Drugs and Polymers for Delivery Systems in OA Joints: Clinical Needs and Opportunities
Abstract
:1. Introduction
1.1. The Osteoarthritic Joint
1.2. Current Treatment
1.3. Clinical Needs
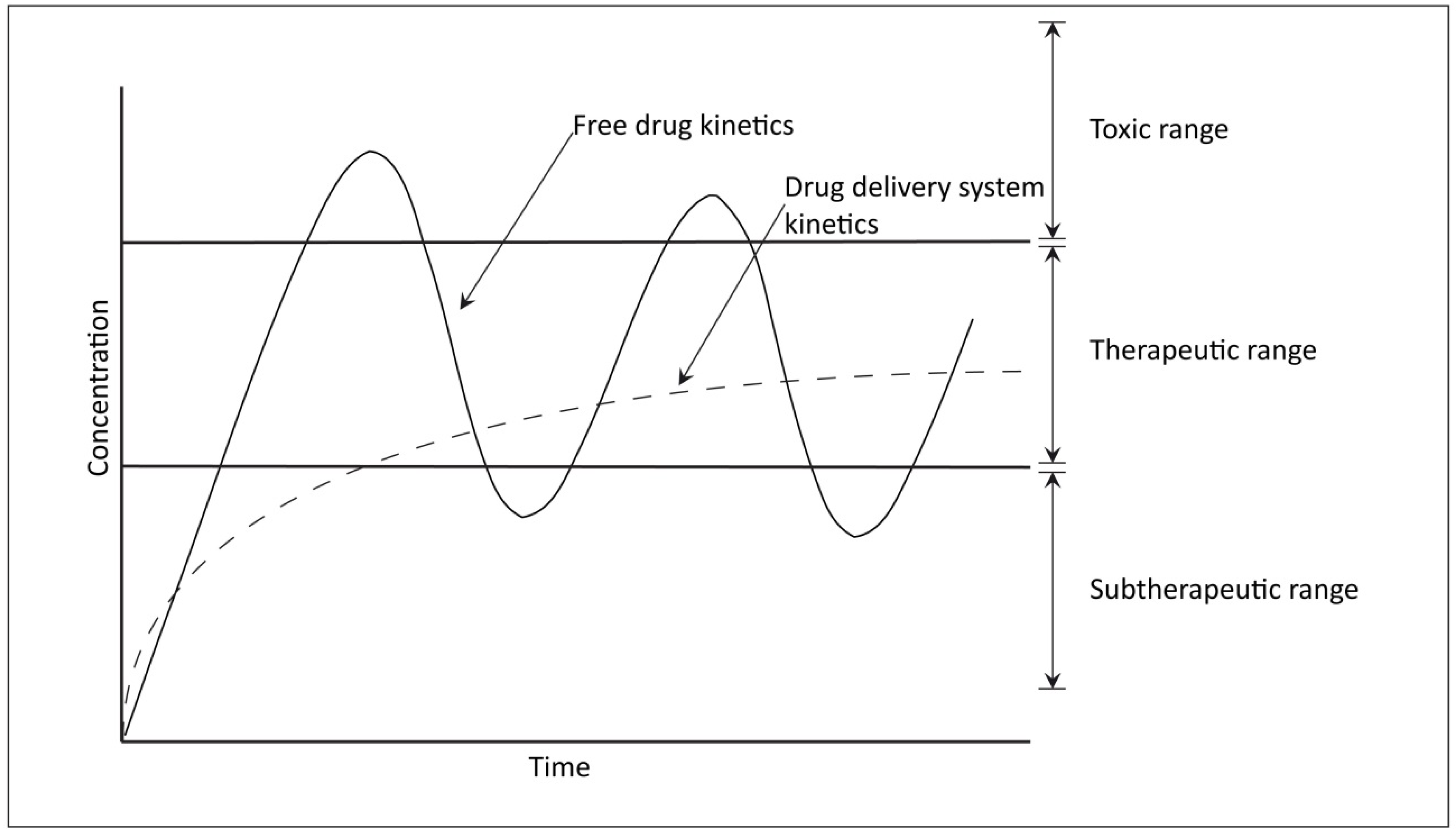
2. Candidate Drugs for OA Treatment
2.1. NSAIDs, Coxibs, Glucocorticoids, and Hyaluronan
| Author | Year | Type DDS | Composition | Drug | Particle Diameter | Model | OA Induction | Outcome |
|---|---|---|---|---|---|---|---|---|
| Ibim | 1998 | Microsphere | PolyPhosphazene | Colchicine | Not stated | in vitro | N.A. | Prolonged release, possible toxicity |
| Brown | 1998 | Microsphere | Gelatin/chondroitin 6-sulfate | 14C-catalase, 14C-albumin, 14C-inulin, 14C-diazepam | 1–60 µm | in vitro/mice | none | partially biocompatible |
| Tuncay | 2000 | Microsphere | PLGA | Diclofenac | 5–10 µm | in vitro/rabbit | Ovalbumin/FCA | No significant difference in inflammation |
| Tuncay | 2000 | Microsphere | Albumin | Diclofenac | ±15 µm | in vitro/rabbit | Ovalbumin/FCA | Promising at day 30 |
| Bozdag | 2001 | Microsphere | PLGA, albumin | Naproxen | 10 µm | in vitro/rabbit | Ovalbumin/FCA | PLGA better than albumin |
| Bragdon | 2001 | Microsphere | PLGA | Paclitaxel | 50 µm | ex vivo horse MCP | none | Biocompatible |
| Horisawa | 2002 | Nano/ microsphere | PLGA | Fluoresceinamine | 265 nm/26.5 µm | Rat | none | Fagocytosis is size dependent |
| Horisawa | 2002 | Nanosphere | PLGA | Betamethasone | 300–490 nm | in vitro/rabbit | Ovalbumin/FCA | Prolonged efficacy |
| Liang | 2003 | Microsphere | PLLA | Methotrexate | 83.7–187.6 µm | in vitro/rabbit | None | mild inflammation, prolonged release |
| Fernández-Carballido | 2004 | Microsphere | PLGA | Ibuprofen, PEG oil (Labrafil) | 39.69 µm | in vitro | N.A. | Labrafil reduces burst release, prolonged release |
| Liggins | 2004 | Microsphere | PLGA, PLA, PCL, Chitosan | Paclitaxel | 1–20 µm, 10–35 µm, 35–105µm | Rabbit | BSA/FCA, Carrageenan | Chitosan not biocompatible, small PLGA particles give greater inflammation. |
| Thakkar | 2004 | Microsphere | Chitosan | Celecoxib | 8 µm | Rat | FCA | Chitosan is biocompatible, improved retention |
| Fernández-Carballido | 2004 | Microsphere | PLGA | Ibuprofen, PEG oil (Labrafil) | 39.31 µm | in vitro | N.A. | Storage of PLGA/Ibuprofen particles does not change characteristics |
| Park | 2005 | Hydrogel | Hyaluronic acid | Hyaluronic acid Ultrasound | 3000 kDA | Rabbit | ACLT/MT | Combination of HYA and US is more effective than monotherapy |
| Betre | 2006 | Aggregate | Elastin-like polypeptides | none | N.A. | rat | None | Biocompatible, prolonged residence time |
| Tsai | 2007 | Nanosphere | Nanogold | none | 5, 13 nm | Rat | Collagen | RA reduction |
| Zhang | 2007 | Micelle | PNIPAAm/EAB-PPP | Indomethacin | Not stated | in vitro/rat | FCA, Carrageenan | Prolonged release/effect |
| Hui | 2007 | Hydrogel | α-CD-EG 4400 | Chondroitin sulfate | N.A. | Rabbit | Chondral defect | Biocompatible, improved biomechanical and histologic properties |
| Lu | 2007 | Microsphere | Gelatin | Flurbiprofen | 2.5–12.3 µm | Rabbit | None | Prolonged residence IA, biocompatibility unclear |
| Thakkar | 2007 | Nanoparticles | Glycerol behenate | Celecoxib | 257 nm | Rat | FCA | Prolonged residence, biocompatible |
| Rothenfluh | 2008 | Nanoparticles | Poly(propylene sulphide) | WYRGRL (Col II-binding peptide) | 38, 96 nm | Mice | None | Retention of the small particles in cartilage matrix |
