Crayfish Carapace Micro-powder (CCM): A Novel and Efficient Adsorbent for Heavy Metal Ion Removal from Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Adsorption Experiments
2.3. Determination of Calcium Level
3. Results and Discussion
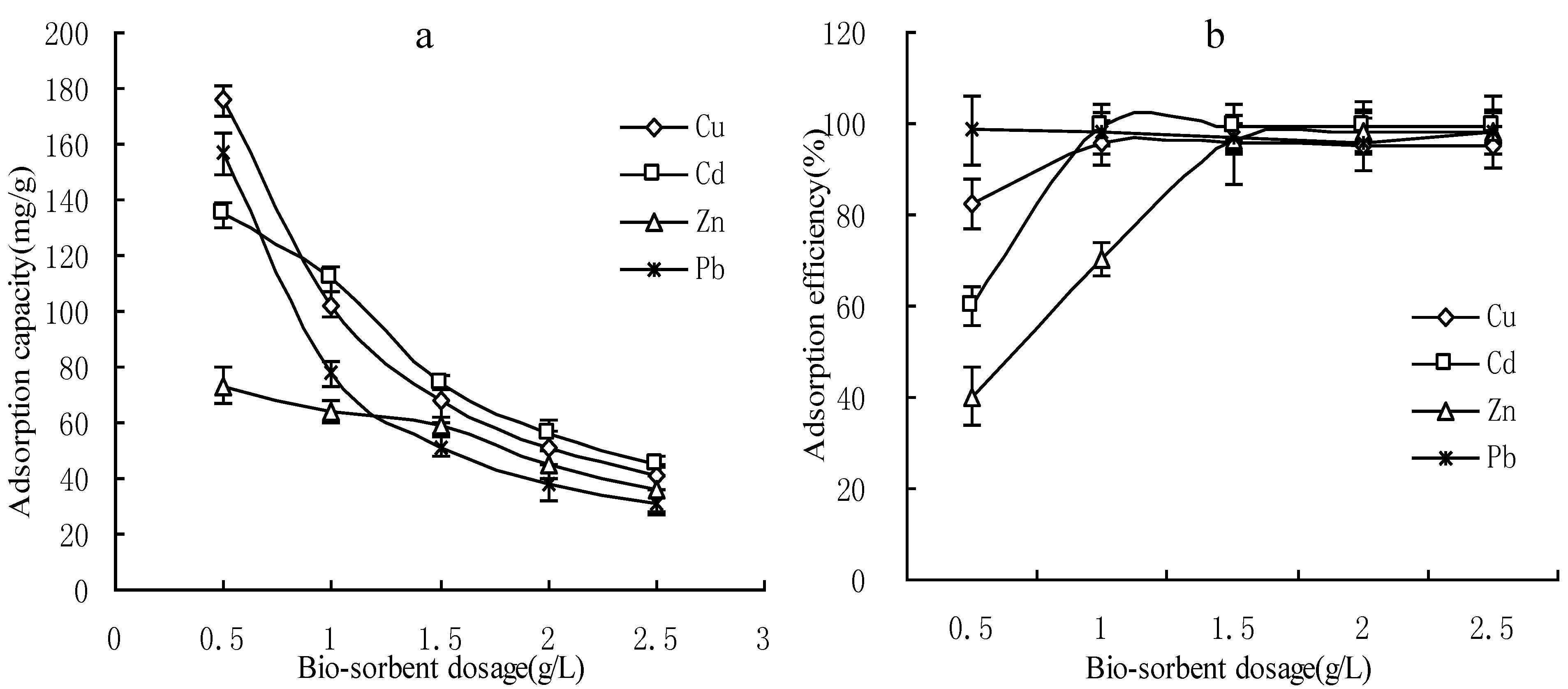
3.1. Effect of Adsorbent Dosages
3.2. Adsorption Kinetics
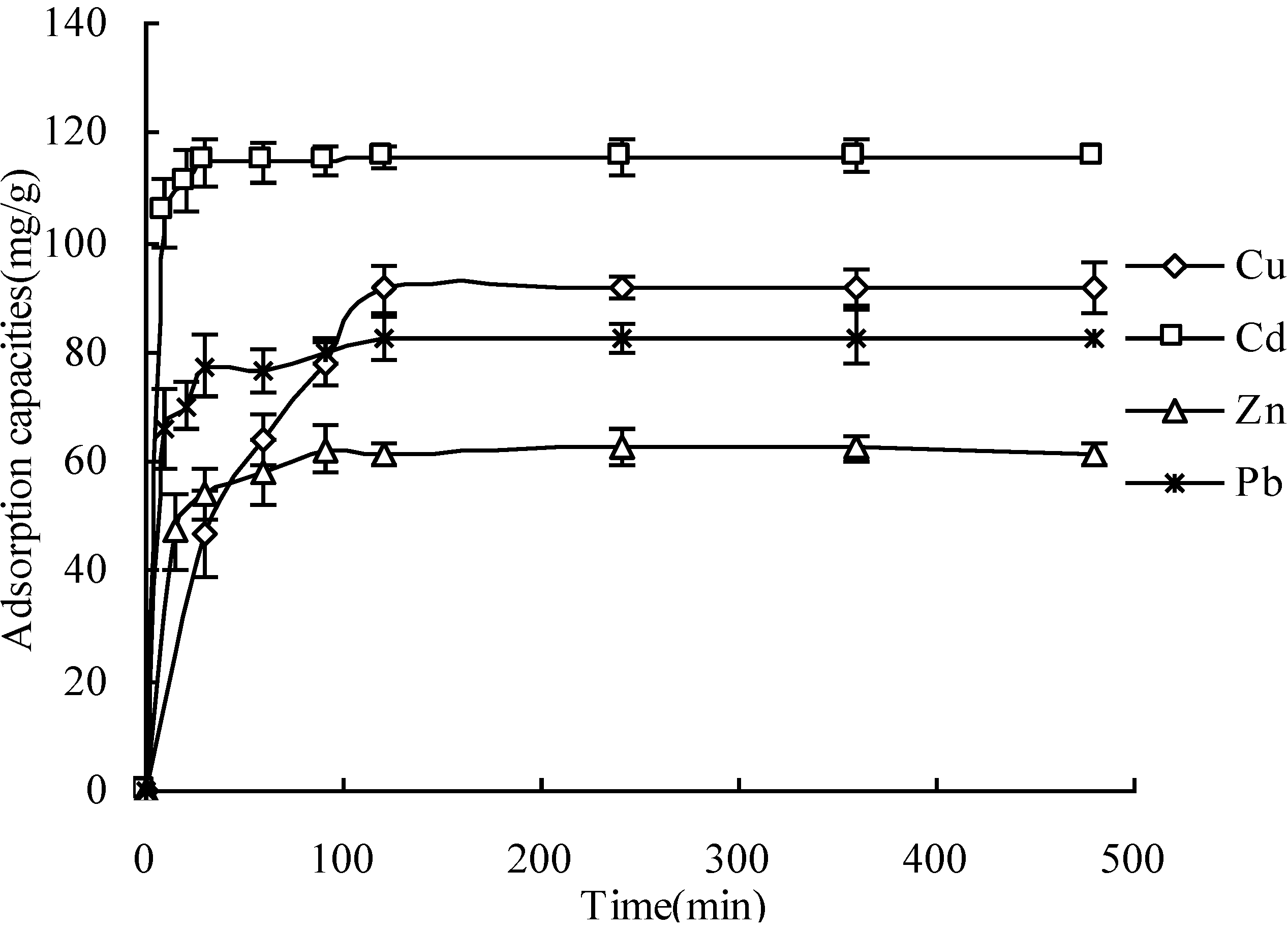
