Polyoxotungstate-Surfactant Layered Crystal toward Conductive Inorganic-Organic Hybrid
Abstract
:1. Introduction
2. Results and Discussion
2.1. Crystal Structure of C16py-W10
| Chemical formula | C84H152N4W10O32 |
| Formula weight | 3568.54 |
| Crystal system | triclinic |
| Space group | P  (No.2) (No.2) |
| a (Å) | 10.7727 (12) |
| b (Å) | 11.3734 (12) |
| c (Å) | 23.982 (3) |
| α (°) | 98.566 (4) |
| β (°) | 95.298 (4) |
| γ (°) | 116.168 (4) |
| V (Å3) | 2566.1 (5) |
| Z | 1 |
| ρcalcd (g cm−3) | 2.309 |
| T (K) | 213 (2) |
| μ (Mo Kα) (mm−1) | 11.230 |
| No. of reflections measured | 41418 |
| No. of independent reflections | 11736 |
| Rint | 0.0635 |
| No. of parameters | 587 |
| R1 (I > 2σ(I)) | 0.0354 |
| wR2 (all data) | 0.0842 |


| H···O (Ǻ) | C···O (Ǻ) | C-H···O (deg) | |
|---|---|---|---|
| C1 i–H1 i···O15 | 2.967(5) | 3.880(9) | 164.3(5) |
| C28 i–H28B i···O8 | 2.410(4) | 3.318(7) | 154.1(4) |
| C28–H28A···O5 | 2.795(3) | 3.730(6) | 159.8(4) |
| C3–H3···O2 | 2.634(4) | 3.550(8) | 164.7(5) |
| C21 iii–H21A iii···O2 | 2.476(4) | 3.380(11) | 155.2(5) |
| C26–H26···O4 | 2.534(4) | 3.448(7) | 164.5(5) |
| C6 ii–H6A ii···O4 | 2.384(5) | 3.278(9) | 151.3(4) |
2.2. Powder X-Ray Diffraction (XRD) Patterns of C16py-W10
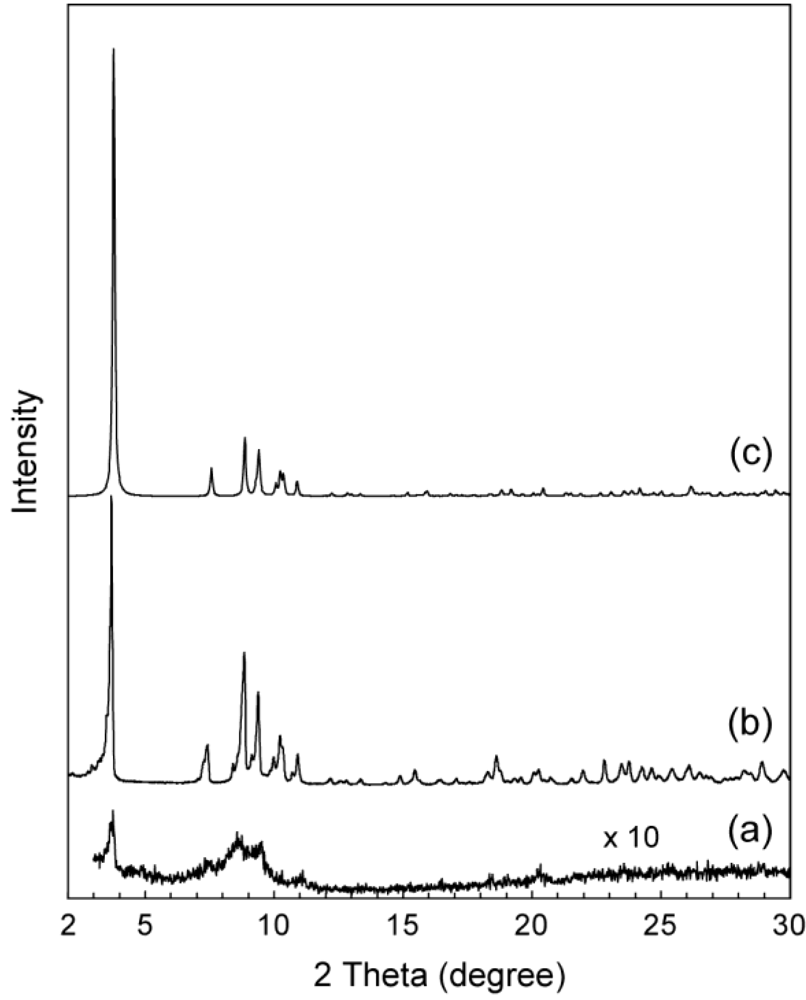
2.3. Conductivity of C16py-W10
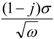 ).
