2.1. Working Hypothesis
Immune monitoring, in general, and ELISPOT in particular, aims at establishing accurately the frequencies of antigen-specific T cells
in vivo. Ideally, T cell assays are performed with freshly isolated PBMC as soon as possible after the blood draw. During the 24 h duration of the ELISPOT assays antigen stimulation does not result in significant
in vitro expansion of the antigen-specific T cells. Therefore, the frequencies measured in freshly isolated cells match the sought after
in vivo frequency. Test results obtained with freshly isolated PBMC, therefore, can be considered as the baseline against which variations of PBMC handling, such as cryopreservation or resting, can be compared. When PBMC are cryopreserved and thawed according to protocols that we have established, the frequencies measured in freshly isolated PBMC “
ex vivo”, and in the thawed PBMC were shown to be identical for CD4 and CD8 cells [
15] which established that PBMC can be cryopreserved without loss of T cell functionality. Notably, this equal performance of non-cryopreserved “
ex vivo” and of cryopreserved PBMC was seen without resting when the cells were plated into the assay right after thawing. In the present study, we selected CEF peptides to stimulate CD8 cells [
28,
29], and mumps antigen to stimulate CD4 cells. The CEF pool consists of 32 immunodominant peptides of cytomegalo-, Epstein Barr- and flu virus.
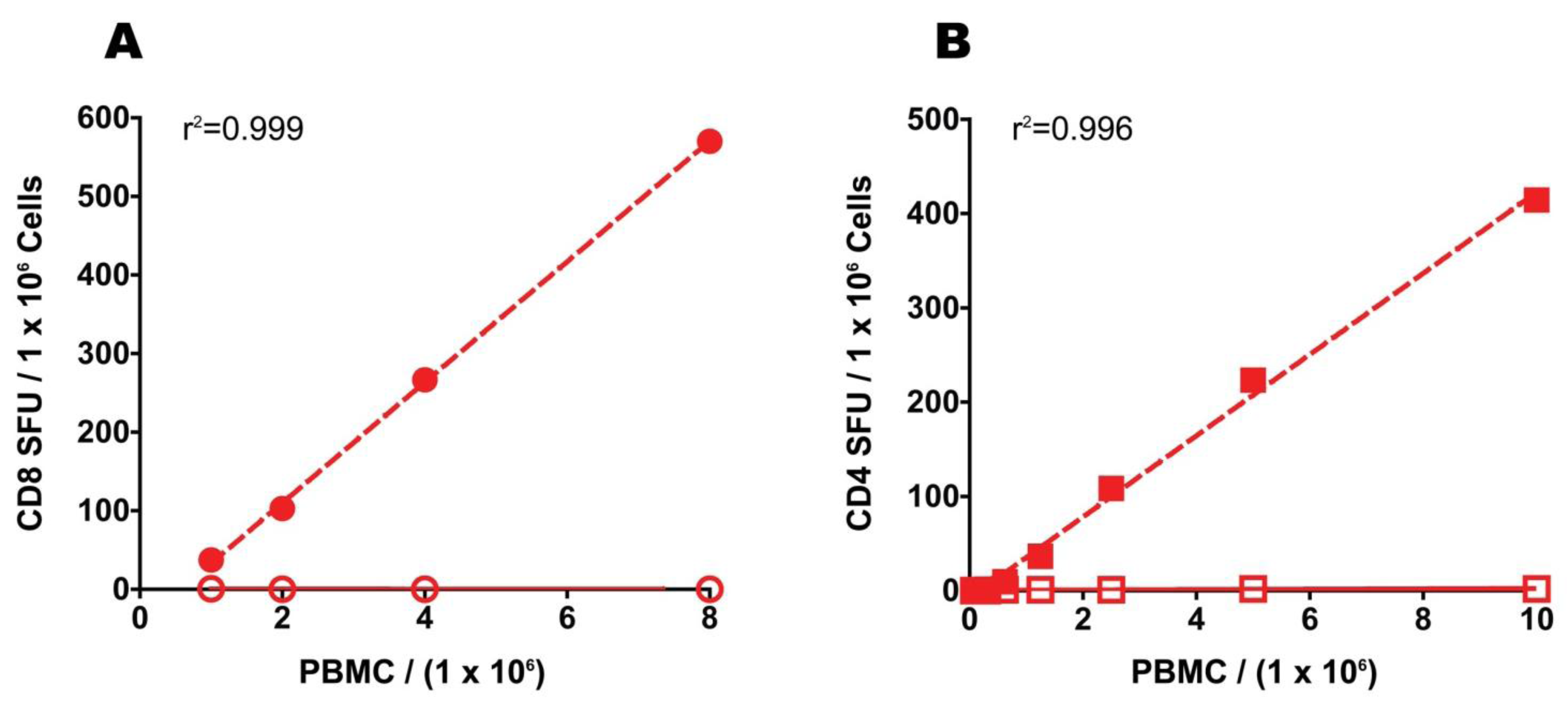
Figure 1A,B shows cell separation experiments that confirm that CEF peptides activate CD8, and mumps antigen activates CD4 cells, respectively.
