Synthesis, Structure and Solid State Properties of Cyclohexanemethylamine Substituted Phenalenyl Based Molecular Conductor
Abstract
:1. Introduction

2. Results and Discussion
2.1. Synthesis of Ligand, Preparation and Electrochemical Properties of Radical 11

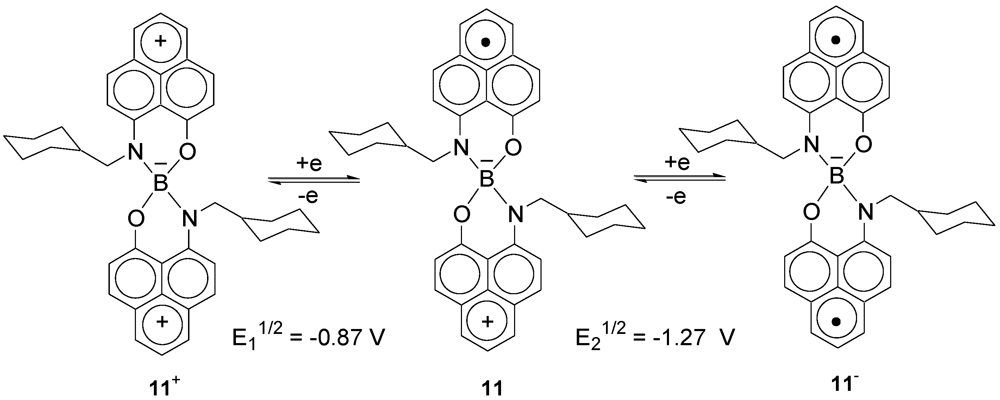
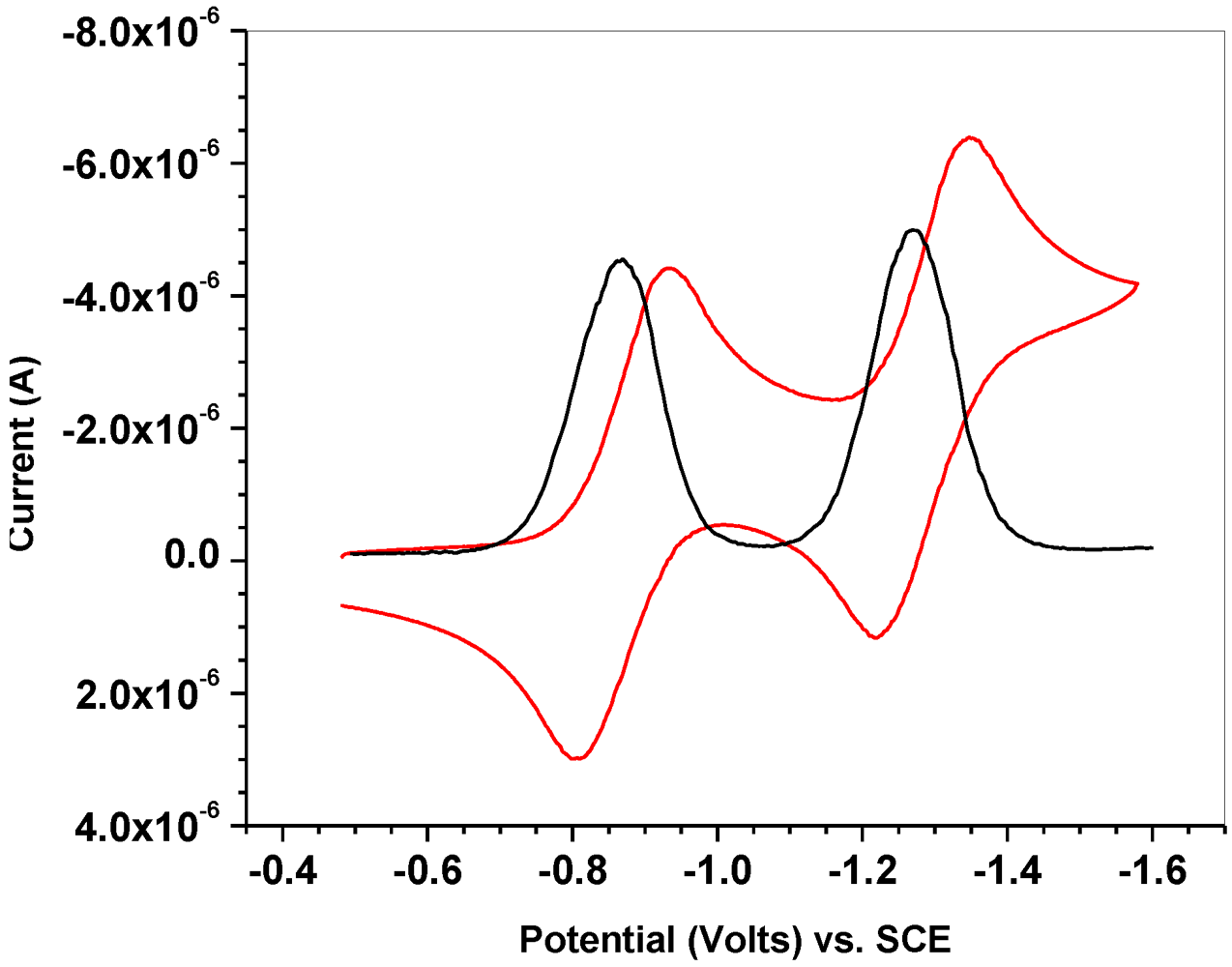
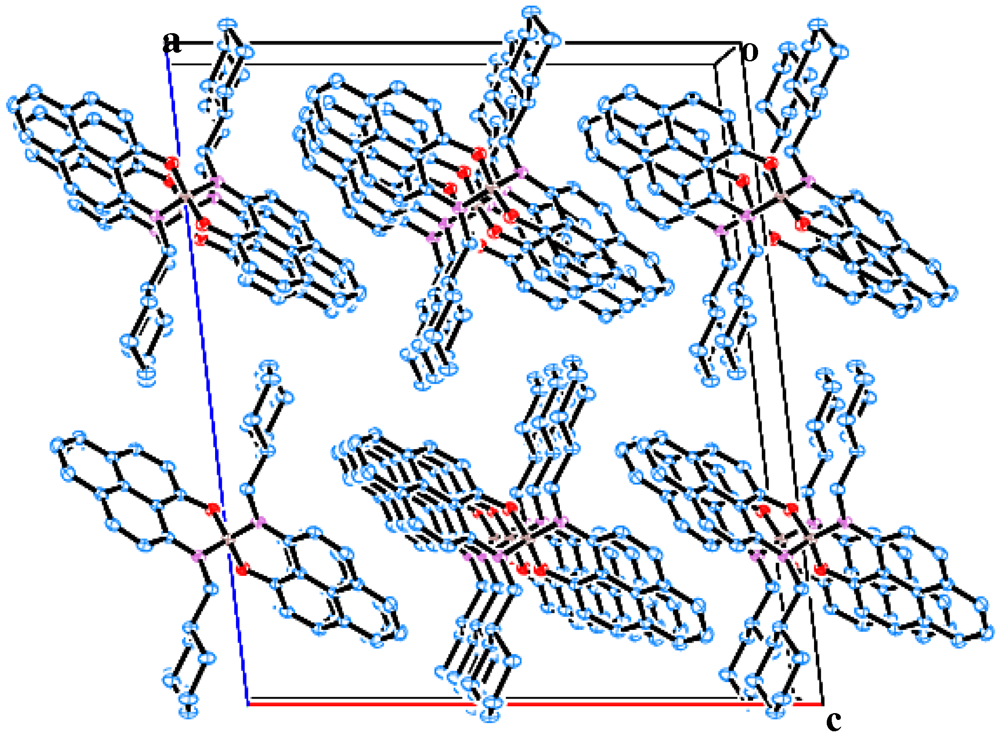
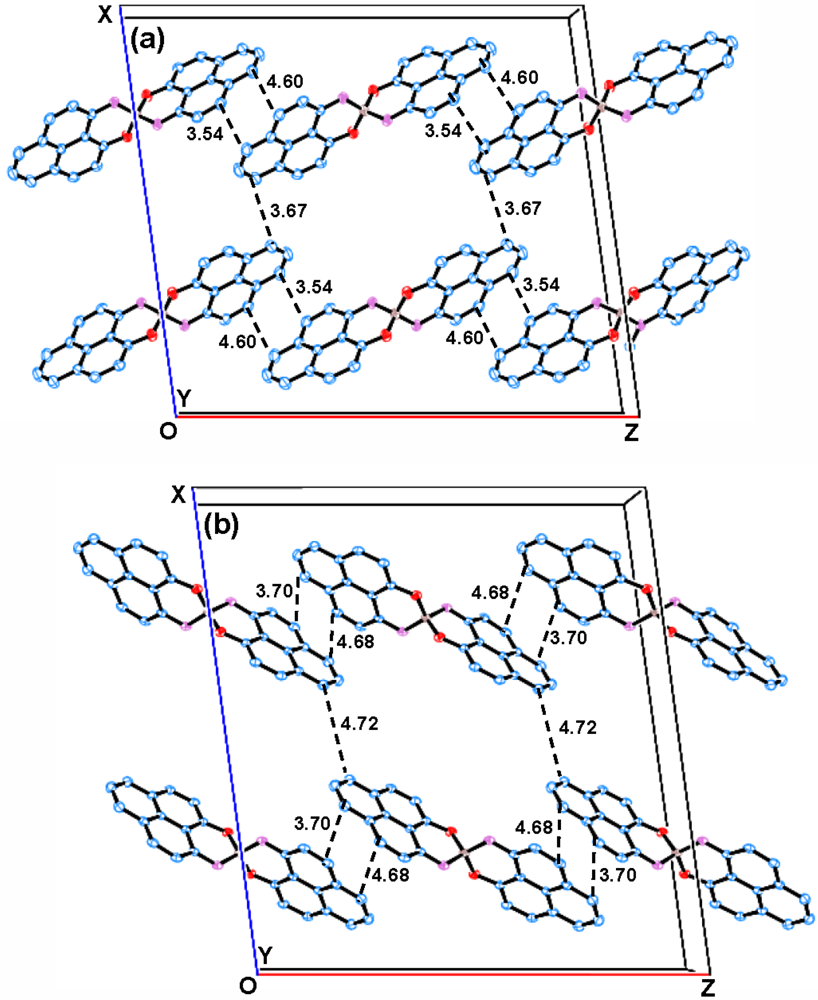
2.2. X-ray Crystal Structure of 11

| Compound | 11 | 7 |
| Temperature | 100(2) K | 223(2) K |
| Formula | C40H40BN2O2 | C40H28BN2O2 |
| Formula Weight | 591.55 | 579.45 |
| Crystal System | Monoclinic | Monoclinic |
| Space group | C2/c | C2/c |
| a, Å | 21.4373(15) | 23.386(2) |
| b, Å | 6.1623(4) | 5.7326(6) |
| c, Å | 23.2775(16) | 20.963(2) |
| α, degree | 90 | 90 |
| β, degree | 97.597 (4) | 97.782 |
| γ, degree | 90 | 90 |
| V, Å3 | 3048(4) | 2784.6(5) |
| Z | 4 | 4 |
| Crystal size, mm3 | 0.40 × 0.39 × 0.15 | 0.55 × 0.19 × 0.17 |
| Theta range for data collection | 1.77 to 27.52° | 1.76 to 26.37° |
| Reflection collected | 19619 | 8590 |
| Independent reflections | 3460 | 2209 |
| Goodness-of-fit on F2 | 1.033 | 1.038 |
| Final R indices [I > 2sigma(I)] | R1 = 0.0401 wR2 = 0.1005 | R1 = 0.0396 wR2 = 0.1027 |
| R indices (all data) | R1 = 0.0607, wR2 = 0.1157 | R1 = 0.0524, wR2 = 0.1142 |
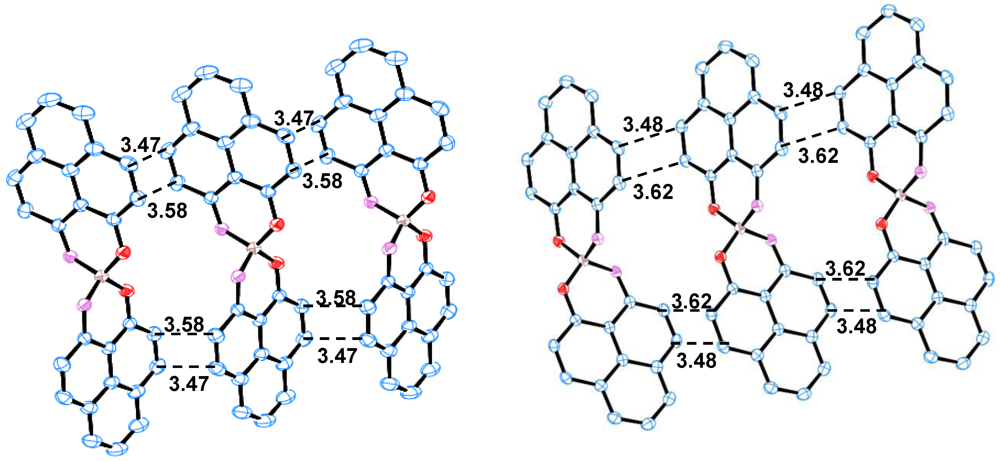
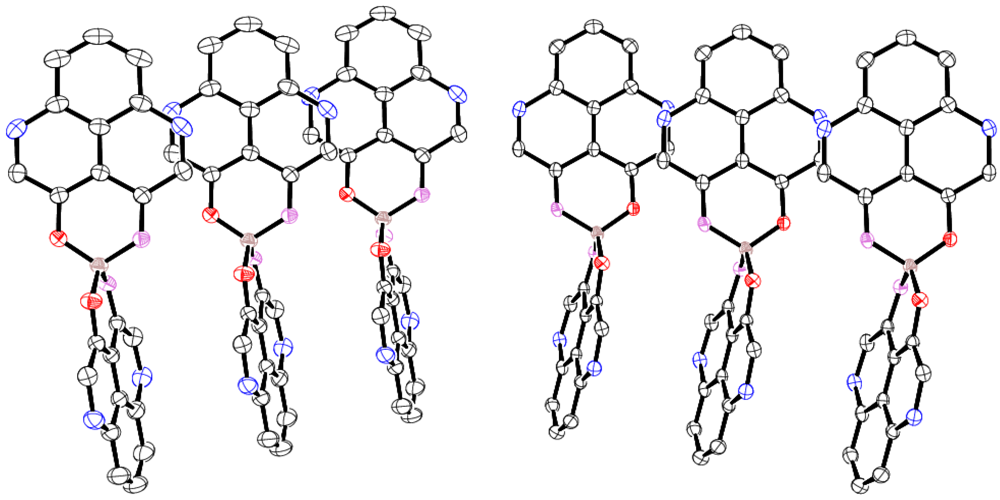
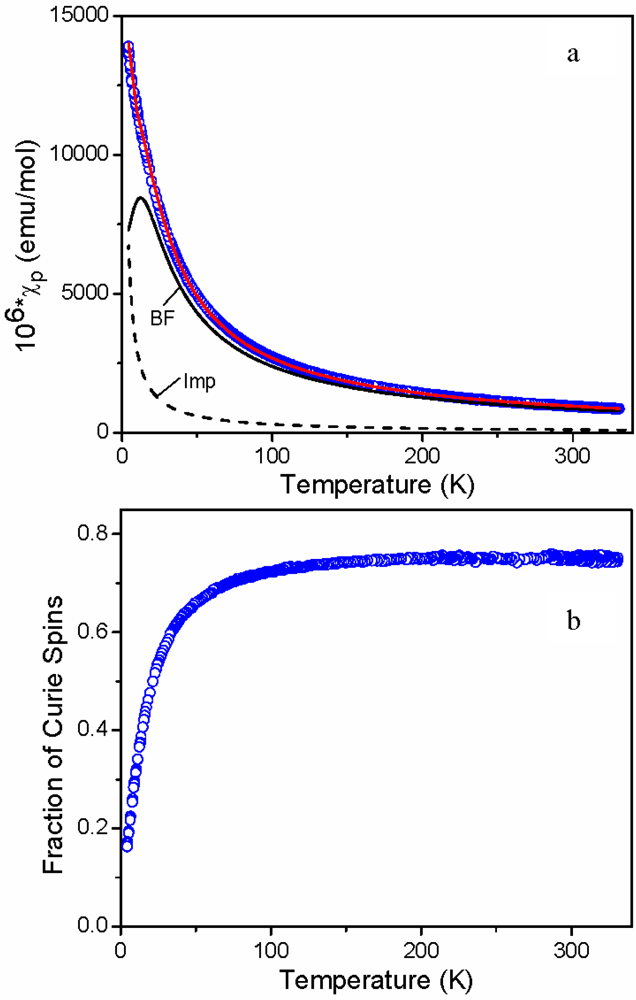
2.3. Magnetic Susceptibility of 11
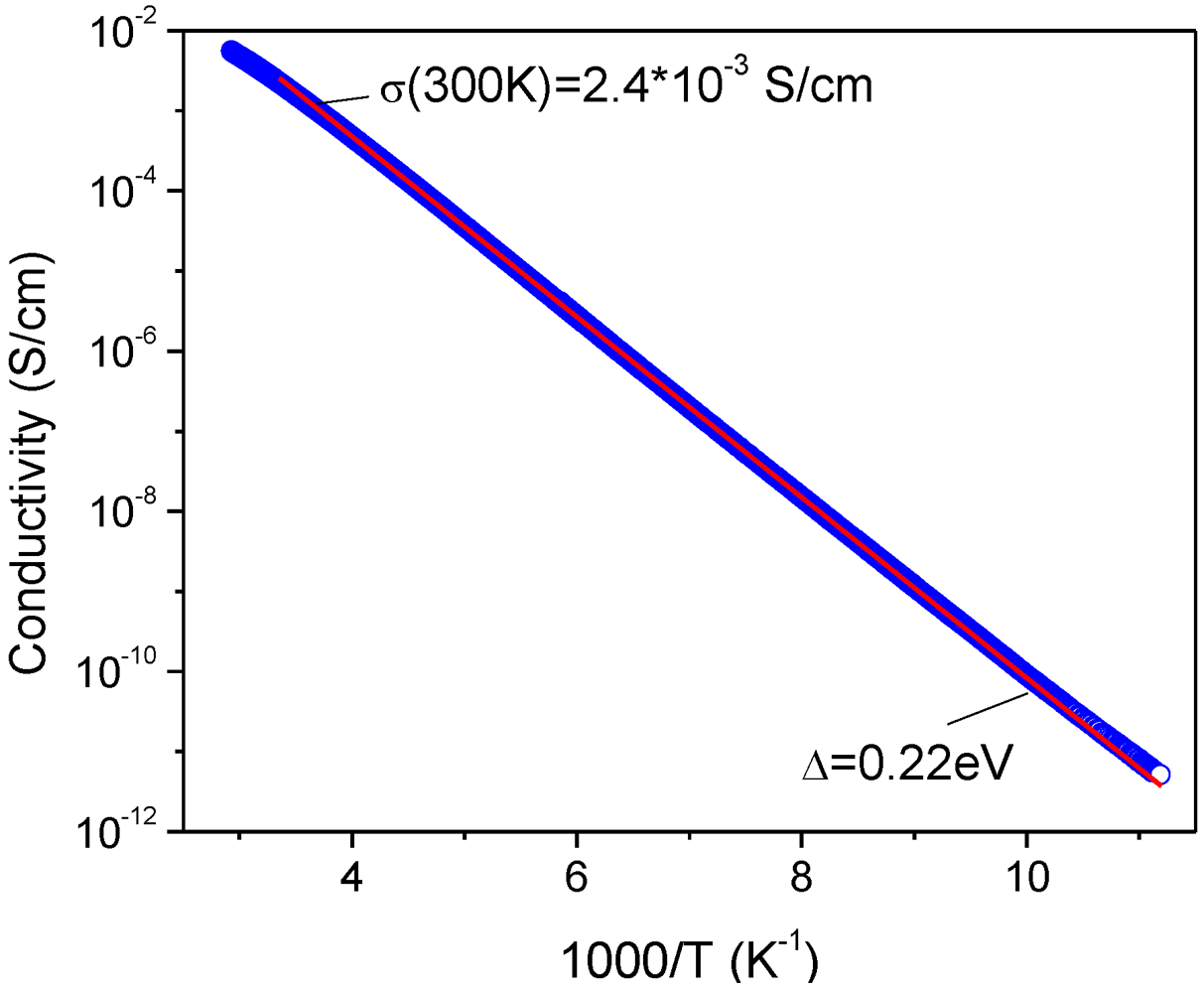
2.4. Conductivity of 11
2.5. Optical Measurement of 11
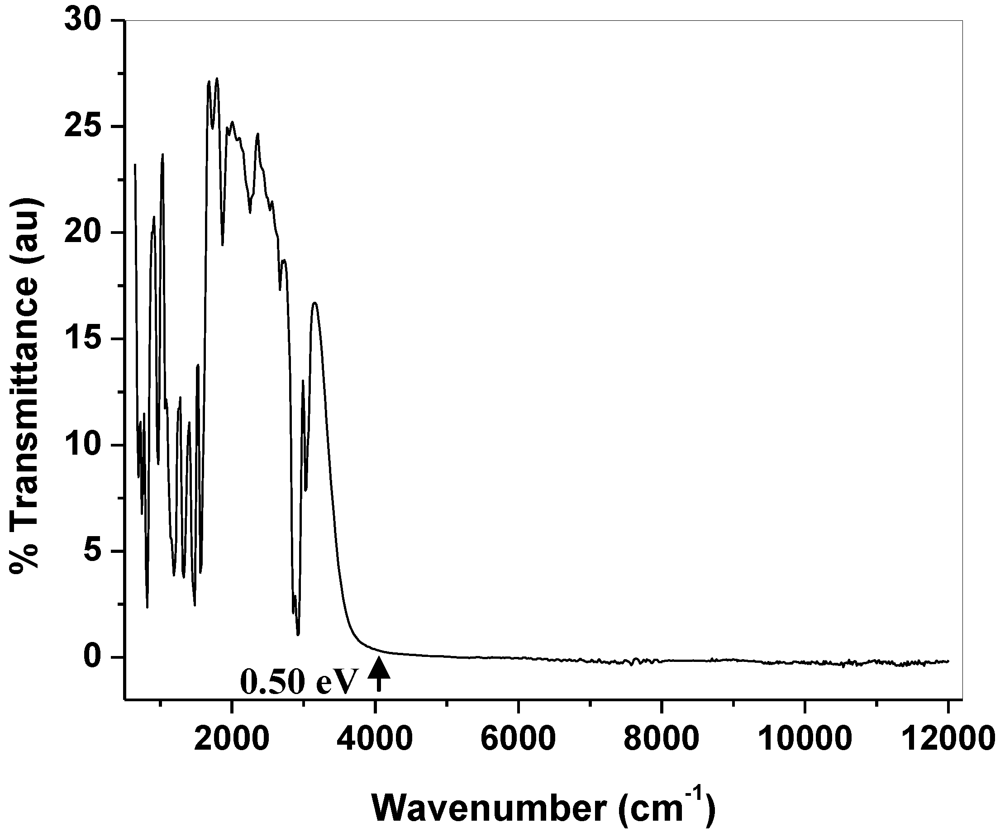
2.6. Band Electronic Structure of 11

3. Experimental Section
3.1. Materials
3.2. Preparation of NCH2CyHx, O-PLY
3.3. Preparation of [(NCH2CyHx,O-PLY)2B]+BPh4− (11+BPh4−)
3.4. Crystallization of [(NCH2CyHx,O-PLY)2B]• (11)
3.5. Cyclic Voltammetry
3.6. X-ray Crystallography
3.7. Magnetic Susceptibility Measurements
3.8. Conductivity Measurements
3.9. Single-Crystal Near- and Mid-Infrared Transmission Spectroscopy
3.10. Band Structure Calculations
3.11. Electron Paramagnetic Resonance Spectroscopy
4. Conclusions
Conflict of Interest
Acknowledgements
Supplementary Files
References
