Comparing Two Operating Configurations in a Full-Scale Arsenic Removal Plant. Case Study: Guatemala
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Area
2.2. Water Quality

| Parameter | Well No. 1 | Well No. 2 | COGUANOR NTG 29 001 | |
|---|---|---|---|---|
| MAL | MPL | |||
| Physical | ||||
| True Color (Pt-Co Units) | <2.0 | 10 | 5 | 35 |
| Turbidity (NTU) | 0.09 | 20 | 5 | 15 |
| Total dissolved solids 1 | 206 | 233 | 500 | 1000 |
| Chemical | ||||
| pH | 6.61 | 7.14 | 7.0–7.5 | 6.5–8.5 |
| Alkalinity 1 | 128 | 134 | - | - |
| Hardnesstotal 1 (CaCO3) | 96.8 | 67.1 | 100 | 500 |
| Chlorides 1 | 5.54 | 3.02 | 100 | 250 |
| Sulphates 1 | 8.24 | 28 | 100 | 250 |
| Nitrates 1 | 1.023 | <0.093 | - | 10 |
| Fluorides 1 | 0.322 | 0.295 | - | 1.7 |
| Iron 1 | <0.05 | <0.05 | 0.1 | 1.0 |
| Manganese 1 | <0.05 | <0.05 | 0.05 | 0.5 |
| Arsenic 1 | 0.1341 | 0.1671 | - | 0.010 |
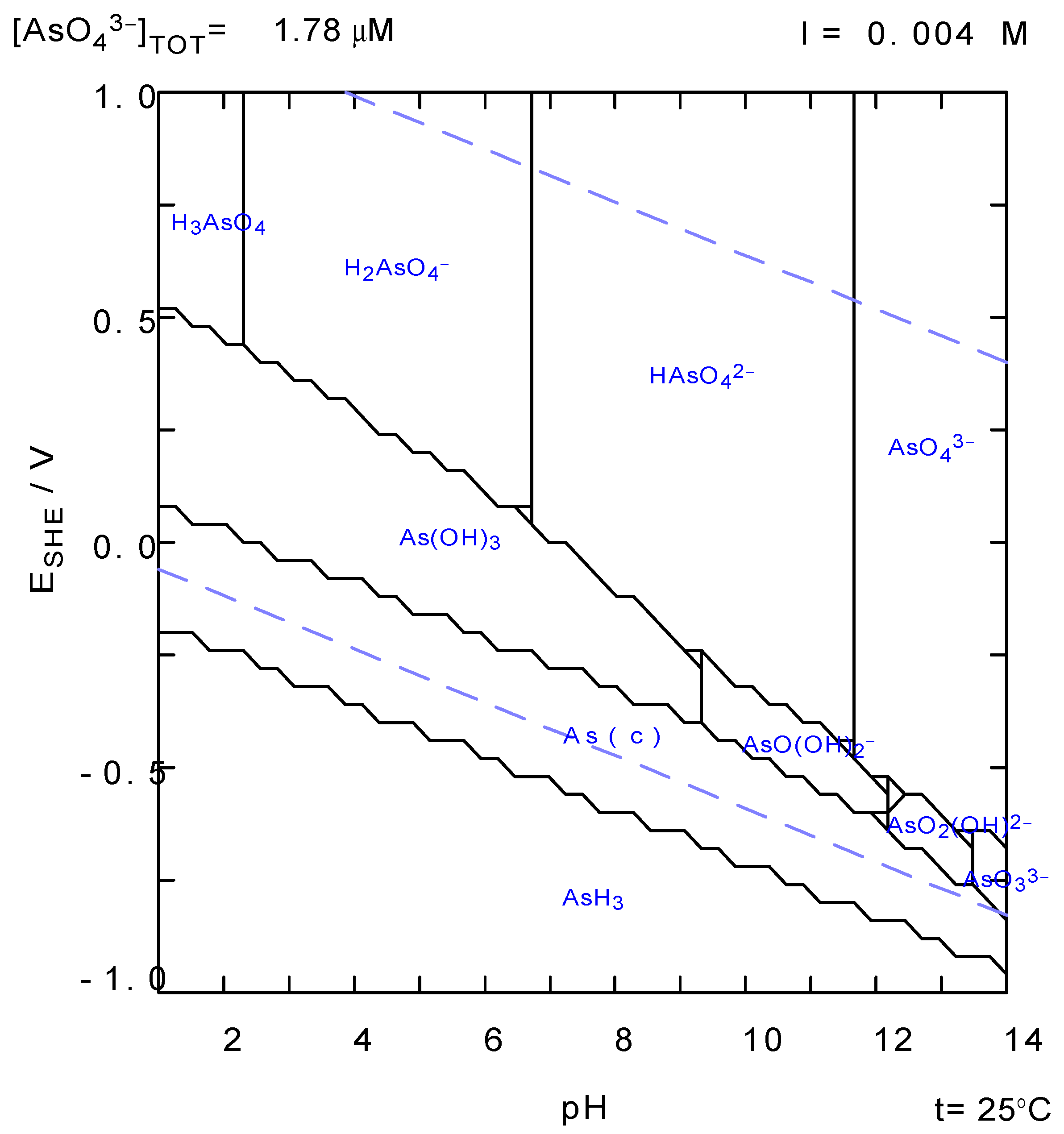

2.3. Laboratory Tests
| Experiment No. | pH Adjustment | Dose (mg L−1) | |
|---|---|---|---|
| NaOCl | Clay | ||
| 1 | 6.2 to 7 | 1.0 | - |
| 2 | None | 0.5 | - |
| 3 | 6.5 | 1.0 | 0.10 |
| 4 | None | 0.5 | 0.05 |
| 5 | None | 0.5 | 0.10 |
| Process | Mixer (rpm) | Time | Gradient (s−1) |
|---|---|---|---|
| Fast mixing | 300 | 5–15 s | 600 |
| Flocculation | (min) | ||
| First stage | 70 | 5 | 80 |
| Second stage | 60 | 5 | 60 |
| Third stage | 44 | 5 | 40 |
| Fourth stage | 33 | 5 | 27 |
| Sedimentation | 0 | 15 | - |
2.4. Full-Scale Water Plant
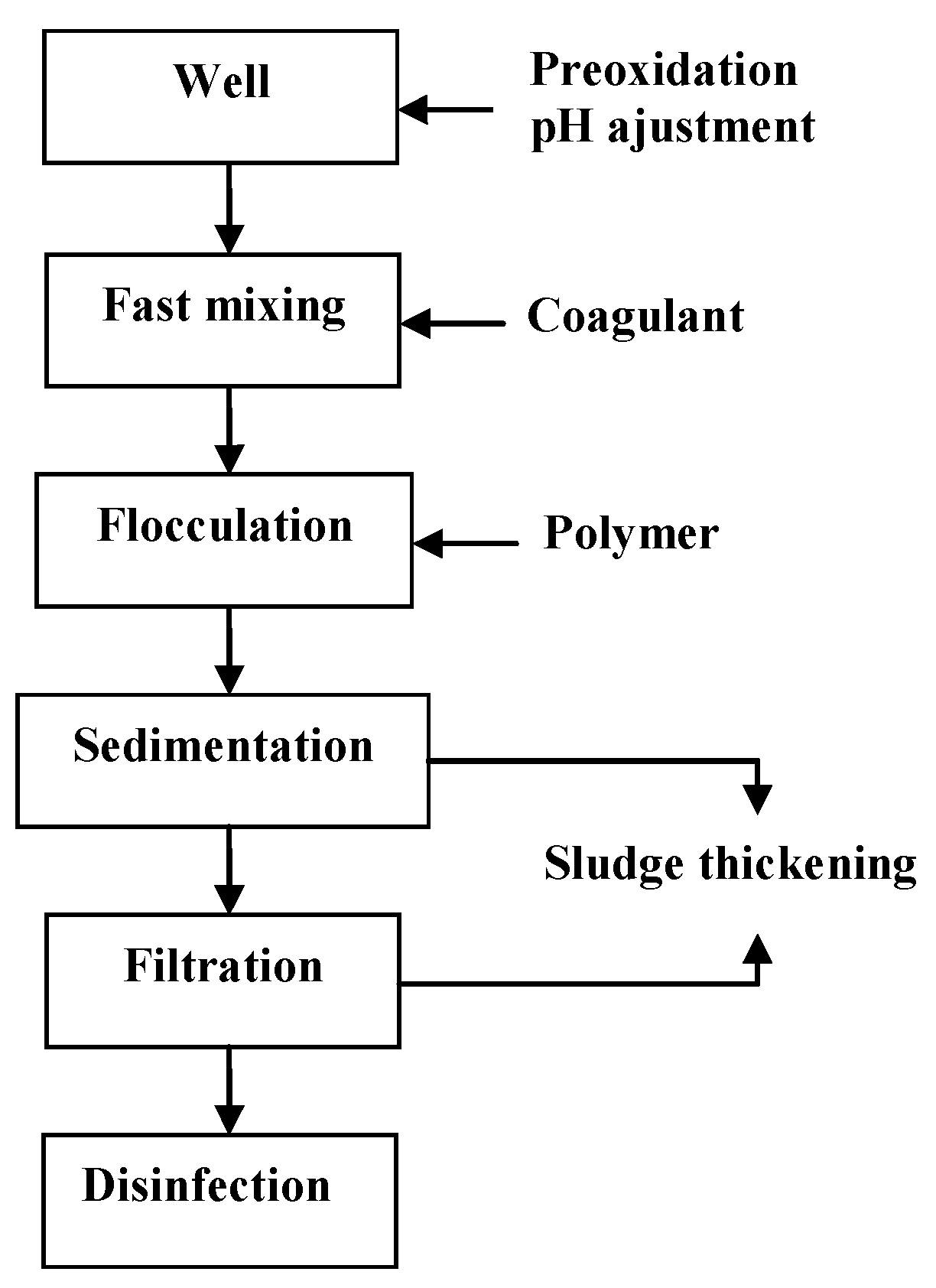
| Process Unit | Ceramic Filter Configuration (A) | Clinoptilolite Filter Configuration (B) |
