Effect of Tidal Cycling Rate on the Distribution and Abundance of Nitrogen-Oxidizing Bacteria in a Bench-Scale Fill-and-Drain Bioreactor
Abstract
:1. Introduction
2. Materials and Methods
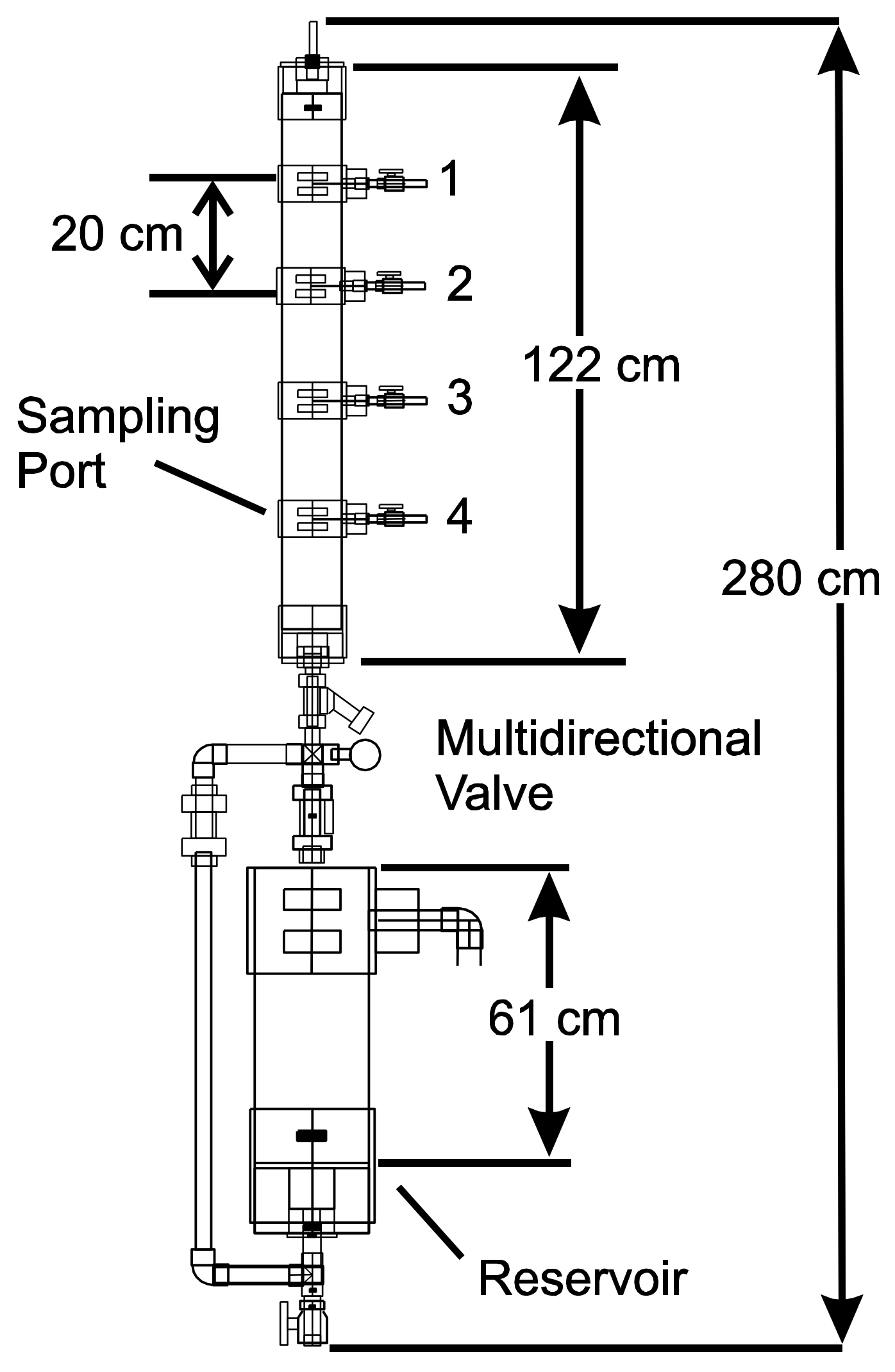
2.1. Reactor Design
2.2. Experiments
2.3. Microbiological Sampling and Analysis
2.4. Water Sampling and Analysis
3. Results