- Wolf, S.A.; Awschalom, D.D.; Buhrman, R.A.; Daughton, J.M.; von Molnar, S.; Roukes, M.L.; Chtchelkanova, A.Y.; Treger, D.M. Spintronics: A spin-based electronics vision for the future. Science 2001, 294, 1488–1495. [Google Scholar]
- Matsushita, M.M.; Kawakami, H.; Kawada, Y.; Sugawara, T. Negative magnetoresistance observed on an ion radical salt of a TTF-based spin polarized donor. Chem. Lett. 2007, 36, 110–111. [Google Scholar]
- Matsushita, M.M.; Kwakami, H.; Sugawara, T.; Ogata, M. Molecule-based system with coexisting conductivity and magnetism and without magnetic inorganic ions. Phys. Rev. B 2008, 77, 195208–195213. [Google Scholar]
- Takahide, Y.; Konoike, T.; Enomoto, K.; Nishimura, M.; Terashima, T.; Uji, S.; Yamamoto, H.M. Large positive magnetoresistance of insulating organic crystals in the non-ohmic region. Phy. Rev. Lett. 2007, 98, 116602–116605. [Google Scholar]
- Dediu, V.A.; Hueso, L.E.; Bergenti, I.; Taliani, C. Spin routes in organic semiconductors. Nat. Mater. 2009, 8, 707–716. [Google Scholar]
- Hicks, R.G. What's new in stable radical chemistry? Org. Biomol. Chem. 2007, 5, 1321–1338. [Google Scholar] [CrossRef]
- Hicks, R.G. A new spin on bistability. Nat. Chem. 2011, 3, 189–191. [Google Scholar]
- Morita, Y.; Nishida, S. Stable radicals: Fundamentals and applied aspects of odd-electron compounds. In Phenalenyls, Cyclopentadienyls, and Other Carbon-Centered Radicals; Hicks, R.G., Ed.; John Wiley & Sons: West Suxxes, UK, 2010; pp. 83–132. [Google Scholar]
- Reid, D.H. Syntheses and properties of the perinaphthyl radical. Chem. Ind. 1956, 1504–1505. [Google Scholar]
- Reid, D.H. The chemistry of the phenalenes. Quart. Rev. 1965, 19, 274–302. [Google Scholar]
- Morita, Y.; Suzuki, S.; Sato, K.; Takui, T. Synthetic organic spin chemistry for structurally well-defined open-shell graphene fragments. Nat. Chem. 2011, 3, 197–204. [Google Scholar]
- Morita, Y.; Suzuki, S.; Fukui, K.; Nakazawa, S.; Kitagawa, H.; Kishida, H.; Okamoto, H.; Naito, A.; Sekine, A.; Ohashi, Y.; et al. Thermochromism in an organic crystal based on the coexistance of σ- and π-dimers. Nat. Mater. 2008, 7, 48–51. [Google Scholar]