|---|---|---|
| Pre-oxidation | NaOCl: 2.7 mg L−1 | NaOCl: 4.95 mg L−1 |
| Coagulation | NaOH: 22.78 mg L−1 FeCl3: 12 mg L−1 (74 µmol·L−1) Poliflocal-CH: 1.0 mg L−1 Velocity gradient, G: 1198 s−1 pH coagulation: 7.23 Hydraulic detention time, t: 10 s | NaOH: 13.90 mg L−1 FeCl3: 10 mg L−1 (61.7 µmol·L−1) Poliflocal-CH: 1.8 mg L−1 MIT03: 0.4 mg L−1 G: 1198 s−1 pH coagulation: 6.98 t: 10 s |
| Flocculation | G: 200-38-26-9 s−1 Hydraulic detention time, t: 20.73 min | G: 187-65-27-7 s−1 t: 20.73 min |
| Sedimentation | Without laminar-flow devices. Surface loading rate, SLR: 72.2 m3·m−2·d−1 Weir loading rate: 199.71 m3·m−1·day−1 Hydraulic detention time, t: 30.51 min | With laminar-flow devices SRL: 150 m3·m−2·day−1 Weir loading rate : 316.97 m3·m−1·day−1 t: 30.51 min |
| Filtration Pressure filters | Ceramic media Area, A: 0.899 m2 Flow for each filter, Q: 6.31 L·s−1 Hydraulic loading rate, v: 25.27 m3·m−2·h−1 Contact time, t: 2.12 min Backwash rate, Ub: 18.95 m3·m−2·h−1 | Clinoptilolite media Area, A: 0.899 m2 Q: 5.89 L·s−1 v: 23.59 m3·m−2·h−1 t: 2.12 min Ub: 16.42 m3·m−2·h−1 |
| Media | Physical Characteristics | |||
|---|---|---|---|---|
| Surface Area (m2 g−1) | Particle Size (mm) | Uniformity Coefficient | Bed Depth (m) | |
| Ceramic | 5–14 | 0.25–0.43 | 1.50 | 0.86 |
| Clinoptilolite | 14–25 | 0.6–1.40 | 1.64 | 0.85 |
- (a)
- Existing infrastructure was considered, such as the water transportation system from the wells to the tanks, filters (included ceramic media) and pumping, and the water treatment plant’s electromechanical equipment located in NCMG.
