Assessment of Groundwater Chemistry and Status in a Heavily Used Semi-Arid Region with Multivariate Statistical Analysis
Abstract
:1. Introduction
2. Study area
2.1. Location
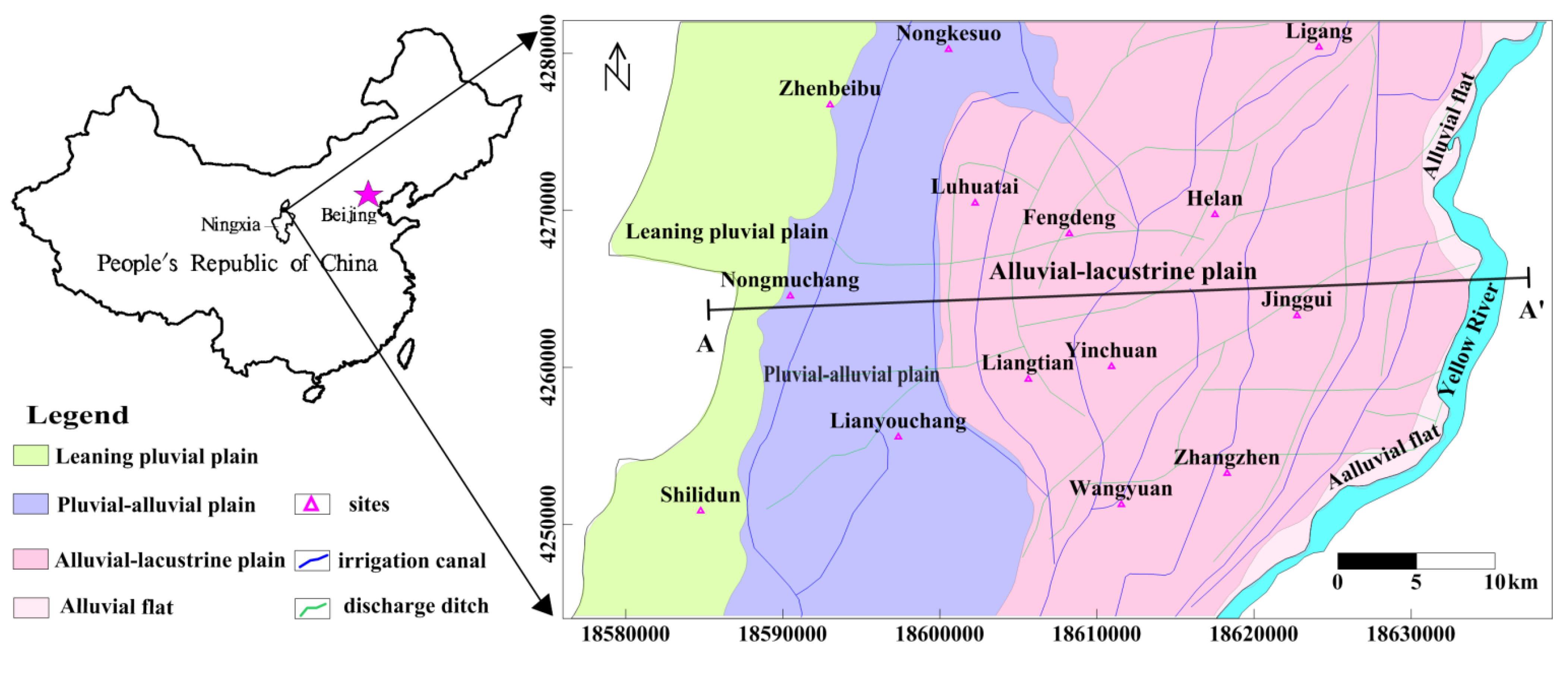
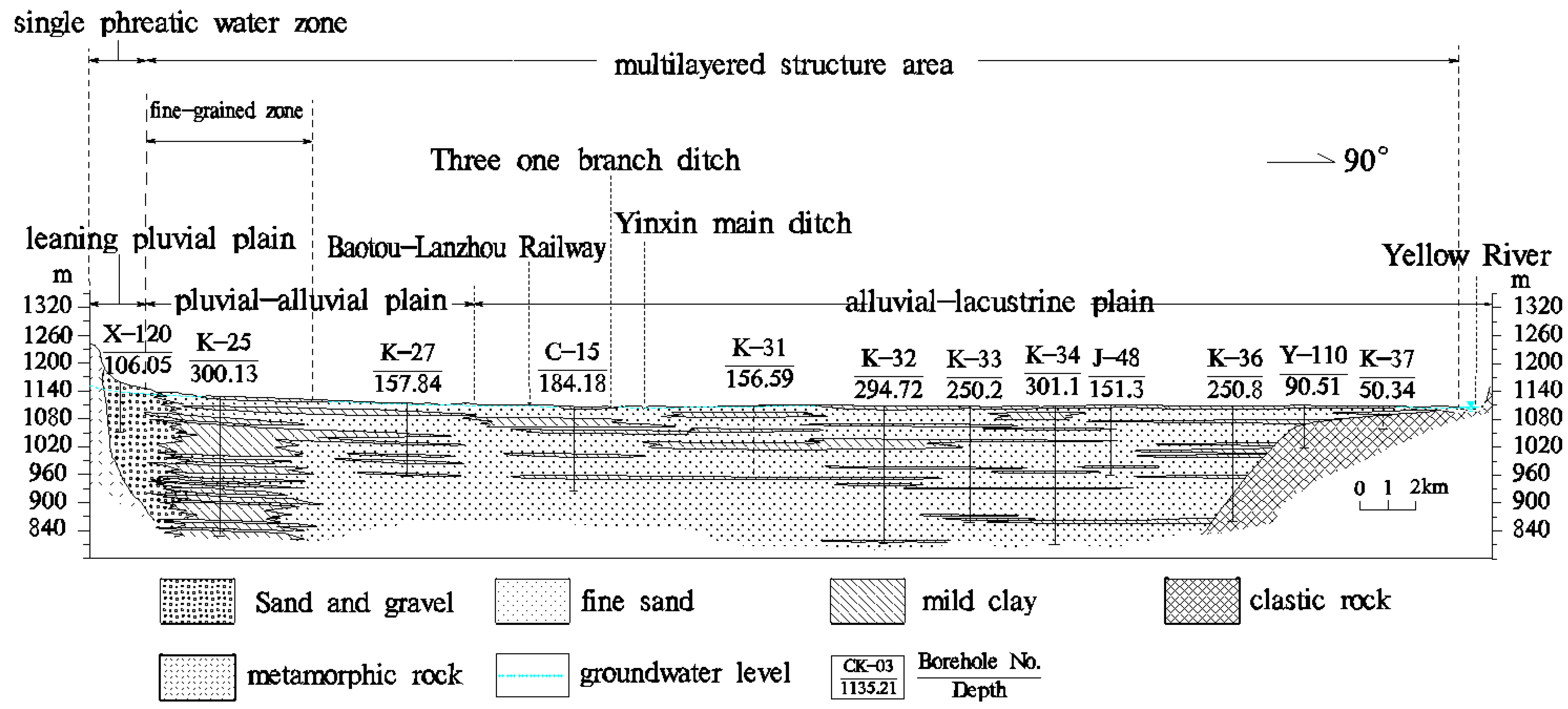
2.2. Hydrogeology

3. Materials and Methods
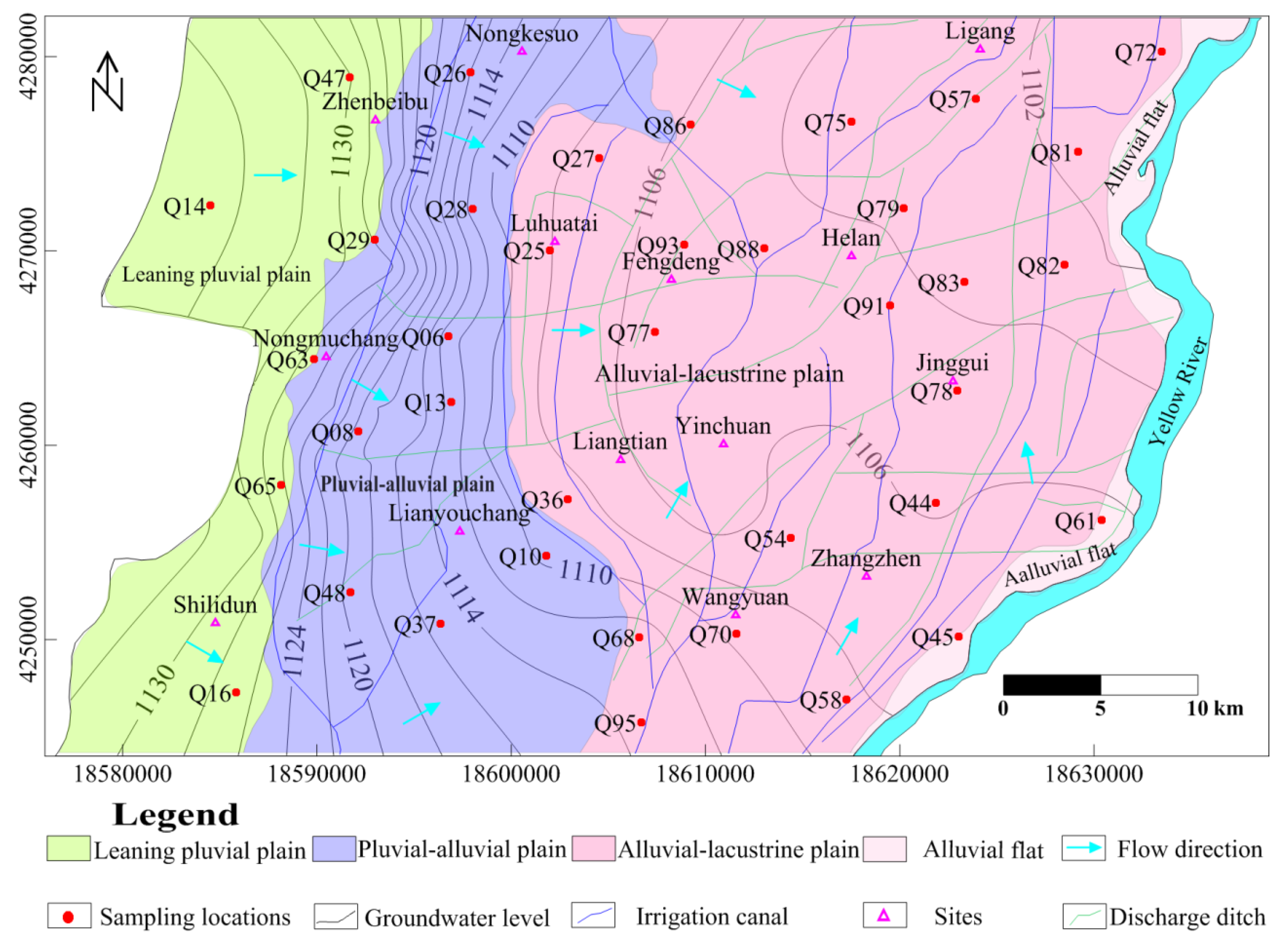
3.1. Sampling Locations and Analytical Procedures
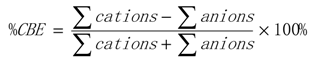
3.2. Multivariate Statistical Analysis
4. Results and Discussion
4.1. Hydrochemical Characteristics
| Parameters | Phreatic water | Yellow river | National standard | ||||
|---|---|---|---|---|---|---|---|
| Category | Minimum | Maximum | Mean | SD | CV | / | / |
| pH | 7.71 | 8.38 | 8.08 | 0.11 | 0.01 | 8.10 | 6.5–8.5 |
| TH | 146 | 2845 | 509 | 417 | 0.8 | 245 | 450 |
| TDS | 272 | 5664 | 1065 | 885 | 0.8 | 386 | 1000 |
| Ca2+ | 23.0 | 439.7 | 83.9 | 66.3 | 0.8 | 53.8 | _ |
| Mg2+ | 23.3 | 509 | 85.7 | 81.4 | 0.9 | 27.9 | _ |
| K+ | 1.5 | 17.0 | 4.9 | 3.4 | 0.7 | 3.0 | _ |
| Na+ | 24.0 | 860 | 181 | 167 | 0.9 | 43.0 | 200 |
| Cl- | 27.6 | 2592 | 222 | 406 | 1.8 | 55.2 | 250 |
| SO42+ | 35.2 | 986 | 214 | 184 | 0.9 | 83.6 | 250 |
| HCO3- | 220 | 1106 | 501 | 210 | 0.4 | 207 | _ |
| NO3- | 0.0 | 21 | 2.1 | 4.4 | 2.0 | _ | 20 |
| NH4+ | 0.0 | 1.0 | 0.1 | 0.2 | 4.1 | _ | 0.20 |
| F- | 0.1 | 1.2 | 0.3 | 0.2 | 0.9 | _ | 1 |
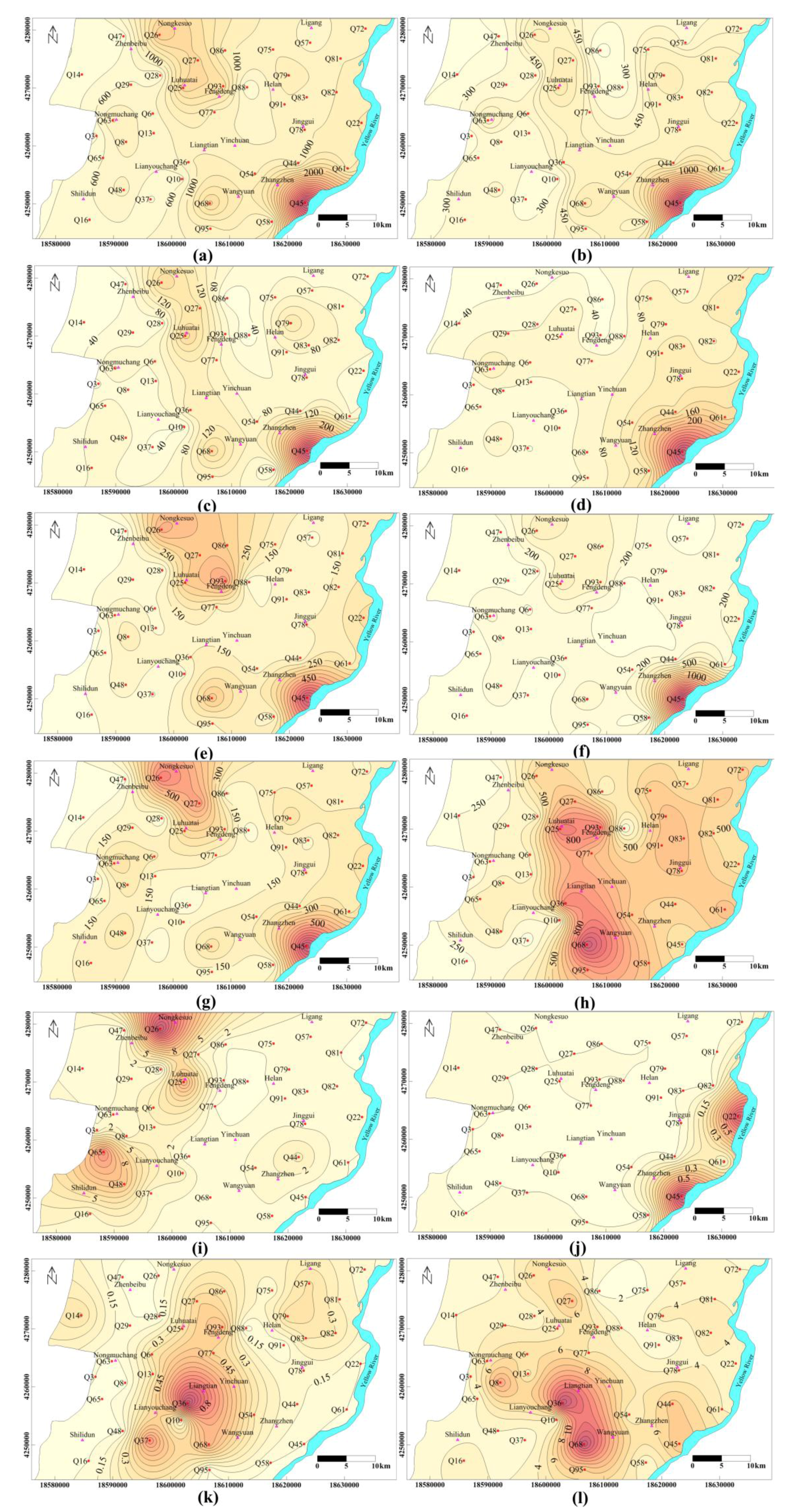
4.2. Correlation among Parameters
| Catalog | pH | TH | TDS | Ca2+ | Mg2+ | K+ | Na+ | Cl− | SO42+ | HCO3− | NO3− | NH4+ | F− |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pH | 1 | ||||||||||||
| TH | −0.50 | 1 | |||||||||||
| TDS | −0.57 | 0.935 | 1 | ||||||||||
| Ca2+ | −0.41 | 0.928 | 0.792 | 1 | |||||||||
| Mg2+ | −0.51 | 0.953 | 0.952 | 0.771 | 1 | ||||||||
| K+ | 0.04 | 0.293 | 0.345 | 0.106 | 0.415 | 1 | |||||||
| Na+ | −0.54 | 0.742 | 0.929 | 0.568 | 0.805 | 0.343 | 1 | ||||||
| Cl− | −0.62 | 0.941 | 0.955 | 0.846 | 0.919 | 0.216 | 0.839 | 1 | |||||
| SO42− | −0.58 | 0.809 | 0.907 | 0.641 | 0.861 | 0.277 | 0.851 | 0.817 | 1 | ||||
| HCO3- | 0.17 | 0.245 | 0.347 | 0.106 | 0.332 | 0.557 | 0.438 | 0.109 | 0.227 | 1 | |||
| NO3− | −0.27 | 0.00 | 0.126 | −0.19 | 0.163 | −0.033 | 0.173 | 0.044 | 0.344 | −0.02 | 1 | ||