- Haddon, R.C. Design of organic metals and superconductors. Nature 1975, 256, 394–396. [Google Scholar]
- Haddon, R.C. Quantum chemical studies in the design of organic metals. III. Odd-alternant hydrocarbons (OAHs): The phenalenyl (PLY) system. Aust. J. Chem. 1975, 28, 2343–2351. [Google Scholar] [CrossRef]
- Haddon, R.C. Quantum chemical studies in the design of organic metals. II. The equilibrium geometries and electronic structure of tetrathiafulvalene (TTF) and tetracyanoquinodimethane (TCNQ) and their uni- and di-valent ions. Aust. J. Chem. 1975, 28, 2333–2342. [Google Scholar] [CrossRef]
- Goto, K.; Kubo, T.; Yamamoto, K.; Nakasuji, K.; Sato, K.; Shiomi, D.; Takui, T.; Kubota, M.; Kobayashi, T.; Yakusi, K.; et al. A stable neutral hydrocarbon radical: Synthesis, crystal structure, and physical properties of 2,5,8-tri-tert-butyl-phenalenyl. J. Am. Chem. Soc. 1999, 121, 1619–1620. [Google Scholar]
- Morita, Y.; Aoki, T.; Fukui, K.; Nakazawa, S.; Tamaki, K.; Suzuki, S.; Fuyuhiro, A.; Yamamoto, K.; Sato, K.; Shiomi, D.; et al. A new trend in phenalenyl chemistry: A persistent neutral radical, 2,5,8-tri-tert-butyl-1,3-diazaphenalenyl, and the excited triplet state of the gable syn-dimer in the crystal of column motif. Angew. Chem. Int. Ed. 2002, 41, 1793–1796. [Google Scholar]
- Morita, Y.; Nishida, S.; kawai, J.; Takui, T.; Nakasuji, K. Oxophenalenoxyl: Novel stable neutral radicals with a unique spin-delocalized nature depending on topological symmetries and redox states. Pure Appl. Chem. 2008, 80, 507–517. [Google Scholar]
- Koutentis, P.A.; Chen, Y.; Cao, Y.; Best, T.P.; Itkis, M.E.; Beer, L.; Oakley, R.T.; Brock, C.P.; Haddon, R.C. Perchlorophenalenyl radical. J. Am. Chem. Soc. 2001, 123, 3864–3871. [Google Scholar]
- Fukui, K.; Sato, K.; Shiomi, D.; Takui, T.; Itoh, K.; Gotoh, K.; Kubo, T.; Yamamoto, K.; Nakasuji, K.; Naito, A. Electronic structure of a stable phenalenyl radical in crystalline state as studied by squid measurements, cw-esr, and 13C cp/mas nmr spectroscopy. Synth. Met. 1999, 103, 2257–2258. [Google Scholar] [CrossRef]
- Small, D.; Zaitsev, V.; Jung, Y.; Rosokha, S.V.; Head-Gordon, M.; Kochi, J.K. Intermolecular π to π bonding between stacked aromatic dyads. Experimental and theoretical binding energies and near-ir optical transitions for phenalenyl radical/radical versus radical/cation dimerizations. J. Am. Chem. Soc. 2004, 126, 13850–13858. [Google Scholar]