| Butoescu | 2008 | Microparticles | PLGA | Dexamethasone/SPIONs | ~10 µm | in vitro | N.A. | Possible to incorporate 2 active substances |
| Butoescu | 2009 | Microparticles | PLGA | Dexamethasone/SPIONs | 1, 10 µm | Mice | None | Biocompatible, uptake of 1 and 10 µm particles, prolonged action of magnetic particles |
| Elron-Gross | 2009 | Collagomers | Collagen:DPPE | Diclofenac | Not stated | Rat | MIA | Better and sustained reduction of inflammation |
| Butoescu | 2009 | Microparticles | PLGA | Dexamethasone/SPIONs | ~10 µm | Mice | N.A. (dorsal air pouch) | Sustained release, first order kinetics |
| Saravanan | 2011 | Microsphere | Gelatin | Diclofenac sodium | 1–60 µm | Rabbit | None | Prolonged release |
| Zille | 2010 | Nanoparticles | PLGA, PLA, HA | FITC–dextran | Not stated | Rat | None | Weak hyperplasia, no inflammation |
| Zhang | 2011 | Microspheres | PLGA | Lornoxicam | 7.47 µm | Rabbit/rat | None | Prolonged retention |
| Panusa | 2011 | Microspheres | PLGA | Methylprednisolone | 3–60 µm | Rat | Carrageenan | Prolonged retention, less inflammation |
| Zarnescu | 2011 | Liposomes | PC:DOPE:cholesterol:stearylamine | Chondroitin sulfate | Not stated | in vitro | N.A. | Interacts with collagen |
| Eswaramoorthy | 2012 | Microspheres | PLGA | Parathyroid hormone | 51–85 µm | Rat | Papain/Cystein | Biocompatible, improved GAG and Col II levels |
| te Boekhorst | 2012 | Nanoparticles | PLGA | siRNA (against RA) | 235–285 nm | Mice | Collagen antibody | Positive effect on RA depending on dose |
| Kawadkar | 2012 | Microspheres | Genipin cross-linked chitosan | Flurbiprofen | 5.18–9.74 µm | Rat | Carrageenan | Biocompatible, prolonged retention |
| Zhang | 2012 | Microspheres | PLGA | Lornoxicam | Not stated | Rat | Papain | Biocompatible, effect comparable with weekly injections of Lornoxicam |
| Whitmire | 2012 | Nanoparticles | TEGM-CHM | Interleukin-1 Ra | 300 nm | Rat | MIA | Prolonged retention, no negative effects on cartilage |
| Gaignaux | 2012 | Microparticles | PLGA | Clonidine | 10–30 µm | in vitro | N.A. | Possible to incorporate small hydrophilic drug in PLGA |
| Présumey | 2012 | Microspheres | PLGA | anti-TNF siRNA | 23.5 µm | Mice | Collagen | Biocompatible, prolonged inhibition of TNA-α |
| Chen | 2012 | Microspheres/hydrogel | Chitosan | Brucine | 0.5–4.5 µm | Rat/rabbit | Collagenase | Prolonged retention of microsphere/hydrogel composite, inhibiting inflammation |
| Morgen | 2012 | Nanoparticles | Dextran propionate/MEH-PPV | Fluorescent labeled peptide | 100–150 nm | Rat | None | Prolonged retention of peptide, biocompatible |
| Kawadkar | 2013 | Microspheres | Genipin cross-linked gelatin | Flurbiprofen | 6.39 µm | Rat | Carrageenan | Biocompatible, prolonged release |
| Ryan | 2013 | Nanocomplex | HA-chitosan | Salmon calcitonin (sCT) | 100–200 nm | Mice | K/BxN serum | sCT-HA-chitosan nanoparticles reduces inflammation and preserves bone and cartilage |
| Ko | 2013 | Microspheres | PLGA | Sulforaphane | 14.5 µm | Rat | ACLT | Prolonged retention, inhibition of inflammation |
| Sandker | 2013 | Hydrogel | PCLA-PEG-PCLA | None | N.A. | Rat | None | Hydrogel degrades after 3+ weeks |
| Bédouet | 2013 | Microsphere | PLGA cross-linked PEG | None | 40–100 µm | Sheep | None | Slow degradation, little inflammation from MS |