| NH4+ | −0.41 | 0.688 | 0.657 | 0.719 | 0.591 | 0.046 | 0.556 | 0.73 | 0.55 | −0.054 | −0.11 | 1 | |
| F− | 0.243 | −0.06 | 0.013 | −0.22 | 0.071 | 0.551 | 0.092 | −0.06 | −0.11 | 0.443 | −0.16 | −0.17 | 1 |
4.3. Principal Component Analysis (PCA)
| Variable | PC1 | PC2 | PC3 | PC4 |
|---|---|---|---|---|
| TH | 0.97 | 0.04 | −0.04 | 0.07 |
| Cl− | 0.97 | 0.06 | 0.03 | −0.13 |
| TDS | 0.97 | 0.16 | 0.15 | 0.07 |
| Mg2+ | 0.93 | 0.21 | 0.16 | 0.08 |
| Ca2+ | 0.90 | −0.18 | −0.28 | 0.06 |
| SO42+ | 0.86 | 0.05 | 0.39 | 0.04 |
| Na+ | 0.84 | 0.25 | 0.27 | 0.09 |
| NH4+ | 0.77 | −0.13 | −0.25 | −0.20 |
| pH | −0.59 | 0.04 | −0.37 | 0.57 |
| F− | −0.12 | 0.92 | −0.13 | 0.04 |
| K+ | 0.24 | 0.79 | 0.03 | 0.26 |
| NO3− | 0.02 | −0.10 | 0.94 | −0.02 |
| HCO3− | 0.21 | 0.49 | 0.09 | 0.76 |
| Eigenvalue | 6.99 | 1.89 | 1.45 | 1.06 |
| % of Variance explained | 53.7 | 14.6 | 11.2 | 8.1 |
| % Cumulative variance | 53.7 | 68.3 | 79.5 | 87.6 |
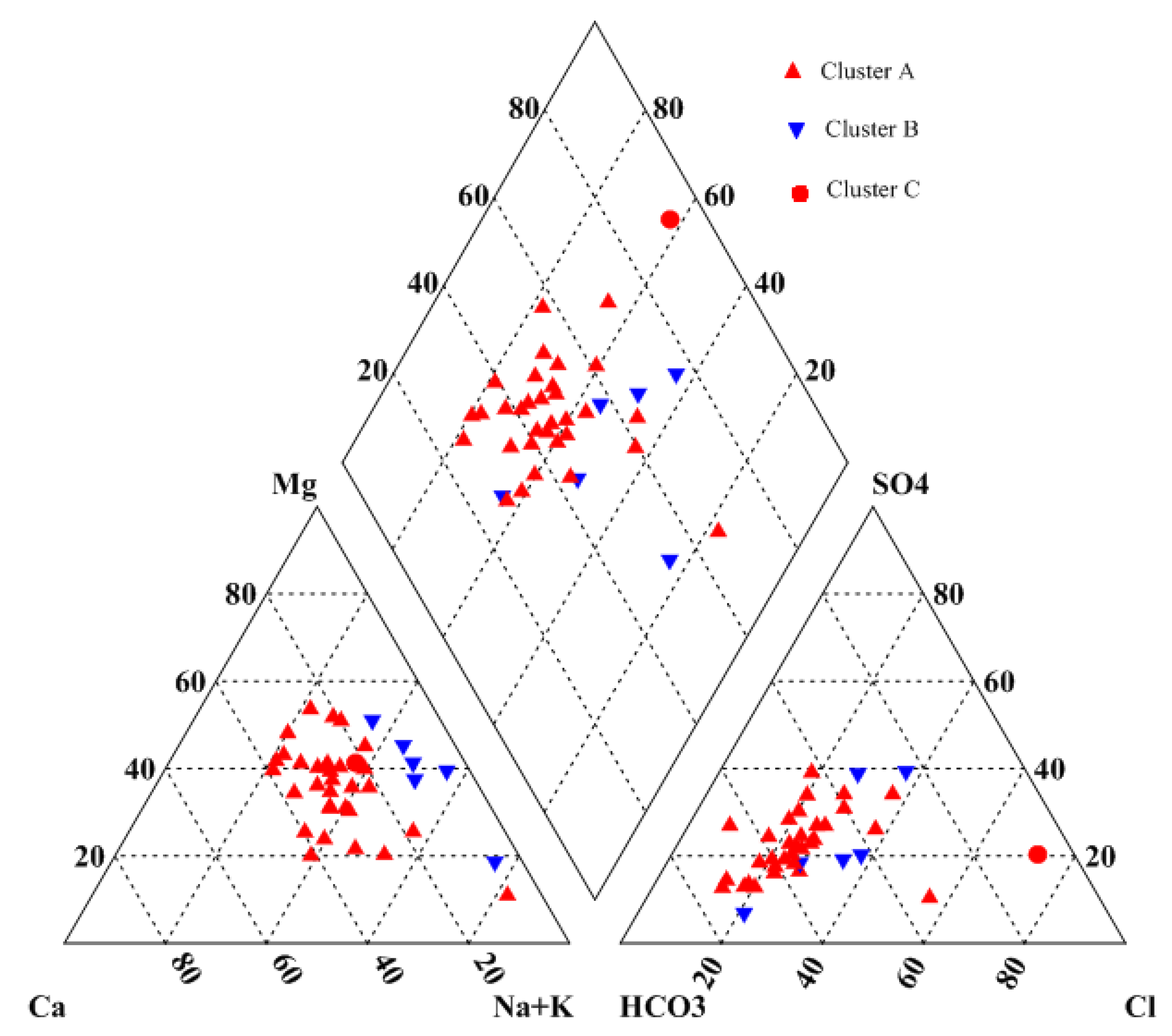
4.4. Cluster Analysis
4.4.1. Q-Mode Cluster Analysis
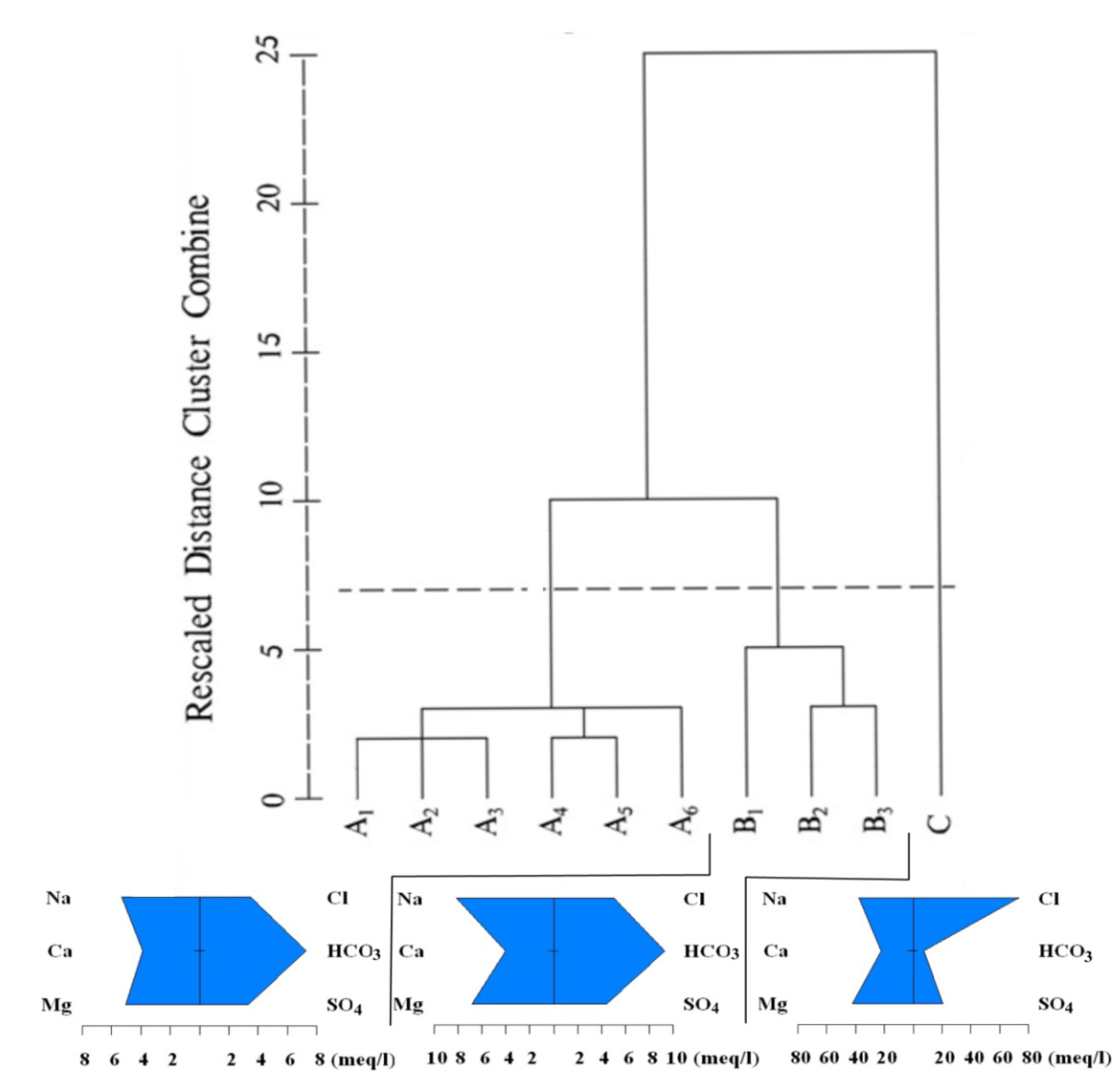
| Cluster | Samples |
|---|---|
| A1 | Q3,Q8,Q10,Q16, Q28,Q29, Q47,Q63,Q88 |
| A2 | Q48, Q65 |
| A3 | Q6, Q13, Q37, Q57, Q77, Q86 |
| A4 | Q14, Q82 |
| A5 | Q44,Q54, Q58,Q61,Q72,Q75,Q78,Q79,Q81,Q83,Q91,Q95 |
| A6 | Q22 |
| B1 | Q36, Q68, |
| B2 | Q25, Q27, Q93 |
| B3 | Q26 |
| C | Q45 |
| Index | Cluster A | Cluster B | Cluster C |
|---|---|---|---|
| pH | 8.10 | 8.06 | 7.71 |
| TH | 417 | 610 | 2845 |
| TDS | 790 | 1765 | 5664 |
| Ca2+ | 78.0 | 56.0 | 439.7 |
| Mg2+ | 60.9 | 147.4 | 508.7 |
| K+ | 3.9 | 9.6 | 8.0 |
| Na+ | 122 | 381 | 860 |
| Cl− | 123 | 351 | 2592 |
| SO42+ | 159 | 378 | 986 |
| HCO3- | 444 | 815 | 440 |
| NO3− | 1.53 | 5.76 | 0 |
| NH4+ | 0.03 | 0.00 | 1.01 |
| F− | 0.21 | 0.55 | 0.10 |
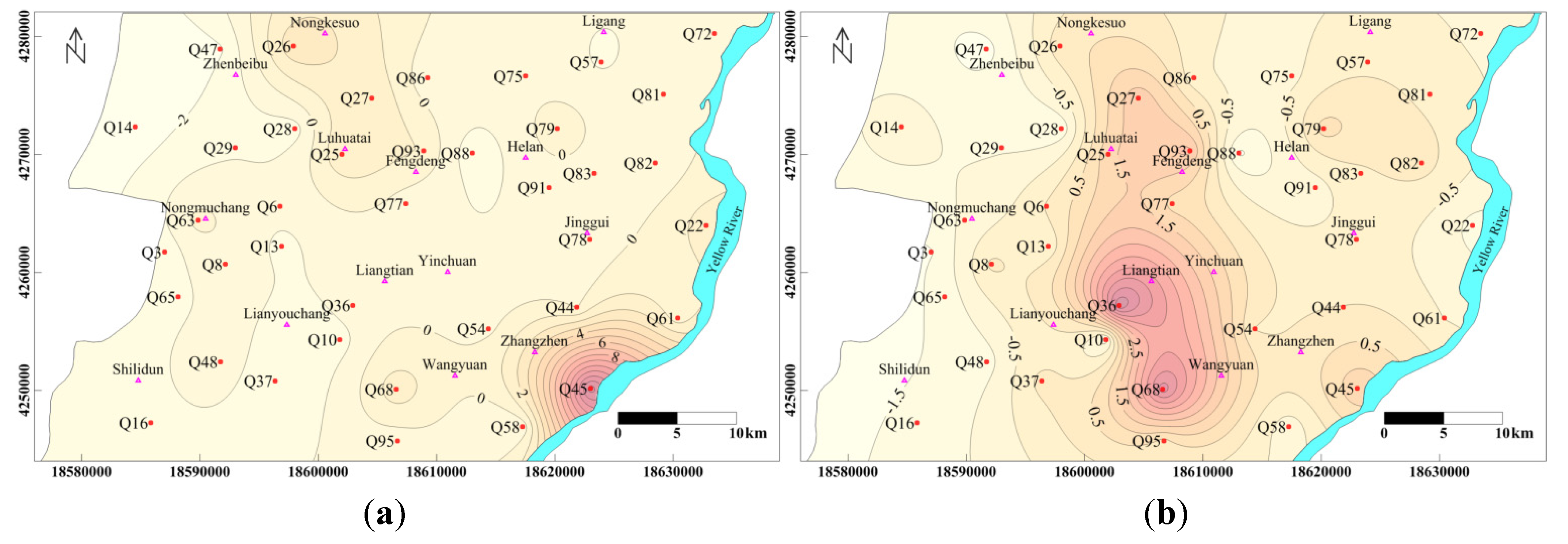
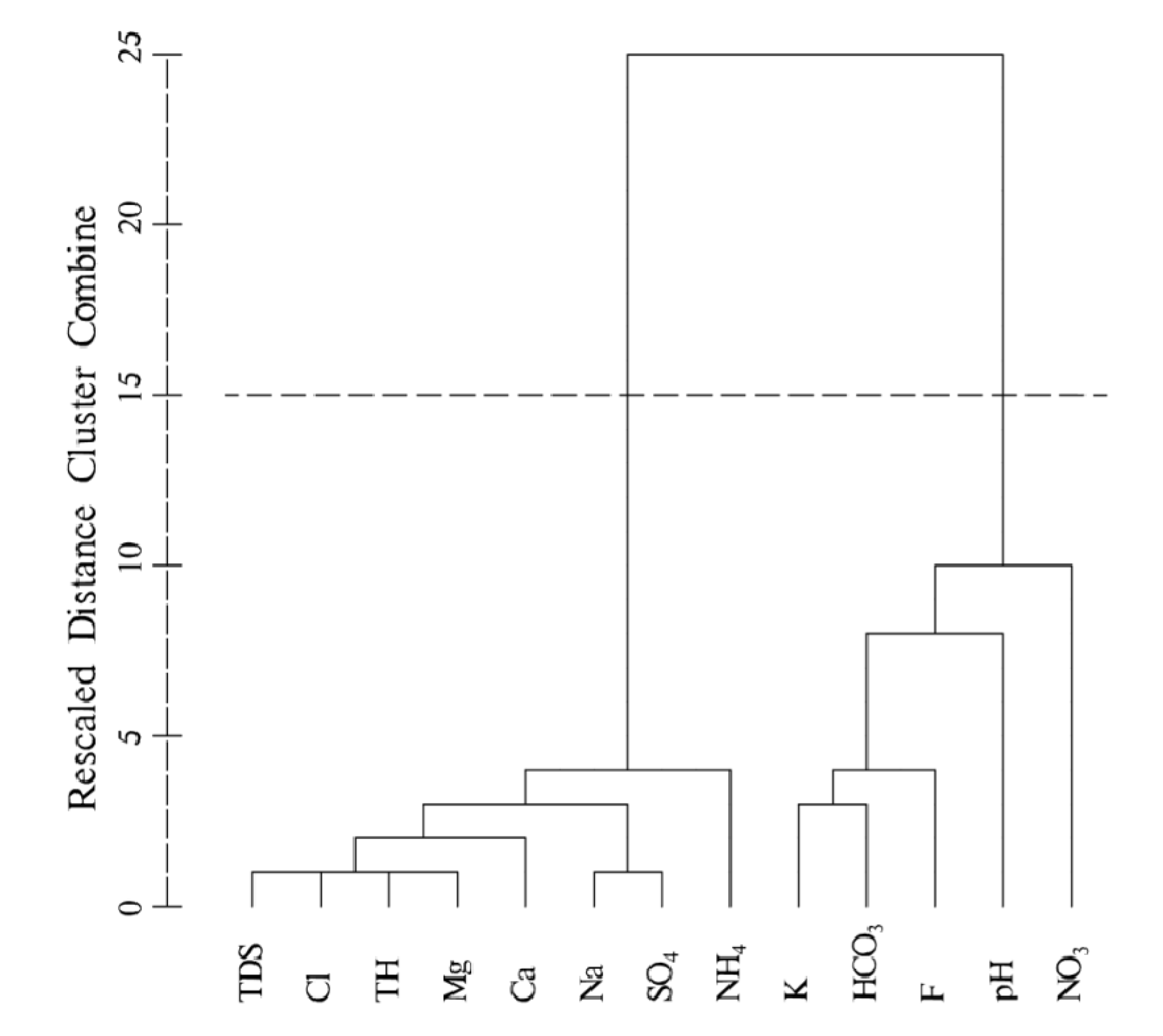
4.4.2. R-Mode Cluster Analysis
5. Conclusions
- The quality of phreatic water varied greatly across the Yinchuan region and its composition changed greatly in alluvial-lacustrine plain, the east of study area. Changes of TDS and concentrations of major ions generally increased along the hydraulic flow path, from west to east. Samples with TDS < 1 g/L were mainly from the west part of the plain, while samples in the central part of the region usually had TDS of 1 to 1.5 g/L. Moderate salt water with TDS > 1.5 g/L was located at three single areas: west of Wangyuan Town, near Luhuatai and Nongkesuo in the north, and east of Zhangzheng, along the Yellow River (Figure 5a).
