Yield Response of Native Warm-Season Forage Grasses to Harvest Intervals and Durations in Mixed Stands
Abstract
:1. Introduction
2. Results and Discussion
2.1. Forage Yield per Plant
2.1.1. Species Cumulative In-Season Yield
| Harvest interval (days) | Yield June–September 2008 | Yield June–September 2009 | ||||
|---|---|---|---|---|---|---|
| Y108‡ | Y207 | P > Fα§ | Y109 | Y208 | p > Fα | |
| g plant−1 | g plant−1 | |||||
| Indiangrass | ||||||
| 120 (2)¶ | 105 | 62 | 0.29 | 111 A# | 39 B | 0.01 |
| 90 (2) | 87 | 69 | 0.57 | 105 | 50 | 0.26 |
| 60 (3) | 167 | 73 | 0.05 | 99 | 58 | 0.16 |
| 40 (4) | 108 | 76 | 0.23 | 70 | 47 | 0.25 |
| 30 (5) | 105 | 88 | 0.55 | 78 A | 36 B | 0.04 |
| CV | 48 | 65 | 59 | 54 | ||
| p > Fα | 0.23 | 0.94 | 0.73 | 0.63 | ||
| Little bluestem | ||||||
| 120 (2) | 16 | 37 | 0.27 | 25 A | 6 B | 0.03 |
| 90 (2) | 21 | 10 | 0.28 | 11 | 7 | 0.33 |
| 60 (3) | 17 | 23 | 0.30 | 21 A | 5 B | 0.05 |
| 40 (4) | 28 | 33 | 0.37 | 18 | 9 | 0.40 |
| 30 (5) | 44 | 38 | 0.64 | 12 | 9 | 0.48 |
| CV | 61 | 66 | 79 | 82 | ||
| p > Fα | 0.06 | 0.14 | 0.45 | 0.68 | ||
2.1.2. Species Post-Season Recovery Yield
| Harvest interval (days) | May 2008 | May 2009 | ||||
|---|---|---|---|---|---|---|
| Y108‡ | Y207 | p > Fα§ | Y109 | Y208 | p > Fα | |
| g plant−1 | g plant−1 | |||||
| Indiangrass | ||||||
| 120 (2)¶ | 235 | 117 | 0.05 | 92 A# | 9 B | 0.01 |
| 90 (2) | 209 | 93 | 0.08 | 79 A | 16 B | <0.01 |
| 60 (3) | 212 A | 111 B | 0.04 | 108 A | 22 B | <0.01 |