| Chen | 2013 | Nanoparticles in microspheres | PLGA-PVA | Brucine | 12.38 µm | Rat | None | Prolonged retention, less burst release |
| Bédouet | 2014 | Microspheres | PEG-hydrogel | Ibuprofen | 40–100 µm | ex vivo sheep | LPS | Prolonged retention, less burst release, inhibition of inflammation |
2.2. DMOADs
2.3. Cytostatic Drugs
| DMOADs | Chemical Nature |
|---|---|
| Enzyme inhibitors | |
| MMP inhibitors (TIMP 1-4) | Protein/Peptide |
| Aggrecanase inhibitors (ADAMTS) | Small molecule |
| Cytokine inhibitors | |
| IL-1 inhibitors (IL-1 Ra) | Protein |
| TNF-α antagonists | Antibody |
| iNOS inhibitors | Various |
| Growth factors | |
| Fibroblast Growth Factor (FGF)-18 | Protein/Peptide |
| Bone morphogenetic protein (BMP)-7 | Protein/Peptide |
| Platelet-rich plasma (PRP) | Plasma |
| Drugs targeting subchondral bone | |
| Calcitonin | Peptide |
| Bisphosphonates | Bisphosphonate |
3. Drug Delivery Systems
3.1. History
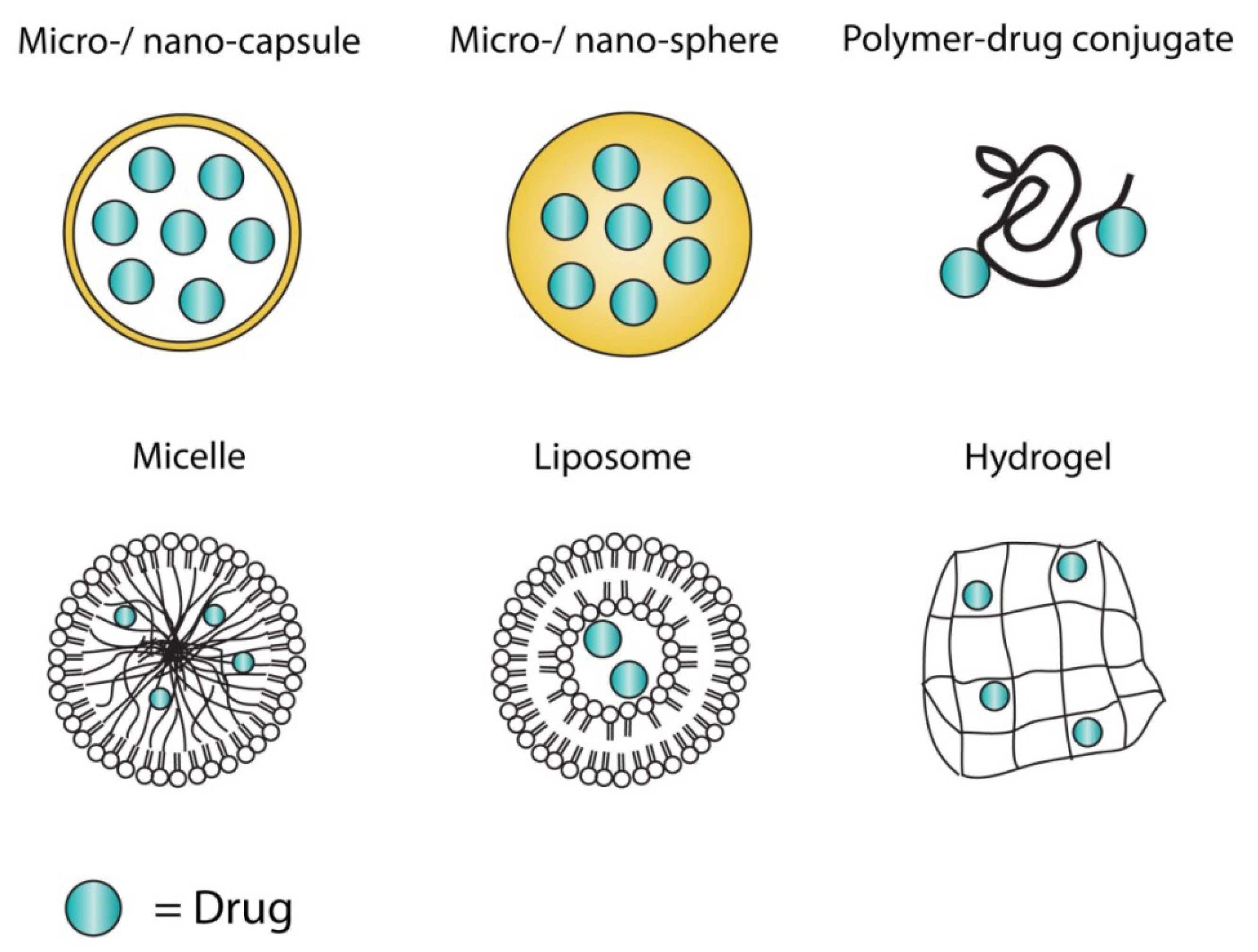
3.2. Hurdles in Drug Delivery System Design
3.3. Polymers
3.4. Liposomes
3.5. Hydrogels
4. Discussion
5. Conclusions
Conflicts of Interest
References
- Zhang, Y.; Jordan, J.M. Epidemiology of osteoarthritis. Clin. Geriatr. Med. 2010, 26, 355–369. [Google Scholar] [CrossRef]
- van Saase, J.L.; van Romunde, L.K.; Cats, A.; Vandenbroucke, J.P.; Valkenburg, H.A. Epidemiology of osteoarthritis: Zoetermeer survey. Comparison of radiological osteoarthritis in a Dutch population with that in 10 other populations. Ann. Rheum. Dis. 1989, 48, 271–280. [Google Scholar] [CrossRef]
- Buckwalter, J.A.; Saltzman, C.; Brown, T. The impact of osteoarthritis: implications for research. Clin. Orthop. Relat. Res. 2004, 427, S6–S15. [Google Scholar] [CrossRef]
- CDC. Prevalence of doctor-diagnosed arthritis and arthritis-attributable activity limitation—United States, 2007–2009. MMWR Morb. Mortal. Wkly. Rep. 2010, 59, 1261–1265. [Google Scholar]
- Haq, I.; Murphy, E.; Dacre, J. Osteoarthritis. Postgrad. Med. J. 2003, 79, 377–383. [Google Scholar] [CrossRef]
- Hermans, J.; Koopmanschap, M.A.; Bierma-Zeinstra, S.M.; van Linge, J.H.; Verhaar, J.A.; Reijman, M.; Burdorf, A. Productivity costs and medical costs among working patients with knee osteoarthritis. Arthritis Care Res. 2012, 64, 853–861. [Google Scholar] [CrossRef]
- Hochberg, M.C.; Altman, R.D.; April, K.T.; Benkhalti, M.; Guyatt, G.; McGowan, J.; Towheed, T.; Welch, V.; Wells, G.; Tugwell, P. American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care Res. 2012, 64, 465–474. [Google Scholar]
- Zhang, W.; Doherty, M.; Arden, N.; Bannwarth, B.; Bijlsma, J.; Gunther, K.P.; Hauselmann, H.J.; Herrero-Beaumont, G.; Jordan, K.; Kaklamanis, P.; Leeb, B.; Lequesne, M.; Lohmander, S.; Mazieres, B.; Martin-Mola, E.; Pavelka, K.; Pendleton, A.; Punzi, L.; Swoboda, B.; Varatojo, R.; Verbruggen, G.; Zimmermann-Gorska, I.; Dougados, M. EULAR evidence based recommendations for the management of hip osteoarthritis: report of a task force of the EULAR Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT). Ann. Rheum. Dis. 2005, 64, 669–681. [Google Scholar] [CrossRef]
- Bitton, R. The economic burden of osteoarthritis. Am. J. Manag. Care 2009, 15, S230–235. [Google Scholar]
- Dixon, T.; Shaw, M.; Ebrahim, S.; Dieppe, P. Trends in hip and knee joint replacement: socioeconomic inequalities and projections of need. Ann. Rheum. Dis. 2004, 63, 825–830. [Google Scholar] [CrossRef]