Ions | Pseudo first-order equation | Pseudo second-order equation | |||||
|---|---|---|---|---|---|---|---|
| Qe (mg/g) | k1 | R2 | Qe (mg/g) | k2 | R2 | ||
| Cu | 93.045 | 0.0294 | 0.8916 | 106.383 | 0.0025 | 0.9651 | |
| Cd | 101.3705 | 0.0469 | 0.8622 | 116.2791 | 0.0881 | 0.9514 | |
| Zn | 61.8682 | 0.0365 | 0.8077 | 63.6943 | 0.0302 | 0.9796 | |
| Pb | 77.2632 | 0.0415 | 0.9139 | 82.6445 | 0.0452 | 0.9314 | |
3.3. Effect of CCM Size


3.4. Effect of Solution pH
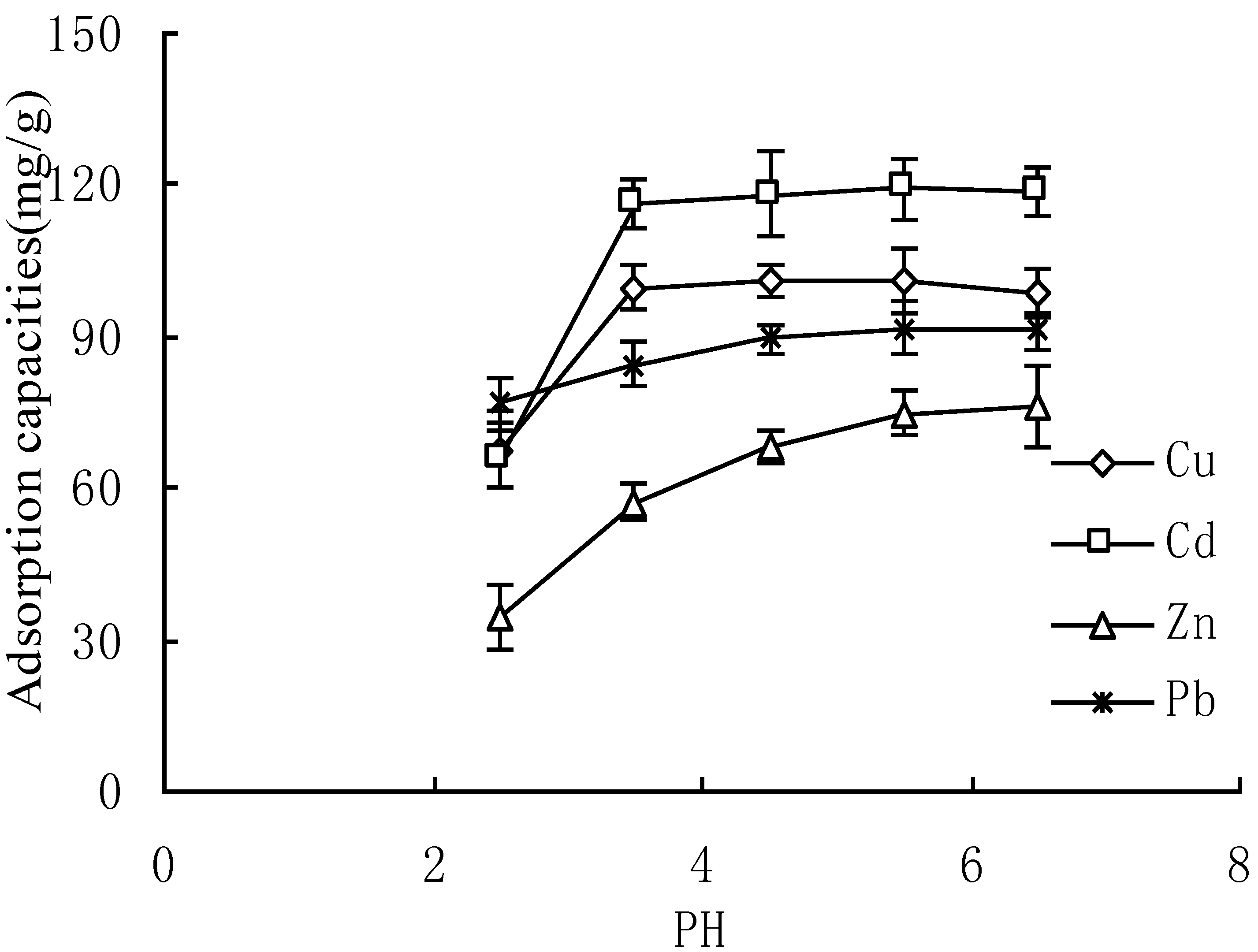
3.5. Thermodynamic Property of Adsorption
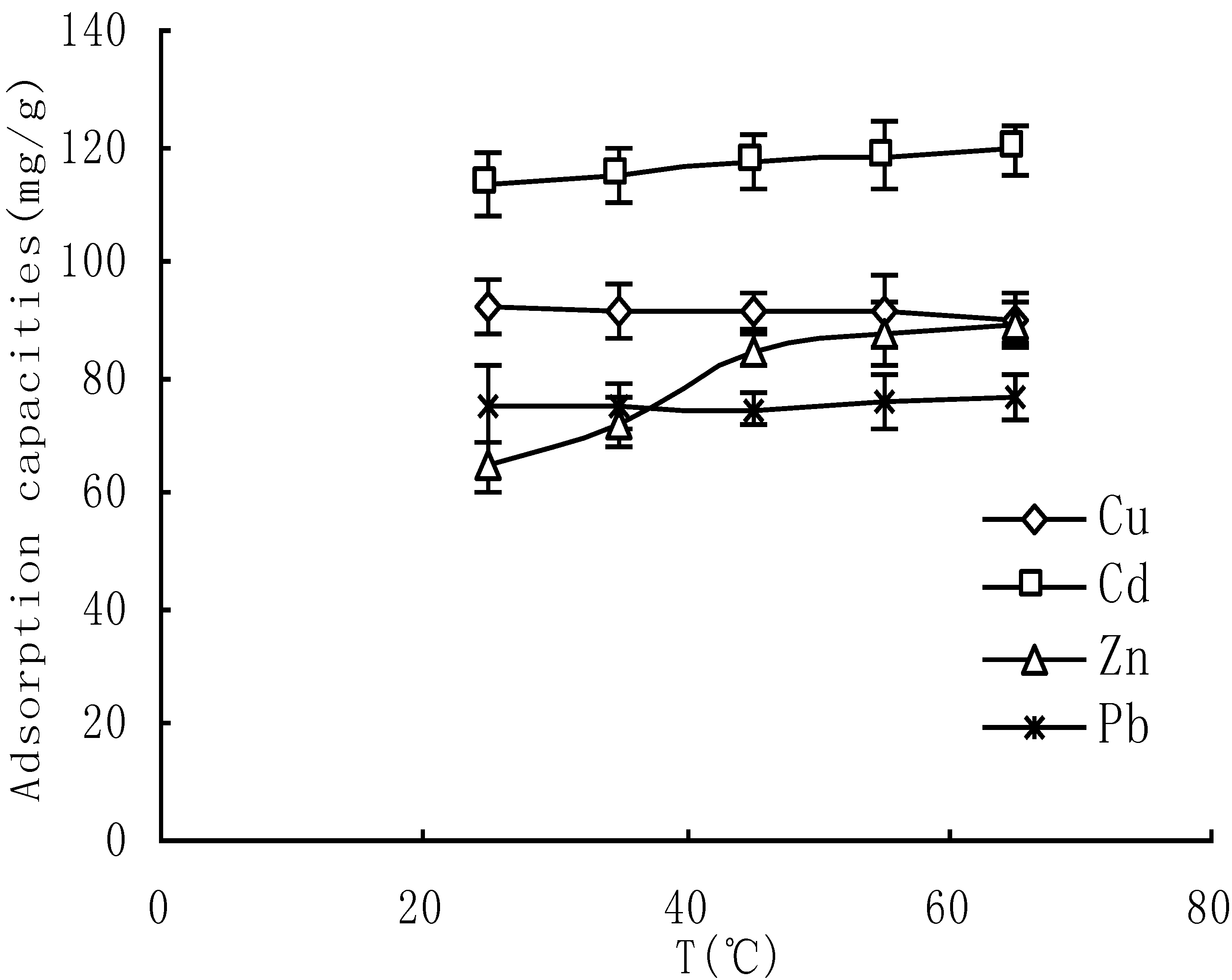
| Metal ions | ΔH (KJ/g) | R2 |
|---|---|---|
| Cu | −2,036.26 | 0.9497 |
| Cd | 4,223.51 | 0.9592 |
| Zn | 54,006.08 | 0.9963 |
| Pb | - | - |
3.6. Adsorption Isotherm

| Ion | Langmuir | Freundlich | |||||
|---|---|---|---|---|---|---|---|
