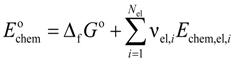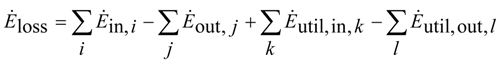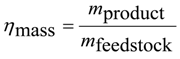2. Methods
In the following, the basic equations for calculation of exergy flows, the system boundary required for the accounting of exergy flows and the according measures are introduced.
The molar exergy content of a material flow is composed of three terms [
16], namely the chemical exergy
Echem, the physical exergy
Ephys, and the mixing exergy
Emix:
In general, exergy is the work that can be obtained by bringing a substance through a reversible process into equilibrium with the environment [
17]. Thus, exergy has to be defined with respect to a reference environment.
Chemical exergy is the exergy content of a substance at reference conditions regarding temperature and pressure. The molar chemical exergy of a component is given by the sum of molar Gibbs energy of formation Δ
fGo and molar standard exergy values
Echem,el,i that depend on the composition of the environment:
where
Nel is the number of different elements and ν
el,i denotes the stoichiometric coefficient of each element in the substance [
17]. Physical exergy is the exergy content of a substance due to differences with the environment regarding temperature and pressure. It can be calculated as a sum of enthalpy and entropy differences with the environment:
where
H is the molar enthalpy and
S the molar entropy of a substance at temperature
T and pressure
p. The subscript 0 denotes the state of the environment. Mixing exergy is usually smaller than chemical and physical exergy and can generally be neglected.
The calculation of exergy depends on a reference environment. In this paper, the reference model of Szargut
et al. is used for the composition of the environment [
17]. Regarding reference temperature and pressure standard conditions are used, because in this case, the Gibbs energy of formation also can be used at standard conditions for calculation of the chemical exergy.
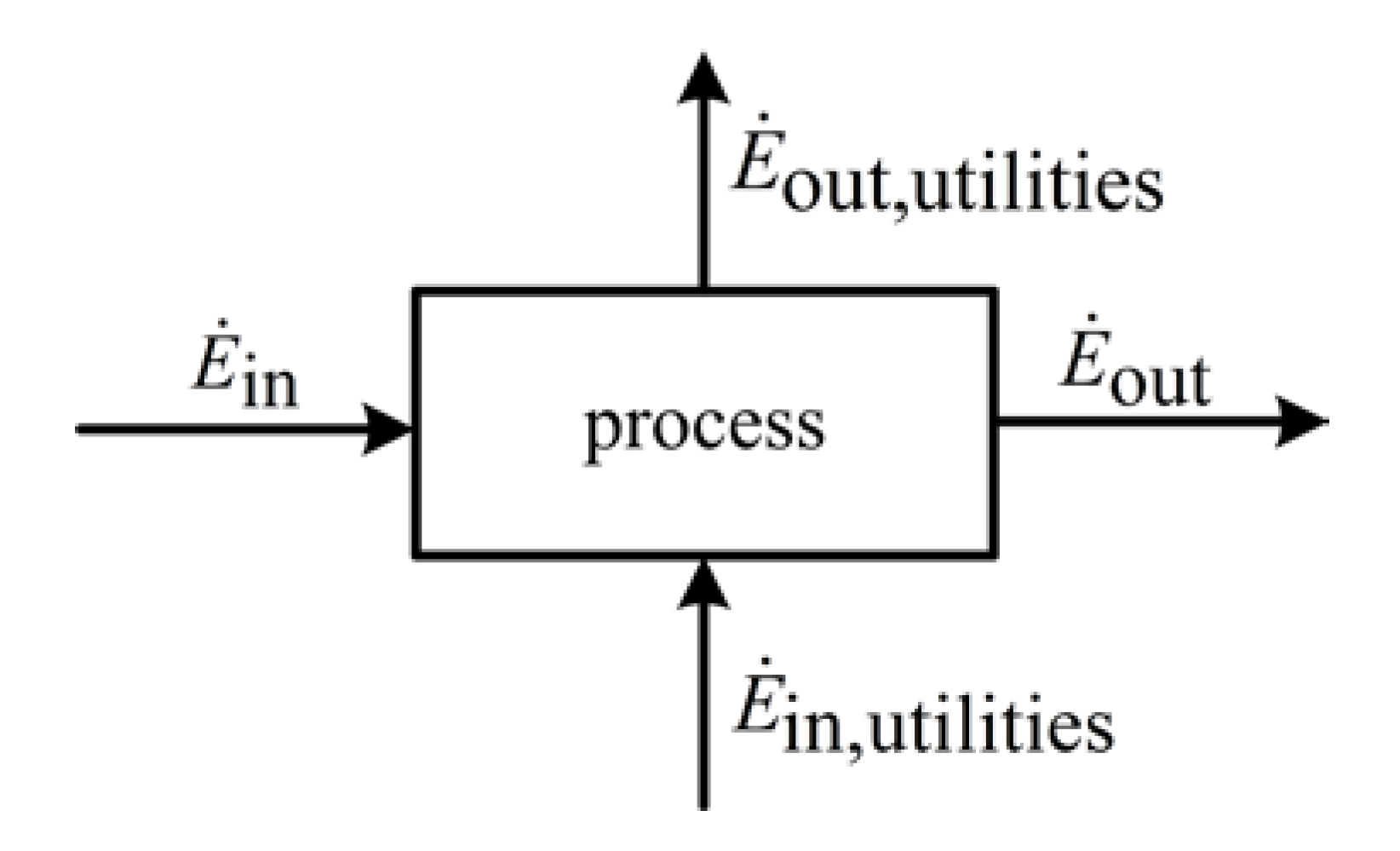
For an assessment of systems with exergy balances, an appropriate measure is required. The measure is based on the system depicted in
Figure 1.
Figure 1.
Entering and leaving exergy flows of a chemical process.
Figure 1.
Entering and leaving exergy flows of a chemical process.
The difference that results in balancing of all entering and leaving exergy flows is denoted as exergy loss
Ėloss:
where
Ėin and
Ėout denote the entering and leaving material flows and
Ėutil,in and
Ėutil,out the entering and leaving utilities.