- Lavernia, C.; Lee, D.J.; Hernandez, V.H. The increasing financial burden of knee revision surgery in the United States. Clin. Orthop. Relat. Res. 2006, 446, 221–226. [Google Scholar] [CrossRef]
- Pelletier, J.P.; Martel-Pelletier, J.; Raynauld, J.P. Most recent developments in strategies to reduce the progression of structural changes in osteoarthritis: Today and tomorrow. Arthritis Res. Ther. 2006, 8. [Google Scholar] [CrossRef]
- Qvist, P.; Bay-Jensen, A.C.; Christiansen, C.; Dam, E.B.; Pastoureau, P.; Karsdal, M.A. The disease modifying osteoarthritis drug (DMOAD): Is it in the horizon? Pharmacol. Res. 2008, 58, 1–7. [Google Scholar]
- King, J.; Zhao, J.; Clingan, P.; Morris, D. Randomised double blind placebo control study of adjuvant treatment with the metalloproteinase inhibitor, Marimastat in patients with inoperable colorectal hepatic metastases: significant survival advantage in patients with musculoskeletal side-effects. Anticancer Res. 2003, 23, 639–645. [Google Scholar]
- Hudson, M.P.; Armstrong, P.W.; Ruzyllo, W.; Brum, J.; Cusmano, L.; Krzeski, P.; Lyon, R.; Quinones, M.; Theroux, P.; Sydlowski, D.; Kim, H.E.; Garcia, M.J.; Jaber, W.A.; Weaver, W.D. Effects of selective matrix metalloproteinase inhibitor (PG-116800) to prevent ventricular remodeling after myocardial infarction: results of the PREMIER (Prevention of Myocardial Infarction Early Remodeling) trial. J. Am. Coll. Cardiol. 2006, 48, 15–20. [Google Scholar] [CrossRef]
- Rudolphi, K.; Gerwin, N.; Verzijl, N.; van der Kraan, P.; van den Berg, W. Pralnacasan, an inhibitor of interleukin-1beta converting enzyme, reduces joint damage in two murine models of osteoarthritis. Osteoar. Cartil. 2003, 11, 738–746. [Google Scholar] [CrossRef]
- Edwards, S.H. Intra-articular drug delivery: the challenge to extend drug residence time within the joint. Vet. J. 2011, 190, 15–21. [Google Scholar] [CrossRef]
- Larsen, C.; Ostergaard, J.; Larsen, S.W.; Jensen, H.; Jacobsen, S.; Lindegaard, C.; Andersen, P.H. Intra-articular depot formulation principles: role in the management of postoperative pain and arthritic disorders. J. Pharm. Sci. 2008, 97, 4622–4654. [Google Scholar] [CrossRef]
- Owen, S.G.; Francis, H.W.; Roberts, M.S. Disappearance kinetics of solutes from synovial fluid after intra-articular injection. Br. J. Clin. Pharmacol. 1994, 38, 349–355. [Google Scholar] [CrossRef]
- Shuid, A.N.; Ibrahim, N.; Mohd Amin, M.C.; Mohamed, I.N. Drug delivery systems for prevention and treatment of osteoporotic fracture. Curr. Drug Targets 2013, 14, 1558–1564. [Google Scholar] [CrossRef]
- Lotz, M.; Martel-Pelletier, J.; Christiansen, C.; Brandi, M.L.; Bruyere, O.; Chapurlat, R.; Collette, J.; Cooper, C.; Giacovelli, G.; Kanis, J.A.; Karsdal, M.A.; Kraus, V.; Lems, W.F.; Meulenbelt, I.; Pelletier, J.P.; Raynauld, J.P.; Reiter-Niesert, S.; Rizzoli, R.; Sandell, L.J.; Van Spil, W.E.; Reginster, J.Y. Value of biomarkers in osteoarthritis: current status and perspectives. Ann. Rheum. Dis. 2013, 72, 1756–1763. [Google Scholar] [CrossRef]
- Committee for Medicinal Products for Human Use. Guideline on clinical investigation of medicinal products used in the treatment of osteoarthritis. 2010. Available online: http://www.ema.europa.eu (accessed on 28 January 2014).