Figure 1C,D also shows that for CEF- and mumps-antigen-elicited CD4 and CD8 cell recall responses similar IFN-γ spot counts were obtained when non-cryopreserved PBMC were tested
ex vivo within 2 h after the blood draw, or after freeze-thawing, without resting. In this paper we refer to non-rested freeze-thawed cells as “fresh” to distinguish them from “rested” and “
ex vivo” PBMC. In extensive subsequent studies we have confirmed for a multitude of other antigens that recall CD4 and CD8 memory cells in non-cryopreserved (“
ex vivo”) PBMC and freeze-thawed PBMC without resting (“fresh”) provide comparable results, and we have shown that in the thawed PBMC the numbers of apoptotic cells were less than 5% when measured within 30 minutes after thawing [
30]. A basic assumption of this paper therefore is that freshly thawed non-rested (“fresh”) PBMC show equal performance to non-cryopreserved
ex vivo PBMC and can be used to test the impact of resting in ELISPOT. The basic question that we address here therefore is, does “overnight resting” reliably improve ELISPOT assay performance relative to test results obtained on freshly thawed PBMC?
Figure 1.
CD8 cells respond to CEF peptides (A), and CD4 cells to mumps antigen (B): these responses seen ex vivo are preserved after freeze thawing (C&D). To determine T cell subsets responding to the CEF and mumps antigens, magnetic affinity bead-based separation was performed depleting more than 95% of CD4 or CD8 T cells, as specified. The PBMC and cell fractions were stimulated with CEF peptides (A) or mumps antigen (B). For each condition, triplicate wells were tested with the mean and standard deviation shown. The data shown are from one of two experiments performed with similar results. Freshly isolated, non-cryopreserved PBMC (ex vivo) and cryopreserved PBMC that were tested directly without resting following thawing (fresh) were tested in an IFN-γ ELISPOT assay against CEF peptides (C) and mumps antigen (D). Data points obtained for individual donors are connected with a line. Each data point represents the mean of triplicate antigen-stimulated wells. Standard deviations are not shown being between 5 and 20% of the mean. For each sample, fresh or freeze-thawed, the medium background was less than 10 spots per well. Nonparametric Wilcoxon signed-rank test was used to compare matched ex vivo vs. fresh responses with a p-value ≤ 0.05 being considered significant.
Figure 1.
