Short-Term Effect of Feedstock and Pyrolysis Temperature on Biochar Characteristics, Soil and Crop Response in Temperate Soils
Abstract
:1. Introduction
2. Materials and Methods
2.1. Soil
2.2. Biochar Characterization
| Biochar type | Time at treatment temperature (min) | Total time in reactor (min) | Moisture (%) | C (%) | H (%) | N (%) | C:N | Volatile matter (%) | Ash (%) | pH-KCl | CEC (cmolc kg−1) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Willow-450 °C | 29 | 89 | 3.2 ± 0.4 | 78.4 ± 0.7 | 2.03 ± 0.04 | 0.82 ± 0.02 | 96 | 11.2 ± 0.6 | 4.3 ± 0.1 | 7.3 ± 0.1 | 33.4 |
| Willow-550 °C | 23 | 100 | 3.2 ± 0.6 | 86.3 ± 0.6 | 1.95 ± 0.10 | 0.85 ± 0.14 | 102 | 6.7 ± 0.7 | 3.2 ± 0.0 | 7.5 ± 0.0 | 42.8 |
| Willow-650 °C | 28 | 118 | 5.3 ± 0.7 | 84.8 ± 0.7 | 1.14 ± 0.02 | 1.00 ± 0.02 | 85 | 6.0 ± 0.4 | 4.9 ± 0.1 | 8.1 ± 0.3 | 59.1 |
| Pine-450 °C | 88 | 151 | 2.6 ± 0.1 | 86.8 ± 1.0 | 2.80 ± 0.09 | 0.19 ± 0.05 | 457 | 12.1 ± 1.6 | 0.9 ± 0.2 | 6.7 ± 0.0 | 38.6 |
| Pine-550 °C | 60 | 159 | 3.3 ± 0.6 | 91.6 ± 1.6 | 2.13 ± 0.01 | 0.19 ± 0.04 | 482 | 7.3 ± 1.7 | 1.0 ± 0.3 | 6.8 ± 0.0 | 52.1 |
| Pine-650 °C | 33 | 148 | 3.4 ± 0.5 | 92.6 ± 0.2 | 1.68 ± 0.01 | 0.15 ± 0.00 | 617 | 6.0 ± 1.7 | 1.1 ± 0.2 | 7.7 ± 0.2 | 68.8 |
2.3. Phytotoxicity Test
2.4. Nitrogen Incubation Experiment
2.5. ILVO Pot Trial
2.6. DTU Pot Trial
2.7. Statistical Analyses
3. Results
3.1. Soil and Biochar Characterization
3.2. Phytotoxicity Test
| Phytotoxicity (%) | ||
|---|---|---|
| 10 g kg−1 | 50:50 v:v | |
| Willow-450 °C | 0.5 ± 1.0 | 1.0 ± 2.9 |
| Willow-550 °C | 1.0 ± 1.6 | 54.0 ± 4.2 |
| Willow-650 °C | 2.0 ± 2.0 | 86.4 ± 14.1 |