- The quality of phreatic water in study area is mainly controlled by the strong evaporation effect caused by the dry climate, dissolution of carbonate minerals and those containing F− and K−, and human activities including the treatment of industrial and municipal wastewater, the discharge of domestic sewage, and utilization of chemical fertilizer. This is confirmed by the results of correlation analysis and principal component analysis. The main hydrogeochemical processes include the strong evaporation effect caused by the dry climate, the dissolution/precipitation of limestone, dolomite and calcite. Cation exchange and dissolution of other minerals, such as fluorite, feldspar and mica, also took place. Mixing with local irrigation water can also explain some of the observed variation of ions.
- Two types of clustering analysis helped to further characterize the water quality. CA in Q-mode identified three clusters, representing a significant gradient from A to C of increasing hydrochemical measures (except Ca2+ and HCO3−), from fresh water to almost saline water, which is consisted with the general flow direction (from west to east). R-mode CA revealed two distinct clusters of variables, related to two distinct sets of factors that are likely influencing Yinchuan region’s phreatic water, which may include natural processes pollution, and/or other human impacts.
- In order to improve and protect the quality of phreatic water, greater importance should be attached to water resources management and planning in the study area. In leaning pluvial plain, as TDS < 1 g/L and the concentrations of other ions are small, groundwater depth is much larger than 3 m (the extinction evaporation depth), so the evaporation effect is weak and the quality is good for drinking, agricultural irrigation and industry. In addition, the area is the recharge zone of groundwater for the whole plain, so water resources management shall be strictly performed in this area for the pollution from industrial enterprises and livestock breeding. However, in other parts, the water