CD8 cells respond to CEF peptides (A), and CD4 cells to mumps antigen (B): these responses seen ex vivo are preserved after freeze thawing (C&D). To determine T cell subsets responding to the CEF and mumps antigens, magnetic affinity bead-based separation was performed depleting more than 95% of CD4 or CD8 T cells, as specified. The PBMC and cell fractions were stimulated with CEF peptides (A) or mumps antigen (B). For each condition, triplicate wells were tested with the mean and standard deviation shown. The data shown are from one of two experiments performed with similar results. Freshly isolated, non-cryopreserved PBMC (ex vivo) and cryopreserved PBMC that were tested directly without resting following thawing (fresh) were tested in an IFN-γ ELISPOT assay against CEF peptides (C) and mumps antigen (D). Data points obtained for individual donors are connected with a line. Each data point represents the mean of triplicate antigen-stimulated wells. Standard deviations are not shown being between 5 and 20% of the mean. For each sample, fresh or freeze-thawed, the medium background was less than 10 spots per well. Nonparametric Wilcoxon signed-rank test was used to compare matched ex vivo vs. fresh responses with a p-value ≤ 0.05 being considered significant.
![Cells 01 00409 g001]()
2.2. PBMC Donors with Low and High Responder Status to CEF Peptide Pool and Mumps Antigen
Cryopreserved PBMC of twenty five donors were randomly selected from the ePBMC donor library provided by Cellular Technology Limited (CTL), Shaker Heights, OH, USA. These PBMC were obtained from healthy donors by leucapheresis, and cells of each donor had been frozen in hundreds of identical aliquots allowing replication of experiments using the very same cell material. Hundreds of HLA-typed and immune characterized donors constitute the library. The PBMC were tested “fresh” for reactivity to CEF peptide pool, and to mumps antigen. For both antigens, the response levels ranged from undetectable (defined as the difference between spot counts in triplicate medium control wells
vs. triplicate antigen-stimulated wells not being significantly different based on t-test evaluation) to high, reaching up to 400 antigen-elicited spots forming units (SFU) per one million PBMC over a background that was typically zero, and in no sample exceeded 10 spots per million. Because the background was negligibly low for all PBMC samples when tested “fresh” or “rested”, in the graphical representation of the data for this paper we will omit the background spots. Such a wide range of recall response levels to individual antigens within different human donors is typically seen for non-cryopreserved PBMC
ex vivo [
14,
15,
18,
20], even when donors are vaccinated at the same time and are tested at a given time point after vaccination [
18]. It is even characteristic for recall responses of inbred mice when all parameters of the immunization, the genetic background and environmental influences are kept constant [
4,
7,
9,
17]. For individual donors, the response levels for CEF and mumps were not linked: donors that displayed a high response level to CEF could be low or non-responders to mumps, and
vice versa (examples are shown in
Figure 4). In addition, donors that were low or non-responders to both antigens, responded vigorously to some others of the over 30 antigens for which all PBMC were tested (see ePBMC data base [
31]—the ePBMC library consists of a large selection of precharacterized PBMC samples with established HLA types and antigen reactivities). Therefore, when low response levels were detected to either CEF peptides or mumps antigen this reflects low frequency of CEF or mumps antigen-reactive T cells in the respective PBMC donor, and not a general lack of functionality in that particular PBMC sample caused by suboptimal freezing, storage, or thawing of the cells.
2.4. Resting of Cryopreserved PBMC from High-Responders Augments CEF-, but not Mumps Specific Responses
PBMC donors responding to antigen stimulation with more than 50 SFU per million PBMC were defined as high responders. For eight of nine CEF high responders resting resulted in increased SFU counts (
Figure 2B). With the exception of one donor for whom the increase was slightly more than two fold, the increase for all other donors was less than two fold. For one of these donors, fresh- and rested PBMC provided unchanged reactivity levels. Overall, the increase seen after resting reached statistical significance as judged by the Wilcoxon signed-rank test for CEF high responders with a
p-value of 0.002.
In measuring CD4 responses directed against mumps among the high responders, resting had no significant benefit (
Figure 3B). Of thirteen mumps high responders, three showed an approximate two-fold increase in mumps-induced SFU, while for the remaining donors, the spot counts were decreased (n = 8) or were similar (n = 2).
With the exception of CEF high responders, resting therefore had a variable and statistically insignificant effect on PBMC sample performance in ELISPOT assays. In occasional donors up to two fold increases were seen, in other donors comparable decreases in spot counts were observed.
