plants at various minimum solution nitrate concentrations. J. Plant Nutr. 1982, 5, 73–89. [Google Scholar] [CrossRef]
- Burns, I.G.; Zhang, K.; Turner, M.K.; Edmondson, R. Iso-Osmotic Regulation of Nitrate Accumulation in Lettuce. J. Plant Nutr. 2010, 34, 283–313. [Google Scholar] [CrossRef]
- Mou, B. Lettuce. In Vegetables I; Springer: New York, NY, USA, 2008; Volumn 1, pp. 75–116. [Google Scholar]
- Waterer, D.; Bertelsen, S. Evaluation of Bibb (Butterhead) Lettuce for Hydroponic Production in Saskatchewan Greenhouses; University of Saskatchewan: Saskatoon, SK, Canada, 2014. [Google Scholar]
- Hambly, A.C.; Arvin, E.; Pedersen, L.-F.; Pedersen, P.B.; Seredyńska-Sobecka, B.; Stedmon, C.A. Characterising organic matter in recirculating aquaculture systems with fluorescence EEM spectroscopy. Water Res. 2015, 83, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Canellas, L.P.; Spaccini, R.; Piccolo, A.; Dobbss, L.B.; Okorokova-Façanha, A.L.; de Araujo Santos, G.; Olivares, F.L.; Façanha, A.R. Relationships Between Chemical Characteristics and Root Growth Promotion of Humic Acids Isolated From Brazilian Oxisols. Soil Sci. 2009, 174, 611–620. [Google Scholar] [CrossRef]
- Mylonas, V.A.; McCants, C.B. Effects of humic and fulvic acids on growth of tobacco I. Root initiation and elongation. Plant Soil 1980, 54, 485–490. [Google Scholar] [CrossRef]
- Pingel, U. The Influence of Phenolic Promotors and Inhibitors of IAA-Oxidase-Activity on the Induction of Adventitious Roots in Tradescantia albiflora. Z. Pflanzenphysiol. 1976, 79, 109–120. [Google Scholar] [CrossRef]
- Wilson, P.J.; Van Staden, J. Rhizocaline, rooting co-factors, and the concept of promotors and inhibitors of adventitius rooting—A review. Ann. Bot. 1990, 66, 479–490. [Google Scholar]
- Haghighi, M.; Kafi, M.; Fang, P. Photosynthetic Activity and N Metabolism of Lettuce as Affected by Humic Acid. Int. J. Veg. Sci. 2012, 18, 182–189. [Google Scholar] [CrossRef]
- Ruzzi, M.; Aroca, R. Plant growth-promoting rhizobacteria act as biostimulants in horticulture. Sci. Hortic. (Amsterdam) 2015, 196, 124–134. [Google Scholar] [CrossRef]
- Mangmang, J.S.; Deaker, R.; Rogers, G. Response of lettuce seedlings fertilized with fish effluent to Azospirillum brasilense inoculation. Biol. Agric. Hortic. 2014, 31, 61–71. [Google Scholar] [CrossRef]
- Vermeulen, N.V.J. Recirculation Aquaculture System (RAS) with Tilapia in a Hydroponic System with Tomatoes. Acta Hortic. 2012, 927. [Google Scholar] [CrossRef]
- Ulrich, A.E.; Frossard, E. On the history of a reoccurring concept: Phosphorus scarcity. Sci. Total Environ. 2014, 490, 694–707. [Google Scholar] [CrossRef] [PubMed]
| System | pH | EC | NO3−-N | NH4+-N | PO43−-P | K+ | Ca2+ | Mg2+ | SO42−-S | Fe2+ |
|---|---|---|---|---|---|---|---|---|---|---|
| mS/cm | mg/L | |||||||||