| Pine-450 °C | 2.5 ± 2.5 | 2.5 ± 1.9 |
| Pine-550 °C | 0.0 ± 2.0 | 2.5 ± 1.9 |
| Pine-650 °C | 0.0 ± 1.2 | 72.7 ± 33.0 |
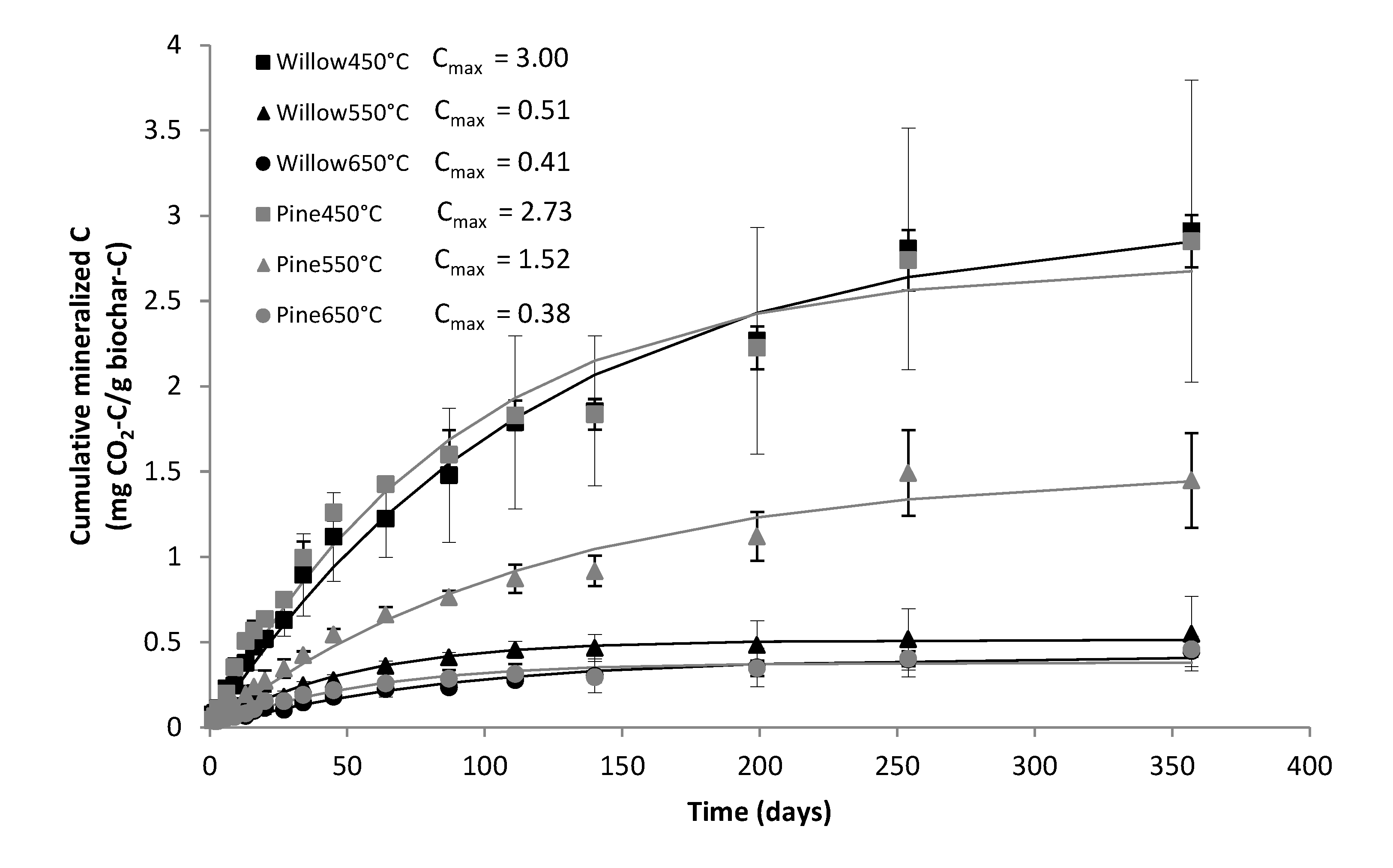
3.3. Nitrogen Incubation Experiment

| Fertilizer dose (kg N ha−1) | |||
|---|---|---|---|
| 0 | 50 | 150 | |
| Control | 16.77 ± 0.39 | 28.18 ± 2.65 | 58.79 ± 2.27 |
| Willow-450 °C | 10.71 ± 0.36 ** | 21.96 ± 1.53 * | 45.02 ± 2.16 * |
| Willow-550 °C | 11.82 ± 3.77 ns | 18.55 ± 1.87 * | 41.95 ± 1.99 * |
| Willow-650 °C | 6.73 ± 0.53 ** | 15.13 ± 1.96 * | 34.79 ± 4.63 * |
| Pine-450 °C | 14.58 ± 0.31 * | 26.28 ± 1.67 ns | 60.86 ± 0.46 ns |
| Pine-550 °C | 12.12 ± 1.36 * | 26.27 ± 1.27 ns | 51.11 ± 3.9 * |
| Pine-650 °C | 8.997 ± 0.37 * | 19.17 ± 2.17 * | 49.58 ± 0.87 * |
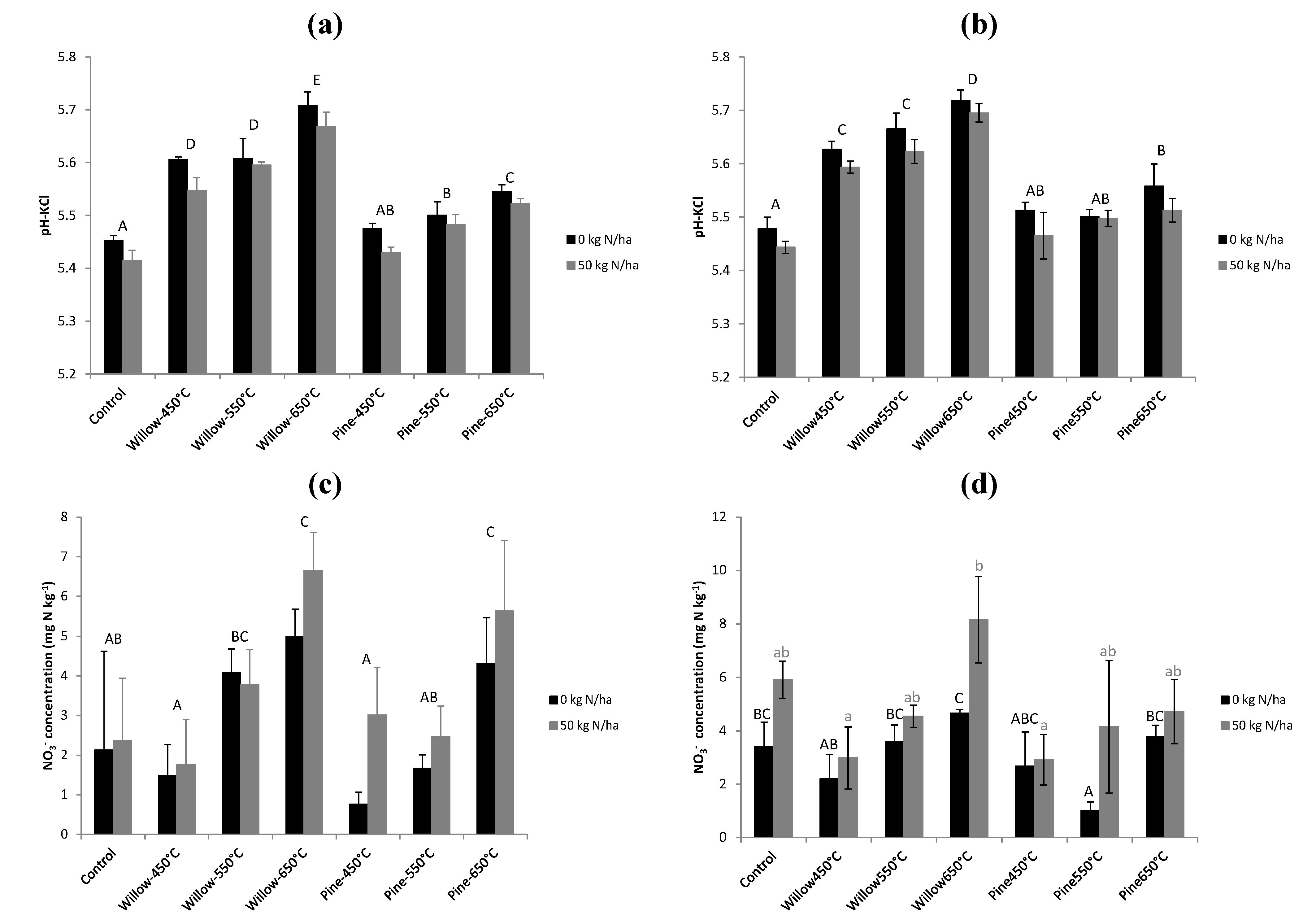
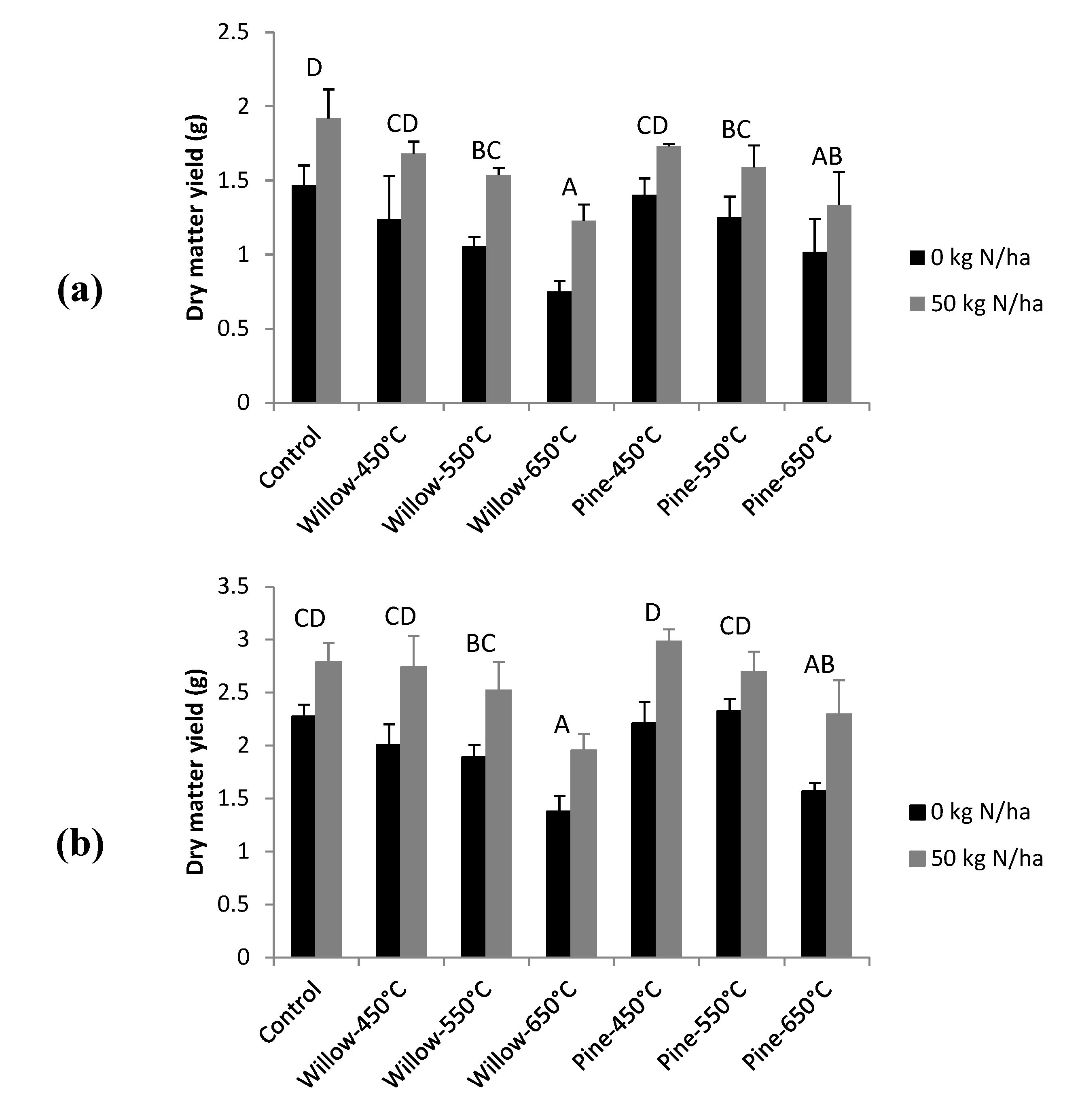
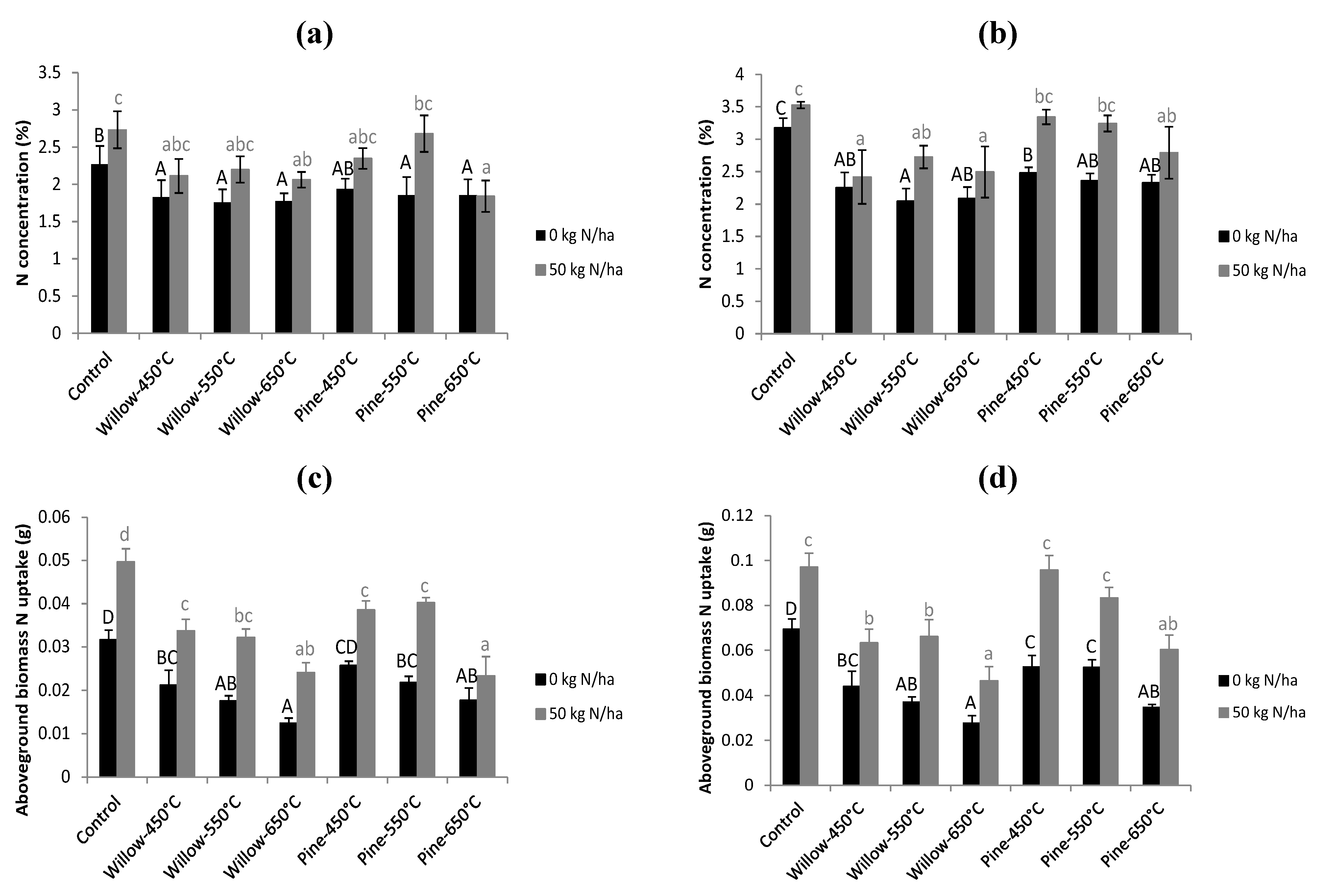
3.4. ILVO Pot Trial

3.5. DTU Pot Trial

4. Discussion
4.1. Biochar and Toxicity
4.2. Mechanisms for Reduced N Availability after Biochar Application
4.3. Crop Growth Effects
5. Conclusions
Acknowledgements
References
- Lehmann, J.; Gaunt, J.; Rondon, M. Bio-char sequestration in terrestrial ecosystems—A review. Mitig. Adapt. Strateg. Glob. Chang. 2006, 11, 403–427. [Google Scholar]
- Sohi, S. Carbon Storage with Benefits. Science 2012, 338, 1034–1035. [Google Scholar] [CrossRef]
- Jeffery, S.; Verheijen, F.G.A.; van der Velde, M.; Bastos, A.C. A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agric. Ecosyst. Environ. 2011, 144, 175–187. [Google Scholar] [CrossRef]