- Conaghan, P.G.; Hunter, D.J.; Maillefert, J.F.; Reichmann, W.M.; Losina, E. Summary and recommendations of the OARSI FDA osteoarthritis Assessment of Structural Change Working Group. Osteoar. Cartil. 2011, 19, 606–610. [Google Scholar] [CrossRef]
- Pelletier, J.P.; Cooper, C.; Peterfy, C.; Reginster, J.Y.; Brandi, M.L.; Bruyere, O.; Chapurlat, R.; Cicuttini, F.; Conaghan, P.G.; Doherty, M.; Genant, H.; Giacovelli, G.; Hochberg, M.C.; Hunter, D.J.; Kanis, J.A.; Kloppenburg, M.; Laredo, J.D.; McAlindon, T.; Nevitt, M.; Raynauld, J.P.; Rizzoli, R.; Zilkens, C.; Roemer, F.W.; Martel-Pelletier, J.; Guermazi, A. What is the predictive value of MRI for the occurrence of knee replacement surgery in knee osteoarthritis? Ann. Rheum. Dis. 2013, 72, 1594–1604. [Google Scholar] [CrossRef]
- Kinds, M.B.; Marijnissen, A.C.; Viergever, M.A.; Emans, P.J.; Lafeber, F.P.; Welsing, P.M. Identifying phenotypes of knee osteoarthritis by separate quantitative radiographic features may improve patient selection for more targeted treatment. J. Rheumatol. 2013, 40, 891–902. [Google Scholar] [CrossRef]
- Serra Moreno, J.; Agas, D.; Sabbieti, M.G.; Di Magno, M.; Migliorini, A.; Loreto, M.A. Synthesis of novel pyrrolyl-indomethacin derivatives. Eur. J. Med. Chem. 2012, 57, 391–397. [Google Scholar] [CrossRef]
- Kawadkar, J.; Jain, R.; Kishore, R.; Pathak, A.; Chauhan, M.K. Formulation and evaluation of flurbiprofen-loaded genipin cross-linked gelatin microspheres for intra-articular delivery. J. Drug Target. 2013, 21, 200–210. [Google Scholar] [CrossRef]
- Kawadkar, J.; Chauhan, M.K. Intra-articular delivery of genipin cross-linked chitosan microspheres of flurbiprofen: preparation, characterization, in vitro and in vivo studies. Eur. J. Pharm. Biopharm. 2012, 81, 563–572. [Google Scholar] [CrossRef]
- Zhang, Z.; Huang, G. Intra-articular lornoxicam loaded PLGA microspheres: enhanced therapeutic efficiency and decreased systemic toxicity in the treatment of osteoarthritis. Drug Deliv. 2012, 19, 255–263. [Google Scholar] [CrossRef]
- Saravanan, M.; Bhaskar, K.; Maharajan, G.; Pillai, K.S. Development of gelatin microspheres loaded with diclofenac sodium for intra-articular administration. J. Drug Target. 2011, 19, 96–103. [Google Scholar] [CrossRef]
- Zhang, Z.; Bi, X.; Li, H.; Huang, G. Enhanced targeting efficiency of PLGA microspheres loaded with Lornoxicam for intra-articular administration. Drug Deliv. 2011, 18, 536–544. [Google Scholar] [CrossRef]
- Elron-Gross, I.; Glucksam, Y.; Biton, I.E.; Margalit, R. A novel Diclofenac-carrier for local treatment of osteoarthritis applying live-animal MRI. J. Control. Release 2009, 135, 65–70. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, G.; Sun, D.; Zhong, Y. Preparation and evaluation of biodegradable flubiprofen gelatin micro-spheres for intra-articular administration. J. Microencapsul. 2007, 24, 515–524. [Google Scholar] [CrossRef]
- Zhang, J.X.; Yan, M.Q.; Li, X.H.; Qiu, L.Y.; Li, X.D.; Li, X.J.; Jin, Y.; Zhu, K.J. Local delivery of indomethacin to arthritis-bearing rats through polymeric micelles based on amphiphilic polyphosphazenes. Pharm. Res. 2007, 24, 1944–1953. [Google Scholar] [CrossRef]
- Bozdag, S.; Calis, S.; Kas, H.S.; Ercan, M.T.; Peksoy, I.; Hincal, A.A. In vitro evaluation and intra-articular administration of biodegradable microspheres containing naproxen sodium. J. Microencapsul. 2001, 18, 443–456. [Google Scholar] [CrossRef]
- Fernandez-Carballido, A.; Herrero-Vanrell, R.; Molina-Martinez, I.T.; Pastoriza, P. Sterilized ibuprofen-loaded poly(D,L-lactide-co-glycolide) microspheres for intra-articular administration: effect of gamma-irradiation and storage. J. Microencapsul. 2004, 21, 653–665. [Google Scholar] [CrossRef]
- Fernandez-Carballido, A.; Herrero-Vanrell, R.; Molina-Martinez, I.T.; Pastoriza, P. Biodegradable ibuprofen-loaded PLGA microspheres for intraarticular administration. Effect of Labrafil addition on release in vitro. Int J. Pharm. 2004, 279, 33–41. [Google Scholar] [CrossRef]
- Tuncay, M.; Calis, S.; Kas, H.S.; Ercan, M.T.; Peksoy, I.; Hincal, A.A. In vitro and in vivo evaluation of diclofenac sodium loaded albumin microspheres. J. Microencapsul. 2000, 17, 145–155. [Google Scholar] [CrossRef]
- Tuncay, M.; Calis, S.; Kas, H.S.; Ercan, M.T.; Peksoy, I.; Hincal, A.A. Diclofenac sodium incorporated PLGA (50:50) microspheres: formulation considerations and in vitro/in vivo evaluation. Int. J. Pharm. 2000, 195, 179–188. [Google Scholar] [CrossRef]
- Thakkar, H.; Kumar Sharma, R.; Murthy, R.S. Enhanced retention of celecoxib-loaded solid lipid nanoparticles after intra-articular administration. Drugs R D 2007, 8, 275–285. [Google Scholar] [CrossRef]