qualities of phreatic water have been polluted more or less due to the lessening groundwater depth and extensive influences of human activities. Therefore, the best solution is reducing the amount of evaporation by increasing pumping groundwater and decreasing canal irrigation water in irrigation period in order to reduce groundwater level into a more proper level.
Acknowledgments
Author Contributions
Appendix
| Samples | pH | TH | TDS | Ca2+ | Mg2+ | K+ | Na+ | Cl− | SO42− | HCO3− | NO3− | NH4+ | F− |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Q3 | 8.0 | 254.5 | 368 | 49.9 | 34.9 | 3.00 | 31.0 | 51.8 | 58.2 | 245.1 | 1.24 | 0.00 | 0.10 |
| Q6 | 8.1 | 405.2 | 727 | 71.0 | 64.0 | 6.00 | 89.2 | 89.8 | 209.6 | 352.0 | 3.62 | 0.00 | 0.36 |
| Q8 | 8.0 | 317.2 | 933 | 55.7 | 50.1 | 10.00 | 197.0 | 158.8 | 235.4 | 377.1 | 1.47 | 0.00 | 0.08 |
| Q10 | 8.1 | 260.3 | 528 | 51.8 | 34.9 | 3.00 | 86.0 | 62.1 | 127.1 | 289.1 | 2.49 | 0.00 | 0.16 |
| Q13 | 8.1 | 334.7 | 621 | 55.7 | 55.9 | 4.50 | 86.0 | 103.6 | 129.6 | 333.1 | 2.71 | 0.00 | 0.30 |
| Q14 | 8.4 | 197.8 | 272 | 40.3 | 25.6 | 3.00 | 24.0 | 27.6 | 36.4 | 251.4 | 0.57 | 0.03 | 0.36 |
| Q16 | 8.1 | 314.1 | 517 | 44.2 | 60.5 | 3.50 | 44.0 | 58.7 | 172.9 | 232.6 | 1.70 | 0.00 | 0.10 |
| Q22 | 8.1 | 513.2 | 1236 | 115.2 | 55.9 | 3.00 | 268.0 | 281.8 | 272.1 | 465.1 | 0.34 | 0.93 | 0.10 |
| Q25 | 8.0 | 789.3 | 1905 | 71.0 | 192.1 | 4.50 | 356.8 | 466.0 | 337.0 | 892.5 | 11.30 | 0.00 | 0.36 |
| Q26 | 7.9 | 635.7 | 2200 | 32.6 | 179.3 | 6.00 | 480.0 | 490.2 | 701.9 | 534.2 | 21.47 | 0.00 | 0.10 |
| Q27 | 8.1 | 661.1 | 1850 | 74.9 | 145.5 | 8.00 | 370.0 | 314.1 | 592.4 | 647.4 | 1.81 | 0.00 | 0.44 |
| Q28 | 8.0 | 305.5 | 439 | 57.6 | 44.2 | 3.00 | 40.0 | 58.7 | 53.1 | 333.1 | 0.00 | 0.00 | 0.10 |
| Q29 | 8.1 | 349.2 | 631 | 61.4 | 54.7 | 4.00 | 83.0 | 79.4 | 164.7 | 333.1 | 1.70 | 0.00 | 0.11 |
| Q36 | 8.2 | 515.2 | 985 | 51.8 | 119.9 | 17.00 | 149.0 | 145.0 | 62.6 | 842.2 | 0.00 | 0.00 | 1.20 |
| Q37 | 8.0 | 213.0 | 359 | 38.4 | 32.6 | 2.00 | 44.0 | 62.1 | 53.0 | 220.0 | 1.47 | 0.00 | 0.76 |
| Q44 | 8.1 | 600.2 | 951 | 126.7 | 73.3 | 7.50 | 107.0 | 141.5 | 205.2 | 534.2 | 3.39 | 0.00 | 0.16 |
| Q45 | 7.7 | 2845.0 | 5664 | 439.7 | 508.7 | 8.00 | 860.0 | 2592.5 | 986.3 | 440.0 | 0.00 | 1.01 | 0.10 |