- Bruun, E.W.; Ambus, P.; Egsgaard, H.; Hauggaard-Nielsen, H. Effects of slow and fast pyrolysis biochar on soil C and N turnover dynamics. Soil Biol. Biochem. 2012, 46, 73–79. [Google Scholar] [CrossRef]
- Kammann, C.I.; Linsel, S.; Göβling, J.W.; Koyro, H.W. Influence of biochar on drought tolerance of Chenopodium quinoa Willd and on soil-plant relations. Plant Soil 2011, 345, 195–210. [Google Scholar] [CrossRef]
- Jones, D.L.; Rousk, J.; Edwards-Jones, G.; DeLuca, T.H.; Murphy, D.V. Biochar-Mediated changes in soil quality and plant growth in a three year field trial. Soil Biol. Biochem. 2012, 45, 113–124. [Google Scholar] [CrossRef]
- Hammond, J.; Shackley, S.; Prendergast-Miller, M.; Cook, J.; Buckingham, S.; Pappa, V.A. Biochar field testing in the UK: Outcomes and implications for use. Carbon Manag. 2013, 4, 159–170. [Google Scholar] [CrossRef]
- Biederman, L.A.; Harpole, W.S. Biochar and its effects on plant productivity and nutrient cycling: A meta-analysis. GCB Bioenergy 2013, 5, 202–214. [Google Scholar] [CrossRef]
- Bruun, E.W.; Müller-Stöver, D.; Ambus, P.; Hauggaard-Nielsen, H. Application of biochar to soil and N2O emissions: Potential effects of blending fast-pyrolysis biochar with anaerobically digested slurry. Eur. J. Soil Sci. 2011, 62, 581–589. [Google Scholar] [CrossRef]
- Ippolito, J.A.; Novak, J.M.; Busscher, W.J.; Ahmedna, M.; Rehrah, D.; Watts, D.W. Switchgrass biochar effects two aridisols. J. Environ. Qual. 2012, 41, 1123–1130. [Google Scholar] [CrossRef]
- Knowles, O.A.; Robinson, B.H.; Contangelo, A.; Clucas, L. Biochar for the mitigation of nitrate leaching from soil amended with biosolids. Sci. Total Environ. 2011, 409, 3206–3210. [Google Scholar] [CrossRef]