| Hydroponics (NFT) [6] | 5.5–5.8 | 1.5–2.0 | 165 | 25 | 50 | 210 | 200 | 40 | 113 | 5 |
| Aquaponics (UVI) [13] | 7.0–7.6 | 0.7–0.8 | 42.2 | 2.2 | 8.2 | 44.9 | 11.9 | 6.5 | 15 | 2.5 |
| Treatment 1 | (N) 2 | Shoot Fresh Weight (g/Plant) 3 | Root Fresh Weight (g/Plant) | Log10 (Shoot:Root) |
|---|---|---|---|---|
| Trial 1 | ||||
| CAP | 26 | 136.28 a | 4.86 a | 1.47 a |
| HP | 26 | 98.17 b | 3.58 b | 1.47 a |
| AP | 25 | 80.55 b | 5.80 a | 1.14 b |
| Significance | *** 4 | * | *** | |
| Trial 2 | ||||
| CAP | 24 | 55.05 a | 1.71 a | 1.52 a |
| HP | 20 | 39.64 b | 1.08 b | 1.53 a |
| AP | 25 | 35.72 b | 1.52 a | 1.39 b |
| Significance | ** | ** | ** | |
| pH 1 | EC (μS/cm) | DO (mg/L) | Water T (°C) | Air T (°C) | Air RH (%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CAP | HP | AP | CAP | HP | AP | CAP | HP | AP | CAP | HP | AP | GH | GH | ||
| Trial 1 | Mean | 5.59 | 5.73 | 7.32 | 2606 | 2453 | 823 | - 4 | - | - | 20.01 | 21.07 | 19.60 | 22.84 | 58.21 |
| SD 2 | 0.69 | 0.45 | 0.50 | 297 | 206 | 163 | - | - | - | 1.46 | 1.28 | 1.43 | 3.78 | 14.69 | |
| (N) 3 | 21 | 14 | 18 | 7 | 7 | 9 | - | - | - | 10 | 10 | 10 | 4461 | 4461 | |
| Min | 4.30 | 4.76 | 6.50 | 2236 | 2189 | 630 | - | - | - | 17.50 | 18.60 | 17.20 | 15.60 | 27.20 | |
| Max | 7.55 | 6.56 | 8.20 | 2945 | 2710 | 1014 | - | - | - | 22.30 | 22.50 | 21.40 | 35.60 | 86.90 | |
| Trial 2 | Mean | 5.87 | 5.77 | 7.50 | 2493 | 2418 | 642 | 7.51 | 7.14 | 7.36 | 20.68 | 20.96 | 22.28 | 22.15 | 71.29 |
| SD | 0.43 | 0.34 | 0.25 | 116 | 140 | 48 | 0.34 | 0.53 | 0.32 | 1.39 | 1.26 | 0.96 | 2.58 | 10.26 | |
| (N) | 19 | 20 | 17 | 9 | 9 | 16 | 10 | 10 | 10 | 15 | 16 | 15 | 1162 | 1162 | |
| Min | 5.24 | 5.32 | 7.10 | 2318 | 2237 | 567 | 6.92 | 6.12 | 6.91 | 19.10 | 19.50 | 20.90 | 18.50 | 37.90 | |
| Max | 6.84 | 6.80 | 7.94 | 2656 | 2672 | 749 | 7.91 | 7.82 | 7.92 | 24.70 | 24.80 | 25.00 | 33.20 | 88.30 | |
| Element | Treatment 1 | (N) 2 | Average | SD 3 | Min | Max |
|---|---|---|---|---|---|---|
| NO3−-N | CAP | 6 | 215.54 | 28.13 | 164.00 | 245.80 |
| HP | 6 | 193.29 | 12.35 | 181.23 | 211.55 | |
| AP | 8 | 50.31 | 1.80 | 46.57 | 52.39 | |
| TAN | CAP | 4 | 25.79 | 3.09 | 22.83 | 29.87 |
| HP | 6 | 23.95 | 2.51 | 20.53 | 26.67 | |
| AP | 8 | 1.82 | 1.35 | 0.25 | 3.32 | |
| PO43−-P | CAP | 6 | 52.66 | 2.42 | 50.03 | 56.27 |
| HP | 5 | 50.93 | 4.47 | 44.20 | 55.57 | |
| AP | 6 | 7.83 | 0.52 | 7.06 | 8.49 | |
| SO42−-S | CAP | 6 | 66.72 | 6.97 | 57.33 | 77.60 |
| HP | 5 | 95.36 | 4.72 | 87.77 | 99.97 | |
| AP | 8 | 10.99 | 1.17 | 9.24 | 12.30 | |
| K+ | CAP | 6 | 219.31 | 39.46 | 169.13 | 260.60 |
| HP | 5 | 242.27 | 36.69 | 212.67 | 295.90 | |
| AP | 8 | 59.51 | 7.89 | 48.87 | 73.03 | |
| Ca2+ | CAP | 6 | 175.09 | 14.87 | 154.43 | 192.63 |
| HP | 4 | 205.68 | 12.58 | 192.30 | 217.27 | |
| AP | 8 | 14.72 | 2.03 | 12.73 | 19.07 | |
| Mg2+ | CAP | 6 | 43.02 | 4.44 | 36.70 | 49.40 |