| 40 (4) | 250 | 115 | 0.07 | 70 A | 12 B | 0.01 |
| 30 (5) | 185 A | 94 B | 0.01 | 115 A | 10 B | <0.01 |
| CV | 47 | 43 | 41 | 64 | ||
| p > Fα | 0.874 | 0.862 | 0.3212 | 0.17 | ||
| Little bluestem | ||||||
| 120 (2) | 96 | 118 | 0.40 | 52 A | 2 B | 0.02 |
| 90 (2) | 137 A | 68 B | <0.01 | 52 A | 4 B | 0.02 |
| 60 (3) | 123 A | 78 B | 0.01 | 62 A | 2 B | 0.02 |
| 40 (4) | 123 | 89 | 0.08 | 63 A | 3 B | 0.01 |
| 30 (5) | 149 A | 87 B | 0.04 | 66 A | 4 B | 0.02 |
| CV | 23 | 36 | 72 | 106 | ||
| p > Fα | 0.09 | 0.18 | 0.97 | 0.75 | ||

| Interval harvests (days) | First, second and third year plots in 2009 | Second vs. third year plots in 2010 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Y109§ | Y208 | Y307 | CV | p > Fα | Y209 | Y308 | CV | p > Fα | |
| g plant−1 | g plant−1 | ||||||||
| Indiangrass | |||||||||
| 120 (2)# | 92 A†† | 9 B | 23 B | 78 | <0.01 | 21 | 18 | 40 | 0.64 |
| 90 (2) | 79 A | 16 B | 15 B | 47 | <0.01 | 16 | 19 | 61 | 0.45 |
| 60 (3) | 108 A | 22 B | 28 B | 32 | <0.01 | 23 | 19 | 39 | 0.44 |
| 40 (4) | 70 A | 12 B | 20 B | 64 | <0.01 | 13 | 21 | 31 | 0.05 |
| 30 (5) | 115 A | 10 B | 18 B | 54 | <0.01 | 13 | 13 | 36 | 0.88 |
| CV | 41 | 64 | 53 | 44 | 42 | ||||
| p > Fα | 0.32 | 0.17 | 0.41 | 0.17 | 0.62 | ||||
| Little bluestem | |||||||||
| 120 (2) | 52 A | 2 B | 4 B | 116 | 0.01 | 3 | 4 | 83 | 0.82 |
| 90 (2) | 52 A | 4 B | 4 B | 111 | 0.01 | 4 | 2 | 128 | 0.59 |
| 60 (3) | 62 A | 2 B | 3 B | 124 | 0.01 | 2 | 3 | 95 | 0.56 |