- Nelissen, V.; Rütting, T.; Huygens, D.; Staelens, J.; Ruysschaert, G.; Boeckx, P. Maize biochars accelerate short-term soil nitrogen dynamics in a loamy sand soil. Soil Biol. Biochem. 2012, 55, 20–27. [Google Scholar] [CrossRef]
- Novak, J.M.; Busscher, W.J.; Watts, D.W.; Laird, D.A.; Ahmedna, M.A.; Niandou, M.A.S. Short-term CO2 mineralization after additions of biochar and switchgrass to a Typic Kandiudult. Geoderma 2010, 154, 281–288. [Google Scholar] [CrossRef]
- Lugato, E.; Vaccari, F.P.; Genesio, L.; Baronti, S.; Pozzi, A.; Rack, M.; Woods, J.; Simonetti, G.; Montanarella, L.; Miglietta, F. An energy-biochar chain involving biomass gasification and rice cultivation in Northern Italy. GCB Bioenergy 2013, 5, 192–201. [Google Scholar] [CrossRef]
- Vaccari, F.P.; Baronti, S.; Lugato, E.; Genesio, L.; Castaldi, S.; Fornasier, F.; Miglietta, F. Biochar as a strategy to sequester carbon and increase yield in durum wheat. Eur. J. Agron. 2011, 34, 231–238. [Google Scholar] [CrossRef]
- Ronsse, F.; Van Hecke, S.; Dickinson, D.; Prins, W. Production and characterization of slow pyrolysis biochar: Influence of feedstock type and pyrolysis conditions. GCB Bioenergy 2013, 5, 104–115. [Google Scholar] [CrossRef]
- American Society for Testing and Materials International, ASTM. Standard Test Method for Chemical Analysis of Wood Charcoal; ASTM D1762-84; American Society for Testing and Materials International: West Conshohocken, PA, USA, 2007. [Google Scholar]
- Chapman, H.D. Cation-Exchange Capacity. In Methods of Soil Analysis: Part 2—Chemical and Microbiological Properties; Black, C.A., Ed.; American Society of Agronomy: Madison, WI, USA, 1965; pp. 891–901. [Google Scholar]