We retested the PBMC of all 25 donors with and without resting to establish whether the gain or loss that we observed after resting for a given sample is an inherent feature of a given PBMC sample, or whether samples respond unpredictably to the 20 h
in vitro culture. In
Figure 4 the results are shown for four donors that are representative of all 25 donors. First, it can be seen that a sample that is a low responder to CEF can be a high responder to mumps, and
vice versa (donor 1
vs. donors 3 and 4). Donor 2, while being a low responder to both CEF and mumps is a high responder to candida and PPD (purified protein derivate) tuberculin (data not shown). Second, if a donor tested “fresh” as high or low responder to CEF or mumps in the first experiment, the same response level reproduced in the repeat experiment for both antigens. The inter-assay variation for the freshly thawed samples was not significant. Third, if after resting there was no significant gain seen for the CEF response in the first experiment, it was also not evident in the repeat experiment (donors 1 and 2), but if resting resulted in a moderate gain in the CEF response in the first experiment, a gain of similar magnitude was also seen in the repeat experiment (donors 2 and 3). Similarly, the decreased mumps response seen after resting for donors 1 and 3 reproduced in the repeat experiment, as did the unchanged mumps response levels for donors 2 and 4. Fourth,
Figure 4 illustrates that the increase for one antigen after resting can result in unchanged or decreased response levels to another antigen. Fifth, this figure illustrates that in the low responder category resting does not predictably increase reactivity levels. Since increases and decreases of the antigen reactivity after resting were reproducible in the repeat experiment, they appear to be an inherent propensity of the respective PBMC.
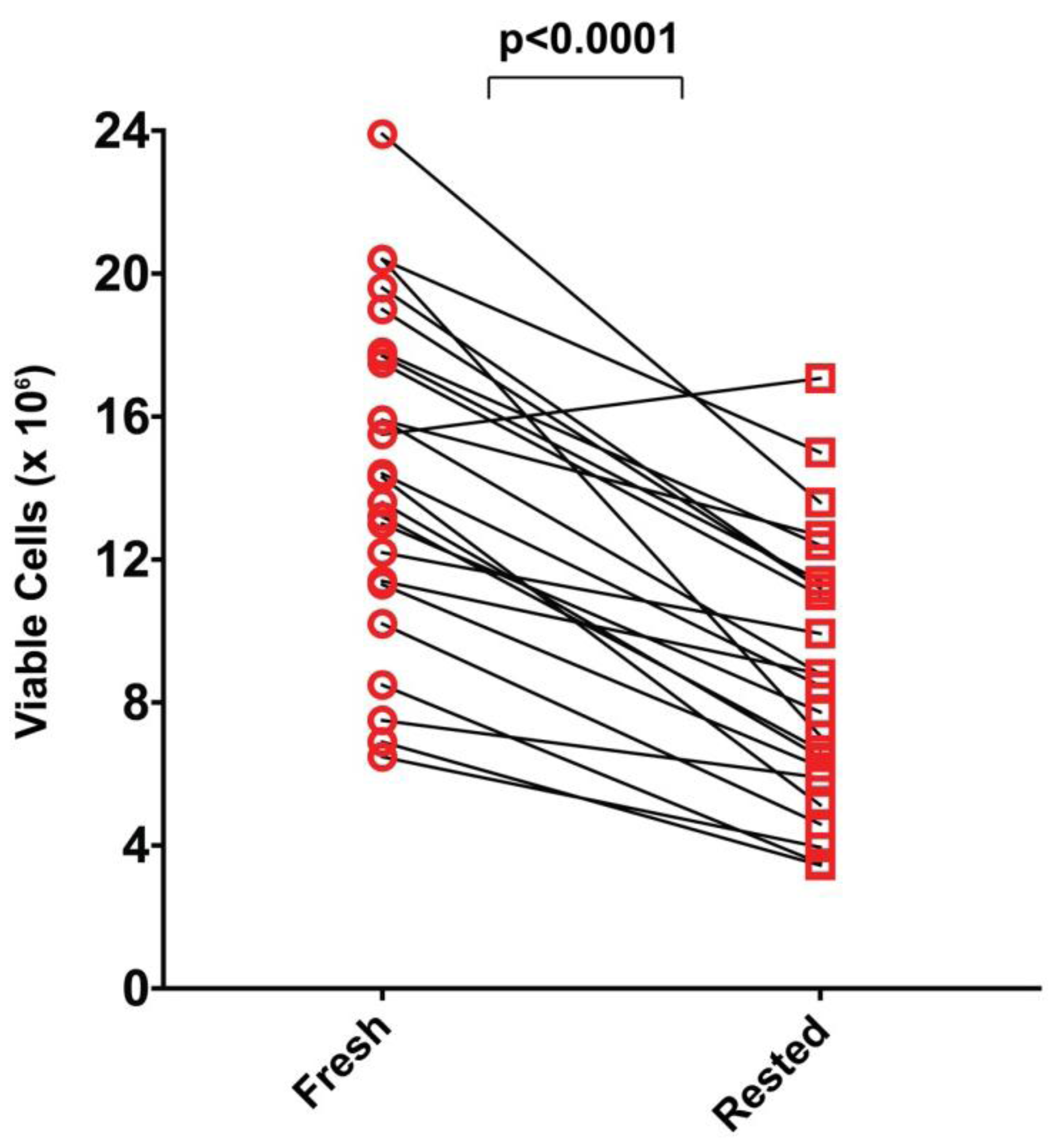
2.5. Resting of PBMC Is Associated with Significant Cell Loss
Without stimulation and exposure to specific growth/survival factors most primary cells die, frequently within a short time, in cell culture. Resting PBMC in medium for 20 h represents such a stimulation-free culture period for the different cell lineages contained within PBMC. It is also conceivable that some cells/cell-lineages that appear viable directly after thawing have been injured during the freeze-thaw process and undergo apoptosis during the resting period. To establish how many cells are lost during resting, we compared viable PBMC counts within 1 h after thawing of the PBMC (“fresh”) to viable cells recovered after the 20 h resting period. Acridine orange/ethidium bromide staining was used to distinguish between live and dead cells. As shown in