| HP | 5 | 43.11 | 3.15 | 39.13 | 45.83 | |
| AP | 8 | 7.36 | 0.64 | 6.76 | 8.56 | |
| Fe3+ | CAP | 6 | 4.40 | 0.20 | 4.19 | 4.69 |
| HP | 5 | 3.83 | 0.29 | 3.39 | 4.11 | |
| AP | 8 | 3.47 | 1.05 | 1.58 | 4.33 | |
| B3+ | CAP | 6 | 0.59 | 0.03 | 0.54 | 0.63 |
| HP | 5 | 0.51 | 0.08 | 0.37 | 0.59 | |
| AP | 8 | 0.47 | 0.13 | 0.24 | 0.60 | |
| Cu+ | CAP | 6 | 0.12 | 0.01 | 0.11 | 0.13 |
| HP | 5 | 0.09 | 0.01 | 0.07 | 0.11 | |
| AP | 8 | 0.09 | 0.03 | 0.05 | 0.12 | |
| Mn2+ | CAP | 6 | 0.66 | 0.06 | 0.58 | 0.73 |
| HP | 5 | 0.64 | 0.10 | 0.48 | 0.75 | |
| AP | 4 | 0.50 | 0.12 | 0.32 | 0.60 | |
| Mo+ | CAP | 6 | 0.33 | 0.02 | 0.29 | 0.35 |
| HP | 5 | 0.32 | 0.04 | 0.25 | 0.36 | |
| AP | 8 | 0.32 | 0.10 | 0.14 | 0.41 | |
| Zn2+ | CAP | 6 | 0.16 | 0.03 | 0.11 | 0.19 |
| HP | 5 | 0.15 | 0.01 | 0.13 | 0.16 | |
| AP | 8 | 0.14 | 0.03 | 0.11 | 0.19 | |
| Na+ | CAP | 6 | 71.67 | 18.24 | 40.20 | 93.53 |
| HP | 5 | 7.95 | 4.52 | 4.22 | 13.77 | |
| AP | 8 | 49.73 | 20.98 | 5.01 | 74.37 |
| Treatment 1 | (N) 2 | P 3 | K | Ca | Mg | S | Na | Fe | B | Cu | Zn | Mn | Mo |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AP | 3 | 5.47 a (0.02) | 24.6 a (0.0) | 6.36 a (0.01) | 2.28 a (0.00) | 1.97 a (0.01) | 3.70 a (0.00) | 739 a (5) | 8.1 a (0.1) | 12.6 a (0.1) | 37.0 a (0.3) | 1343 a (3) | 26.5 a (0.1) |
| CAP | 3 | 9.25 b (0.01) | 29.8 b (0.1) | 11.3 b (0.0) | 3.36 b (0.01) | 2.75 b (0.01) | 2.80 b (0.01) | 935 b (4) | 19.4 b (0.1) | 20.2 b (0.2) | 69.1 b (0.8) | 208 b (2) | 19.8 b (0.3) |
| HP | 3 | 8.56 c (0.02) | 24.7 a (0.1) | 10.8 c (0.0) | 3.00 c (0.01) | 2.56 c (0.01) | 0.40 c (0.00) | 1511 c (4) | 19.3 b (0.1) | 15.3 c (0.1) | 102 c (0) | 202 c (1) | 19.0 c (0.1) |
| Significance | *** 4 | *** | *** | *** | *** | *** | *** | *** | *** | *** | *** | *** |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delaide, B.; Goddek, S.; Gott, J.; Soyeurt, H.; Jijakli, M.H. Lettuce (Lactuca sativa L. var. Sucrine) Growth Performance in Complemented Aquaponic Solution Outperforms Hydroponics. Water 2016, 8, 467. https://doi.org/10.3390/w8100467
Delaide B, Goddek S, Gott J, Soyeurt H, Jijakli MH. Lettuce (Lactuca sativa L. var. Sucrine) Growth Performance in Complemented Aquaponic Solution Outperforms Hydroponics. Water. 2016; 8(10):467. https://doi.org/10.3390/w8100467
Chicago/Turabian StyleDelaide, Boris, Simon Goddek, James Gott, Hélène Soyeurt, and M. Haissam Jijakli. 2016. "Lettuce (Lactuca sativa L. var. Sucrine) Growth Performance in Complemented Aquaponic Solution Outperforms Hydroponics" Water 8, no. 10: 467. https://doi.org/10.3390/w8100467
APA StyleDelaide, B., Goddek, S., Gott, J., Soyeurt, H., & Jijakli, M. H. (2016). Lettuce (Lactuca sativa L. var. Sucrine) Growth Performance in Complemented Aquaponic Solution Outperforms Hydroponics. Water, 8(10), 467. https://doi.org/10.3390/w8100467