| Q47 | 8.1 | 201.5 | 290 | 34.4 | 32.6 | 2.50 | 24.0 | 34.5 | 35.2 | 226.3 | 0.36 | 0.00 | 0.08 |
| Q48 | 8.0 | 436.4 | 896 | 76.8 | 68.7 | 3.00 | 138.0 | 148.4 | 256.6 | 358.3 | 8.50 | 0.00 | 0.10 |
| Q54 | 8.1 | 571.5 | 969 | 111.4 | 79.2 | 4.00 | 128.0 | 155.3 | 187.6 | 565.7 | 1.47 | 0.00 | 0.30 |
| Q57 | 8.1 | 380.9 | 673 | 65.3 | 61.7 | 3.50 | 86.0 | 34.5 | 159.1 | 471.4 | 0.23 | 0.00 | 0.36 |
| Q58 | 8.1 | 474.6 | 810 | 121.0 | 37.3 | 3.00 | 130.4 | 96.7 | 100.5 | 603.4 | 0.90 | 0.00 | 0.10 |
| Q61 | 8.1 | 634.4 | 1157 | 149.8 | 61.7 | 4.00 | 185.0 | 189.9 | 231.6 | 628.5 | 0.00 | 0.08 | 0.10 |
| Q63 | 8.0 | 547.5 | 1025 | 99.8 | 82.6 | 5.00 | 138.0 | 231.3 | 295.8 | 308.0 | 1.36 | 0.00 | 0.08 |
| Q65 | 8.1 | 332.8 | 600 | 42.2 | 68.7 | 3.00 | 72.0 | 65.6 | 130.3 | 383.4 | 13.56 | 0.00 | 0.10 |
| Q68 | 8.2 | 753.2 | 1886 | 73.0 | 178.1 | 17.00 | 388.0 | 310.7 | 290.1 | 1106.2 | 0.00 | 0.00 | 0.64 |
| Q72 | 8.1 | 438.0 | 915 | 101.8 | 44.2 | 3.00 | 174.0 | 145.0 | 146.4 | 553.1 | 0.00 | 0.00 | 0.16 |
| Q75 | 8.1 | 511.3 | 988 | 101.8 | 68.7 | 2.00 | 164.0 | 138.1 | 199.5 | 590.8 | 0.11 | 0.00 | 0.16 |
| Q77 | 8.1 | 462.3 | 934 | 69.1 | 85.0 | 5.00 | 153.0 | 110.5 | 156.3 | 653.6 | 0.00 | 0.00 | 0.44 |
| Q78 | 8.2 | 555.1 | 1002 | 115.2 | 69.8 | 4.00 | 153.0 | 151.9 | 181.3 | 609.7 | 0.68 | 0.00 | 0.16 |
| Q79 | 8.0 | 706.0 | 1217 | 92.2 | 143.2 | 4.50 | 138.0 | 200.2 | 294.9 | 641.1 | 0.57 | 0.00 | 0.40 |
| Q81 | 8.1 | 558.6 | 988 | 115.2 | 71.0 | 4.00 | 153.0 | 145.0 | 188.2 | 565.7 | 0.00 | 0.00 | 0.24 |
| Q82 | 8.4 | 467.9 | 1003 | 76.8 | 79.2 | 5.00 | 172.0 | 169.2 | 233.6 | 490.2 | 0.14 | 0.00 | 0.30 |
| Q83 | 8.1 | 577.5 | 894 | 107.5 | 85.0 | 3.00 | 98.4 | 131.2 | 132.4 | 615.9 | 0.00 | 0.00 | 0.30 |
| Q86 | 8.0 | 145.9 | 1048 | 23.0 | 25.6 | 1.50 | 344.0 | 359.0 | 95.9 | 364.5 | 0.23 | 0.00 | 0.36 |
| Q88 | 8.0 | 242.6 | 397 | 57.6 | 23.3 | 3.00 | 57.0 | 55.2 | 65.2 | 238.8 | 0.00 | 0.00 | 0.08 |
| Q91 | 8.2 | 547.3 | 976 | 105.6 | 76.8 | 2.00 | 144.0 | 127.7 | 168.6 | 647.4 | 0.00 | 0.00 | 0.11 |
| Q93 | 8.0 | 307.4 | 1765 | 32.6 | 69.8 | 5.00 | 540.0 | 379.7 | 282.6 | 867.3 | 0.00 | 0.00 | 0.56 |
| Q95 | 8.1 | 477.7 | 908 | 61.4 | 97.8 | 7.00 | 145.0 | 82.9 | 122.3 | 735.4 | 0.00 | 0.00 | 0.10 |
Conflicts of Interest
References