By applying Equation (4), a synthesis pathway or a process can be judged based on a single number. In order to allow comparability, the measure is given specifically based on a functional unit. In this paper, the functional unit is one kilogram of product. According to Kim and Overcash, in 20% of the processes, valuable by-products are produced [
18]. In these cases, the exergy losses have to be allocated to main and by-products. This is a typical issue in life cycle assessments (LCA) according to DIN 14,044, so that solutions can be found in this field [
19,
20,
21,
22,
23]. As suggested by Marvuglia
et al. and Schmidt, the allocation should be carried out by causal connections that are based on physical or chemical principles [
24,
25]. Hence, a casual allocation is possible if the exergy losses of a single unit operation can be assigned distinctly to the product. If both product and by-products occur in one unit operation, a causal-based allocation is not possible without deeper understanding. In this case, a non-casual allocation is chosen based on the respective mass flows [
26,
27,
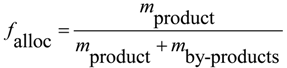
28]. To this end, the allocation factor
fallocis is defined, with which the exergy losses are weighted according to weight fraction of the product:
where
mproduct denotes the mass of product and
mby-product the mass of by-products that leave the process.
The exergy analyses based on the descriptions above are carried out in the simulation toolbox called ExergySim we developed. To this end, the underlying processes of the synthesis pathways have been modelled in this toolbox. The process models are based on process descriptions taken from literature. Essential assumptions and the associated references of the process models are depicted in
Table 1. Processes that already have been developed years ago are usually well described in Ullmann’s Encyclopedia of Industrial Chemistry [
29]. In these cases, detailed information about reaction conditions, conversions, feed compositions and downstream processing can be found. For missing information like the provision of heat, exergetically advantageous assumptions have been made. Some processes are in a stage of development in which no complete process descriptions are publicly available. If the basic technical feasibility of a process has already been demonstrated, this information is often available in patents. These patents usually describe only a certain segment of a process, like a catalyst of the reaction or a suitable downstream processing. For missing information, exergetically advantageous assumptions have also been made or assumptions have been adopted from similar processes. In an even earlier stage of development of a process, only information about the reactions is available. For these reactions that are usually only tested in laboratories, process flow sheets usually do not exist. Nevertheless, in order to carry out an evaluation, hypothetical processes with typical unit operations for separation of the reaction mixture are assumed. To ensure a general comparability of the considered processes, heat and material flows are integrated identically e.g., with respect to temperature differences for heat transfer as well as purge flows. Properties of the pure components are taken from the Design Institute for Physical Properties database [
30].
Table 1.
Basic data and references for the modeled processes.
Table 1.
Basic data and references for the modeled processes.
| Process | Reaction | References |
|---|
| glucose → ethanol | C6H12O6 → C2H6O + 2CO2 | [31] |
| Xethanol = 1 |
| Tr = 308 K |
| pr = 1 bar |
| waq = 0.8 kg/kg |
| ethanol → ethylene | C2H6O → C2H4 + H2O | [32] |
| 2C2H6O → (C2H5)2O + H2O |
| Xethanol = 0.98 |
| Sethylene = 0.97 |
| Tr = 625 K |
| pr = 40 bar |
| ethylene → ethylene oxide | C2H4+0.5 O2 → C2H4O | [33,34] |
| CH4 + 2 O2→ CO2 + 2H2O |
| Xoxygen = 0.35 |
| Sethyleneoxide = 0.80 |
| Tr = 501 K |
| pr = 16 bar |
| ethylene oxide → 1,2-ethanediol | C2H4O + H2O → C2H6O2 | [35] |
| Xethyleneoxide = 1 |
| Tr = 363 K |
| pr = 20 bar |
| glucose → 1,2-ethanediol | C6H12O6 + 3H2 → C2H6O2 | [36] |
| C6H12O6 + H2 → C6H14O6 |
| Xglucose = 0.72 |
| S1,2-ethanediol = 0.06 |
| Tr = 433 K |
| pr = 50 bar |
| waq = 0.8 kg/kg |
| glucose → lactic acid | C6H12O6 → C3H6O3 | [37] |
| Xlacticacid = 0.95 |
| Tr = 323 K |
| pr = 1 bar |
| waq = 0.8 kg/kg |
| plant oil → ethylene | Xplantoil = 0.945 | [38] |
| Sethylene = 0.41 |
| Tr = 773 K |
| pr = 1 bar |
| other by-products (mass fractions): propylene (0.14), carbon monoxide (0.05), carbon (0.06), methane (0.08), ethane (0.03), n-butane (0.13), p-xylene (0.04), benzene (0.04), hydrogen (0.01) degrees of freedom: carbon dioxide, water |
| plant oil → ethylene | C57H104O6 + 9 H2 → 27 C2H4 + C3H8O3 + 3 H2O | structure-destroying (simplified) |
| Xplantoil = 1 |
| Tr = 500 K |
| pr = 1 bar |
| plant oil → 1-octadecene | C57H104O6 + 9 H2 → 3 C18H36 + C3H8O3 + 3 H2O | structure-preserving (simplified) |
| Xplantoil = 1 |
| Tr = 370 K |
| pr = 1 bar |
3. Selection of Representative Synthesis Pathways
In principle, many different bio-based synthesis pathways are conceivable. The synthesis pathways differ in the type of bio-based feedstock, their intermediates as well as their products. This raises the question as to which combination of feedstock, intermediates and products is advantageous in terms of economic potential. As basis for such an evaluation, some representative synthesis pathways are chosen that will be evaluated.
The properties of biomass and crude oil differ significantly, especially in their chemical composition. Crude oil and other fossil raw materials consist mainly of carbon and hydrogen and essentially no oxygen. The element ratios of conventional products like the bulk plastics polyethylene, polypropylene, polystyrene are very similar to their fossil raw material and contain mostly no oxygen as well. In contrast to that, the oxygen content of biomass is up to roughly 50 wt %. To produce conventional products from biomass, the oxygen content, i.e., the O:C-ratio has to be adjusted along the synthesis pathway. The material adjustment is less, if products and raw materials are similar in element composition. Two approaches are conceivable in order to design synthesis pathways with little material adjustments: On the one hand, alternative bio-based raw materials can be used, the oxygen content of which is low and, thus, their composition is similar to today’s conventional products. On the other hand, new products can be targeted with higher oxygen content and element ratios more similar to typical bio-based raw materials.
Alternative raw materials of the first approach are lignin or plant oil. The oxygen content of both lignin and plant oil is low and, thus, they differ only slightly from today’s conventional products in this aspect. However, the depolymerisation of lignin and the selective usage of defined fragments as raw material are difficult because of its complex structure [
39]. Plant oil consists of triglycerides which are already used today in large quantities for the production of bio-diesel [
40] and chemical products like detergents.