- (b)
- The topographic slope was used for gravity flow, as well as to save energy and avoid the use of pumping equipment and prevent large volumes of soil movement.
- (c)
- It was attempted to avoid separation between structures so as to reduce the length of the interconnection lines (as well as hydraulic head loss), leaving enough space for the installation of piping, meters, platforms and access stairs to elevated structures.
- (d)
- The pipes were situated in order to not unnecessarily increase their lengths and so that deflections, due to changes in direction, corresponded to angles for which special commercial parts are available.
- (e)
- Treated water from the arsenic removal plant was directed toward the property’s low area, from where it flowed by gravity to four storage cisterns.
- (f)
- A booth was situated for the placement of pH control panels, dispensers for sodium hydroxide, ferric chloride and polymer, and variable-frequency drives to set the rotational speed of the agitators to fast and slow mixing.
2.5. Analytical Methods
3. Results and Discussion
3.1. Laboratory Test
3.1.1. Optimum Dose
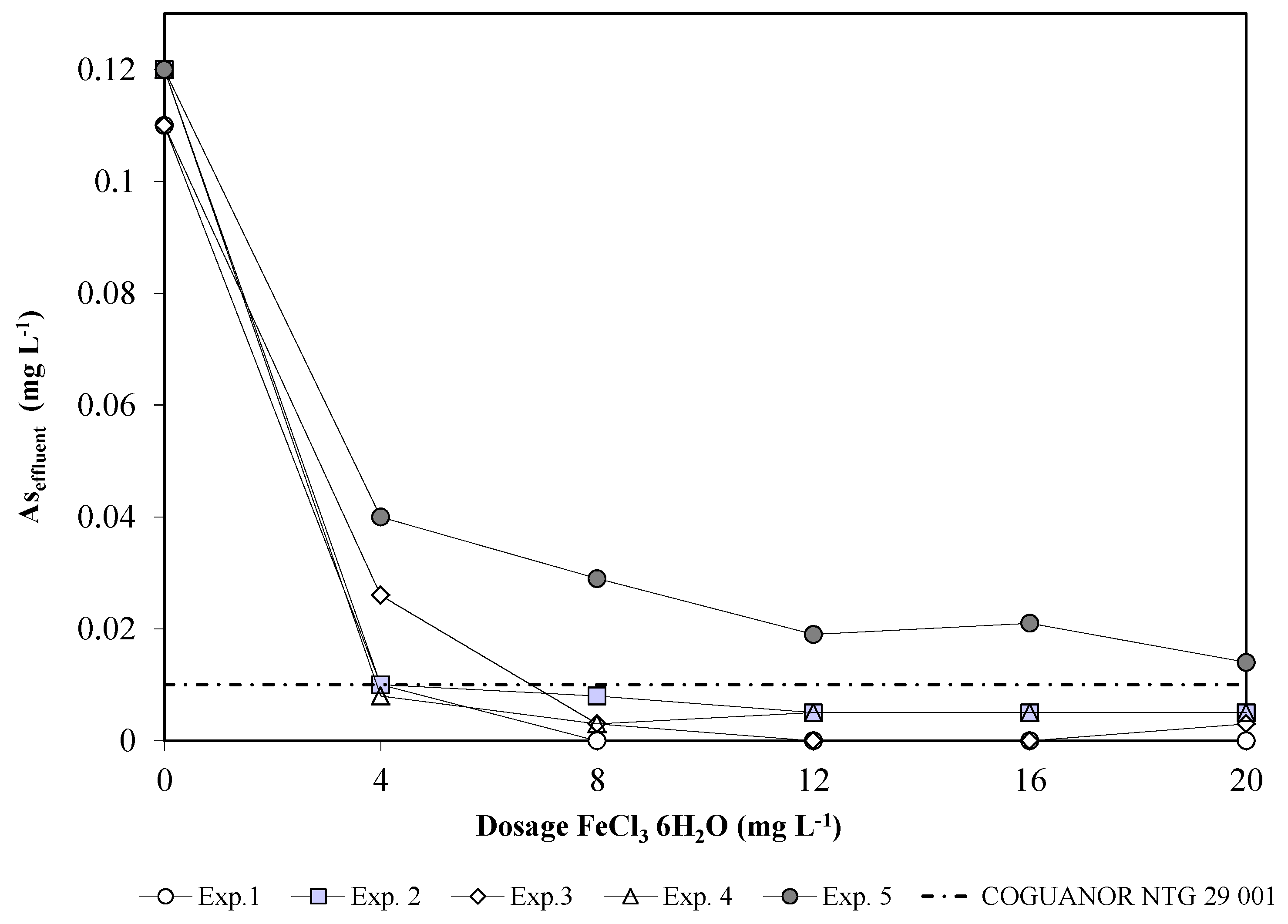