- Brunke, M.; Gonser, T. The ecological significance of exchange processes between rivers and groundwater. Freshw. Biol. 1997, 37, 1–33. [Google Scholar]
- Dillon, P.J.; Kirchner, W.B. The effects of geology and land use on the export of phosphorous from watersheds. Water Res. 1975, 9, 125–148. [Google Scholar] [CrossRef]
- Sophocleous, M. Interactions between groundwater and surface water: The state of the science. Hydrogeology 2002, 10, 52–67. [Google Scholar] [CrossRef]
- Vega, M.; Pardo, R.; Barrado, E.; Deban, L. Assessment of seasonal and polluting effects on the quality of river water by exploratory data analysis. Water Res. 1998, 32, 3581–3592. [Google Scholar] [CrossRef]
- Qian, H.; Li, P.Y. Hydrochemical characteristics of groundwater in Yinchuan plain and their control factors. Asian J. Chem. 2011, 23, 2927–2938. [Google Scholar]
- Jin, X.; Wan, L.; Zhang, Y.; Xue, Z.; Yin, Y. A study of the relationship between vegetation growth and groundwater in the Yinchuan Plain. Earth Sci. Front. 2007, 14, 197–203. [Google Scholar]
- Andradea, E.M.; Palacio, H.A.Q.; Souzab, I.H.; Leaoa, R.A.O.; Guerreiroc, M.J. Land use effects in groundwater composition of an alluvial aquifer (Trussu River, Brazil) by multivariate techniques. Environ. Res. 2008, 106, 170–177. [Google Scholar] [CrossRef]
- Reisenhofer, E.; Adami, G.; Barbieri, P. Using chemical and physical parameters to define the quality of karstic freshwaters (Timavo River, Northeastern Italy): A chemometric approach. Wat. Res. 1998, 32, 1193–1203. [Google Scholar] [CrossRef]
- Qian, H.; Wu, J.; Zhou, Y.H.; Li, P.Y. Stable oxygen and hydrogen isotopes as indicators of lake water recharge and evaporation in the lakes of the Yinchuan Plain. Hydrol. Process. 2013. [Google Scholar] [CrossRef]
- Liu, P.G.; Fan, S.X.; Li, X.J. The geochemical element characteristics and paleo-sedimentary environment of the Quaternary deposits in Yinchuan Basin. J. Geomech. 2000, 6, 43–50. [Google Scholar]
- Zhang, L.; Wang, L. Groundwater Resources in Ningxia; Ningxia People Publishing House: Yinchuan, China, 2003. [Google Scholar]
- Wu, X.H.; Qian, H.; Yu, D.M. Investigation and Assessment of Rational Allocation of Groundwater Resources in the Yinchuan Plain; Geology Publishing House: Beijing, China, 2008. [Google Scholar]