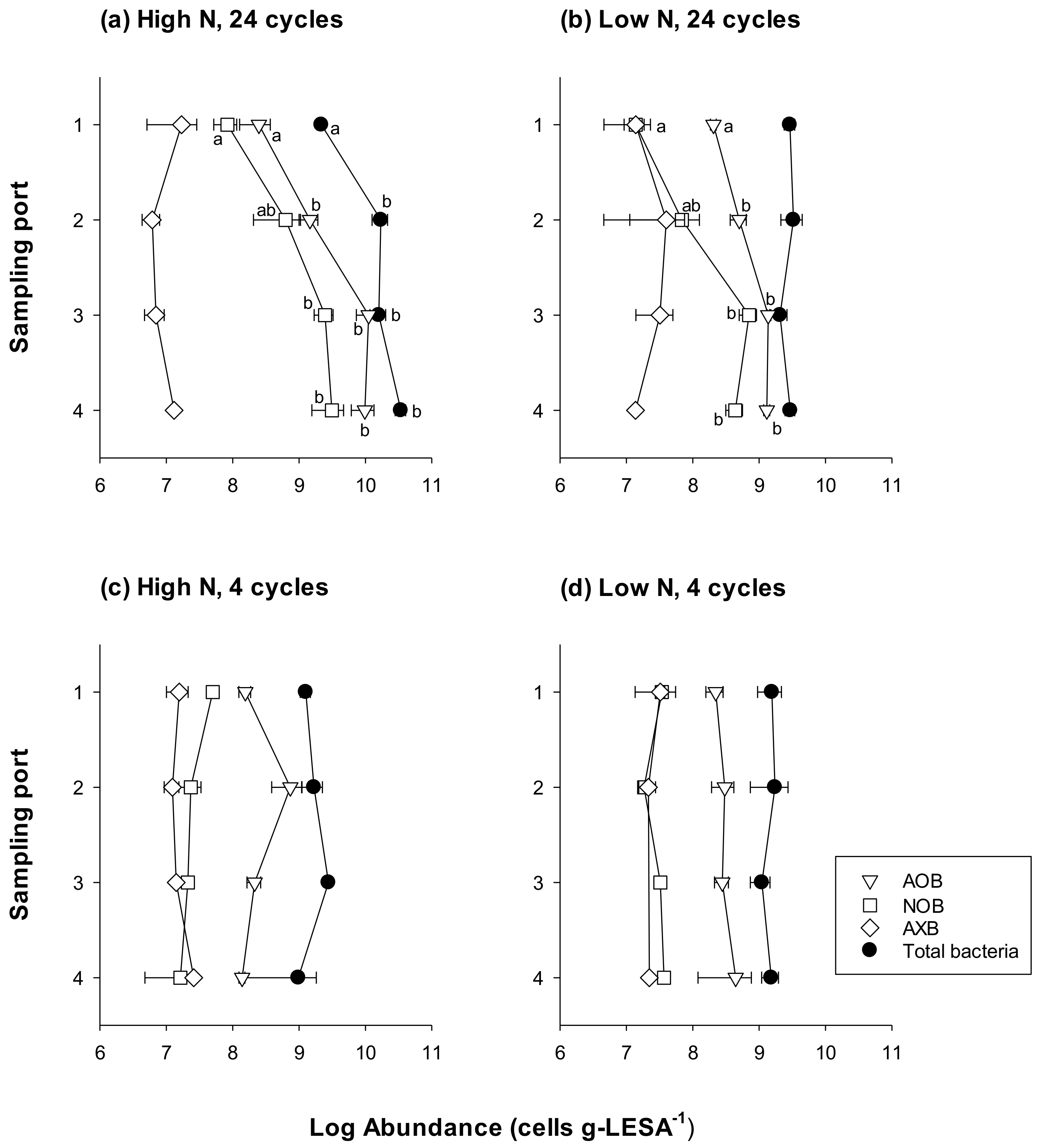
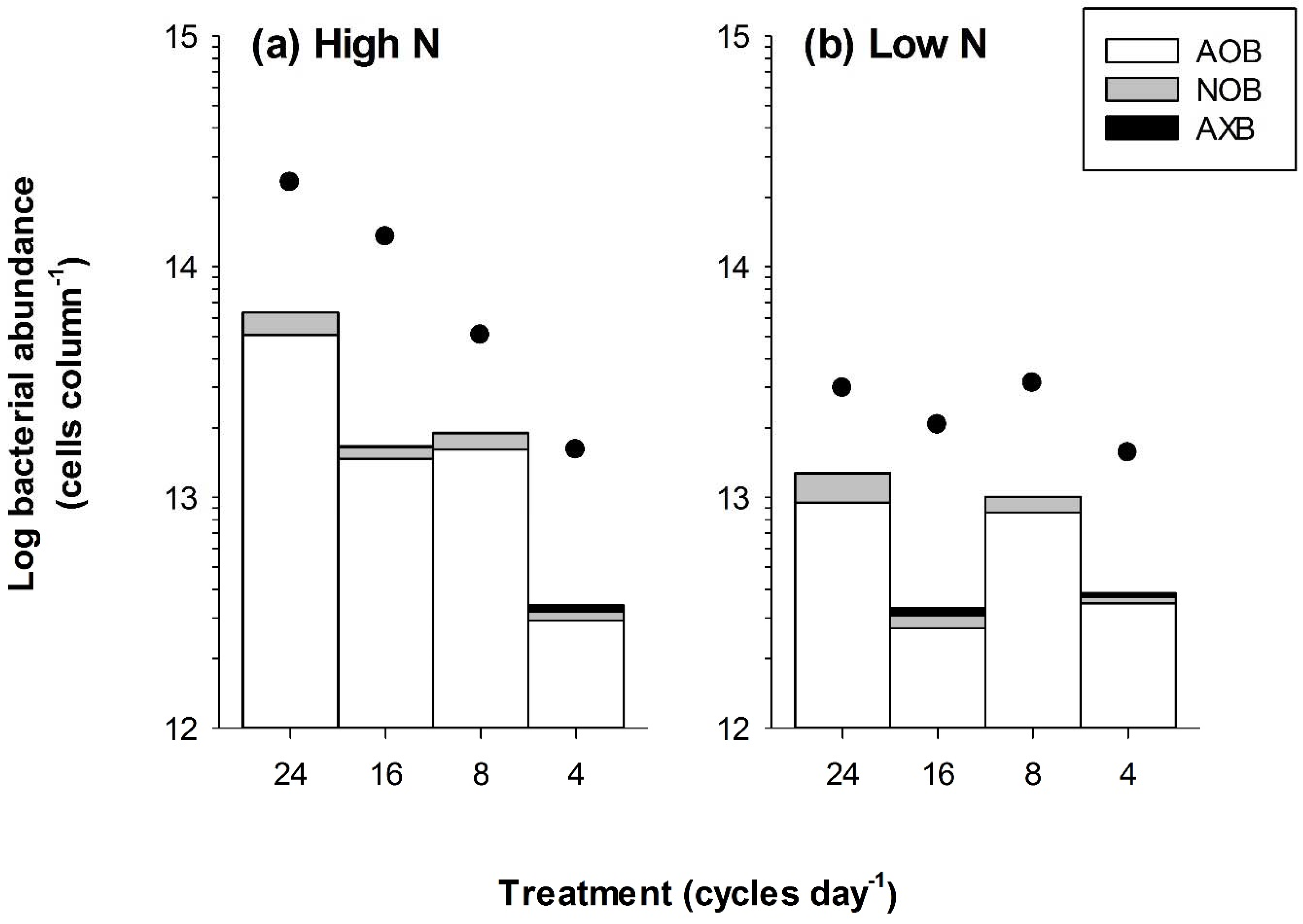
3.1. Bacterial Abundance
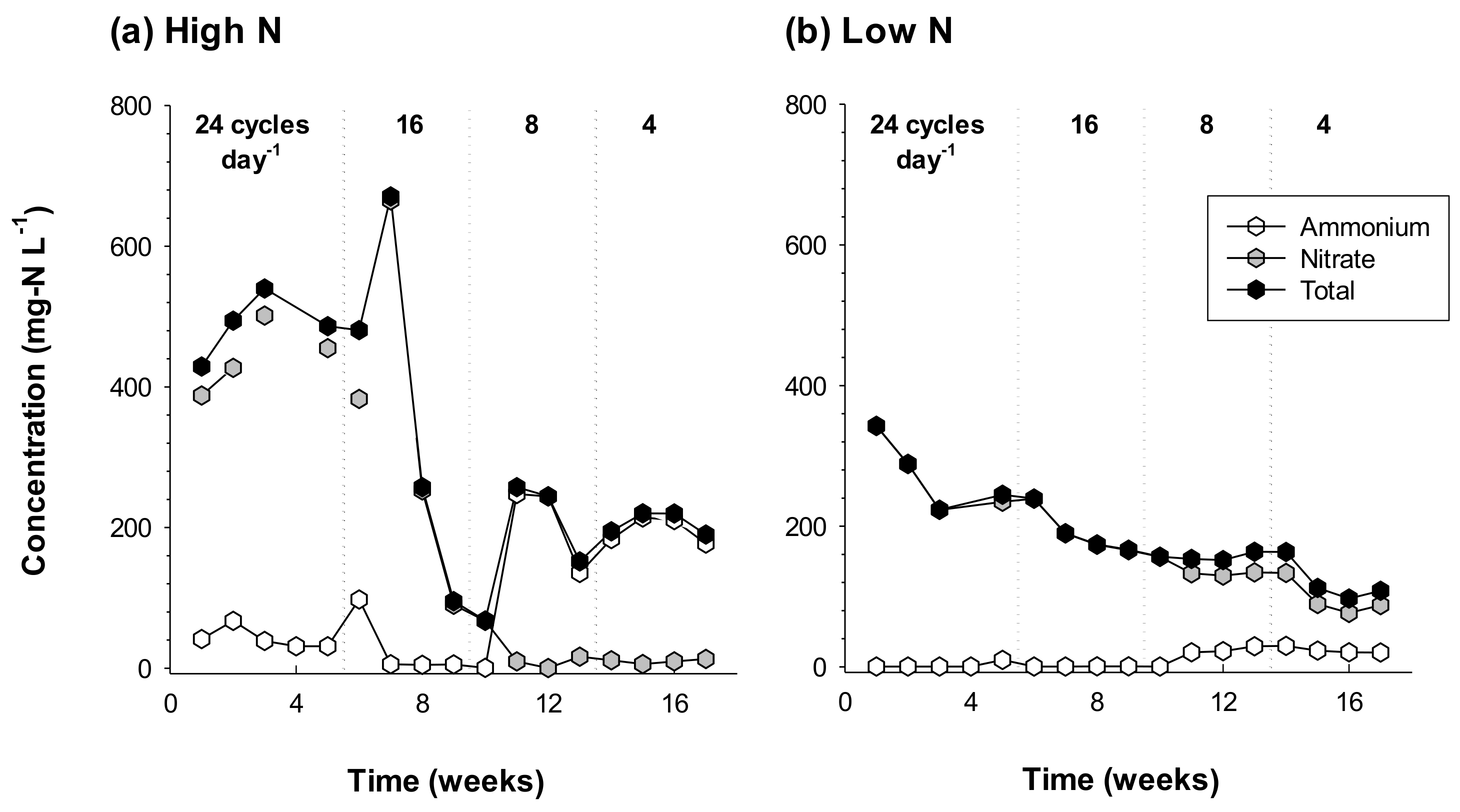
3.2. Water Chemistry
3.3. Nitrogen Removal Efficiency
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gleick, P.H. Water use. Annu. Rev. Environ. Resour. 2003, 28, 275–314. [Google Scholar] [CrossRef]
- Galloway, J.N.; Cowling, E.B. Reactive nitrogen and the world: 200 years of change. Ambio 2002, 31, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Vitousek, P.M.; Aber, J.D.; Howarth, R.W.; Likens, G.E.; Matson, P.A.; Schindler, D.W.; Schlesinger, W.H.; Tilman, D. Human alteration of the global nitrogen cycle: Sources and consequences. Ecol. Appl. 1997, 7, 737–750. [Google Scholar] [CrossRef]
- Zhu, G.; Peng, Y.; Li, B.; Guo, J.; Yang, Q.; Wang, S. Biological removal of nitrogen from wastewater. In Reviews of Environmental Contamination and Toxicology; Whitacre, D.M., Ed.; Springer: New York, NY, USA, 2008; pp. 159–195. [Google Scholar]
- United States Environmental Protection Agency (EPA). Manual for Nitrogen Control; EPAl625/R-; USEPA: Washington, DC, USA, 1993.