Figure 5, losses were seen for all but one of the 25 PBMC samples, and overall losses occurred at a similar rate, with about half of the cells lost after resting. The diminution seen was highly significant (
p < 0.0001) as determined by paired Wilcoxon signed-rank sum test. Because less than half of the originally seeded PBMC were recovered after resting for several samples, it is advisable to plan experiments that involve rested PBMC with two to three fold excess of PBMC than what is to be plated into the assay itself. Since PBMC are limiting for most clinical studies, these losses of PBMC need to be considered
vs. possible benefits of assay results obtained through resting.
Figure 4.
Gain or loss of function after resting is an inherent feature of PBMC samples. Responses to CEF peptide pool and mumps antigen were assessed with and without resting in two independent experiments for each donor. Data are shown for four donors who are fully representative of all the 25 donors tested. Black bars represent responses from freshly thawed cells while grey bars represent responses elicited by rested PBMC. Error bars depict the SD of the SFU from triplicate wells.
Figure 4.
Gain or loss of function after resting is an inherent feature of PBMC samples. Responses to CEF peptide pool and mumps antigen were assessed with and without resting in two independent experiments for each donor. Data are shown for four donors who are fully representative of all the 25 donors tested. Black bars represent responses from freshly thawed cells while grey bars represent responses elicited by rested PBMC. Error bars depict the SD of the SFU from triplicate wells.
Figure 5.
Comparison of viable PBMC recovered before and after resting. PBMC of each donor were counted by acridine orange/ethidium bromide staining within 1 h after thawing, and after a 20 h resting period. Live cell counts for each donor before and after resting are connected by a line. The data are from one experiment in which all 25 donors were tested, and are fully representative of the repeat experiment. The significance of the change was determined by the paired Wilcoxon signed-rank sum test.