).
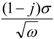 ).
).
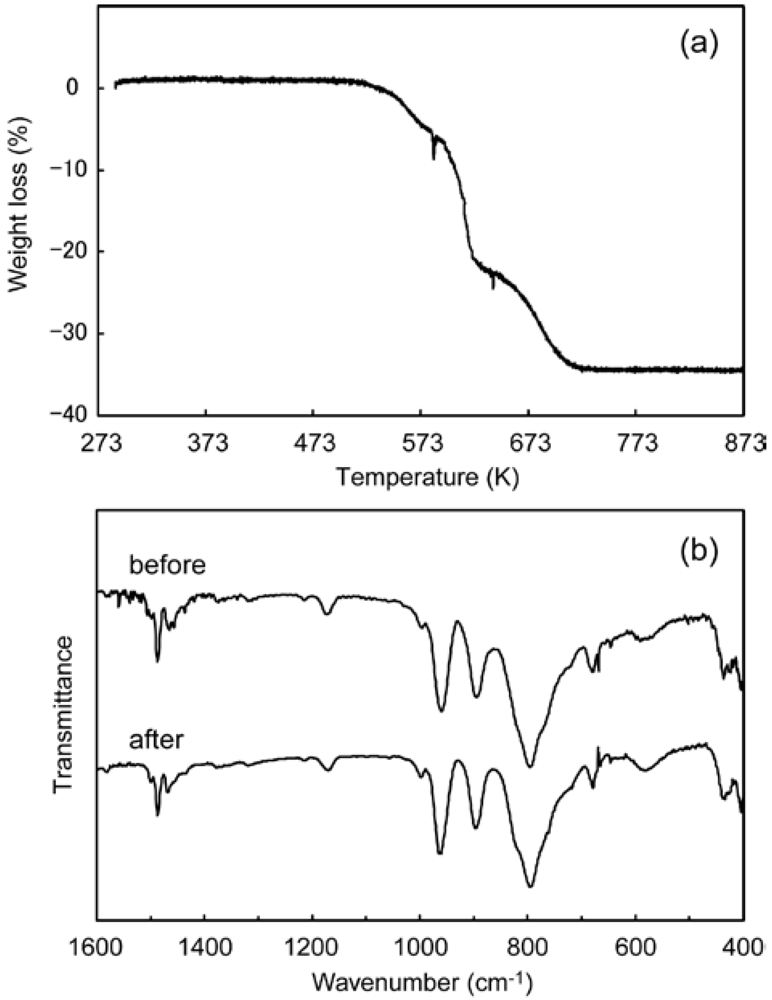
3. Experimental Section
3.1. Syntheses and Methods
3.2. X-ray Diffraction Measurements
4. Conclusions
Acknowledgments
References and Notes
- Batail, P. Molecular Conductors. In Chemical Reviews; Josef, Michl, Ed.; American Chemical Society: Washington, DC, USA, 2004; Volume 104, pp. 4887–5782. [Google Scholar]
- Coronado, E.; Gómez-García, C.J. Polyoxometalate-based molecular materials. Chem. Rev. 1998, 98, 273–296. [Google Scholar]
- Coronado, E.; Giménez-Saiz, C.; Gómez-García, C.J. Recent advances in polyoxometalate-containing molecular conductors. Coord. Chem. Rev. 2005, 249, 1776–1796. [Google Scholar]
- Kato, R. Conductive copper salts of 2,5-disubstituted N,N'-dicyanobenzoquinonediimines (DCNQIs): structural and physical properties. Bull. Chem. Soc. Jpn. 2000, 73, 515–534. [Google Scholar] [CrossRef]
- Casciola, M. Ionic Conductivity in Layered Materials. In Comprehensive Supramolecular Chemistry; Atwood, J.L., Davies, J.E.D., MacNicol, D.D., Vögtle, F., Eds.; Elsevier Science: Oxford, UK, 1996; Volume 7, pp. 355–378. [Google Scholar]
- Wu, C.-G.; DeGroot, D.C.; Marcy, H.O.; Schindler, J.L.; Kannewurf, C.R.; Liu, Y.-J.; Hirpo, W.; Kanatzidis, M.G. Redox intercalative polymerization of aniline in V2O5 xerogel. the postintercalative intralamellar polymer growth in polyaniline/metal oxide nanocomposites is facilitated by molecular oxygen. Chem. Mater. 1996, 8, 1992–2004. [Google Scholar]
- Ruiz-Hitzky, E.; Aranda, P.; Darder, M.; Ogawa, M. Hybrid and biohybrid silicate based materials: Molecular vs. block-assembling bottom–up processes. Chem. Soc. Rev. 2011, 40, 801–828. [Google Scholar] [CrossRef]
- Cao, M.; Djerdj, I.; Jagličić, Z.; Antonietti, M.; Niederberger, M. Layered hybrid organic–inorganic nanobelts exhibiting a field-inducedmagnetic transition. Phys. Chem. Chem. Phys. 2009, 11, 6166–6172. [Google Scholar]
- Song, Y.-F.; McMillan, N.; Long, D.-L.; Thiel, J.; Ding, Y.; Chen, H.; Gadegaard, N.; Cronin, L. Design of hydrophobic polyoxometalate hybrid assemblies beyond surfactant encapsulation. Chem. Eur. J. 2008, 14, 2349–2354. [Google Scholar]