| 40 (4) | 63 A | 3 B | 4 B | 100 | <0.01 | 2 | 4 | 69 | 0.14 |
| 30 (5) | 66 A | 4 B | 2 B | 114 | 0.01 | 2 | 3 | 53 | 0.25 |
| CV | 72 | 106 | 69 | 129 | 77 | ||||
| p > Fα | 0.97 | 0.75 | 0.67 | 0.81 | 0.80 | ||||
2.1.3. Implied Species Yield Sustainability
2.2. Forage Yield per Area
2.2.1. Cumulative Whole-Plot Yield
| Harvest interval (days) | Yield June–September 2008 | Yield June–September 2009 | ||||
|---|---|---|---|---|---|---|
| Y108‡ | Y207 | p > Fα§ | Y109 | Y208 | p > Fα | |
| kg ha−1 | kg ha−1 | |||||
| 120 (2)¶ | 4094 b # | 3433 b | 0.83 | 4270 | 2834 | 0.14 |
| 90 (2) | 3760 b | 3111 b | 0.48 | 4813 A | 3495 B | 0.02 |
| 60 (3) | 5115 b | 4161 b | 0.52 | 4030 A | 2759 B | 0.03 |
| 40 (4) | 6325 ab | 5053 ab | 0.26 | 3411 | 2823 | 0.44 |
| 30 (5) | 8472 a | 7627 a | 0.67 | 4219 A | 2395 B | 0.02 |
| CV | 42 | 47 | 25 | 36 | ||
| p > Fα | 0.03 | 0.03 | 0.34 | 0.57 | ||
2.2.2. Post-Season Whole-Plot Recovery
| Harvest interval (days) | Mid-May 2008 | Mid-May 2009 | ||||
|---|---|---|---|---|---|---|
| Y108§ | Y207 | p > Fα¶ | Y109 | Y208 | p > Fα | |
| kg ha−1 | kg ha−1 | |||||
| 120 (2)# | 4596 | 2813 | 0.0609 | 4198 A | 1330 B | 0.0062 |
| 90 (2) | 5184 A†† | 2049 B | 0.0050 | 3315 A | 1401 B | 0.0005 |
| 60 (3) | 5342 A | 2541 B | 0.0001 | 3309 A | 1107 B | 0.0002 |
| 40 (4) | 5284 A | 1849 B | 0.0023 | 3481 A | 1136 B | 0.0046 |
| 30 (5) | 4364 A | 1800 B | 0.0015 | 3675 A | 809 B | 0.0001 |
| CV | 26 | 39 | 31 | 30 | ||
| p > Fα | 0.661 | 0.295 | 0.707 | 0.106 | ||