Figure 5.
Comparison of viable PBMC recovered before and after resting. PBMC of each donor were counted by acridine orange/ethidium bromide staining within 1 h after thawing, and after a 20 h resting period. Live cell counts for each donor before and after resting are connected by a line. The data are from one experiment in which all 25 donors were tested, and are fully representative of the repeat experiment. The significance of the change was determined by the paired Wilcoxon signed-rank sum test.
2.6. Frequencies of CD4 and CD8 Cells Are Unchanged between Fresh and Rested PBMC
For CEF high-responders, we observed in rested samples increased frequencies of CD8 antigen-specific responses (see above,
Figure 2B), which could partly explain published claims [
24,
25]. This finding could be explained by a preferential survival of CD8 cells in such PBMC samples over the resting period while other cell types could have died more rapidly leading to a CD8 cell enrichment in the rested sample. Increased responses to CEF in conjunction with decreased mumps responses after resting (e.g., donor 3 in
Figure 4) might result from more rapid dying off of CD4 cells. To formally test this hypothesis, we used flow cytometry to monitor potential changes in the frequency of CD4
+ and CD8
+ T cell lineages following resting.
Phenotypic characterization of the freshly thawed and rested PBMC by flow cytometry did not show any statistically significant changes in CD8
+ or CD4
+ T cell subsets due to resting (
Figure 6). Importantly, no correlation existed between the moderate changes in the percentage of CD8
+ T cells in a given PBMC sample and the CEF peptide-elicited SFU response; similarly, slight increases/decreases in frequencies of CD4 cells after resting did not result in a proportional increase/decrease in the mumps recall response (data not shown). The changed T cell responses seen after resting, therefore, resulted from changes in T cell functionality, not from altered T cell frequencies within the tested sample.
Figure 6.
Frequencies of T cells and the CD4/CD8 subpopulations in PBMC before and after 20 h resting determined by flow cytometry. For all 25 donors, the percentages of CD3+, CD3+/CD4+, and CD3+/CD8+ cells were established once before (black bars), and once after (grey bars), resting. The mean and SD for the 25 PBMC of both groups are shown.
Figure 6.
Frequencies of T cells and the CD4/CD8 subpopulations in PBMC before and after 20 h resting determined by flow cytometry. For all 25 donors, the percentages of CD3+, CD3+/CD4+, and CD3+/CD8+ cells were established once before (black bars), and once after (grey bars), resting. The mean and SD for the 25 PBMC of both groups are shown.
2.7. Increasing the PBMC Numbers Plated in ELISPOT, Unlike Resting, Predictably Increases the Signal-to-Noise Performance of the Assay
In spite of the increased work load and the cell losses, resting has been advocated because it is presumed to improve the signal-to-noise performance of ELISPOT assays. As shown above, resting does not predictably have such an impact on low responders. Worse, for the low responders, rested samples could provide decreased antigen-induced spot counts, in particular for CD4 cells (e.g., donor 3 in
Figure 4).
Due to the unpredictable outcome of resting, and the need for two to three times more PBMC to accommodate the cell loss, we tested the hypothesis that the desired increase in signal-to-noise performance of the PBMC can be accomplished simply by increasing the numbers of freshly thawed PBMC plated into the ELISPOT assay. To evaluate this hypothesis directly, we plated unrested PBMC in two-fold serial dilutions and asked if observed CEF and mumps antigen-specific SFU show a linear increase proportional to the number of cells assayed. The data presented in
Figure 7 suggest that in the 0.1 to 0.8 million PBMC per well range there is a high degree of linearity between the numbers of PBMC plated and the observed SFU both in the CD8 (Panel A) and CD4 (Panel B) compartments. This linear relationship between spot counts and PBMC numbers (in the range of 100,000–800,000 cells/well) was confirmed in nine independent experiments using different antigens and PBMC of different donors (data not shown). Therefore, by doubling or tripling the numbers of freshly thawed PBMC plated into an ELISPOT assay one can predictably accomplish what resting does not, and in spite of the increased cell numbers needed, this strategy does not require more cells than resting does.