- Zheng, S.; Lan, J.; Khan, S.I.; Rubin, Y. Synthesis, characterization, and coordination chemistry of the 2-azaphenalenyl radical. J. Am. Chem. Soc. 2003, 125, 5786–5791. [Google Scholar]
- Zheng, S.; Thompson, J.D.; Tontcheva, A.; Khan, S.I.; Rubin, Y. Perchloro-2,5,8-triazaphenalenyl radical. Org. Lett. 2005, 7, 1861–1863. [Google Scholar]
- Haddon, R.C.; Wudl, F.; Kaplan, M.L.; Marshall, J.H.; Cais, R.E.; Bramwell, F.B. 1,9-dithiophenalenyl system. J. Am. Chem. Soc. 1978, 100, 7629–7633. [Google Scholar]
- Beer, L.; Reed, R.W.; Robertson, C.M.; Oakley, R.T.; Tham, F.S.; Haddon, R.C. Tetrathiophenalenyl radical and its disulfide-bridged dimer. Org. Lett. 2008, 10, 3121–3123. [Google Scholar]
- Beer, L.; Mandal, S.K.; Reed, R.W.; Oakley, R.T.; Tham, F.S.; Donnadieu, B.; Haddon, R.C. The first electronically stabilized phenalenyl radical: Effect of substituents on solution chemistry and solid state structure. Cryst. Growth Design 2007, 7, 802–809. [Google Scholar]
- Parkin, S.S.P.; Engler, E.M.; Schumaker, R.R.; Lagier, R.; Lee, V.Y.; Scott., J.C.; Greene, R.L. Superconductivity in a new family of organic conductors. Phys. Rev. Lett. 1983, 50, 270–273. [Google Scholar]
- Haddon, R.C.; Hebard, A.F.; Rosseinsky, M.J.; Murphy, D.W.; Duclos, S.J.; Lyons, K.B.; Miller, B.; Rosamilia, J.M.; Fleming, R.M.; Kortan, A.R.; et al. Conducting films of C60 and C70 by alkali metal doping. Nature 1991, 350, 320–322. [Google Scholar]
- Pal, S.K.; Itkis, M.E.; Tham, F.S.; Reed, R.W.; Oakley, R.T.; Haddon, R.C. Resonating valence-bond ground state in a phenalenyl-based neutral radical conductor. Science 2005, 309, 281–284. [Google Scholar]
- Bag, P.; Itkis, M.E.; Pal, S.K.; Donnadieu, B.; Tham, F.S.; Park, H.; Schleuter, J.A.; Siegrist, T.; Haddon, R.C. Resonating valence bond and sigma-charge density wave phases in a benzannulated phenalenyl radical. J. Am. Chem. Soc. 2010, 132, 2684–2694. [Google Scholar]