2.2.3. Implications on Yield Sustainability

3. Experimental Section
3.1. Study Location and Field Layout
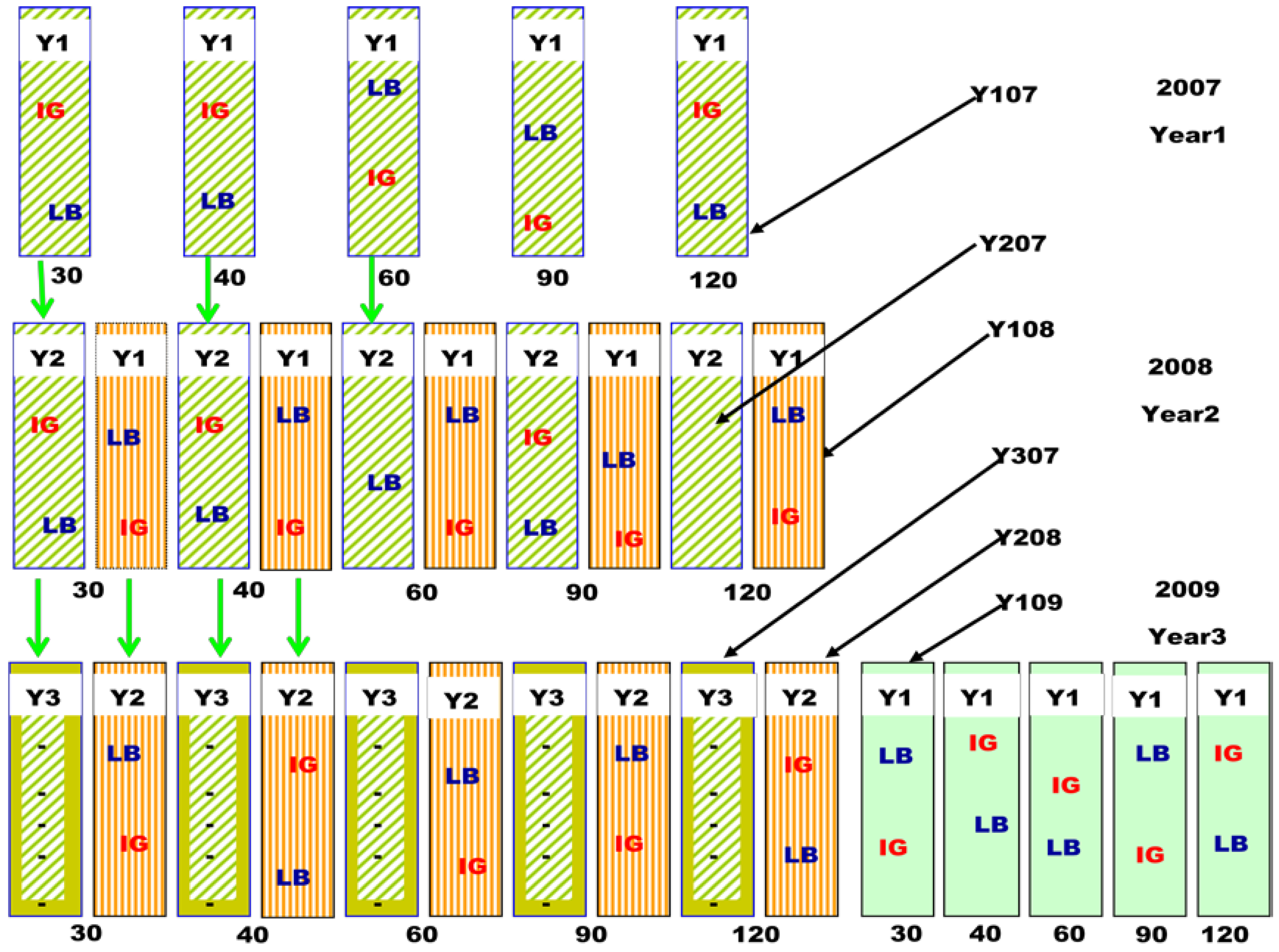
3.2. Measurements of Forage Yield
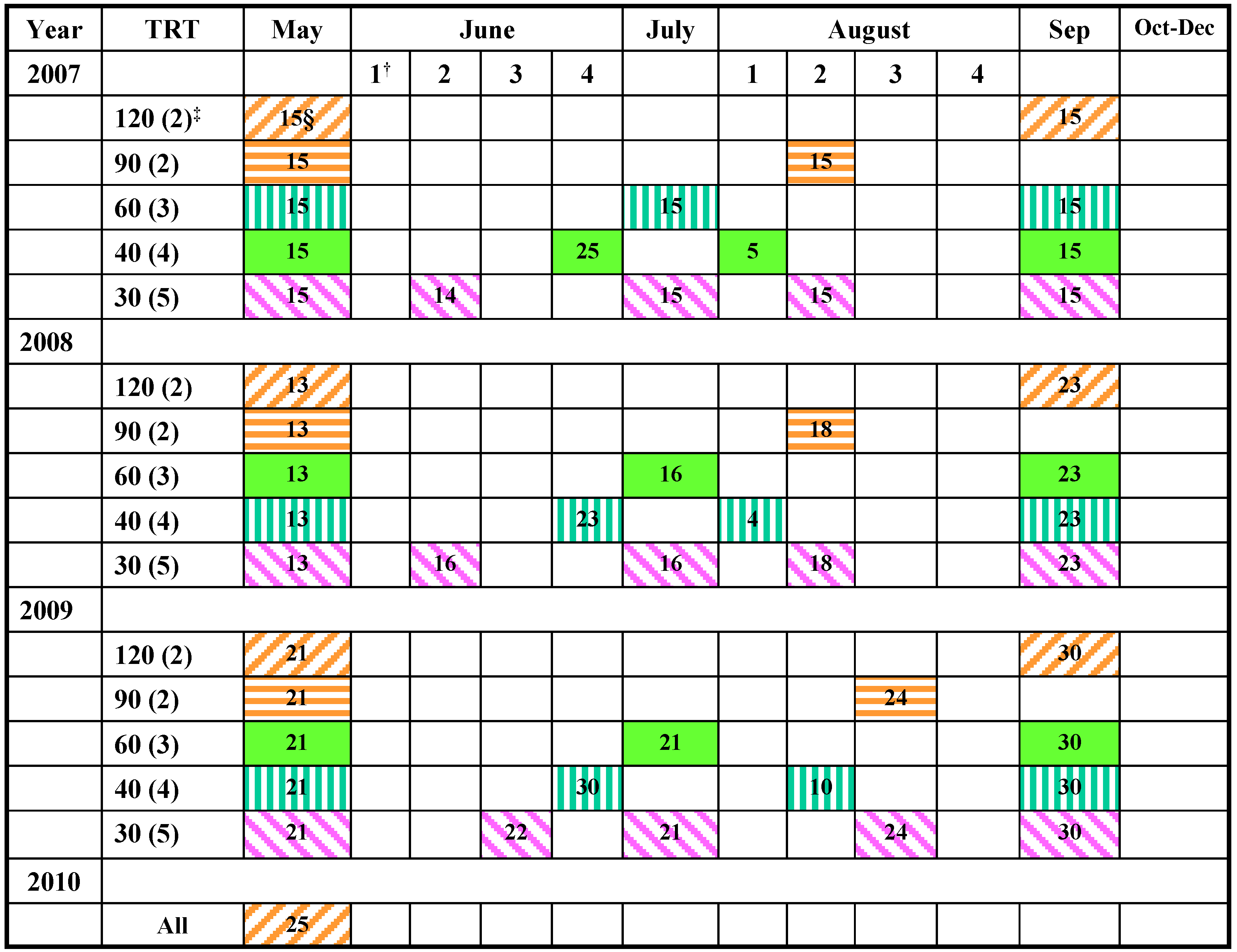
3.3. Data Analyses
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Shea, A.B. The Return of Native Grasses to Tennessee. 1999. Available online: http://www.tn.gov/environment/conservationist/archive/grass.htm (accessed on 12 August 2009).
- Jones, J.; Coggin, D.S.; Cummins, J.L.; Hill, J. Restoring and Managing Native Prairies; A Handbook for Mississippi Landowners; Wildlife Mississippi: Starkville, MS, USA, 2007. [Google Scholar]
- Kiss, Z.; Fieldsend, A.F.; Wolf, D.D. Yield of switchgrass (Panicum virgatum L.) as influenced by cutting management. Acta Agron. Hung. 2007, 55, 227–233. [Google Scholar] [CrossRef]
- Mulkey, V.R.; Owens, V.N.; Lee, D.K. Management of warm-season grass mixtures for biomass production in South Dakota USA. Bioresour. Technol. 2008, 99, 609–617. [Google Scholar] [CrossRef]
- Jung, G.A.; Griffin, J.L.; Kocher, R.E.; Shaffer, J.A.; Gross, C.F. Performance of switchgrass and bluestem cultivars mixed with cool-season species. Agron. J. 1985, 77, 846–850. [Google Scholar] [CrossRef]
- Angima, S.D.; Kallenbach, R.L.; Riggs, W.W. Optimizing hay yield under lower nitrogen rates for selected warm-season forages. J. Integr. Biosci. 2009, 7, pp. 1–6. Available online: http://altweb.astate.edu/electronicjournal/Articles/Angima.pdf (accessed on 20 July 2010).