- Thakkar, H.; Sharma, R.K.; Mishra, A.K.; Chuttani, K.; Murthy, R.S. Celecoxib incorporated chitosan microspheres: in vitro and in vivo evaluation. J. Drug Target. 2004, 12, 549–557. [Google Scholar] [CrossRef]
- Panusa, A.; Selmin, F.; Rossoni, G.; Carini, M.; Cilurzo, F.; Aldini, G. Methylprednisolone-loaded PLGA microspheres: A new formulation for sustained release via intra-articular administration. A comparison study with methylprednisolone acetate in rats. J. Pharm. Sci. 2011, 100, 4580–4586. [Google Scholar] [CrossRef]
- Butoescu, N.; Jordan, O.; Burdet, P.; Stadelmann, P.; Petri-Fink, A.; Hofmann, H.; Doelker, E. Dexamethasone-containing biodegradable superparamagnetic microparticles for intra-articular administration: physicochemical and magnetic properties, in vitro and in vivo drug release. Eur. J. Pharm. Biopharm. 2009, 72, 529–538. [Google Scholar] [CrossRef]
- Butoescu, N.; Jordan, O.; Petri-Fink, A.; Hofmann, H.; Doelker, E. Co-encapsulation of dexamethasone 21-acetate and SPIONs into biodegradable polymeric microparticles designed for intra-articular delivery. J. Microencapsul. 2008, 25, 339–350. [Google Scholar] [CrossRef]
- Butoescu, N.; Seemayer, C.A.; Foti, M.; Jordan, O.; Doelker, E. Dexamethasone-containing PLGA superparamagnetic microparticles as carriers for the local treatment of arthritis. Biomaterials 2009, 30, 1772–1780. [Google Scholar] [CrossRef]
- Horisawa, E.; Hirota, T.; Kawazoe, S.; Yamada, J.; Yamamoto, H.; Takeuchi, H.; Kawashima, Y. Prolonged anti-inflammatory action of DL-lactide/glycolide copolymer nanospheres containing betamethasone sodium phosphate for an intra-articular delivery system in antigen-induced arthritic rabbit. Pharm. Res. 2002, 19, 403–410. [Google Scholar] [CrossRef]
- Pavanetto, F.; Genta, I.; Giunchedi, P.; Conti, B.; Conte, U. Spray-dried albumin microspheres for the intra-articular delivery of dexamethasone. J. Microencapsul. 1994, 11, 445–454. [Google Scholar] [CrossRef]
- Morgen, M.; Tung, D.; Boras, B.; Miller, W.; Malfait, A.M.; Tortorella, M. Nanoparticles for improved local retention after intra-articular injection into the knee joint. Pharm. Res. 2013, 30, 257–268. [Google Scholar] [CrossRef]
- Park, S.R.; Park, S.H.; Jang, K.W.; Cho, H.S.; Cui, J.H.; An, H.J.; Choi, M.J.; Chung, S.I.; Min, B.H. The effect of sonication on simulated osteoarthritis. Part II: alleviation of osteoarthritis pathogenesis by 1 MHz ultrasound with simultaneous hyaluronate injection. Ultrasound Med. Biol. 2005, 31, 1559–1566. [Google Scholar] [CrossRef]
- Zille, H.; Paquet, J.; Henrionnet, C.; Scala-Bertola, J.; Leonard, M.; Six, J.L.; Deschamp, F.; Netter, P.; Verges, J.; Gillet, P.; Grossin, L. Evaluation of intra-articular delivery of hyaluronic acid functionalized biopolymeric nanoparticles in healthy rat knees. Biomed. Mater. Eng. 2010, 20, 235–242. [Google Scholar]
- Jotanovic, Z.; Mihelic, R.; Sestan, B.; Dembic, Z. Role of interleukin-1 inhibitors in osteoarthritis: an evidence-based review. Drugs Aging 2012, 29, 343–358. [Google Scholar] [CrossRef]
- Lee, A.S.; Ellman, M.B.; Yan, D.; Kroin, J.S.; Cole, B.J.; van Wijnen, A.J.; Im, H.J. A current review of molecular mechanisms regarding osteoarthritis and pain. Gene 2013, 527, 440–447. [Google Scholar] [CrossRef]
- Weinans, H.; Siebelt, M.; Agricola, R.; Botter, S.M.; Piscaer, T.M.; Waarsing, J.H. Pathophysiology of peri-articular bone changes in osteoarthritis. Bone 2012, 51, 190–196. [Google Scholar] [CrossRef]
- Intema, F.; Hazewinkel, H.A.; Gouwens, D.; Bijlsma, J.W.; Weinans, H.; Lafeber, F.P.; Mastbergen, S.C. In early OA, thinning of the subchondral plate is directly related to cartilage damage: results from a canine ACLT-meniscectomy model. Osteoar. Cartil. 2010, 18, 691–698. [Google Scholar] [CrossRef]
- Tan, M.L.; Choong, P.F.; Dass, C.R. Recent developments in liposomes, microparticles and nanoparticles for protein and peptide drug delivery. Peptides 2010, 31, 184–193. [Google Scholar] [CrossRef]
- Gupta, S.; Jain, A.; Chakraborty, M.; Sahni, J.K.; Ali, J.; Dang, S. Oral delivery of therapeutic proteins and peptides: a review on recent developments. Drug Deliv. 2013, 20, 237–246. [Google Scholar] [CrossRef]
- Jain, A.; Gulbake, A.; Shilpi, S.; Hurkat, P.; Jain, S.K. Peptide and protein delivery using new drug delivery systems. Crit. Rev. Ther. Drug Carrier. Syst. 2013, 30, 293–329. [Google Scholar] [CrossRef]
- Laslett, L.L.; Kingsbury, S.R.; Hensor, E.M.; Bowes, M.A.; Conaghan, P.G. Effect of bisphosphonate use in patients with symptomatic and radiographic knee osteoarthritis: Data from the Osteoarthritis Initiative. Ann. Rheum. Dis. 2013. [Google Scholar]
- Saito, M.; Takahashi, K.A.; Arai, Y.; Inoue, A.; Sakao, K.; Tonomura, H.; Honjo, K.; Nakagawa, S.; Inoue, H.; Tabata, Y.; Kubo, T. Intraarticular administration of platelet-rich plasma with biodegradable gelatin hydrogel microspheres prevents osteoarthritis progression in the rabbit knee. Clin. Exp. Rheumatol. 2009, 27, 201–207. [Google Scholar]