- Lu, D.M. History of Diverting Yellow River Water for Irrigation in Ningxia; China Water Power Press: Beijing, China, 1987. [Google Scholar]
- Ministry of Health of the People's Republic of China. Standards for drinking water quality; GB 5749-2006; Standards Press of China: Beijing, China, 2007. [Google Scholar]
- Kaufman, L. Finding Groups in Data: An Introduction to Cluster Analysis; Wiley: New York, NY, USA, 1990. [Google Scholar]
- Tang, Q.Y. DPS© Data Processing System: Experimental Design, Statistical Analysis and Data Mining, 2nd ed.; Science Press: Beijing, China, 2010. (In Chinese) [Google Scholar]
- Helena, B.; Pardo, R.; Vega, M.; Barrado, E.; Fernandez, J.M.; Fernandez, L. Temporal evolution of groundwater composition in an alluvial aquifer (Pisuerga River, Spain) by principal component analysis. Water Res. 2000, 34, 807–816. [Google Scholar] [CrossRef]
- Willet, P. Similarity and Clustering in Chemical Information Systems; Research Studies Press, Wiley: New York, NY, USA, 1987. [Google Scholar]
- Li, P.Y.; Qian, H.; Wu, J.H.; Zhang, Y.Q.; Zhang, H.B. Major ion chemistry of shallow groundwater in the Dongsheng Coalfield, Ordos Basin, China. Mine Water Environ. 2013, 32, 195–206. [Google Scholar] [CrossRef]
- Brown, C. Applied Multivariate Statistics in Geohydrology and Related Science, 1st ed.; Springer: Berlin, Germany, 1998; pp. 1–248. [Google Scholar]
- Hopke, P.K.; Gladney, E.S.; Gordon, G.E.; Zoller, W.H.; Jones, A.G. The use of multivariate analysis to identify sources of selected elements in the Boston urban aerosol. Atmos. Environ. 1976, 10, 1015–1025. [Google Scholar] [CrossRef]
- Voudouris, K.; Panagopolous, A.; Koumanatakis, J. Multivariate statistical analysis in the assessment of hydrochemistry of the Northern Korinthia Prefecture alluvial system (Peloponnese, Greece). Nat. Resour. Res. 2000, 9, 135–146. [Google Scholar] [CrossRef]
- Hussein, M.T. Hydrochemical evaluation of groundwater in the Blue Nile Basin, eastern Sudan, using conventional and multivariate techniques. Hydrogeol. J. 2004, 12, 12–144. [Google Scholar] [CrossRef]
- Mahlknecht, J.; Steinich, B.; Navarro de Leon, I. Groundwater chemistry and mass transfers in the Independence aquifer, central Mexico, by using multivariate statistics and mass-balance models. Environ. Geol. 2004, 45, 781–795. [Google Scholar] [CrossRef]