- Robertson, C.M.; Leitch, A.A.; Cvrkalj, K.; Myles, D.J.T.; Reed, R.W.; Dube, P.A.; Oakley, R.T. Ferromagnetic ordering in bisthiaselenazoyl radicals: Variations on a tetragonal theme. J. Am. Chem. Soc. 2008, 130, 14791–14801. [Google Scholar]
- Leitch, A.A.; Lekin, K.; Winter, S.M.; Downie, L.E.; Tsuruda, H.; Tse, J.S.; Mito, M.; Desgreniers, S.; Dube, P.A.; Zhang, S.; et al. From magnets to metals: The response of tetragonal bisdiselenazolyl radicals to pressure. J. Am. Chem. Soc. 2011, 133, 6051–6060. [Google Scholar]
- Winter, S.M.; Datta, S.; Hill, S.; Oakley, R.T. Magnetic anisotropy in a heavy atom radical ferromagnet. J. Am. Chem. Soc. 2011, 133, 8126–8129. [Google Scholar]
- Yu, X.; Mailman, A.; Dube, P.A.; Assoud, A.; Oakley, R.T. The first semiquinone-bridged bisdithiazolyl radical conductor: A canted antiferromagnet displaying a spin-flop transition. Chem. Commun. 2011, 47, 4655–4657. [Google Scholar]
- Yu, X.; Mailman, A.; Lekin, K.; Assoud, A.; Robertson, C.M.; Noll, B.C.; Campana, C.F.; Howard, J.A.K.; Dube, P.A.; Oakley, R.T. Semiquinone-bridged bisdithiazoyl radicals as neutral radical conductors. J. Am. Chem. Soc. 2012, 134, 2264–2275. [Google Scholar]
- Chi, X.; Itkis, M.E.; Patrick, B.O.; Barclay, T.M.; Reed, R.W.; Oakley, R.T.; Cordes, A.W.; Haddon, R.C. The first phenalenyl-based neutral radical molecular conductor. J. Am. Chem. Soc. 1999, 121, 10395–10402. [Google Scholar]
- Mandal, S.K.; Itkis, M.E.; Chi, X.; Samanta, S.; Lidsky, D.; Reed, R.W.; Oakley, R.T.; Tham, F.S.; Haddon, R.C. New family of aminophenalenyl-based neutral radical molecular conductors: Synthesis, structure, and solid state properties. J. Am. Chem. Soc. 2005, 127, 8185–8196. [Google Scholar]
- Chi, X.; Itkis, M.E.; Kirschbaum, K.; Pinkerton, A.A.; Oakley, R.T.; Cordes, A.W.; Haddon, R.C. Dimeric phenalenyl-based neutral radical molecular conductors. J. Am. Chem. Soc. 2001, 123, 4041–4048. [Google Scholar]
- Itkis, M.E.; Chi, X.; Cordes, A.W.; Haddon, R.C. Magneto-opto-electronic bistability in a phenalenyl-based neutral radical. Science 2002, 296, 1443–1445. [Google Scholar]
- Miller, J.S. Bistable electrical, optical, and magnetic behavior in molecule-based material. Angew. Chem. Int. Ed. 2003, 42, 27–29. [Google Scholar] [CrossRef]