- Yan, Y.; Wang, H.; Li, B.; Hou, G.; Yin, Z.; Wu, L.; Yam, V.W.W. Smart self-assemblies based on a surfactant-encapsulated photoresponsive polyoxometalate complex. Angew. Chem. Int. Ed. 2010, 49, 9233–9236. [Google Scholar]
- Inumaru, K.; Ishihara, T.; Kamiya, Y.; Okuhara, T.; Yamanaka, S. Water-tolerant, highly active solid acid catalysts composed of the Keggin-type polyoxometalate H3PW12O40 immobilized in hydrophobic nanospaces of organomodified mesoporous silica. Angew. Chem. Int. Ed. 2007, 46, 7625–7628. [Google Scholar]
- Pope, M.T. Heteropoly and Isopoly Oxometalates; Springer: Berlin, Germany, 1983. [Google Scholar]
- Long, D.-L.; Burkholder, E.; Cronin, L. Polyoxometalate clusters, nanostructures and materials: From self assembly to designer materials and devices. Chem. Soc. Rev. 2007, 36, 105–121. [Google Scholar]
- Proust, A.; Thouvenot, R.; Gouzerh, P. Functionalization of polyoxometalates: Towards advanced applications in catalysis and materials science. Chem. Commun. 2008, 1837–1852. [Google Scholar]
- Okuhara, T.; Mizuno, N.; Misono, M. Catalytic chemistry of heteropoly compounds. Adv. Catal. 1996, 41, 113–252. [Google Scholar]
- Sadakane, M.; Steckhan, E. Electrochemical properties of polyoxometalates as electrocatalysts. Chem. Rev. 1998, 98, 219–237. [Google Scholar]
- Huo, Q.; Margolese, D.I.; Ciesla, U.; Demuth, D.G.; Feng, P.; Gier, T.E.; Sieger, P.; Firouzi, A.; Chmelka, B.F.; Schüth, F.; et al. Organization of organic molecules with inorganic molecular species into nanocomposite biphase arrays. Chem. Mater. 1994, 6, 1176–1191. [Google Scholar] [CrossRef]
- Kanatzidis, M.G. Beyond silica: Nonoxidic mesostructured materials. Adv. Mater. 2007, 19, 1165–1181. [Google Scholar]
- Yamauchi, Y.; Kuroda, K. Rational design of mesoporous metals and related nanomaterials by a soft-template approach. Chem. Asian J. 2008, 3, 664–676. [Google Scholar]
- Stein, A.; Fendorf, M.; Jarvie, T.P.; Mueller, K.T.; Benesi, A.J.; Mallouk, T.E. Salt-gel synthesis of porous transition-metal oxides. Chem. Mater. 1995, 7, 304–313. [Google Scholar]