- Huang, B. Roots and drought resistance. Golf Course Manag. 1997, 65, 55–59. [Google Scholar]
- Dahl, B.E. Developmental Morphology of Plants. In Wildland Plants: Physiological Ecology and Developmental Morphology; Bedunan, D., Sosobee, R., Eds.; Soc. Range Manage: Denver, CO, USA, 1995; pp. 22–58. [Google Scholar]
- Coughenour, M.B. Graminoid responses to grazing by large herbivores: Adaptations, exaptations, and interacting processes. Ann. Mo. Bot. Gard. 1985, 72, pp. 852–863. Available online: http://www.jstor.org/stable/2399227 (accesses on 8 January 2014).
- Boschma, S.P.; Hill, M.J.; Scott, J.M.; Rapp, G.G. The response to moisture and defoliation stresses, and traits for resilience of perennial grasses on the Northern Tablelands of New South Wales, Australia. Aust. J. Agric. Res. 2003, 54, 903–916. [Google Scholar] [CrossRef]
- Chaves, M.M.; Maroco, J.P.; Pereira, J.S. Understanding plant responses to drought—From genes to the whole plant. Funct. Plant Biol. 2003, 30, 239–264. [Google Scholar] [CrossRef]
- Craine, J.M.; Ocheltree, T.W.; Nippert, J.B.; Towne, E.G.; Skibbe, A.M.; Kembel, S.W.; Fargione, J.E. Global diversity of drought tolerance and grassland climate-change resilience. Nat. Clim. Chang. 2012, 3, pp. 63–67. Available online: http://www.readcube.com/articles/10.1038/NCLIMATE1634 (accessed on 8 January 2014).
- Manske, L.L. General Description of Grass Growth and Development and Defoliation Resistance Mechanisms; Range Management Report DREC 98-1022; North Dakota State University, Dickinson Research Extension Center: Dickinson, ND, USA, 1998. [Google Scholar]
- Noy-Meir, I. Compensating growth of grazed plants and its relevance to the use of rangelands. Ecol. Appl. 1993, 3, 32–34. [Google Scholar] [CrossRef]
- Belsky, A.J.; Carson, W.P.; Jensen, C.L.; Fox, G.A. Overcompensation by plants: Herbivore optimization or red herring? Evol. Ecol. 1993, 7, 109–121. [Google Scholar] [CrossRef]
- Garay, A.H.; Matthew, C.; Hodgson, J. Tiller size/density compensation in perennial ryegrass miniature swards subject to differing defoliation heights and a proposed productivity index. Grass Forage Sci. 1999, 54, 347–356. [Google Scholar] [CrossRef]
- Ferraro, D.O.; Oesterheld, M. Effect of defoliation on grass growth. A quantitative review. Oikos 2002, 98, 125–133. [Google Scholar] [CrossRef]
- Mullahey, J.J.; Waller, S.S.; Moser, L.E. Defoliation effects on production and morphological development of little bluestem. J. Range Manag. 1990, 43, 497–500. [Google Scholar] [CrossRef]
- Forwood, J.R.; Magai, M.M. Clipping frequency and intensity effects on big bluestem yield, quality, and persistence. J. Range Manag. 1992, 45, 554–559. [Google Scholar] [CrossRef]
- Anderson, V.J.; Briske, D.D. Herbivore-induced species replacement in grasslands: Is it driven by herbivory tolerance or avoidance? Ecol. Appl. 1995, 5, 1014–1024. [Google Scholar] [CrossRef]
- Oesterheld, M.; McNaughton, S.J. Effect of stress and time for recovery on the amount of compensatory growth after grazing. Oecologia (Berlin) 1991, 85, 305–313. [Google Scholar] [CrossRef]
- Lee, W.G.; Fenner, M.; Loughnan, A.; Lloyd, K.M. Long-term effects of defoliation: Incomplete recovery of a New Zealand alpine tussock grass, Chionochloa pallens, after 20 years. J. Appl. Ecol. 2000, 37, 348–355. [Google Scholar] [CrossRef]
- Turner, C.L.; Seastedt, T.R.; Dyer, M.I. Maximization of aboveground grassland production: The role of defoliation frequency, intensity, and history. Ecol. Appl. 1993, 3, 175–186. [Google Scholar] [CrossRef]