3.1.2. Qualitative Evaluation (Floc Size and Formation Time)
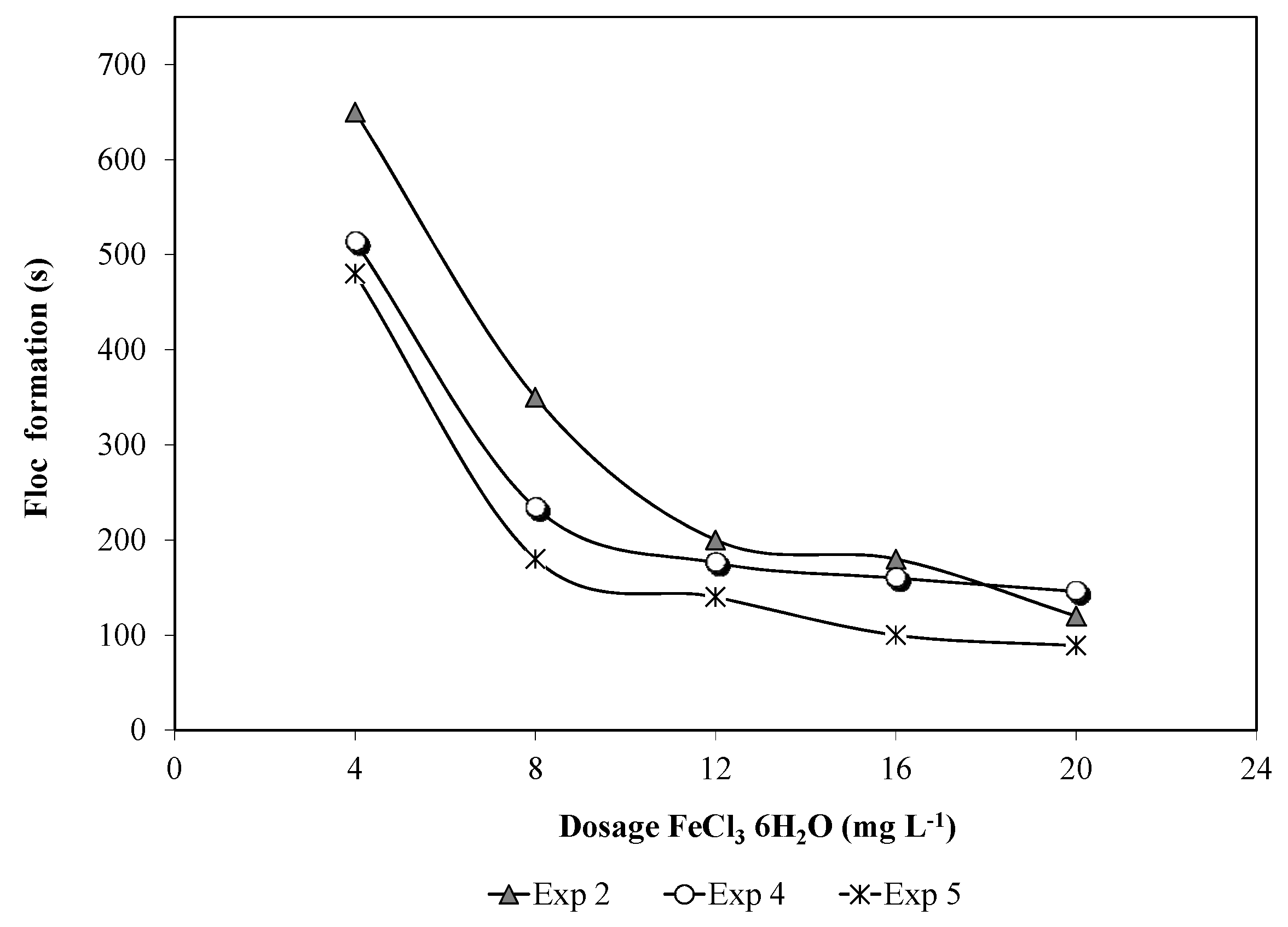
3.1.3. Optimum pH
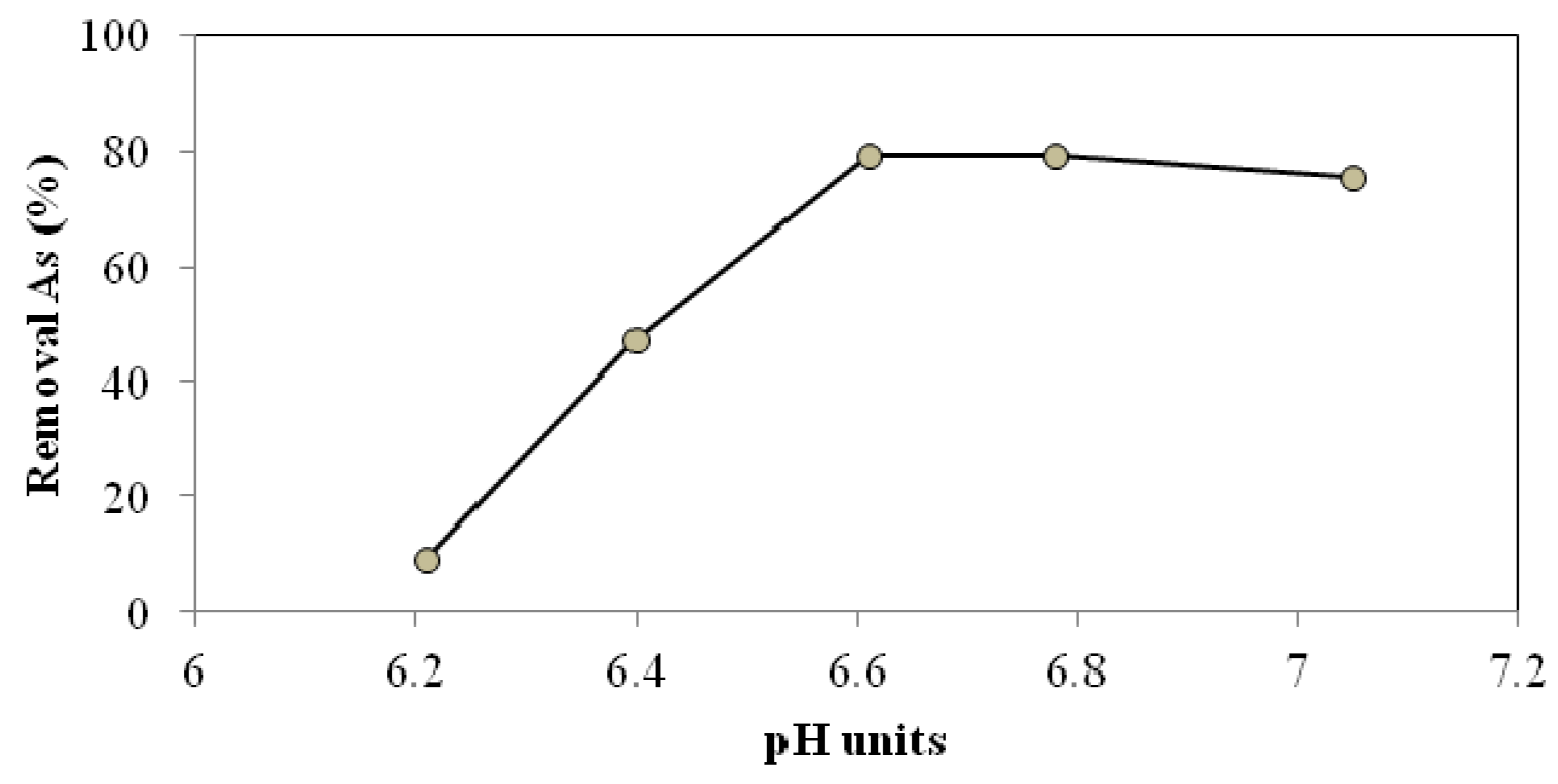
3.2. Evaluation of Plant Performance Using Two Configurations to Remove Arsenic
| Process Water | Configuration A, Ceramic Filter | Configuration B, Clinoptilolite Filter | ||||||
|---|---|---|---|---|---|---|---|---|
| pH | Color (UPt-Co) | Fetotal (mg L−1) | As(V) (mg·L−1) | pH | Color (UPt-Co) | Fetotal (mg·L−1) | As(V) (mg·L−1) | |
| Raw water | 7.23 | 17.8 | 0.72 | 0.092 | 6.98 | 35.0 | 1.48 | 0.171 |
| Raw water + Fe(III) dosage | - | - | 2.78 | - | - | - | 3.96 | - |
| Settled water | 7.08 | 37.8 | 1.96 | 0.038 | 6.84 | 6.33 | 0.75 | 0.028 |
| Unfiltered water | 7.13 | 55.4 | 2.18 | 0.068 | 7.10 | 12.33 | 0.88 | 0.019 |
| Filtered water | 7.22 | 4.0 | 0.56 | 0.026 | 7.12 | 3.66 | 0.35 | 0.015 |
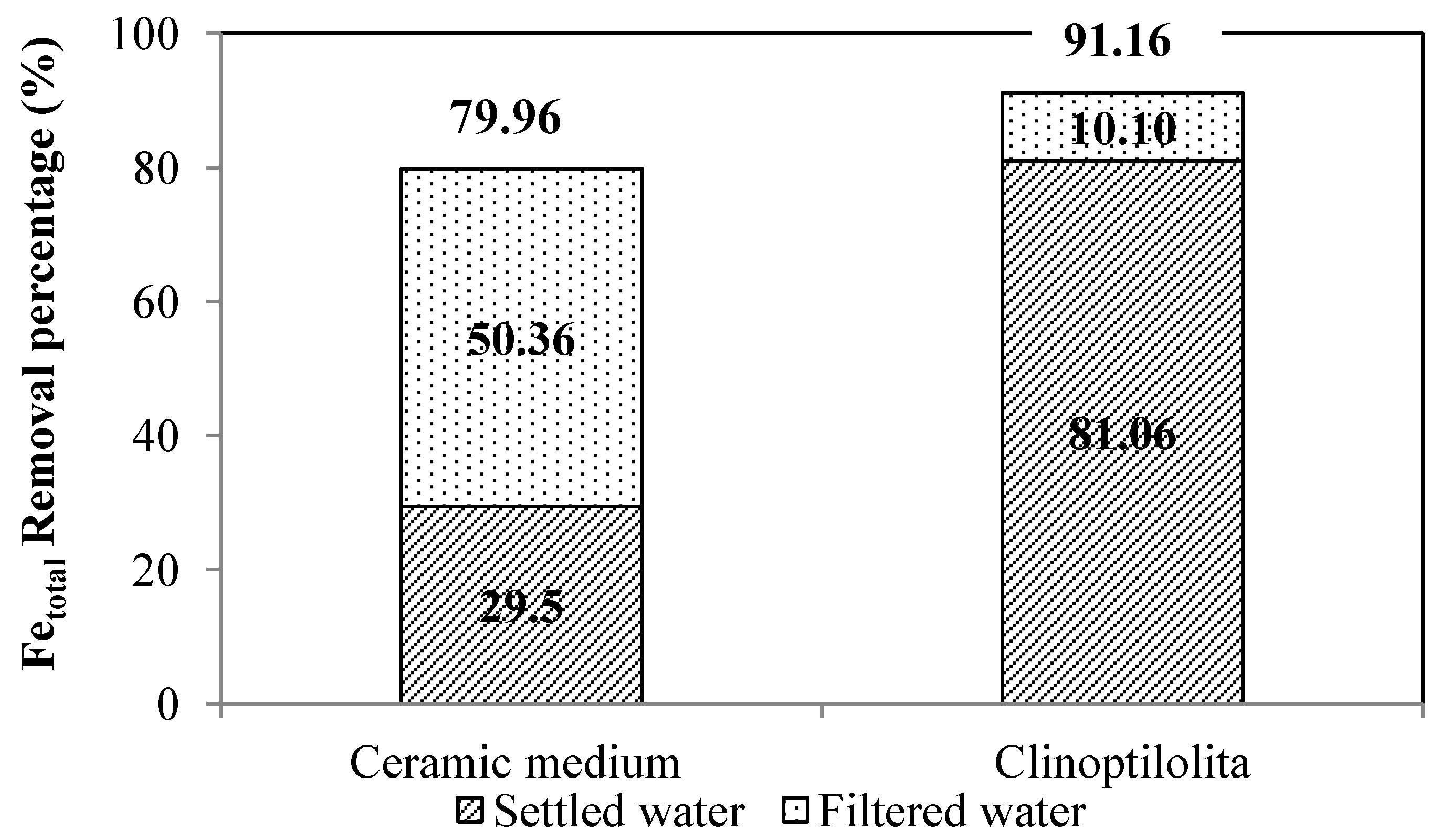
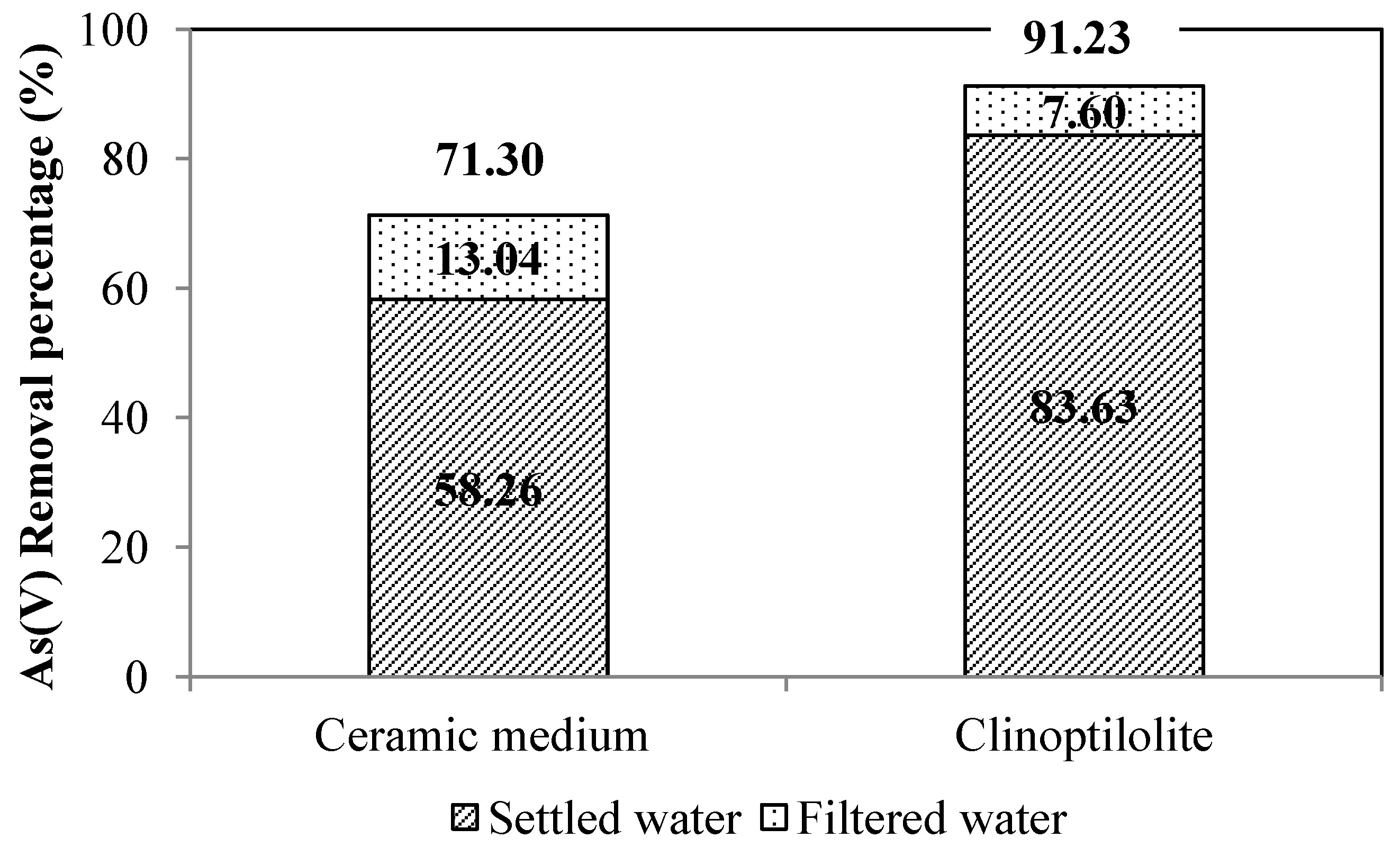

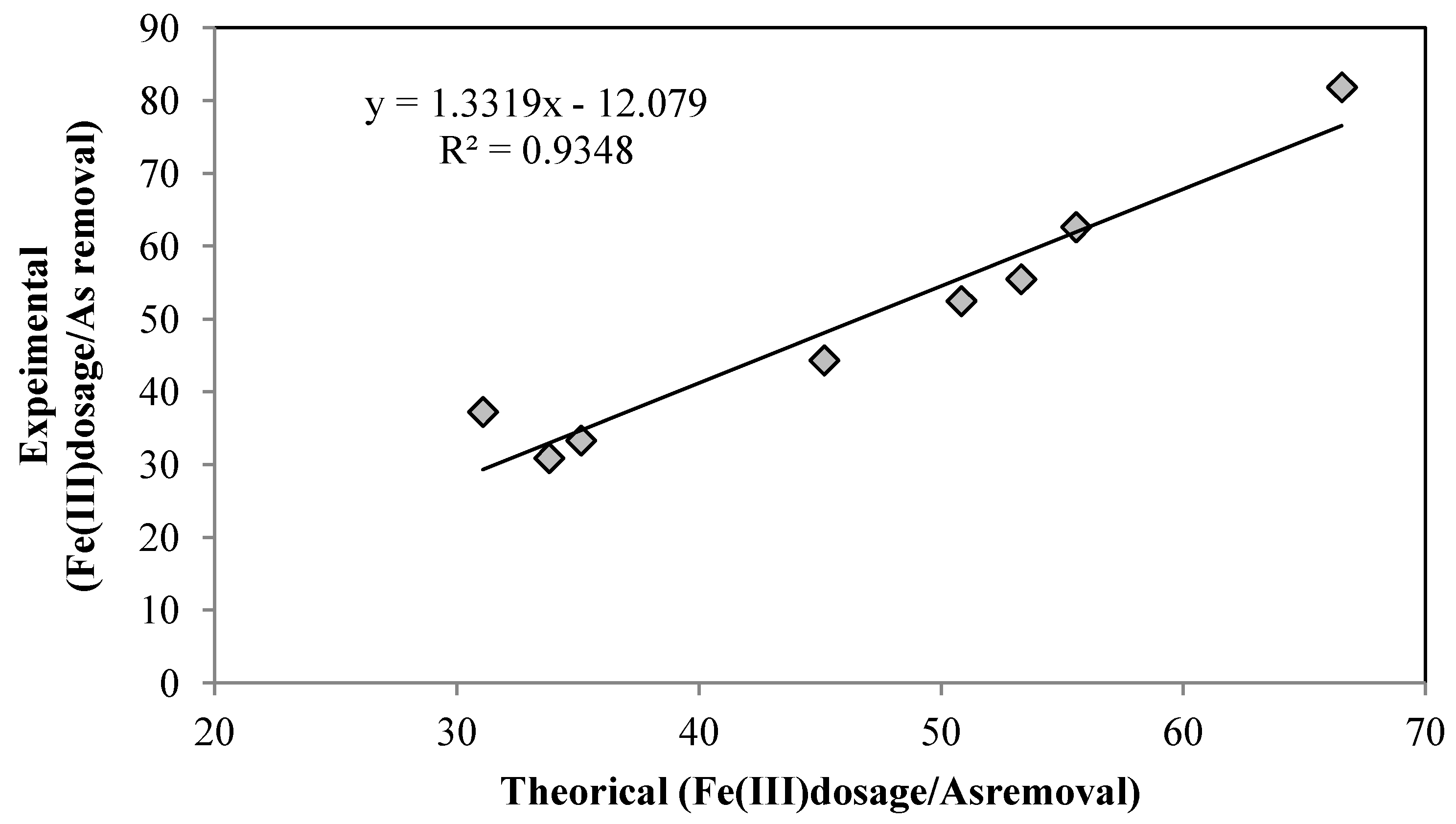

4. Conclusions
References
- Tseng, W. Effects and dosage-response relationships of skin cancer and blackfoot disease with arsenic. Environ. Health Perspect. 1977, 19, 109–119. [Google Scholar] [CrossRef]