- Bragdon, B.; Bertone, A.L.; Hardy, J.; Simmons, E.J.; Weisbrode, S.E. Use of an isolated joint model to detect early changes induced by intra-articular injection of paclitaxel-impregnated polymeric microspheres. J. Invest. Surg. 2001, 14, 169–182. [Google Scholar] [CrossRef]
- Liggins, R.T.; Cruz, T.; Min, W.; Liang, L.; Hunter, W.L.; Burt, H.M. Intra-articular treatment of arthritis with microsphere formulations of paclitaxel: biocompatibility and efficacy determinations in rabbits. Inflamm. Res. 2004, 53, 363–372. [Google Scholar]
- Liang, L.S.; Jackson, J.; Min, W.; Risovic, V.; Wasan, K.M.; Burt, H.M. Methotrexate loaded poly(L-lactic acid) microspheres for intra-articular delivery of methotrexate to the joint. J. Pharm. Sci. 2004, 93, 943–956. [Google Scholar] [CrossRef]
- Hoffman, A.S. The origins and evolution of “controlled” drug delivery systems. J. Control. Release 2008, 132, 153–163. [Google Scholar] [CrossRef]
- Ratcliffe, J.H.; Hunneyball, I.M.; Wilson, C.G.; Smith, A.; Davis, S.S. Albumin microspheres for intra-articular drug delivery: investigation of their retention in normal and arthritic knee joints of rabbits. J. Pharm. Pharmacol. 1987, 39, 290–295. [Google Scholar] [CrossRef]
- Liechty, W.B.; Kryscio, D.R.; Slaughter, B.V.; Peppas, N.A. Polymers for drug delivery systems. Annu. Rev. Chem. Biomol. 2010, 1, 149–173. [Google Scholar] [CrossRef]
- Butoescu, N.; Jordan, O.; Doelker, E. Intra-articular drug delivery systems for the treatment of rheumatic diseases: A review of the factors influencing their performance. Eur J. Pharm. Biopharm. 2009, 73, 205–218. [Google Scholar] [CrossRef]
- Sinha, V.R.; Trehan, A. Biodegradable microspheres for protein delivery. J. Control. Release 2003, 90, 261–280. [Google Scholar] [CrossRef]
- Grund, S.; Bauer, M.; Fischer, D. Polymers in Drug Delivery—State of the Art and Future Trends. Adv. Eng. Mater. 2011, 13, B61–B87. [Google Scholar] [CrossRef]
- Wang, L.; Liu, Y.; Zhang, W.; Chen, X.; Yang, T.; Ma, G. Microspheres and microcapsules for protein delivery: strategies of drug activity retention. Curr. Pharm. Des. 2013, 19, 6340–6352. [Google Scholar] [CrossRef]
- Ryan, S.M.; McMorrow, J.; Umerska, A.; Patel, H.B.; Kornerup, K.N.; Tajber, L.; Murphy, E.P.; Perretti, M.; Corrigan, O.I.; Brayden, D.J. An intra-articular salmon calcitonin-based nanocomplex reduces experimental inflammatory arthritis. J. Control. Release 2013, 167, 120–129. [Google Scholar] [CrossRef]
- Brown, K.E.; Leong, K.; Huang, C.H.; Dalal, R.; Green, G.D.; Haimes, H.B.; Jimenez, P.A.; Bathon, J. Gelatin/chondroitin 6-sulfate microspheres for the delivery of therapeutic proteins to the joint. Arthritis Rheum. 1998, 41, 2185–2195. [Google Scholar] [CrossRef]
- Bedouet, L.; Moine, L.; Pascale, F.; Nguyen, V.N.; Labarre, D.; Laurent, A. Synthesis of hydrophilic intra-articular microspheres conjugated to ibuprofen and evaluation of anti-inflammatory activity on articular explants. Int. J. Pharm. 2014, 459, 51–61. [Google Scholar] [CrossRef]
- Bedouet, L.; Pascale, F.; Moine, L.; Wassef, M.; Ghegediban, S.H.; Nguyen, V.N.; Bonneau, M.; Labarre, D.; Laurent, A. Intra-articular fate of degradable poly(ethyleneglycol)-hydrogel microspheres as carriers for sustained drug delivery. Int. J. Pharm. 2013, 456, 536–544. [Google Scholar] [CrossRef]
- Gaignaux, A.; Reeff, J.; Siepmann, F.; Siepmann, J.; De Vriese, C.; Goole, J.; Amighi, K. Development and evaluation of sustained-release clonidine-loaded PLGA microparticles. Int. J. Pharm. 2012, 437, 20–28. [Google Scholar] [CrossRef]
- Sandker, M.J.; Petit, A.; Redout, E.M.; Siebelt, M.; Muller, B.; Bruin, P.; Meyboom, R.; Vermonden, T.; Hennink, W.E.; Weinans, H. In situ forming acyl-capped PCLA-PEG-PCLA triblock copolymer based hydrogels. Biomaterials 2013, 34, 8002–8011. [Google Scholar] [CrossRef]
- Chen, Z.; Liu, D.; Wang, J.; Wu, L.; Li, W.; Chen, J.; Cai, B.C.; Cheng, H. Development of nanoparticles-in-microparticles system for improved local retention after intra-articular injection. Drug Deliv. 2013. [Google Scholar] [CrossRef]
- Eswaramoorthy, R.; Chang, C.C.; Wu, S.C.; Wang, G.J.; Chang, J.K.; Ho, M.L. Sustained release of PTH(1–34) from PLGA microspheres suppresses osteoarthritis progression in rats. Acta Biomater. 2012, 8, 2254–2262. [Google Scholar] [CrossRef]
- Horisawa, E.; Kubota, K.; Tuboi, I.; Sato, K.; Yamamoto, H.; Takeuchi, H.; Kawashima, Y. Size-dependency of DL-lactide/glycolide copolymer particulates for intra-articular delivery system on phagocytosis in rat synovium. Pharm. Res. 2002, 19, 132–139. [Google Scholar] [CrossRef]