- United States Environmental Protection Agency (EPA). Toxicity Reduction Evaluation Guidance for Municipal Wastewater Treatment Plants; USEPA: Washington, DC, USA, 1999.
- Hatzenpichler, R. Diversity, physiology, and niche differentiation of ammonia-oxidizing archaea. Appl. Environ. Microbiol. 2012, 78, 7501–7510. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Sun, Q.; Wen, D.; Tang, X. Abundance of ammonia-oxidizing bacteria and archaea in industrial and domestic wastewater treatment systems. FEMS Microbiol. Ecol. 2012, 80, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Jetten, M.S.M.; Wagner, M.; Fuerst, J.; van Loosdrecht, M.; Kuenen, G.; Strous, M. Microbiology and application of the anaerobic ammonium oxidation (’anammox’) process. Curr. Opin. Biotechnol. 2001, 12, 283–288. [Google Scholar] [CrossRef]
- Tsushima, I.; Ogasawara, Y.; Kindaichi, T.; Satoh, H.; Okabe, S. Development of high-rate anaerobic ammonium-oxidizing (anammox) biofilm reactors. Water Res. 2007, 41, 1623–1634. [Google Scholar] [CrossRef] [PubMed]
- Van de Graaf, A.A.; Mulder, A.; Debruijn, P.; Jetten, M.S.M.; Robertson, L.A.; Kuenen, J.G. Anaerobic oxidation of ammonium is a biologically mediated process. Appl. Environ. Microbiol. 1995, 61, 1246–1251. [Google Scholar] [PubMed]
- Brix, H. Gas-exchange through the soil atmosphere interphase and through dead culms of Phragmites australis in a constructed reed bed receiving domestic sewage. Water Res. 1990, 24, 259–266. [Google Scholar] [CrossRef]
- Luederitz, V.; Eckert, E.; Lange-Weber, M.; Lange, A.; Gersberg, R.M. Nutrient removal efficiency and resource economics of vertical flow and horizontal flow constructed wetlands. Ecol. Eng. 2001, 18, 157–171. [Google Scholar] [CrossRef]
- Pan, J.; Zhang, H.; Li, W.; Ke, F. Full-scale experiment on domestic wastewater treatment by combining artificial aeration vertical- and horizontal-flow constructed wetlands system. Water Air Soil Pollut. 2012, 223, 5673–5683. [Google Scholar] [CrossRef]
- Tuncsiper, B. Nitrogen removal in a combined vertical and horizontal subsurface-flow constructed wetland system. Desalination 2009, 247, 466–475. [Google Scholar] [CrossRef]
- Austin, D.; Nivala, J. Energy requirements for nitrification and biological nitrogen removal in engineered wetlands. Ecol. Eng. 2009, 35, 184–192. [Google Scholar] [CrossRef]
- Sun, G.; Gray, K.R.; Biddlestone, A.J.; Cooper, D.J. Treatment of agricultural wastewater in a combined tidal flow-downflow reed bed system. Water Sci. Technol. 1999, 40, 139–146. [Google Scholar]
- Austin, D. Influence of cation exchange capacity (CEC) in a tidal flow, flood and drain wastewater treatment wetland. Ecol. Eng. 2006, 28, 35–43. [Google Scholar] [CrossRef]
- Austin, D.; Lohan, E.; Verson, E. Nitrification and denitrification in a tidal vertical flow wetland plot. In Proceedings of the Water Environment Technical Conference; Water Environment Federation: Los Angeles, CA, USA, 2003. [Google Scholar]
- United States Environmental Protection Agency (EPA). Method 9081—Cation Exchange Capacity of Soils; EPA: Washington, DC, USA, 1986.