- Kumar-Mandal, B.; Suzuki, T. Arsenic round the world: A review. Talanta 2002, 58, 201–235. [Google Scholar] [CrossRef]
- Farias, S.; Casa, V.; Vazquez, C.; Ferpozzi, L.; Pucci, G.; Cohen, I. Natural contamination with arsenic and other trace elements in ground waters of Argentine Pampean Plain. Sci. Total Environ. 2003, 309, 187–199. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Verification of Arsenic Mitigation Technologies and Field Test Methods. In Proceedings of Intercountry Consultation, Kolkata, India, 9–12 December 2002; WHO Regional Office for South-East Asia: New Delhi, India, 2004.
- Bundschuh, J.; Litter, M.; Ciminelli, V.S.T.; Morgada, M.E.; Cornejo, L.; Hoyos, S.G.; Hoinkis, J.; Alarcon-Herrera, M.T.; Armienta, M.A.; Bhattacharya, P. Emerging mitigation needs and sustainable options for solving the arsenic problems of rural and isolated urban areas in Latin America—A critical analysis. Water Res. 2010, 44, 5828–5845. [Google Scholar] [CrossRef]
- Garrido, S.; Avilés, M.; Ramírez, A.; Calderón, C.; Ramírez-Orozco, A.; Nieto, A.; Shelp, G.; Seed, L.; Cebrian, M.; Vera, E. Arsenic Removal from Water of Huautla, Morelos, Mexico Using Capacitive Deionization. In Proceedings of the International Congress on Natural Arsenic in Groundwaters of Latin America, Mexico City, Mexico, 20–24 June 2006; pp. 665–676.
- Amy, G.; Edwards, M.; Brandhuber, P.; McNeill, L.; Benjamin, M.; Vagliasindi, F.; Carlson, K.; Chwirka, J. Arsenic Treatability Options and Evaluation of Residuals Management Issues; 90771; American Water Works Association: Denver, CO, USA, 1999. [Google Scholar]
- >Environmental Protection Agency (EPA), Technologies and Costs for Removal of Arsenic from Drinking Water; EPA 815-R-00-028; Environmental Protection Agency: Washington, DC, USA, 2000.
- Shen, Y.S. Study of arsenic removal from drinking water. J. Am. Water Works Assoc. 1973, 65, 543–547. [Google Scholar]
- Ahmed, K.; Bhattacharya, P.; Hasan, M.; Akhter, S.; Alam, S.M.; Bhuyian, M.A.; Imam, M.B.; Khan, A.; Sracek, O. Arsenic enrichment in groundwater of the alluvial aquifers in Bangladesh: An overview. Appl. Geochem. 2004, 19, 181–200. [Google Scholar] [CrossRef]
- Pande, S.P.; Deshpande, L.S.; Patni, P.M. Arsenic removal studies in some ground waters of West Bengal, India. J. Environ. Sci. Health 1997, A32, 1981–1987. [Google Scholar]