- Nelissen, V.; Rütting, T.; Huygens, D.; Ruysschaert, G.; Boeckx, P. Temporal evolution of biochar’s impact on soil nitrogen processes—A 15N tracing study. GCB Bioenergy 2014, in press. [Google Scholar]
- Nelissen, V.; Ruysschaert, G. Institute for Agricultural and Fisheries Research, Merelbeke, Belgium. Unpublished work. 2013. [Google Scholar]
- Compendium voor monsterneming en analyse (CMA). Anorganische analysemethoden: Compost—Fytotoxiciteit; CMA/2/IV/12; EMIS, energie-en milieu-informatiesysteem voor het Vlaamse Gewest: Mol, Belgium, 2005. (in Dutch)
- Müller-Stöver, D.; Hauggaard-Nielsen, H. Department of Chemical and Biochemical Engineering, Technical University of Denmark, Denmark. Unpublished work. 2013. [Google Scholar]
- Van Zwieten, L.; Kimber, S.; Morris, S.; Chan, K.Y.; Downie, A.; Rust, J.; Joseph, S.; Cowie, A. Effects of biochar from slow pyrolysis of papermill waste on agronomic performance and soil fertility. Plant Soil 2010, 327, 235–246. [Google Scholar] [CrossRef]
- Rogovska, N.; Laird, D.; Cruse, R.M.; Trabue, S.; Heaton, E. Germination tests for assessing biochar quality. J. Environ. Qual. 2012, 41, 1014–1022. [Google Scholar] [CrossRef]
- Oleszczuk, P.; Rycaj, M.; Lehmann, J.; Cornelissen, G. Influence of activated carbon and biochar on phytotoxicity of air-dried sewage sludges to Lepidium sativum. Ecotox. Environ. Safe. 2012, 80, 321–326. [Google Scholar]
- White, R.E. Principles and Practice of Soil Science: The Soil as a Natural Resource, 4th ed.; Blackwell Publishing: Oxford, UK, 2006; pp. 34–58. [Google Scholar]