Our literature research has identified only two original studies in which a beneficial effect of resting has been described directly. In one study [
24], fresh and rested PBMC of four patients with Stage III and IV follicular lymphoma were compared; in addition to bearing a large tumor burden, these patients were under chemotherapy. In the second study [
25], data are shown for four healthy donors, who were assessed for responses to CEF and CMV. Of these, only one (D4) was in the low responder category by our definition—no benefit of resting was seen for this donor. The remaining three donors were CEF or CMV high responders by our definition—an approximately two-fold increase was seen in two high responders. Janetzki
et al. [
25] also refer to a study by Kierstead
et al. that suggests overnight resting is essential to achieve optimal T cell responses comparable to those from unfrozen PBMC though details of the comparison experiments are not provided and the data are listed as “not shown” in the study [
32]. In another report resting was positively correlated with the detection of low frequency CD8 cells [
26], but this study was inconclusive about resting itself because resting was just one of several assay parameters in which the compared results differed. Increasing the numbers of cells plated was another assay parameter in which the results differed, which in itself suffices to predictably improve the signal to noise performance of the assay (
Figure 7 in our study). Overall, in that study PBMC of 7 donors were tested for reactivity to CMV and flu peptides of which three possessed CMV-specific CD8 cells, and five responded to the flu peptides. Results were compared between thirteen centers in which “ELISPOT analysis was performed according to 11 more or less different protocols”, and the influence of different parameters on the number of positive responses was studied.
Figure 7.
Relationship between the numbers of freshly thawed PBMC added per well and observed SFU in ELISPOT. CEF peptide (A) or mumps antigen-elicited SFU (B) plotted against the number of PBMC added per well. The PBMC were plated in serial dilution with antigen (closed symbols) or without (medium alone, open symbols) keeping all other assay variables constant. The regression for antigen-induced SFU was calculated and is shown. The data are from one experiment.
Figure 7.
Relationship between the numbers of freshly thawed PBMC added per well and observed SFU in ELISPOT. CEF peptide (A) or mumps antigen-elicited SFU (B) plotted against the number of PBMC added per well. The PBMC were plated in serial dilution with antigen (closed symbols) or without (medium alone, open symbols) keeping all other assay variables constant. The regression for antigen-induced SFU was calculated and is shown. The data are from one experiment.
In our study presented here, the benefit of resting was tested systematically in a larger cohort of 25 subjects while maintaining all other assay parameters constant. Resting caused a predictable gain in assay sensitivity for CD8 responses of high-magnitude alone. This gain was on average less than two-fold, and while statistically significant, it might have limited significance in immune diagnostic terms as the responses that are enhanced through resting are already clear-cut without resting. Also, the added labor and increased need for sample material might not be justified. Importantly, the responses seen with freshly isolated (non-cryopreserved) PBMC correspond with responses seen with freshly thawed, unrested PBMC (
Figure 1 C,D). Therefore the enhancement obscures the sought after
ex vivo frequencies of antigen-reactive T cells. For low magnitude CD8 responses, and for CD4 reactivity of all magnitudes, resting had an unpredictable effect: decreased or unchanged reactivity was seen just as frequently as instances of increased reactivity. In contrast, doubling the number of freshly thawed PBMC predictably doubles the signal-to-noise performance of ELISPOT assays. Therefore, by doubling the number of freshly thawed PBMC plated in an ELISPOT assay one can reliably accomplish accurate monitoring of both high- and low magnitude responses derived from CD8 and CD4 cells alike, with less labor, and with the same amount of cell material.
Our studies were done with PBMC of healthy donors that were cryopreserved, importantly, according to optimized protocols that permit full recovery of CD4 and CD8 T cell reactivity after optimal thawing, as we have previously shown [
15] and as confirmed in
Figure 1 A,B for the CEF and mumps responses that we have studied in this paper. We can envision several scenarios in which resting benefits T cell monitoring.