Examples for the second approach, i.e., the adjustments of the product to its raw material, are polyethylenterephthalat and new bio-based polymers like polylactic acid and polyhydroxybutyrate, polymers with high oxygen content similar to that of glucose.
In order to evaluate the economic potential of these different routes, exergy balances can be used according to the exergoeconomic approach. As a first step, the often largest contribution to exergy, namely the chemical exergy, is regarded. Chemical exergy is a measure not only of the effects due to chemical composition but also of the effects of the molecular structure. Because the chemical exergy characterizes the value of components, it can be taken as a first indicator for evaluating the material adjustments within a synthesis pathway.
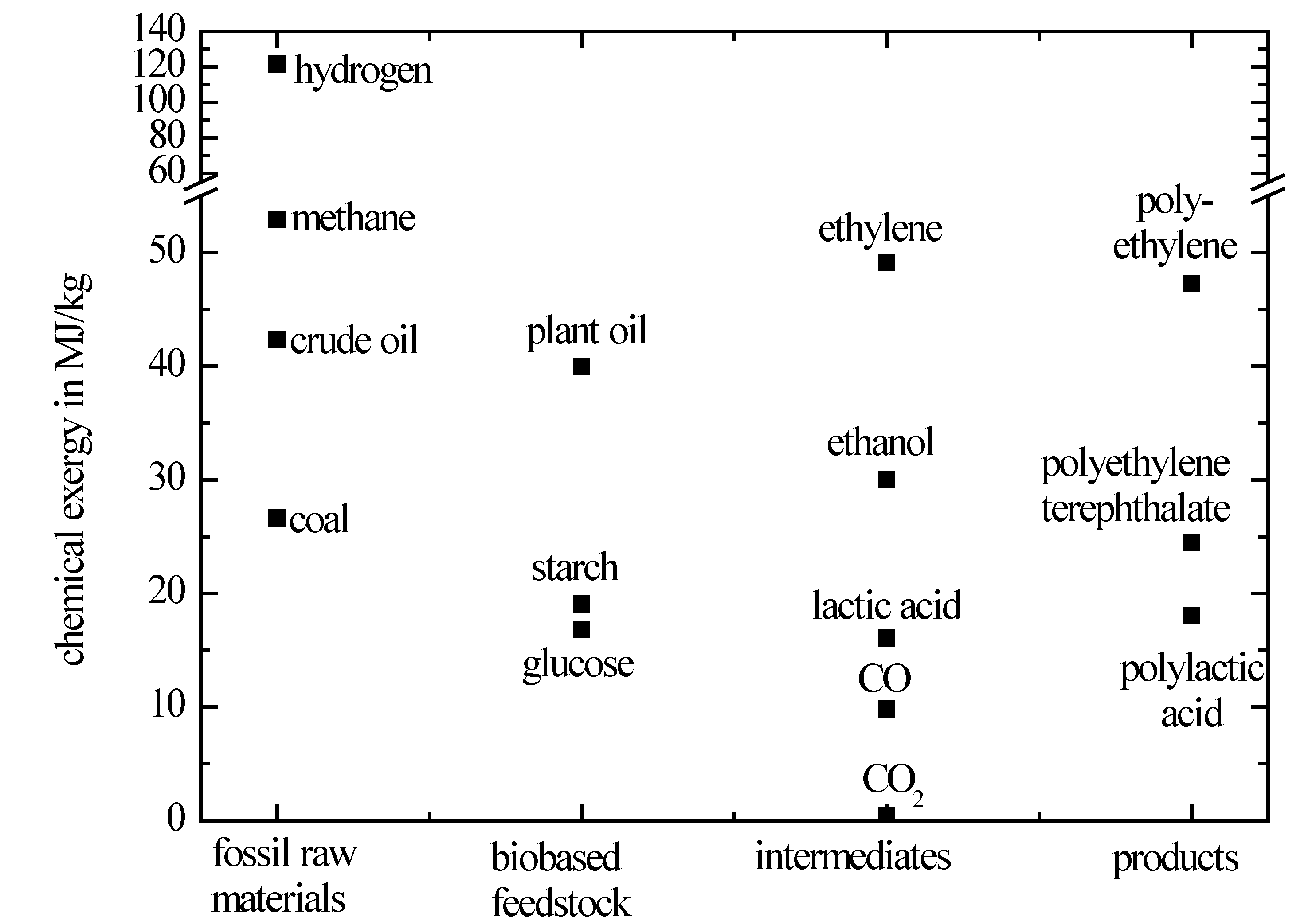
In
Figure 2, the chemical exergy of crude oil, bio-based raw materials and conventional as well as bio-based polymers is depicted. Additionally, the chemical exergy of further reagents and by-products is shown.
Figure 2.
Chemical exergy of fossil and biobased raw materials, intermediates and conventional and biobased polymers.
Figure 2.
Chemical exergy of fossil and biobased raw materials, intermediates and conventional and biobased polymers.
It is evident that the chemical exergy of components is high, if their oxygen content is low and
vice versa. Because of its low oxygen content, the chemical exergy of crude oil is relatively high. Likewise, ethylene that is a typical crude-oil based intermediate has a high chemical exergy as well as the conventional polymers depicted in
Figure 2. Therefore, the chemical exergy of the main components involved in typical synthesis pathways of petrochemistry is relatively similar. In contrast to that, the chemical exergy of bio-based precursors like glucose or starch is significantly lower. Processes for conventional products with a low oxygen content based on biobased feedstock thus have to make up for this difference. If, on the other hand, polymers with a chemical exergy similar to glucose are produced from biomass, the synthesis pathways would again be horizontal like classical crude-oil based synthesis pathways.
Thus, different routes of synthesis pathways are possible that can be characterized by the chemical exergy of the components involved. Four characteristic routes can be identified:
Horizontal routes: Chemical exergy and oxygen content of all main components involved is nearly the same.
Routes increasing in chemical exergy: Chemical exergy of the product is higher, and oxygen content is lower compared to the feedstock.
Routes decreasing in chemical exergy: Chemical exergy of the product is lower, and oxygen content is higher compared to the feedstock.
Routes first increasing, then decreasing or first decreasing, then increasing in chemical exergy: Chemical exergy and oxygen content of the product and the feedstock is similar, but chemical exergy of intermediates is higher and their oxygen content is lower or vice versa.