- Petkova, V.; Rivera, L.; Aviles, M.; Piña, M.; Martin, A. Remoción de Arsénico de Agua Para Consumo Humano; Reporte final; Instituto Mexicano de Tecnología del Agua: Jiutepec, Mexico, 1997. [Google Scholar]
- Ngo, H.; Vigneswaran, S.; Hu, J.; Thirunavukkarasu, O.; Viraraghavan, T. A comparison of conventional and non-conventional treatment technologies on arsenic removal from water. Water Sci. Technol. Water Supply. 2002, 2, 119–125. [Google Scholar]
- Gulledge, J.H.; O’Connor, J.T. Removal of arsenic(V) from water by adsorption on aluminum and ferric hydroxides. J. Am. Water Works Assoc. 1973, 65, 548–552. [Google Scholar]
- Sorg, T.J.; Logsdon, G.S. Treatment technology to meet the interin primary drinking water regulations for inorganics: Part 2. J. Am. Water Works Assoc. 1978, 70, 379–393. [Google Scholar]
- Logsdon, G.S.; Sorg, T.J.; Symons, M. Removal of Heavy Metals by Conventional Treatment. In Proceedings of the 16th Water Quality Conference, College of Engineering, University of Illinois, Urbana-Champaign, IL, USA, 12–13 February 1974; Volume 71, No. 108. pp. 111–133.