- Huang, J.; Kertesz, M. Spin crossover of spiro-biphenalenyl neutral radical molecular conductors. J. Am. Chem. Soc. 2003, 125, 13334–13335. [Google Scholar]
- Huang, J.; Kertesz, M. Theoritical analysis of intermolecular covalent π-π bonding and magnetic properties of phenalenyl and spiro-biphenalenyl radical pi-dimers. J. Phys. Chem. A 2007, 111, 6304–6315. [Google Scholar]
- Chi, X.; Itkis, M.E.; Reed, R.W.; Oakley, R.T.; Cordes, A.W.; Haddon, R.C. Conducting pathways in organic solids: A phenalenyl-based neutral radical of low conductivity. J. Phys. Chem. B 2002, 106, 8278–8287. [Google Scholar]
- Chi, X.; Itkis, M.E.; Tham, F.S.; Oakley, R.T.; Cordes, A.W.; Haddon, R.C. Synthesis, structure and physical properties of a new phenalenyl-based neutral radical crystal: Correlation between structure and transport properties in carbon-based molecular conductors. Int. J. Quant. Chem. 2003, 95, 853–865. [Google Scholar]
- Liao, P.; Itkis, M.E.; Oakley, R.T.; Tham, F.S.; Haddon, R.C. Light-mediated c-c sigma-bond driven crystallization of a phenalenyl radical dimer. J. Am. Chem. Soc. 2004, 126, 14297–14302. [Google Scholar]
- Pal, S.K.; Itkis, M.E.; Tham, F.S.; Reed, R.W.; Oakley, R.T.; Haddon, R. Trisphenalenyl-based neutral radical molecular conductor. J. Am. Chem. Soc. 2008, 130, 3942–3951. [Google Scholar]
- Pal, S.K.; Itkis, M.E.; Tham, F.S.; Reed, R.W.; Oakley, R.T.; Donnadieu, B.; Haddon, R.C. Phenalenyl-based neutral radical molecular conductors: Substituent effects on solid-state structures and properties. J. Am. Chem. Soc. 2007, 129, 7163–7174. [Google Scholar]
- Pal, S.K.; Itkis, M.E.; Reed, R.W.; Oakley, R.T.; Cordes, A.W.; Tham, F.S.; Siegrist, T.; Haddon, R.C. Synthesis, structure and physical properties of the first one-dimensional phenalenyl-based neutral radical conductor. J. Am. Chem. Soc. 2004, 126, 1478–1484. [Google Scholar]
- Pauling, L. The metallic state. Nature 1948, 161, 1019–1020. [Google Scholar]
- Anderson, P.W. Resonating valence bonds: A new kind of insulator? Mater. Res. Bull. 1973, 8, 153–160. [Google Scholar] [CrossRef]
- Mandal, S.K.; Samanta, S.; Itkis, M.E.; Jensen, D.W.; Reed, R.W.; Oakley, R.T.; Tham, F.S.; Donnadieu, B.; Haddon, R.C. Resonating valence bond ground state in oxygen-functionalized phenalenyl-based neutral radical conductors. J. Am. Chem. Soc. 2006, 128, 1982–1994. [Google Scholar]