To investigate the effect of the different routes, the role of the chemical exergy is examined. According to Equation (2), the chemical exergy of a component is closely linked to the Gibbs energy of formation, from which the Gibbs energy of reaction can be calculated for a given stoichiometry. Frenzel
et al. have shown that the Gibbs energy of reaction needs to be negative or at most slightly positive in order to obtain processes with low overall exergy losses, because otherwise chemical equilibrium will reach only a low yield leading to high separation effort and large recycle streams [
41]. For synthesis pathways, where the chemical exergy of the product is higher than that of the feedstock, either a further exergy-rich reactant has to be added or a low-exergy by-product has to be eliminated. Both options are associated with disadvantages: To generate exergy-rich reactants like hydrogen, additional exergy is required. By elimination of low-exergy by-products like carbon dioxide or water, only a part of the feedstock is available for the product. In the opposite case, where the chemical exergy of the product is lower compared to the feedstock, either a further low-exergy reactant has to be added or an exergy-rich by-product has to be eliminated and, therefore, the amount of the main feedstock required is reduced. To account for the effects of elimination of by-products and addition of further reactants a mass efficiency is defined, which shows how well the feedstock is utilized in the main product:
where
mfeedstock is the mass of the feedstock and
mproduct that of the main product.
Thus, with reactions with additional products or reactants, it is possible to develop synthesis pathways where the exergy of the essential step is increasing or decreasing without thermodynamic limitations and where the mass efficiency allows judging the effect of the additional components. In the following, representative synthesis pathways are chosen, which correspond to the routes mentioned above.
The bio-based feedstock of the considered synthesis pathways are glucose and plant oil. As end products, polyethylene, polyethylene terephthalate and polylactic acid are chosen. Polyethylene is a plastic with the highest production volume worldwide and without any oxygen. Polyethylene terephthalate is a polyester with a high production volume as well but with a significant oxygen content. Polylactic acid is also an oxygen containing polyester which can substitute polyethylene terephthalate [
42]. The polymers polyethylene and polyethylene terephthalate are based on monomers which are conventionally produced from crude oil. Because the bio- and crude-oil based synthesis pathways compete with each other, corresponding synthesis pathways from crude oil to these polymers are chosen as a benchmark.
The chosen precursors and polymers cover substances with different exergy and oxygen content, so that the characteristic routes of the synthesis pathways discussed above can be mapped in principle. The structure of the synthesis pathways which are examined is depicted in
Table 2. The chemical composition of crude oil is assumed to be 86.5 wt % carbon, 12.5 wt % hydrogen and 1 wt % oxygen [
43]. Plant oil is approximated with triolein, a triglyceride with three oleic acid side groups.
Table 2.
Structure of the synthesis pathways.
Table 2.
Structure of the synthesis pathways.
| Feedstock | Products |
|---|
| PE | PET | PLA |
|---|
| crude oil | horizontal | decreasing | - |
| plant oil | horizontal | - | - |
| glucose | decreasing | horizontal, first increasing and then decreasing | horizontal |
4. Results and Discussion
In the following, the exergy losses of the entire synthesis pathways will be discussed. The discussion addresses several goals: First, the effects of the different characteristic routes of the synthesis pathways are indicated. Structures of synthesis pathways are identified that promise a high exergetic potential. Secondly, particularities of individual processes are indicated, where the insights based on exergy losses can be transferred to other processes. Thirdly, the applicability of exergy balances as an analysis tool is examined for processes that are in an early stage of development.
The first end product is polyethylene for which four synthesis pathways are considered. The structures of the synthesis pathways are depicted by the chemical exergy of the raw materials, intermediates and the end product in
Figure 3. Each arrow represents a process that is modelled separately.
Polyethylene contains no oxygen and, therefore, its chemical exergy is high. The typical monomer of polyethylene is ethylene, on which three of the four synthesis pathways are based. Today, ethylene is typically obtained from crude oil. The chemical exergy of the main components of this synthesis pathway is nearly identical. Therefore, the course of this synthesis pathway is horizontal. The situation is similar to synthesis pathways starting from plant oil, the chemical exergy of which is high as well. On the one hand, the alkyl substituents of plant oil can be cracked to ethylene by gasification. Therefore, the course of this synthesis pathway is horizontal. On the other hand, it has been assumed that the alkyl substituents of the triglycerides can be preserved and converted to polyethylene directly. Because the chemical exergy of the intermediate 1-octadecene is also high, the course of this synthesis pathway is horizontal as well. The fourth synthesis pathway starting from glucose has an increasing course because the chemical exergy of glucose is significantly lower than that of ethylene or polyethylene.
Figure 3.
Structure of the considered synthesis pathways to polyethylene.
Figure 3.
Structure of the considered synthesis pathways to polyethylene.