- Instituto Nacional de Estadística (INE), Anuario Estadístico de Guatemala; Instituto Nacional de Estadística: Guatemala, Guatemala, 2002.
- Cebrián, M.E.; Albores, A.; García-Vargas, G.; Del Razo, L.M. Chronic Arsenic Poisoning in Humans: The Case of Mexico. In Arsenic in the Environment. Part II: Human Health and Ecosystem Effects; Wiley Interscience: New York, NY, USA, 1994; pp. 94–97. [Google Scholar]
- American Society for Testing and Materials (ASTM), Standard Practice for Coagulation-Flocculation Jar Test of Water E1-1994 R(1995), D 2035-80; Annual book of ASTM standards; ASTM International: Whashington, DC, USA; Volume 11.02, 1995.
- Arboleda, J. Teoría y Práctica de la Purificación del Agua, 3th ed.; Arboleda Valencia, J., Ed.; Mac Graw Hill.: Bogotá, Colombia, 2000; pp. 169–170. [Google Scholar]
- Environmental Protection Agency (EPA), Removal of Arsenic in Drinking Water. Kinetico® Incorporated Macrolite Coagulation and Filtration System; Model CPS100CPT, 2001. NSF 01/23EPADW395; Environmental Technology Verification Report; Environmental Protection Agency: Washington, DC, USA, 2001.
- Standard Methods for Examination of Water and Waste Water, 20th ed.; American Public Health Association: New York, NY, USA, 1998.
- HACH™ Company, Procedures Manual DR/2010, Spectrophotometer; HACH Company: Loveland, CO, USA, 1998.
- Erickson, B.E. Fields kits fail to provide accurate measure of arsenic in groundwater. Environ. Sci. Technol. 2003, 1, 35–38. [Google Scholar] [CrossRef]
- Scott, K.N.; Green, J.F.; Do, H.D.; McLean, S.J. Arsenic removal by coagulation. J. Am. Water Works Assoc. 1995, 87, 114–126. [Google Scholar]
- Chen, H.W.; Frey, M.M.; Clifford, D.; McNeill, L.S.; Edwards, M. Arsenic treatment considerations. J. Am. Water Works Assoc. 1999, 91, 74–85. [Google Scholar]
- Hering, J.G.; Chen, P.Y.; Wilkie, J.A.; Elimelech, M. Arsenic removal from drinking water during coagulation. ASCE J. Environ. Eng. 1997, 123, 800–807. [Google Scholar]
- Laky, D.; László, B.; Licskó, I. Effects of several water quality parameters on arsenic removal by coagulation: Laboratory experiments and a pilot-scale study. Water Pract. Tecnol. 2008, 3, 1–8. [Google Scholar]
- Garrido Hoyos, S.E. Análisis de la información técnica y evaluación del funcionamiento de la planta para remover arsénico, Mixco, Guatemala; Informe final; Instituto Mexicano de Tecnología del Agua: Jiutepec, México, 2008; p. 125. [Google Scholar]
- Garrido Hoyos, S.E. Seguimiento del funcionamiento de la planta convencional con filtración kinetico para remoción de arsénico y capacitación de personal en el funcionamiento de la planta, Mixco, Guatemala; Reporte final; Instituto Mexicano de Tecnología del Agua: Jiutepec, México, 2010; p. 72. [Google Scholar]
- Garrido, S.; Piña, M.; López, I.; De La O, D.; Rodríguez, R. Behavior of two filters media for to remove arsenic from drinking water. Filtration 2013, 13, 21–26. [Google Scholar]
- Dahi, E.; Liang, Q. Arsenic Removal in Hand Pump Connected Iron Removal Plants in Noakhali, Bangladesh. In Proceedings of the International Conference on Arsenic Pollution of Ground Water in Bangaldesh: Causes, Effect and Remedies, Dhaka, Bangladesh, 8–12 February 1998.
- Eriksen-Hamel, N.; Zinia, K.N. A Study of Arsenic Treatment Technologies and Leaching Characteristics of Arsenic Contaminated Sludge. In Technologies for Arsenic Removal from Drinking Water; Ahmed, M.F., Ali, M.A., Adeel, Z., Eds.; Bangladesh University of Engineering & Technology and United Nations University: Dhaka, Bangladesh, 2001; pp. 207–213. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Garrido Hoyos, S.E.; Avilés Flores, M.; Ramírez Gonzalez, A.; Grajeda Fajardo, C.; Cardoso Zoloeta, S.; Velásquez Orozco, H. Comparing Two Operating Configurations in a Full-Scale Arsenic Removal Plant. Case Study: Guatemala. Water 2013, 5, 834-851. https://doi.org/10.3390/w5020834
Garrido Hoyos SE, Avilés Flores M, Ramírez Gonzalez A, Grajeda Fajardo C, Cardoso Zoloeta S, Velásquez Orozco H. Comparing Two Operating Configurations in a Full-Scale Arsenic Removal Plant. Case Study: Guatemala. Water. 2013; 5(2):834-851. https://doi.org/10.3390/w5020834
Chicago/Turabian StyleGarrido Hoyos, Sofía E., Martha Avilés Flores, A. Ramírez Gonzalez, Celia Grajeda Fajardo, Saúl Cardoso Zoloeta, and Hayron Velásquez Orozco. 2013. "Comparing Two Operating Configurations in a Full-Scale Arsenic Removal Plant. Case Study: Guatemala" Water 5, no. 2: 834-851. https://doi.org/10.3390/w5020834
APA StyleGarrido Hoyos, S. E., Avilés Flores, M., Ramírez Gonzalez, A., Grajeda Fajardo, C., Cardoso Zoloeta, S., & Velásquez Orozco, H. (2013). Comparing Two Operating Configurations in a Full-Scale Arsenic Removal Plant. Case Study: Guatemala. Water, 5(2), 834-851. https://doi.org/10.3390/w5020834