| Qm (mg/g) | B (mL/mg) | R2 | Kf (mg/g) | n | R2 | ||
| Cu | 200 | 0.0852 | 0.9772 | 30.481 | 2.3207 | 0.9683 | |
| Cd | 217.39 | 0.6053 | 0.9924 | 99.315 | 5.0428 | 0.9427 | |
| Zn | 80 | 0.0945 | 0.9722 | 34.519 | 6.6445 | 0.9461 | |
| Pb | 322.58 | 0.1148 | 0.9992 | 42.06 | 1.6841 | 0.9822 | |
3.7. Effect of Calcium Level of CCM

3.8. Comparison of Adsorption between CCM, Chitin and Chitosan
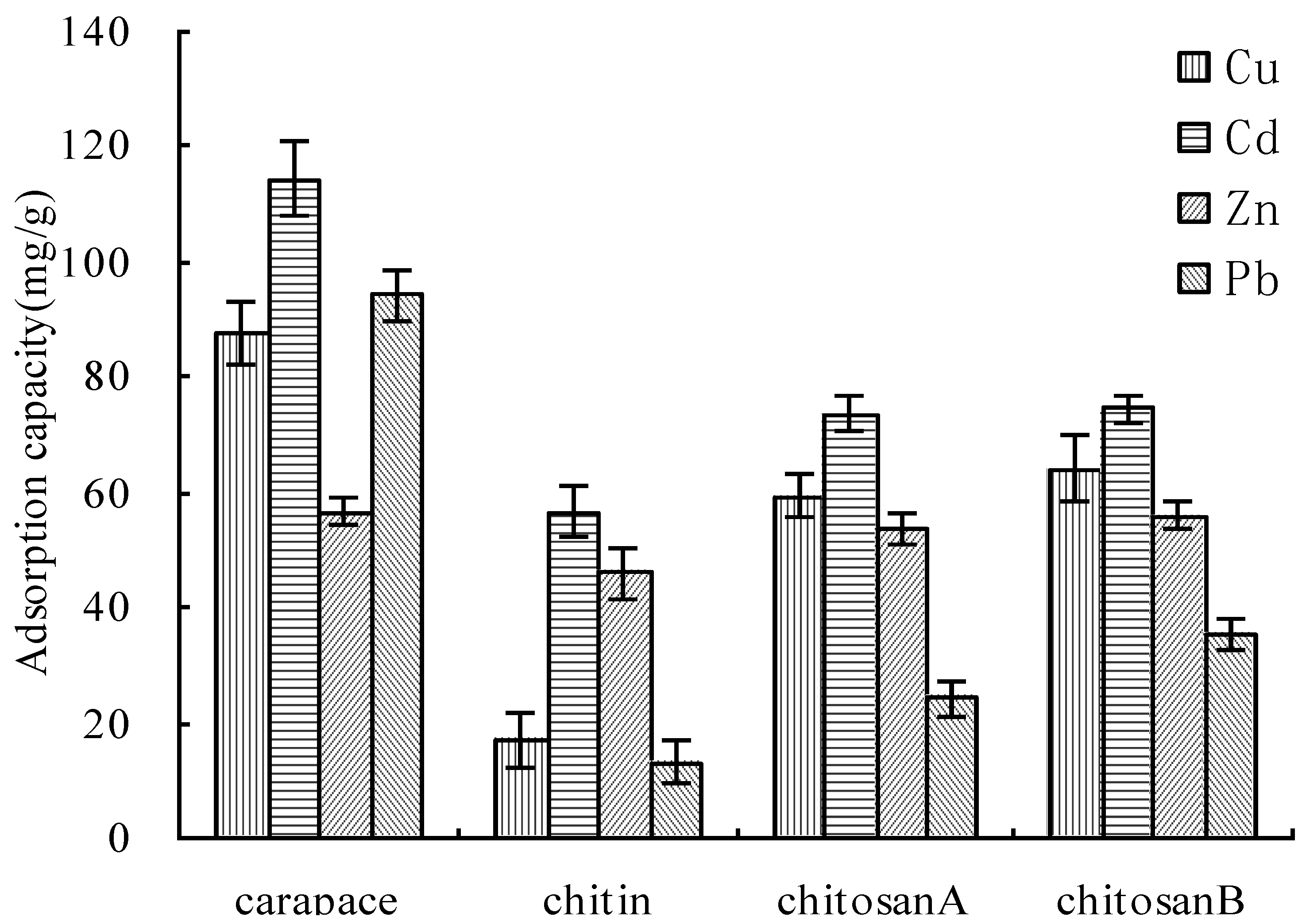
4. Conclusions
Acknowledgements
References
- Sun, S.L.; Wang, Q.; Wang, A.Q. Adsorption properties of Cu(II) ions onto N-succinyl-chitosan and crosslinked N-succinyl-chitosan template resin. Biochem. Eng. J. 2007, 36, 131–138. [Google Scholar] [CrossRef]
- Integrated Wastewater Discharge Standard GB 8978-1996; Ministry of Environmental Protection: Beijing, China, 1998.