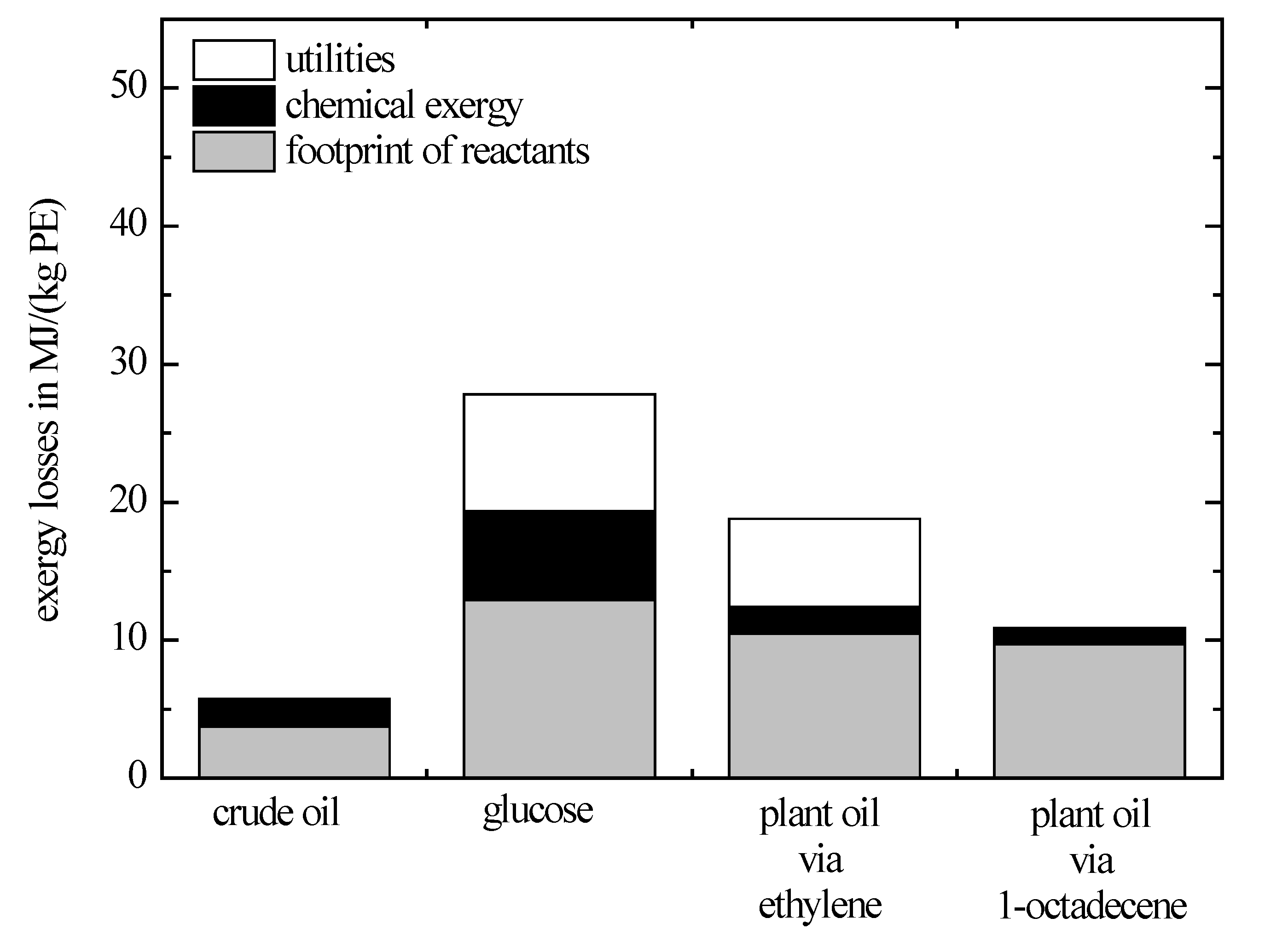
The exergy losses related to polyethylene for the whole synthesis pathways are depicted in
Figure 4. The total exergy losses are separated into three parts: exergy losses regarding chemical exergy, use of utilities, and provision of raw materials and reactants that are indicated as footprints. The values for the footprints are taken from Frenzel
et al. [
44].
Figure 4.
Specific exergy losses of the considered synthesis pathways to polyethylene.
Figure 4.
Specific exergy losses of the considered synthesis pathways to polyethylene.
The conventional synthesis pathway starting from crude oil has the lowest exergy losses of all considered synthesis pathways. In this case, the footprint refers to obtaining ethylene from crude oil. In the subsequent polymerisation, chemical exergy is destroyed in an exothermal reaction. In this exothermal reaction, sufficient heat is released to cover the external heat demand in this process.
The exergy losses of the glucose-route are significantly higher compared to the crude-oil route. The reason for that is the large input of raw materials required to obtain one unit of polyethylene. According to
Figure 3, the synthesis pathway from glucose to ethylene consists of two separate processes. In both processes, the specific chemical exergy of each product is increased compared to their respective reagents. As mentioned above, this is achieved either by adding exergy-rich reactants or low-exergy by-product has to be eliminated. Here, in the first process, carbon dioxide is released during the fermentation of glucose to ethanol, and in the second process, the production of ethylene from ethanol water is eliminated. These effects are described by the mass efficiency according to Equation (6). For the synthesis pathway of glucose to ethylene, the mass efficiency is only 0.27. That means to obtain one kilogram of ethylene, 3.7 kg of glucose are required associated with a correspondingly high footprint.
Besides the footprint of the raw materials, further exergy losses occur within the two processes. Regarding exergy losses in biotechnology processes, two aspects can be identified for the production of ethanol from glucose. On the one hand, biotechnology processes are usually carried out at moderate temperatures and ambient pressure. Therefore, the exergy losses associated with adjustment of reaction conditions are typically low. On the other hand, the product needs to be separated from a diluted aqueous solution. In this case, the downstream processing caused exergy losses of 3.2 MJ/(kg ethanol) and, hence, more than half of the total exergy losses of this process. In context of the entire synthesis pathway from glucose to polyethylene, this exergy loss is even magnified, because the mass efficiency from the intermediate ethanol to polyethylene is significantly lower than unity. Thus, not only the footprints of the primary raw materials like glucose are multiplied, but also the losses of the previous process steps within a synthesis pathway. These effects are characteristic for synthesis pathways where the exergy is increasing and, therefore, transferable to other similar synthesis pathways.
The exergy losses of the horizontal synthesis pathway starting out from plant oil are significantly lower compared to the synthesis pathway starting from glucose. The reason for this is the better utilization of the raw materials and the improved associated mass efficiency according to Equation (6). Starting from plant oil, two horizontal synthesis pathways are considered. The difference between these synthesis pathways is how well the structure of the plant oil is preserved. In the first synthesis pathway, plant oil is cracked to the C2-building block ethylene. Because polyethylene consists of long carbon chains, it seems reasonable to utilize and, therefore, preserve the alkyl substituents instead of cracking them. This is achieved if 1-octadecene is used exemplarily as intermediate in the second synthesis pathway from plant oil. The exergy losses depicted in
Figure 4 for this synthesis pathway are based on a hypothetical process and represent a lower limit. A direct comparison of both synthesis pathways shows that the exergy losses regarding raw-material footprint and chemical exergy are similar. Because both the minimal reaction temperature required for a high conversion and the intensity of the underlying endothermic reaction are significantly lower, if the alkyl substituents are preserved, the exergy losses regarding the utilities are lower compared to the synthesis pathway via ethylene. Today, no suitable catalyst is available for this kind of reaction. In order to use these savings, potential new catalysts need to be developed in the future that allow a direct, structure-preserving conversion of the feedstock.
Thus, all synthesis pathways considered above leading to an end product—the chemical exergy of which is higher than that of the starting material—can be associated with high exergy losses.
The second end product considered here is polyethylene terephthalate, a high-volume polyester. Because of its high oxygen content of 33%, the chemical exergy of polyethylene terephthalate is significantly lower compared to polyethylene. Polyethylene terephthalate is based on two monomers: 1,2-ethanediol and terephthalic acid.