- Tokunaga, T.K.; Olson, K.R.; Wan, J. Moisture characteristics of Hanford gravels: Bulk, grain-surface, and intragranular components. Vadose Zone J. 2003, 2, 322–329. [Google Scholar] [CrossRef]
- Abou-Elela, S.I.; Hellal, M.S. Municipal wastewater treatment using vertical flow constructed wetlands planted with Canna, Phragmites and Cyprus. Ecol. Eng. 2012, 47, 209–213. [Google Scholar] [CrossRef]
- Boursier, H.; Beline, F.; Paul, E. Piggery wastewater characterisation for biological nitrogen removal process design. Bioresour. Technol. 2005, 96, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Battistelli, J.M. Distribution of Nitrifying Bacteria under Fluctuating Environmental Conditions. Ph.D. Dissertation, University of Virginia, Charlottesville, VA, USA, 2012. [Google Scholar]
- Wright, S.F. Serology and Conjugation of Antibodies. In Methods of Soil Analysis: Part 2—Microbiological and Biochemical Properties; SSSA Book Series SV–5.2; Soil Science Society of America: Madison, WI, USA, 1994; pp. 593–618. [Google Scholar]
- Higgins, C. The Potential for Eutrophication Mitigation from Aquaculture of the Native Oyster Crassostrea virginica, in Chesapeake Bay: Quantitative Assessment of an Ecosystem Service. Ph.D. Dissertation, Virginia Commonwealth University, Richmond, VA, USA, 2011. [Google Scholar]
- Krasnits, E.; Friedler, E.; Sabbah, I.; Beliavski, M.; Tarre, S.; Green, M. Spatial distribution of major microbial groups in a well established constructed wetland treating municipal wastewater. Ecol. Eng. 2009, 35, 1085–1089. [Google Scholar] [CrossRef]
- Woznica, A.; Nowak, A.; Beimfohr, C.; Karczewski, J.; Bernas, T. Monitoring structure and activity of nitrifying bacterial biofilm in an automatic biodetector of water toxicity. Chemosphere 2010, 78, 1121–1128. [Google Scholar] [CrossRef] [PubMed]
- Schmid, M.; Walsh, K.; Webb, R.; Rijpstra, W.I.C.; van de Pas-Schoonen, K.; Verbruggen, M.J.; Hill, T.; Moffett, B.; Fuerst, J.; Schouten, S.; et al. Candidatus “Scalindua brodae”, sp. nov., Candidatus “Scalindua wagneri”, sp. nov., two new species of anaerobic ammonium oxidizing bacteria. Syst. Appl. Microbiol. 2003, 26, 529–538. [Google Scholar] [CrossRef] [PubMed]
- Neef, A.; Amann, R.; Schlesner, H.; Schleifer, K.H. Monitoring a widespread bacterial group: In situ detection of planctomycetes with 16S rRNA-targeted probes. Microbiology 1998, 144, 3257–3266. [Google Scholar] [CrossRef] [PubMed]
- Brandsma, J.; van de Vossenberg, J.; Risgaard-Petersen, N.; Schmid, M.C.; Engstrom, P.; Eurenius, K.; Hulth, S.; Jaeschke, A.; Abbas, B.; Hopmans, E.C.; et al. A multi-proxy study of anaerobic ammonium oxidation in marine sediments of the Gullmar Fjord, Sweden. Environ. Microbiol. Rep. 2011, 3, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Hao, X.; Wang, Q.; Zhang, X.; Cao, Y.; van Mark Loosdrecht, C.M. Experimental evaluation of decrease in bacterial activity due to cell death and activity decay in activated sludge. Water Res. 2009, 43, 3604–3612. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.P.; Rector, T.; Reid-Black, K.; Hummerick, M.; Strayer, R.; Birmele, M.; Roberts, M.S.; Garland, J.L. Redox control bioreactor: A unique biological water processor. Biotechnol. Bioeng. 2008, 99, 830–845. [Google Scholar] [CrossRef] [PubMed]
- Norton, J.M. Diversity and environmental distribution of ammonia-oxidizing bacteria. In Nitrification; Ward, B.B., Arp, D.J., Klotz, M.G., Eds.; ASM Press: Washington, DC, USA, 2011; pp. 39–56. ISBN 9781843395461. [Google Scholar]