- Juang, R.S.; Shao, H.J. A simplified equilibrium model for sorption of heavy metal ions from aqueous solutions on chitosan. Water Res. 2002, 36, 2999–3008. [Google Scholar] [CrossRef] [PubMed]
- Baliley, S.E.; Olin, T.J.; Bricka, R.M.; Adria, D.D. A review of potentially low-cost sorbents for heavy metals. Water Res. 1999, 33, 2469–2479. [Google Scholar] [CrossRef]
- Mohan, D.; Chander, S. Single component and multi-component adsorption of metal ions by activated carbons. Colloid. Surface. A 2001, 177, 183–196. [Google Scholar] [CrossRef]
- Guibal, E. Interactions of metal ions with chitosan-based sorbents: A review. Sep. Purif. Technol. 2004, 38, 43–74. [Google Scholar] [CrossRef]
- Li, N.; Bai, R.B. A novel amine-shielded surface crosslinking of chitosan hydrogel beads for enhanced metal adsorption performance. Ind. Eng. Chem. Res. 2005, 44, 6692–6700. [Google Scholar] [CrossRef]
- Lee, M.Y.; Hong, K.J.; Kajiuchi, T.; Yang, J.W. Determination of the efficiency and removal mechanism of cobalt by crab carapace particles. J. Chem. Technol. Biot. 2004, 79, 1388–1394. [Google Scholar] [CrossRef]
- Liu, C.X.; Bai, R.B. Adsorptive removal of copper ions with highly porous chitosan/cellulose acetate blend hollow fiber membranes. J. Membrane Sci. 2006, 284, 313–322. [Google Scholar] [CrossRef]
- Volesky, B. Advances in biosorption of metals: Selection of biomass types FEMS. Microbiol. Rev. 1994, 14, 291–302. [Google Scholar]
- Varma, A.J.; Deshpande, S.V.; Kennedy, J.F. Metal complexation by chitosan and its derivatives: A review. Carbohyd. Polym. 2004, 55, 77–93. [Google Scholar] [CrossRef]
- Rodde, R.H.; Einbu, A.; Varum, K.M. A seasonal study of the chemical composition and chitin quality of shrimp shells obtained from northern shrimp (Pandalus borealis). Carbohyd. Polym. 2008, 71, 388–393. [Google Scholar] [CrossRef]
- Ehrlich, H.; Koutsoukos, P.G.; Demadis, K.D.; Pokrovsky, O.S. Principles of demineralization: Modern strategies for the isolation of organic frameworks. Part II. Decalcification. Micron 2009, 40, 169–193. [Google Scholar]
- Shittu, T.A.; Badmus, B.A. Statistical correlations between mineral element composition, product information and retail price of powdered cocoa beverages in Nigeria. J. Food Compos. Anal. 2009, 22, 212–217. [Google Scholar] [CrossRef]
- Vijayaraghavan, K.; Palanivelu, K.; Velan, M. Biosorption of copper(II) and cobalt(II) from aqueous solutions by crab carapace particles. Bioresource Technol. 2006, 97, 1411–1419. [Google Scholar] [CrossRef]
- Pradhan, S.; Shukla, S.S.; Dorris, K.L. Removal of nickel from aqueous solutions using crab carapaces. J. Hazard. Mater. 2005, 125, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Wan Ngah, W.S.; Kamari, A.; Koay, Y.J. Equilibrium and kinetics studies of adsorption of copper(II) on chitosan and chitosan/PVA beads. Int. J. Biol. Macromol. 2004, 34, 155–161. [Google Scholar] [CrossRef]