- Sarkar, A.; Pal, S.K.; Itkis, M.E.; Liao, P.; Tham, F.S.; Donnadieu, B.; Haddon, R.C. Methoxy-substituted phenalenyl-based neutral radical molecular conductor. Chem. Mater. 2009, 21, 2226–2237. [Google Scholar]
- Sarkar, A.; Itkis, M.E.; Tham, F.S.; Haddon, R.C. Synthesis, structure, and physical properties of a partial pi-stacked phenalenyl-based neutral radical molecular conductor. Chem. Eur. J. 2011, 17, 11576–11584. [Google Scholar] [CrossRef]
- Robbins, J.L.; Edelstein, N.; Spencer, B.; Smart, J.C. Syntheses and electronic structure of decamethylmetallocenes. J. Am. Chem. Soc. 1982, 104, 1882–1893. [Google Scholar]
- Bonner, J.C.; Fisher, M.E. Linear magnetic chains with anisotropic coupling. Phys. Rev. 1964, 135, A640–A658. [Google Scholar]
- Estes, W.E.; Gavel, D.P.; Hatfield, W.E.; Hodgson, D.J. Magnetic and structural characterization of dibromo- and dichlorobis(thiazole)copper(II). Inorg. Chem. 1978, 17, 1415–1421. [Google Scholar]
- Mott, N.F. Metal-insulator transitions, 2nd ed; CRC Press: Boca Raton, FL, USA, 1990. [Google Scholar]
- Yoneyama, N.; Miyazaki, A.; Enoki, T.; Saito, G. Magnetic properties of ttf-type charge transfer salts in mott insulator regime. Bull. Chem. Soc. Jpn. 1999, 72, 639–651. [Google Scholar]
- Whangbo, M.-H.; Hoffmann, R.; Woodward, R.B. Conjugated one and two dimensional polymers. Proc. Royal Soc. Lond. A 1979, 366, 23–46. [Google Scholar]
- Haddon, R.C.; Chichester, S.V.; Mayo, S.L. Direct amination of 9-hydroxyphenalenone to produce 9-aminophenalenone and related compounds. Synthesis 1985, 639–641. [Google Scholar]
- Apex 2, 2009.5-1 version, Bruker AXS Inc.: Madison, WI, USA, 2009.
- Saint Software Reference Manual, V7.23A; Bruker Analytical X-Ray System Inc: Madison, WI, USA, 2003.
- Shelxtl, 2008/4 version, Bruker AXS Inc.: Madison, WI, USA, 2008.
- Sadabs, 2008/1 version, Bruker AXS Inc.: Madison, WI, USA, 2008.
- Hoffmann, R. An extended Hückel theory.I. Hydrocarbons. J. Chem. Phys. 1963, 39, 1397–1412. [Google Scholar] [CrossRef]
- Hofmann, R. Solids and surfaces; Wiley-VCH: Minnesota, MN, USA, 1988. [Google Scholar]