- Geets, J.; Boon, N.; Verstraete, W. Strategies of aerobic ammonia-oxidizing bacteria for coping with nutrient and oxygen fluctuations. FEMS Microbiol. Ecol. 2006, 58, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Dytczak, M.A.; Londry, K.L.; Oleszkiewicz, J.A. Activated sludge operational regime has significant impact on the type of nitrifying community and its nitrification rates. Water Res. 2008, 42, 2320–2328. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Zhang, D.; Austin, D.; Dong, R.; Pang, C. Evaluation of a lab-scale tidal flow constructed wetland performance: Oxygen transfer capacity, organic matter and ammonium removal. Ecol. Eng. 2011, 37, 1789–1795. [Google Scholar] [CrossRef]
- Blackburne, R.; Yuan, Z.; Keller, J. Partial nitrification to nitrite using low dissolved oxygen concentration as the main selection factor. Biodegradation 2008, 19, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Wyffels, S.; Van Hulle, S.W.H.; Boeckx, P.; Volcke, E.I.P.; Van Cleemput, O.; Vanrolleghem, P.A.; Verstraete, W. Modeling and simulation of oxygen-limited partial nitritation in a membrane-assisted bioreactor (MBR). Biotechnol. Bioeng. 2004, 86, 531–542. [Google Scholar] [CrossRef] [PubMed]
- Strous, M.; Kuenen, J.G.; Jetten, M.S.M. Key physiology of anaerobic ammonium oxidation. Appl. Environ. Microbiol. 1999, 65, 3248–3250. [Google Scholar] [PubMed]
- Dapena-Mora, A.; Fernandez, I.; Campos, J.L.; Mosquera-Corral, A.; Mendez, R.; Jetten, M.S.M. Evaluation of activity and inhibition effects on anammox process by batch tests based on the nitrogen gas production. Enzyme Microb. Technol. 2007, 40, 859–865. [Google Scholar] [CrossRef]
- Fux, C.; Huang, D.; Monti, A.; Siegrist, H. Difficulties in maintaining long-term partial nitritation of ammonium-rich sludge digester liquids in a moving-bed biofilm reactor (MBBR). Water Sci. Technol. 2004, 49, 53–60. [Google Scholar] [PubMed]
- Jin, R.-C.; Yang, G.-F.; Yu, J.-J.; Zheng, P. The inhibition of the anammox process: A review. Chem. Eng. J. 2012, 197, 67–79. [Google Scholar] [CrossRef]
- Lackner, S.; Gilbert, E.M.; Vlaeminck, S.E.; Joss, A.; Horn, H.; van Loosdrecht, M.C.M. Full-scale partial nitritation/anammox experiences–An application survey. Water Res. 2014, 55, 292–303. [Google Scholar] [CrossRef] [PubMed]
- Tal, Y.; Watts, J.E.M.; Schreier, H.J. Anaerobic ammonium-oxidizing (anammox) bacteria and associated activity in fixed-film biofilters of a marine recirculating aquaculture system. Appl. Environ. Microbiol. 2006, 72, 2896–2904. [Google Scholar] [CrossRef] [PubMed]
- Daverey, A.; Chei, P.C.; Dutta, K.; Lin, J.-G. Statistical analysis to evaluate the effects of temperature and pH on anammox activity. Int. Biodeterior. Biodegrad. 2015, 102, 89–93. [Google Scholar] [CrossRef]
- Liu, S.T.; Horn, H.; Mueller, E. A systematic insight into a single-stage deammonification process operated in granular sludge reactor with high-loaded reject-water: Characterization and quantification of microbiological community. J. Appl. Microbiol. 2013, 114, 339–351. [Google Scholar] [CrossRef] [PubMed]
- Monballiu, A.; Desmidt, E.; Ghyselbrecht, K.; De Clippeleir, H.; Van Hulle, S.W.H.; Verstraete, W.; Meesschaert, B. Enrichment of anaerobic ammonium oxidizing (anammox) bacteria from OLAND and conventional sludge: Features and limitations. Sep. Purif. Technol. 2013, 104, 130–137. [Google Scholar] [CrossRef]
- Okamoto, H.; Kawamura, K.; Nishiyama, T.; Fujii, T.; Furukawa, K. Development of a fixed-bed anammox reactor with high treatment potential. Biodegradation 2013, 24, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Strous, M.; VanGerven, E.; Zheng, P.; Kuenen, J.G.; Jetten, M.S.M. Ammonium removal from concentrated waste streams with the anaerobic ammonium oxidation (anammox) process in different reactor configurations. Water Res. 1997, 31, 1955–1962. [Google Scholar] [CrossRef]