- Meyer, G.A. Mechanisms promoting recovery from defoliation in goldenrod (Solidago altissima). Botany 1998, 76, 450–459. [Google Scholar] [CrossRef]
- N’Guessan, M.; Hartnett, D.C. Differential responses to defoliation frequency in little bluestem (Schizachyrium scoparium) in tallgrass prairie: Implications for herbivory tolerance and avoidance. Plant Ecol. 2011, 212, 1275–1285. [Google Scholar] [CrossRef]
- Vinton, M.A.; Hartnett, D.C. Effects of bison grazing on Andropogon gerardii and Panicum virgatum in burned and unburned tallgrass paririe. Oecologia (Berlin) 1992, 90, 374–382. [Google Scholar] [CrossRef]
- Oesterheld, M. Effect of defoliation intensity on aboveground and belowground relative growth rates. Oecologia (Berlin) 1992, 92, 313–316. [Google Scholar] [CrossRef]
- Donaghy, D.J.; Fulkerson, W.J. Priority for allocation of water-soluble carbohydrate reserves during regrowth of Lolium perenne. Grass Forage Sci. 1998, 53, 211–218. [Google Scholar]
- Hannaway, D.B.; Hannaway, K.J.; Sohn, P.; Griffith, S.; Wycoff, H. Grass Growth and Regrowth for Improved Management. 2000. Available online: http://www.fsl.orst.edu/forages/projects/regrowth/main.cfm?PageID=7 (accessed on 30 August 2008).
- Havlin, J.L.; Beaton, J.D.; Tisdale, S.L. Soil Fertility and Fertilizers: An Introduction to Nutrient Management, 7th ed.; Pearson Education, Inc.: Upper Saddle River, NJ, USA, 2005. [Google Scholar]
- Brady, N.C.; Weil, R.R. The Nature and Properties of Soils, 14th ed.; Pearson Education, Inc.: Upper Saddle River, NJ, USA, 2008; pp. 1–31. [Google Scholar]
- Scheneiter, O.; Matthew, C.; Rimieri, P. The effect of defoliation management on tiller dynamics of prairie grass. Rev. Argent. Prod. Anim. 2008, 28, 7–20. [Google Scholar]
- Archer, S. The effects of defoliation and competition on regrowth of tillers of two North American mixed-grass prairie graminoids. Oikos 1984, 43, 351–357. [Google Scholar] [CrossRef]
- Anderson, B.; Matches, A.G. Forage yield, quality, and persistence of switchgrass and caucasian bluestem. Agron. J. 1983, 75, 119. [Google Scholar] [CrossRef]
- Christensen, N.L. Fire and soil-plant nutrient relations in a pine-wiregrass savanna on the coastal plain of North Carolina. Oecologia (Berlin) 1977, 31, 27–44. [Google Scholar] [CrossRef]
- Davies, K.W.; Bates, J.D.; Miller, R.F. Short-term effects of burning Wyoming big sagebrush steppe in southeast Oregon. Rangel. Ecol. Manag. 2007, 60, 515–522. [Google Scholar] [CrossRef]
- Bremer, D.J.; Ham, J.M. Effect of spring burning on the surface energy balance in a tallgrass prairie. Agric. For. Meteorol. 1999, 97, 43–54. [Google Scholar] [CrossRef]
- SAS Institute. SAS Version 9.2, SAS Institute: Cary, NC, USA, 2007.
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Temu, V.W.; Rude, B.J.; Baldwin, B.S. Yield Response of Native Warm-Season Forage Grasses to Harvest Intervals and Durations in Mixed Stands. Agronomy 2014, 4, 90-107. https://doi.org/10.3390/agronomy4010090
Temu VW, Rude BJ, Baldwin BS. Yield Response of Native Warm-Season Forage Grasses to Harvest Intervals and Durations in Mixed Stands. Agronomy. 2014; 4(1):90-107. https://doi.org/10.3390/agronomy4010090
Chicago/Turabian StyleTemu, Vitalis W., Brian J. Rude, and Brian S. Baldwin. 2014. "Yield Response of Native Warm-Season Forage Grasses to Harvest Intervals and Durations in Mixed Stands" Agronomy 4, no. 1: 90-107. https://doi.org/10.3390/agronomy4010090