- Lehmann, J.; Rillig, M.C.; Thies, J.; Masiello, C.A.; Hockaday, W.C.; Crowley, D. Biochar effects on soil biota—A review. Soil Biol. Biochem. 2011, 43, 1812–1836. [Google Scholar] [CrossRef]
- DeLuca, T.H.; MacKenzie, M.D.; Gundale, M.J.; Holben, W. Wildfire-Produced charcoal directly influences nitrogen cycling in ponderosa pine forests. Soil Sci. Soc. Am. J. 2006, 70, 448–453. [Google Scholar] [CrossRef]
- Spokas, K.A.; Novak, J.M.; Venterea, R.T. Biochar’s role as an alternative N-fertilizer: Ammonia capture. Plant Soil 2012, 350, 35–42. [Google Scholar] [CrossRef]
- Taghizadeh-Toosi, A.; Clough, T.J.; Sherlock, R.R.; Condron, L.M. A wood based low-temperature biochar captures NH3-N generated from ruminant urine-N, retaining its bioavailability. Plant Soil 2012, 353, 73–84. [Google Scholar] [CrossRef]
- Major, J.; Rondon, M.; Molina, D.; Riha, S.; Lehmann, J. Maize yield and nutrition during 4 years after biochar application to a Colombian savanna oxisol. Plant Soil 2010, 333, 117–128. [Google Scholar] [CrossRef]
- Lehmann, J.; Czimczik, C.; Laird, D.; Sohi, S. Stability of Biochar in the Soil. In Biochar for Environmental Management; Lehmann, J., Joseph, S., Eds.; Earthscan: London, UK, 2009; pp. 183–205. [Google Scholar]
- Laird, D.A.; Brown, R.C.; Amonette, J.E.; Lehmann, J. Review of the pyrolysis platform for coproducing bio-oil and biochar. Biofuels Bioprod. Bioref. 2009, 3, 547–562. [Google Scholar] [CrossRef]
- Yao, Y.; Gao, B.; Zhang, M.; Inyang, M.; Zimmerman, A.R. Effect of biochar amendment on sorption and leaching of nitrate, ammonium, and phosphate in a sandy soil. Chemosphere 2012, 89, 1467–1471. [Google Scholar] [CrossRef]