The considered synthesis pathways to 1,2-ethanediol are depicted in
Figure 5. As in
Figure 4, the structure of the synthesis pathways is depicted following the chemical exergy of the main components.
Figure 5.
Structure of the considered synthesis pathways to 1,2-ethanediol that is one monomer of polyethylene terephthalate.
Figure 5.
Structure of the considered synthesis pathways to 1,2-ethanediol that is one monomer of polyethylene terephthalate.
Today, 1,2-ethanediol is produced from crude oil via ethylene. Subsequently, ethylene is oxidized, first with oxygen to ethylene oxide and afterwards with water, because of which the exergy of this synthesis pathway is decreasing. A further synthesis pathway starts from glucose and leads to 1,2-ethanediol via ethylene as well. Because the chemical exergy of ethylene is higher than that of glucose and 1,2-ethanediol, the exergy along this synthesis pathway is first increasing and then decreasing. In the following, this synthesis pathway is indicated as an indirect synthesis pathway. An alternative is the hydrogenation of glucose to 1,2-ethanediol. Because 1,2-ethanediol is obtained from glucose directly, this pathway is indicated as a direct synthesis pathway.
For these synthesis pathways to polyethylene terephthalate, it is assumed that the second monomer—terephthalic acid—is obtained in all cases from crude-oil based p-xylene because this is the usual process for its production today.
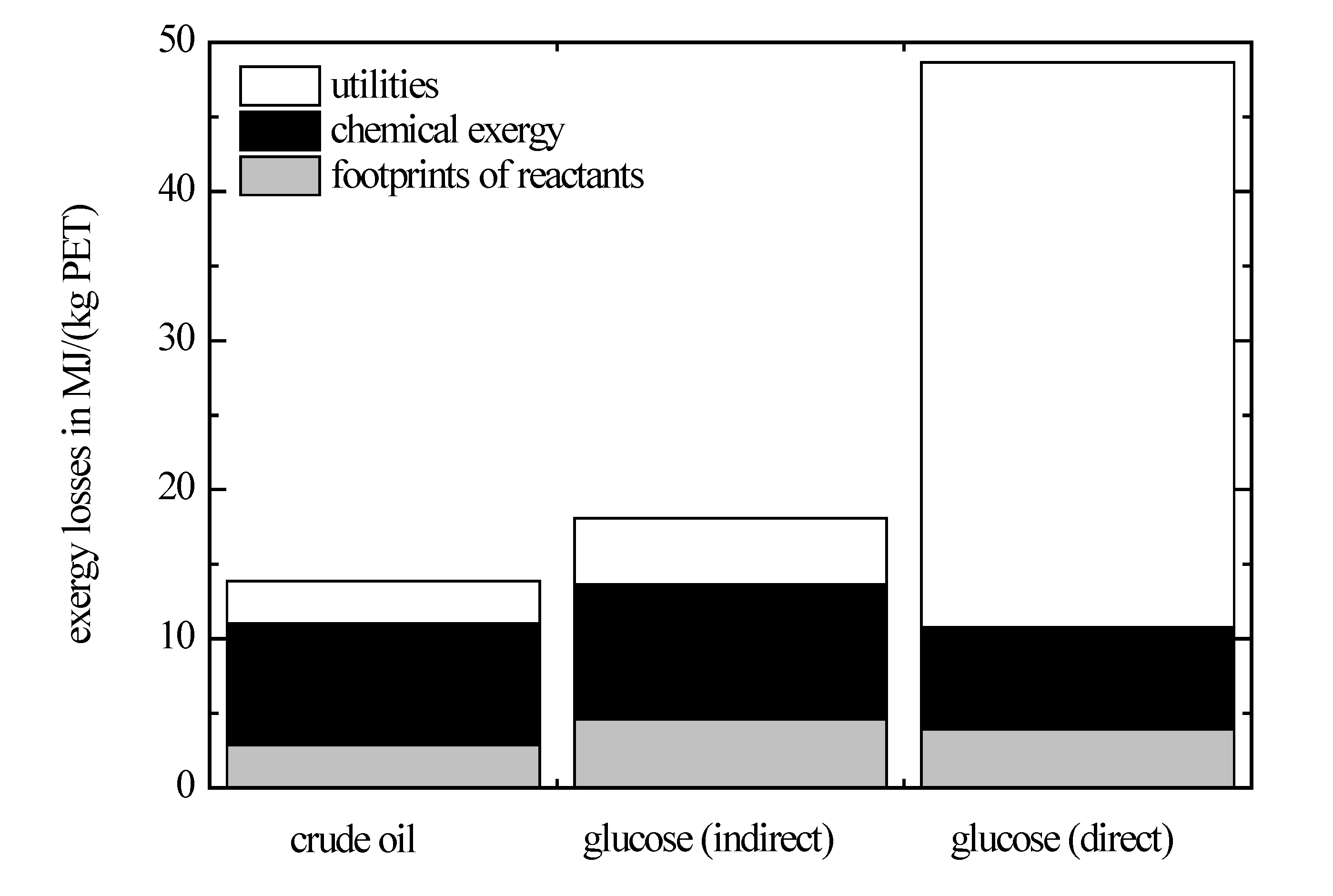
The exergy losses related to polyethylene terephthalate for these synthesis pathways are depicted in
Figure 6. Today’s synthesis pathway for production of polyethylene terephthalate leads to the lowest exergy losses. The exergy losses of the indirect synthesis pathway starting from glucose are of the same order of magnitude. In contrast to that, the exergy losses of the direct synthesis pathway from glucose are significantly higher due to the high exergy losses to produce 1,2-ethanediol from glucose where the data have been taken from literature. This process is currently in an early stage of development, and especially, conversion and selectivity achieved by the catalyst are low. According to Palkovits
et al., conversion of glucose of 72%, and selectivity to 1,2-ethanediol of only 6% have been reached [
36]. Besides 1,2-ethanediol, further by-products occur like glycerol, xylitol or sorbitol. The by-product with the highest fraction is sorbitol, which is why here sorbitol is taken as placeholder for all by-products occurring for simplicity. The exergy losses are allocated between the main product 1,2-ethanediol and this by-product based on their mass flows. Because of this allocation, the associated feedstock footprint is low for 1,2-ethandiol, although due to the low selectivity, only a small part is converted to the desired product.
Figure 6.