- Sun, S.L.; Wang, A.Q. Adsorption kinetics of Cu(II) ions using N, O-carboxymethyl-chitosan. J. Hazard. Mater. 2006, 131, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Rosa, S.; Laranjeira, C.M.; Riela, H.G.; Favere, V.T. Cross-linked quaternary chitosan as an adsorbent for the removal of the reactive dye from aqueous solutions. J. Hazard. Mater. 2008, 155, 253–260. [Google Scholar] [CrossRef]
- Rojasa, G.; Silvaa, J.; Floresb, J.A.; Rodriguezb, A.L.; Lyc, M.; Maldonadob, H. Adsorption of chromium onto cross-linked chitosan. Sep. Purif. Technol. 2005, 44, 31–36. [Google Scholar] [CrossRef]
- Qi, L.F.; Xu, Z.R. Lead sorption from aqueous solutions on chitosan nanoparticles. Colloid. Surface. A 2004, 251, 183–190. [Google Scholar] [CrossRef]
- Lu, S.G.; Gibb, S.W.; Cochrane, E. Effective removal of zinc ions from aqueous solutions using crab carapace biosorbent. J. Hazard. Mater. 2007, 149, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.C.; Zhu, Z.S.; Zhang, Q.M. Mechanism study of adsorbtion Co2+ on cross-linked chitosan. Ion. Exch. Adsorpt. 2005, 21, 452–460. [Google Scholar]
- Vasconcelos, H.L.; Camargo, T.P.; Gonffcalves, N.S.; Neves, A.; Laranjeira, C.M.; Favere, V.T. Chitosan crosslinked with a metal complexing agent: Synthesis, characterization and copper(II) ions adsorption. React. Funct. Polym. 2008, 68, 572–579. [Google Scholar] [CrossRef]
- Vieira, R.S.; Guibal, E.; Silva, E.A.; Beppu, M.M. Adsorption and desorption of binary mixtures of copper and mercury ions on natural and cross-linked chitosan membranes. Adsorption 2007, 13, 603–611. [Google Scholar] [CrossRef]
- Wan Ngah, W.S.; Ghani, S.A.; Kamari, A. Adsorption behaviour of Fe(II) and Fe(III) ions in aqueous solution on chitosan and cross-linked chitosan beads. Bioresource Technol. 2005, 96, 443–450. [Google Scholar] [CrossRef]
- Tong, H.; Yao, S.N. Study on micro-morphology and structure of crab and crayfish shell. J. Anal. Sci. 1997, 13, 206–209. [Google Scholar]
- Tudor, E.A.; Gryte, C.C.; Harris, C. Seashell: Detoxifying agents for metal contamination waters. Water Air Soil Pollut. 2006, 173, 209–242. [Google Scholar] [CrossRef]
- An, H.K.; Park, B.Y.; Kim, D.S. Crab shell for the removal of heavy metals from aqueous solution. Water Res. 2001, 35, 3551–3556. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zheng, X.; Li, B.; Zhu, B.; Kuang, R.; Kuang, X.; Xu, B.; Ma, M. Crayfish Carapace Micro-powder (CCM): A Novel and Efficient Adsorbent for Heavy Metal Ion Removal from Wastewater. Water 2010, 2, 257-272. https://doi.org/10.3390/w2020257
Zheng X, Li B, Zhu B, Kuang R, Kuang X, Xu B, Ma M. Crayfish Carapace Micro-powder (CCM): A Novel and Efficient Adsorbent for Heavy Metal Ion Removal from Wastewater. Water. 2010; 2(2):257-272. https://doi.org/10.3390/w2020257
Chicago/Turabian StyleZheng, Xiaodong, Bin Li, Bo Zhu, Rui Kuang, Xuan Kuang, Baoli Xu, and Meihu Ma. 2010. "Crayfish Carapace Micro-powder (CCM): A Novel and Efficient Adsorbent for Heavy Metal Ion Removal from Wastewater" Water 2, no. 2: 257-272. https://doi.org/10.3390/w2020257