- Downie, A.; Crosky, A.; Munroe, P. Physical Properties of Biochar. In Biochar for Environmental Management; Lehmann, J., Joseph, S., Eds.; Earthscan: London, UK, 2009; pp. 13–32. [Google Scholar]
- Major, J.; Steiner, C.; Downie, A.; Lehmann, J. Biochar Effects on Nutrient Leaching. In Biochar for Environmental Management; Lehmann, J., Joseph, S., Eds.; Earthscan: London, UK, 2009; pp. 271–287. [Google Scholar]
- Deenik, J.L.; McClellan, T.; Uehara, G.; Antal, M.J.; Campbel, S. Charcoal volatile matter content influences plant growth and soil nitrogen transformations. Soil Sci. Soc. Am. J. 2010, 74, 1259–1270. [Google Scholar]
- Chan, K.Y.; Van Zwieten, L.; Meszaros, I.; Downie, A.; Joseph, S. Agronomic values of greenwaste biochar as a soil amendment. Aust. J. Soil Res. 2007, 45, 629–634. [Google Scholar] [CrossRef]
- Van Zwieten, L.; Kimber, S.; Downie, A.; Morris, S.; Petty, S.; Rust, J.; Chan, K.Y. A glasshouse study on the interaction of low mineral ash biochar with nitrogen in a sandy soil. Aust. J. Soil Res. 2010, 48, 569–576. [Google Scholar] [CrossRef]
Supplementary
| Al | Cu | Fe | Mn | Ni | Si | Ti | Zn | |
|---|---|---|---|---|---|---|---|---|
| mg kg−1 | ||||||||
| Willow-450 °C | 64.1 | 20.8 | 59.8 | 34.5 | 1.1 | 293 | 0.5 | 80.3 |
| Willow-550 °C | 208 | 20.1 | 121 | 46.3 | 3.2 | 494 | 2.8 | 95.5 |
| Willow-650 °C | 133 | 25.6 | 154 | 44.7 | 8.6 | 227 | 3.9 | 145 |
| Pine-450 °C | 191 | 5.4 | 173 | 44.5 | 2.0 | 321 | 5.0 | 1.0 |
| Pine-550 °C | 370 | 10.4 | 368 | 35.6 | 3.2 | 198 | 11.2 | 2.2 |