Resting is likely to benefit T cell monitoring if the number of apoptotic cells is high in thawed samples. The number of apoptotic cells might be high if the PBMC were not frozen or thawed under optimized conditions. In this situation resting might help to rescue a result, but for successful T cell work attention should be drawn to optimized freezing and thawing conditions. Following such protocols, PBMC of healthy donors should yield more than 95% viable, and less than 5% apoptotic/dead cells. It will be important for the field to adopt optimized cryopeservation/thawing conditions.
Resting might also be beneficial for PBMC of subjects with certain diseases, or receiving medication/treatments that increases apoptosis of T cells or APC. The PBMC of the patients with Stage III and IV follicular lymphoma that in addition received chemotherapy [
24] are likely to fall into this category. Furthermore, the numbers of apoptotic cells can be elevated if the samples were shipped under suboptimal conditions or requiring long transit periods, or were stored under suboptimal conditions before freezing. In our experience, human PBMC should never be chilled but shipped (as heparinized full blood) and processed (washed) warm. If apoptotic cells are present in PBMC in significant numbers, these, being still alive, will be stained and counted as live cells, but they will be dead by the time the functional assay is performed. Therefore, without accounting for apoptotic cells, one would plate fewer functional PBMC than the live cell count suggests, and the frequency of SFU will be accordingly decreased. After resting, with the apoptotic population gone, the relative frequency of functional T cells will be increased, and subsequently also the spot count. Apoptotic cells can be selectively stained by dyes such as Jo-Pro. Therefore, we support the CIMT (Association for Cancer Immunotherapy) suggestion to include counting of apoptotic cells into the immune monitoring routine [
26].
Presently it is not well established whether apoptotic cells are just non-functional bystander cells, or whether they exert inhibitory effects in functional T cell assays in addition. If they are passive, adjusting the counts of freshly thawed cells to functional cells (viable minus apoptotic) might accomplish in a simpler way what resting does. If apoptotic cells are inhibitory, however, selective depletion of such cells or resting will be required to improve the performance of PBMC samples that contain significant numbers of apoptotic cells. Unlike necrosis that triggers inflammation, apoptosis is an intrinsically non-phlogistic process [
33]. Apoptotic cells can suppress proinflammatory responses of neutrophils and mononuclear phagocytes, and can elicit an anti-inflammatory response by these cells [
34,
35]. The active secretion of lactoferrin by cells undergoing apoptosis is one of the key mechanisms by which apoptotic cells instruct the innate immune system [
36]. Lactoferrin binds to receptors on phagocytes and inhibits their pro-inflammatory responses via NF-κB [
37]. Thus, lactoferrin can inhibit production of pro-inflammatory mediators such as TNF-α and IL-6 [
37,
38], and can induce the secretion of anti-inflammatory cytokines including TGF-β, IL-4 and IL-10 [
39,
40]. It will need to be established experimentally whether and how the presence of cells undergoing apoptosis in PBMC affect T cells in functional assays such as ELISPOT or ICS.
Resting might also benefit T cell monitoring if the serum of the donor contains immune suppressive factors or drugs, for example in cancer patients or HIV-infected subjects. When isolated from such donor’s own serum, and cultured overnight in a neutral medium, such PBMC might recover functionality. Similarly, resting might be of benefit if the serum contains cytokines that stimulate cells of the innate immune system to produce the analyte. For IFN-γ assays, for example, IL-2 is a potent stimulator of IFN-γ production in macrophages, NK and dendritic cells and several other lineages. In such cases, the background in the medium control will be high. IFN-γ spots produced by antigen‑stimulated T cells are typically larger than IFN-γ spots produced by cells of the innate immune system and therefore can be identified by proper gating strategies during ELISPOT analysis. Because several of the above cell lineages of the innate system are plastic adherent, these will be depleted during the resting step, resulting in a lower background. Additionally, the stimulatory effect of the serum cytokines that acted in vivo will wane during resting in neutral test medium in vitro.