Specific exergy losses of the considered synthesis pathways to polyethylene terephthalate.
Figure 6.
Specific exergy losses of the considered synthesis pathways to polyethylene terephthalate.
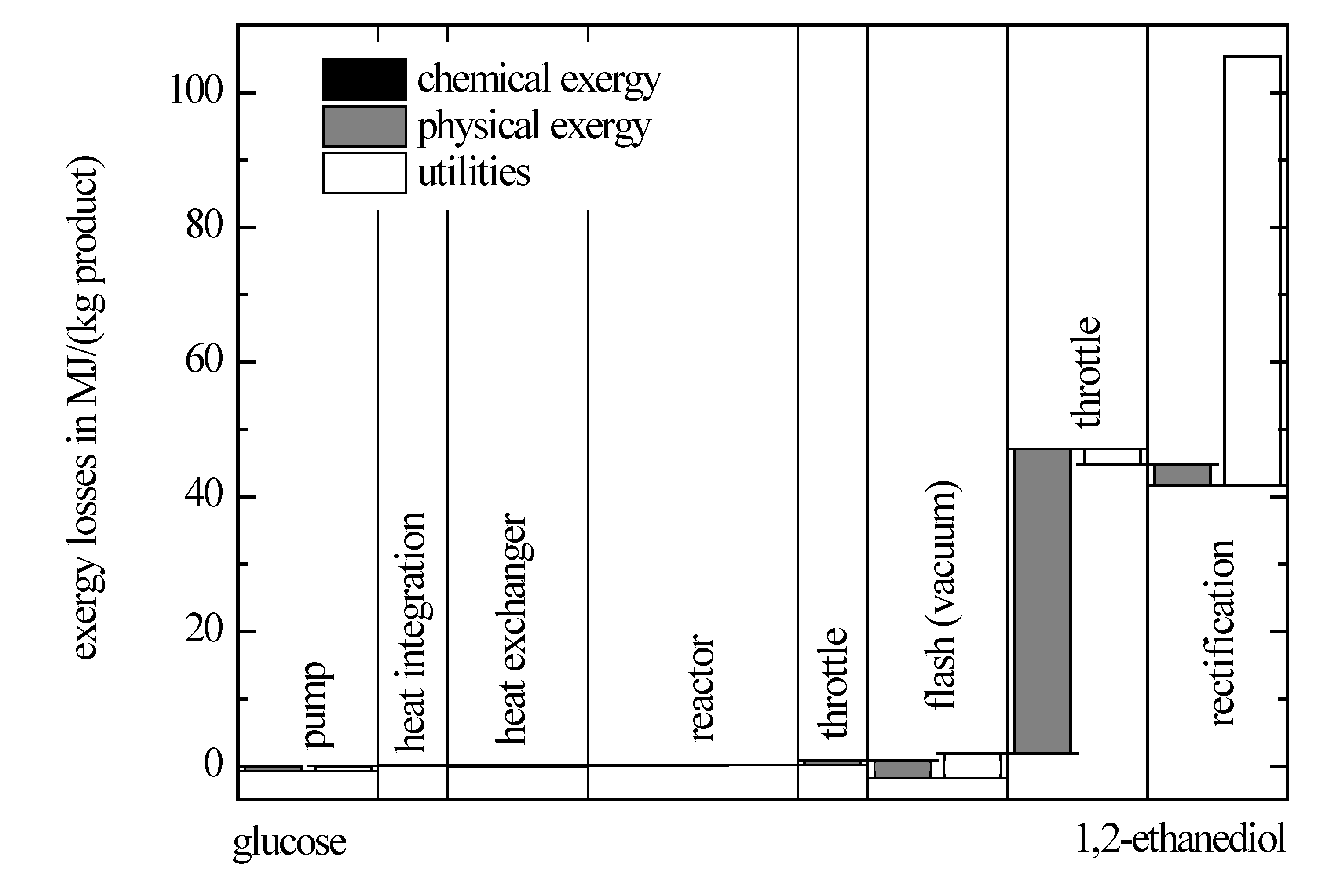
Besides the exergy losses caused by the feedstock, additional losses occur in the chemical process that are depicted in
Figure 7 for the respective exergy contributions according to Equation (1) and each unit operation. To improve the clarity of the figure, only exergy changes above 0.1 MJ/(kg product) are shown.
Figure 7.
Exergy losses caused in the process for the production of 1,2-ethanediol from glucose.
Figure 7.
Exergy losses caused in the process for the production of 1,2-ethanediol from glucose.
The exergy losses caused by setting of the reaction conditions like the temperature of 433 K and the pressure of 50 bar and carrying out of the reaction itself are actually relatively low. Because the reaction is carried out in a diluted aqueous solution, the separation of the reaction mixture is associated with high exergy losses. In a vacuum flash, glucose that is not converted is separated from water and 1,2-ethanediol. A subsequent separation of water and 1,2-ethanediol is carried out in a distillation column, where 1,2-ethanediol is the bottom product. The exergy losses can be reduced, if a higher product concentration would be achieved, for which a catalyst would be required that allows a higher selectivity or conversion. In order to formulate specific objectives for the catalyst development, the saving potential can be estimated from the corresponding exergy balances.