- Ko, J.Y.; Choi, Y.J.; Jeong, G.J.; Im, G.I. Sulforaphane-PLGA microspheres for the intra-articular treatment of osteoarthritis. Biomaterials 2013, 34, 5359–5368. [Google Scholar] [CrossRef]
- Mountziaris, P.M.; Sing, D.C.; Chew, S.A.; Tzouanas, S.N.; Lehman, E.D.; Kasper, F.K.; Mikos, A.G. Controlled release of anti-inflammatory siRNA from biodegradable polymeric microparticles intended for intra-articular delivery to the temporomandibular joint. Pharm. Res. 2011, 28, 1370–1384. [Google Scholar] [CrossRef]
- Mountziaris, P.M.; Sing, D.C.; Mikos, A.G.; Kramer, P.R. Intra-articular microparticles for drug delivery to the TMJ. J. Dent. Res. 2010, 89, 1039–1044. [Google Scholar] [CrossRef]
- Mountziaris, P.M.; Tzouanas, S.N.; Sing, D.C.; Kramer, P.R.; Kasper, F.K.; Mikos, A.G. Intra-articular controlled release of anti-inflammatory siRNA with biodegradable polymer microparticles ameliorates temporomandibular joint inflammation. Acta Biomater. 2012, 8, 3552–3560. [Google Scholar] [CrossRef]
- Presumey, J.; Salzano, G.; Courties, G.; Shires, M.; Ponchel, F.; Jorgensen, C.; Apparailly, F.; De Rosa, G. PLGA microspheres encapsulating siRNA anti-TNFalpha: efficient RNAi-mediated treatment of arthritic joints. Eur. J. Pharm. Biopharm. 2012, 82, 457–464. [Google Scholar] [CrossRef]
- te Boekhorst, B.C.; Jensen, L.B.; Colombo, S.; Varkouhi, A.K.; Schiffelers, R.M.; Lammers, T.; Storm, G.; Nielsen, H.M.; Strijkers, G.J.; Foged, C.; Nicolay, K. MRI-assessed therapeutic effects of locally administered PLGA nanoparticles loaded with anti-inflammatory siRNA in a murine arthritis model. J. Control. Release 2012, 161, 772–780. [Google Scholar] [CrossRef]
- Giteau, A.; Venier-Julienne, M.C.; Aubert-Pouessel, A.; Benoit, J.P. How to achieve sustained and complete protein release from PLGA-based microparticles? Int. J. Pharm. 2008, 350, 14–26. [Google Scholar] [CrossRef]
- Allison, S.D. Analysis of initial burst in PLGA microparticles. Expert Opin. Drug Deliv. 2008, 5, 615–628. [Google Scholar]
- Castaldo, L.; Corbo, P.; Maglio, G.; Palumbo, R. Synthesis and preliminary characterization of polyesteramides containing enzymatically degradable amide bonds. Polym. Bull. 1992, 28, 301–307. [Google Scholar] [CrossRef]
- Dias, A.J.A.A.; Petit, A. Microparticles comprising a crosslinked polymer. Patent WO2007107358, 27 September 2007. [Google Scholar]
- Dias, A.J.A.A.; Plum, B.J.M.; Quaedvlieg, P.J.L.M.; Wiertz, R.W. Carbamate, thiocarbamate or carbamide comprising a biomolecular moiety. Patent WO2008055666, 15 May 2008. [Google Scholar]
- Svelte Medical Systems. Available online: http://www.sveltemedical.com/news.php?pid=39&article=113 (accessed on 12 March 2014).
- Trif, M.; Guillen, C.; Vaughan, D.M.; Telfer, J.M.; Brewer, J.M.; Roseanu, A.; Brock, J.H. Liposomes as possible carriers for lactoferrin in the local treatment of inflammatory diseases. Exp. Biol. Med. 2001, 226, 559–564. [Google Scholar]
- Elron-Gross, I.; Glucksam, Y.; Margalit, R. Liposomal dexamethasone-diclofenac combinations for local osteoarthritis treatment. Int. J. Pharm. 2009, 376, 84–91. [Google Scholar] [CrossRef]
- Dong, J.; Jiang, D.; Wang, Z.; Wu, G.; Miao, L.; Huang, L. Intra-articular delivery of liposomal celecoxib-hyaluronate combination for the treatment of osteoarthritis in rabbit model. Int. J. Pharm. 2013, 441, 285–290. [Google Scholar] [CrossRef]
- Kopecek, J. Hydrogel biomaterials: A smart future? Biomaterials 2007, 28, 5185–5192. [Google Scholar] [CrossRef]
- Chen, Z.P.; Liu, W.; Liu, D.; Xiao, Y.Y.; Chen, H.X.; Chen, J.; Li, W.; Cai, H.; Cai, B.C.; Pan, J. Development of brucine-loaded microsphere/thermally responsive hydrogel combination system for intra-articular administration. J. Control. Release 2012, 162, 628–635. [Google Scholar] [CrossRef]
- Athanasiou, K.A.; Niederauer, G.G.; Agrawal, C.M. Sterilization, toxicity, biocompatibility and clinical applications of polylactic acid/polyglycolic acid copolymers. Biomaterials 1996, 17, 93–102. [Google Scholar] [CrossRef]
- ClinicalTrials. Available online: http://clinicaltrials.gov/ (accessed on 4 March 2014).
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Janssen, M.; Mihov, G.; Welting, T.; Thies, J.; Emans, P. Drugs and Polymers for Delivery Systems in OA Joints: Clinical Needs and Opportunities. Polymers 2014, 6, 799-819. https://doi.org/10.3390/polym6030799
Janssen M, Mihov G, Welting T, Thies J, Emans P. Drugs and Polymers for Delivery Systems in OA Joints: Clinical Needs and Opportunities. Polymers. 2014; 6(3):799-819. https://doi.org/10.3390/polym6030799
Chicago/Turabian StyleJanssen, Maarten, George Mihov, Tim Welting, Jens Thies, and Pieter Emans. 2014. "Drugs and Polymers for Delivery Systems in OA Joints: Clinical Needs and Opportunities" Polymers 6, no. 3: 799-819. https://doi.org/10.3390/polym6030799