- Hussain, M.; Ahmed, S.M.; Abderrahman, W. Cluster analysis and quality assessment of logged water at an irrigation project, eastern Saudi Arabia. J. Environ. Manag. 2008, 86, 297–307. [Google Scholar] [CrossRef]
- Qian, H.; Zhang, X.D.; Li, P.Y. Calculation of CaCO3 solubility (precipitability) in natural waters. Asian J. Chem. 2012, 24, 668–672. [Google Scholar]
- Li, P.Y.; Qian, H.; Wu, J.H. Hydrochemical characteristics and evolution laws of drinking groundwater in Pengyang County, Ningxia, Northwest China. E J. Chem. 2011, 8, 565–575. [Google Scholar] [CrossRef]
- Lucas, L.; Jauzein, M. Use of principal component analysis to profile temporal and spatial variations of chlorinated solvent concentration in groundwater. Environ. Pollut. 2008, 151, 205–212. [Google Scholar] [CrossRef]
- Wu, J.H.; Li, P.Y.; Qian, H.; Duan, Z.; Zhang, X.D. Using correlation and multivariate statistical analysis to identify hydrogeochemical processes affecting the major ion chemistry of waters: a case study in Laoheba phosphorite mine in Sichuan, China. Arab. J. Geosci. 2013. [Google Scholar] [CrossRef]
- Rapin, F. Anthropogenic Effects on Sediment from Bay of Nice and Villefranche-sur-Mer on the French Coast. Ph.D. Dissertation, University of Geneva, Geneva, Switzerland, 1980; p. 221. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, X.; Qian, H.; Chen, J.; Qiao, L. Assessment of Groundwater Chemistry and Status in a Heavily Used Semi-Arid Region with Multivariate Statistical Analysis. Water 2014, 6, 2212-2232. https://doi.org/10.3390/w6082212
Zhang X, Qian H, Chen J, Qiao L. Assessment of Groundwater Chemistry and Status in a Heavily Used Semi-Arid Region with Multivariate Statistical Analysis. Water. 2014; 6(8):2212-2232. https://doi.org/10.3390/w6082212
Chicago/Turabian StyleZhang, Xuedi, Hui Qian, Jie Chen, and Liang Qiao. 2014. "Assessment of Groundwater Chemistry and Status in a Heavily Used Semi-Arid Region with Multivariate Statistical Analysis" Water 6, no. 8: 2212-2232. https://doi.org/10.3390/w6082212
APA StyleZhang, X., Qian, H., Chen, J., & Qiao, L. (2014). Assessment of Groundwater Chemistry and Status in a Heavily Used Semi-Arid Region with Multivariate Statistical Analysis. Water, 6(8), 2212-2232. https://doi.org/10.3390/w6082212