- Third, K.A.; Paxman, J.; Schmid, M.; Strous, M.; Jetten, M.S.M.; Cord-Ruwisch, R. Enrichment of anammox from activated sludge and its application in the CANON process. Microb. Ecol. 2005, 49, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, I.; Dosta, J.; Fajardo, C.; Campos, J.L.; Mosquera-Corral, A.; Mendez, R. Short- and long-term effects of ammonium and nitrite on the anammox process. J. Environ. Manag. 2012, 95, S170–S174. [Google Scholar] [CrossRef] [PubMed]
- Gisvold, B.; Ødegaard, H.; Füllesdal, M. Enhanced removal of ammonium by combined nitrification/adsorption in expanded clay aggregate filters. Water Sci. Technol. 2000, 41, 409–416. [Google Scholar]
- Yan, A.; Liu, C.; Liu, Y.; Xu, F. Effect of ion exchange on the rate of aerobic microbial oxidation of ammonium in hyporheic zone sediments. Environ. Sci. Pollut. Res. 2018, 25, 8880–8887. [Google Scholar] [CrossRef] [PubMed]
- Bellucci, M.; Ofiţeru, I.D.; Graham, D.W.; Head, I.M.; Curtis, T.P. Low-dissolved-oxygen nitrifying systems exploit ammonia-oxidizing bacteria with unusually high yields. Appl. Environ. Microbiol. 2011, 77, 7787–7796. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-D.; Wells, G.F.; Bae, H.; Criddle, C.S.; Francis, C.A. Occurrence of ammonia-oxidizing archaea in wastewater treatment plant bioreactors. Appl. Environ. Microbiol. 2006, 72, 5643–5647. [Google Scholar] [CrossRef] [PubMed]
- Sauder, L.A.; Peterse, F.; Schouten, S.; Neufeld, J.D. Low-ammonia niche of ammonia-oxidizing archaea in rotating biological contactors of a municipal wastewater treatment plant. Environ. Microbiol. 2012, 14, 2589–2600. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.Y.; Tsang, Y.F.; Chua, H.; Sin, S.N.; Cui, L.H. Performance study of vegetated sequencing batch coal slag bed treating domestic wastewater in suburban area. Bioresour. Technol. 2008, 99, 3774–3781. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Feng, J.; Ouyang, Y.; Deng, P. Removal of nutrients from septic effluent with re-circulated hybrid tidal flow constructed wetland. Ecol. Eng. 2012, 46, 112–115. [Google Scholar] [CrossRef]
- Hu, Y.; Zhao, Y.; Rymszewicz, A. Robust biological nitrogen removal by creating multiple tides in a single bed tidal flow constructed wetland. Sci. Total Environ. 2014, 470, 1197–1204. [Google Scholar] [CrossRef] [PubMed]

| Microbial Group | 24 Cycles Day−1 | 4 Cycles Day−1 | ||
|---|---|---|---|---|
| High N | Low N | High N | Low N | |
| Total bacteria | <0.001 * | 0.57 | 0.37 | 0.96 |
| AOB | 0.001 * | <0.001 * | 0.34 | 0.94 |
| NOB | 0.003 * | 0.004 * | 0.34 | 0.90 |
| AXB | 0.60 * | 0.94 | 0.49 | 0.53 |
| Treatment (Cycles Day−1) | Nitrogen Loading | |
|---|---|---|
| High N | Low N | |
| 24 | 79.3 ± 3.4 | 63.3 ± 4.5 |
| 16 | 84.9 ± 8.0 | 70.0 ± 0.5 |
| 8 | 87.4 ± 4.0 | 70.5 ± 0.6 |
| 4 | 86.6 ± 1.3 | 80.3 ± 1.5 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Battistelli, J.M.; Franklin, R.B.; Mills, A.L. Effect of Tidal Cycling Rate on the Distribution and Abundance of Nitrogen-Oxidizing Bacteria in a Bench-Scale Fill-and-Drain Bioreactor. Water 2018, 10, 492. https://doi.org/10.3390/w10040492
Battistelli JM, Franklin RB, Mills AL. Effect of Tidal Cycling Rate on the Distribution and Abundance of Nitrogen-Oxidizing Bacteria in a Bench-Scale Fill-and-Drain Bioreactor. Water. 2018; 10(4):492. https://doi.org/10.3390/w10040492
Chicago/Turabian StyleBattistelli, Joseph M., Rima B. Franklin, and Aaron L. Mills. 2018. "Effect of Tidal Cycling Rate on the Distribution and Abundance of Nitrogen-Oxidizing Bacteria in a Bench-Scale Fill-and-Drain Bioreactor" Water 10, no. 4: 492. https://doi.org/10.3390/w10040492