- Whangbo, M.H. Mott-hubbard condition for electronic localization in the hartee-fock band theory. J. Chem. Phys. 1979, 70, 4963–4966. [Google Scholar]
- Haddon, R.C.; Siegrist, T.; Fleming, R.M.; Bridenbaugh, P.M.; Laudise, R.A. Band structures of organic thin-film transistor materials. J. Mater. Chem. 1995, 5, 1719–1724. [Google Scholar]
- Haddon, R.C.; Chi, X.; Itkis, M.E.; Anthony, J.E.; Eaton, D.L.; Siegrist, T.; Mattheus, C.C.; Palstra, T.T.M. Band electronic structure of one- and two-dimensional pentacene molecular crystals. J. Phys. Chem. B 2002, 106, 8288–8292. [Google Scholar]
- Andrews, M.P.; Cordes, A.W.; Douglass, D.C.; Fleming, R.M.; Glarum, S.H.; Haddon, R.C.; Marsh, P.; Oakley, R.T.; Palstra, T.T.M.; Schneemeyer, L.F.; et al. One-dimensional stacking of bifunctional dithia- and diselena-diazolyl radicals: Preparation and structural and electronic properties of 1,3-[(E2N2C)C6H4(CN2E2)](E = S, Se). J. Am. Chem. Soc. 1991, 113, 3559–3568. [Google Scholar]
- Cordes, A.W.; Haddon, R.C.; Oakley, R.T.; Schneemeyer, L.F.; Waszczak, J.V.; Young, K.M.; Zimmerman, N.M. Molecular semiconductors from bifunctional dithia- and diselenadiazolyl radicals. Preparation and solid-state structural and electronic properties of 1,4-[(E2N2C)C6H4(CN2E2)](E = S, Se). J. Am. Chem. Soc. 1991, 113, 582–588. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bag, P.; Itkis, M.E.; Pal, S.K.; Bekyarova, E.; Donnadieu, B.; Haddon, R.C. Synthesis, Structure and Solid State Properties of Cyclohexanemethylamine Substituted Phenalenyl Based Molecular Conductor. Crystals 2012, 2, 446-465. https://doi.org/10.3390/cryst2020446
Bag P, Itkis ME, Pal SK, Bekyarova E, Donnadieu B, Haddon RC. Synthesis, Structure and Solid State Properties of Cyclohexanemethylamine Substituted Phenalenyl Based Molecular Conductor. Crystals. 2012; 2(2):446-465. https://doi.org/10.3390/cryst2020446
Chicago/Turabian StyleBag, Pradip, Mikhail E. Itkis, Sushanta K. Pal, Elena Bekyarova, Bruno Donnadieu, and Robert C. Haddon. 2012. "Synthesis, Structure and Solid State Properties of Cyclohexanemethylamine Substituted Phenalenyl Based Molecular Conductor" Crystals 2, no. 2: 446-465. https://doi.org/10.3390/cryst2020446