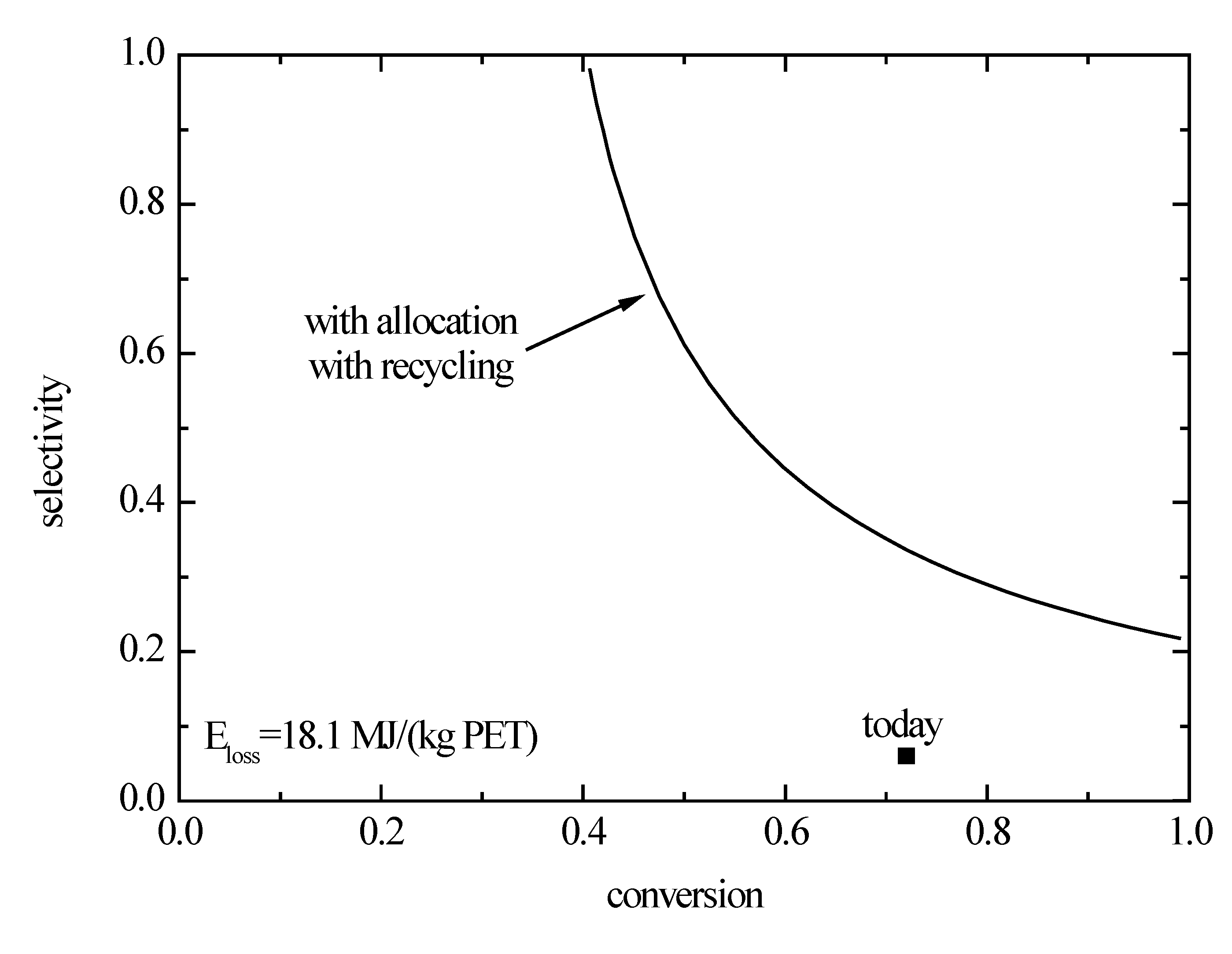
Here, a combination of selectivity and conversion is used to characterize the improvement. In the corresponding diagram (
Figure 8), it can be indicated at which combination of these variables the exergy losses of the direct synthesis pathway are identical to those of the indirect synthesis pathway. The exergy losses are based on allocation of the by-products and the recycling of the reactants that are not converted in the reactor. By allocation, the product-specific exergy losses can be reduced according to Equation (5). A further reduction of exergy losses caused by the footprint of the reactants is achieved if non-converted reagents are recycled.
Figure 8.
Selectivity and conversion for the direct synthesis pathway from glucose to polyethylene terephthalate at which the exergy losses are identical to that of the indirect pathway. The value for today is based on Palkovits
et al. [
36].
Figure 8.
Selectivity and conversion for the direct synthesis pathway from glucose to polyethylene terephthalate at which the exergy losses are identical to that of the indirect pathway. The value for today is based on Palkovits
et al. [
36].
On the one hand, this parameter study shows that the minimal selectivity to 1,2-ethanediol needs to be 20% if a complete conversion is possible in order to achieve the exergy losses of the benchmark. On the other hand, the minimal conversion needs to be 40% if a catalyst is found that avoid any side reactions. In reality, some combinations of intermediate values on these lines would need to be achieved.
As a case study, it was assumed that only the selectivity is modified as to reach the break even, i.e., a selectivity of 35% and a conversion of 72%. A comparison of the separate exergy contributions shows that the exergy losses stemming from the footprint are then nearly identical for both variants. In contrast to that, the losses according to the chemical exergy are lower for the direct variant. These results are specific to a comparison of direct and indirect synthesis pathways if the overall stoichiometry is different. Assuming an ideal stoichiometry, three molecules of 1,2-ethanediol can be obtained from glucose if the direct synthesis pathway is followed. For the indirect pathway, the yield amounts only to two molecules of the product desired.
The minimal exergy losses of the direct synthesis pathway for a complete conversion and avoiding of any side reactions is 14.1 MJ/(kg PET). This value is of the same order of magnitude as the exergy losses of the crude-oil route. Therefore, in principle, it is possible to carry out bio-based synthesis pathways with exergy losses being comparable to today’s crude-oil pathways. From this, a need for further research can be derived, with the goal to optimize catalysts for bio-based synthesis pathways, because from the exergetical point of view, significant potential has been identified. Polyethylene terephthalate and its monomers are typical products of the petrochemistry; thus, the conversions and selectivities of the crude-oil route are high and the associated exergy losses are low as expected.
The third product considered in this study is polylactic acid, a typical bio-based polymer. Polylactic acid is a polyester, the oxygen content of which is high and the chemical exergy low. In this paper, only one synthesis pathway to polylactic acid is considered. Polylactic acid is obtained from glucose via lactic acid that is obtained by fermentation. The fermentation of glucose to lactic acid is very efficient; hence, the conversion is nearly unity. By-products occur only in small quantities with less than 0.5%, which is why they can be neglected in this consideration [
37]. According to
Figure 2, the chemical exergy of the main components involved in this synthesis pathway is nearly the same and, therefore, the course of this synthesis pathway is horizontal. The exergy losses caused for the production of one kilogram of polylactic acid including the footprint of the feedstock amounts to approximately 10 MJ. This low value is mainly related to the high conversion and selectivity of the reaction which have already been achieved today; thus, the feedstock is well utilized and the separation of the reaction mixture is relatively simple. Because both polylactic acid and polyethylene terephthalate are polyesters, the properties of which are similar, they can substitute each other in principle [
42]. Compared to bio-based synthesis pathways to polyethylene terephthalate, the exergy losses of the pathway to polylactic acid are lower assuming today’s achievable conditions. Thus, by utilizing appropriately adapted monomers, bio-based synthesis pathways are possible today, the exergy losses of which can compete with their crude-oil based counterpart.