| Pine-650 °C | 205 | 16.9 | 135 | 40.7 | 1.9 | 153 | 6.1 | 5.1 |
| V | Ba | Na | Ca | Mg | K | Sr | B | |
| mg kg−1 | ||||||||
| Willow-450 °C | 2.7 | 11.2 | 627 | 9,755 | 1617 | 1858 | 81.6 | 15.1 |
| Willow-550 °C | 5.7 | 12.6 | 748 | 12,384 | 1863 | 3392 | 114 | 19.1 |
| Willow-650 °C | <2.6 | 11.9 | 512 | 13,618 | 2093 | 4446 | 142 | 21.7 |
| Pine-450 °C | <2.6 | 9.1 | 35.4 | 3,454 | 517 | 67.0 | 18.4 | 2.3 |
| Pine-550 °C | <2.6 | 12.3 | 60.6 | 3,438 | 674 | 239 | 18.9 | 2.9 |
| Pine-650 °C | <2.6 | 14.2 | 85.2 | 2,863 | 541 | 379 | 16.4 | 4.8 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nelissen, V.; Ruysschaert, G.; Müller-Stöver, D.; Bodé, S.; Cook, J.; Ronsse, F.; Shackley, S.; Boeckx, P.; Hauggaard-Nielsen, H. Short-Term Effect of Feedstock and Pyrolysis Temperature on Biochar Characteristics, Soil and Crop Response in Temperate Soils. Agronomy 2014, 4, 52-73. https://doi.org/10.3390/agronomy4010052
Nelissen V, Ruysschaert G, Müller-Stöver D, Bodé S, Cook J, Ronsse F, Shackley S, Boeckx P, Hauggaard-Nielsen H. Short-Term Effect of Feedstock and Pyrolysis Temperature on Biochar Characteristics, Soil and Crop Response in Temperate Soils. Agronomy. 2014; 4(1):52-73. https://doi.org/10.3390/agronomy4010052
Chicago/Turabian StyleNelissen, Victoria, Greet Ruysschaert, Dorette Müller-Stöver, Samuel Bodé, Jason Cook, Frederik Ronsse, Simon Shackley, Pascal Boeckx, and Henrik Hauggaard-Nielsen. 2014. "Short-Term Effect of Feedstock and Pyrolysis Temperature on Biochar Characteristics, Soil and Crop Response in Temperate Soils" Agronomy 4, no. 1: 52-73. https://doi.org/10.3390/agronomy4010052