- Janauer, G.G.; Dobley, A.; Guo, J.; Zavalij, P.; Whittingham, M.S. Novel tungsten, molybdenum, and vanadium oxides containing surfactant ions. Chem. Mater. 1996, 8, 2096–2101. [Google Scholar] [CrossRef]
- Taguchi, A.; Abe, T.; Iwamoto, M. Non-silica-based mesostructured materials: Hexagonally mesostructured array of surfactant micelles and 11-tungstophosphoric heteropoly anions. Adv. Mater. 1998, 10, 667–669. [Google Scholar]
- Do, J.; Jacobson, A.J. Mesostructured lamellar phases containing six-membered vanadium borophosphate cluster anions. Chem. Mater. 2001, 13, 2436–2440. [Google Scholar]
- Polarz, S.; Smarsly, B.; Antonietti, M. Colloidal organization and clusters: self-assembly of polyoxometalate-surfactant complexes towards three-dimensional organized structures. ChemPhysChem 2001, 457–461. [Google Scholar]
- Zhang, G.; Ke, H.; He, T.; Xiao, D.; Chen, Z.; Yang, W.; Yao, J. Synthesis and characterization of new layered polyoxometallates-1,10-decanediamine intercalative nanocomposites. J. Mater. Res. 2004, 19, 496–500. [Google Scholar]
- Janauer, G.G.; Dobley, A.D.; Zavalij, P.Y.; Whittingham, M.S. Evidence for decavanadate clusters in the lamellar surfactant ion phase. Chem. Mater. 1997, 9, 647–649. [Google Scholar]
- Spahr, M.E.; Nesper, R. Anhydrous octamolybdate with trimethyl hexadecyl ammoniumu cations. Z. Anorg. Allg. Chem. 2001, 627, 2133–2138. [Google Scholar]
- Nyman, M.; Ingersoll, D.; Singh, S.; Bonhomme, F.; Alam, T.M.; Brinker, C.J.; Rodriguez, M.A. Comparative study of inorganic cluster-surfactant arrays. Chem. Mater. 2005, 17, 2885–2895. [Google Scholar]
- Nyman, M.; Rodriguez, M.A.; Anderson, T.M.; Ingersoll, D. Two structures toward understanding evolution from surfactant-polyoxometalate lamellae to surfactant-encapsulated polyoxometalates. Cryst. Growth Des. 2009, 9, 3590–3597. [Google Scholar]
- Ito, T.; Sawada, K.; Yamase, T. Crystal structure of bis(dimethyldioctadecylammonium) hexamolybdate:A molecular model of Langmuir–Blodgett films. Chem. Lett. 2003, 32, 938–939. [Google Scholar]
- Ito, T.; Yamase, T. Inorganic-organic hybrid layered crystal composed of polyoxomolybdate and surfactant with π electrons. Chem. Lett. 2009, 38, 370–371. [Google Scholar] [CrossRef]
- Ito, T.; Mikurube, K.; Abe, Y.; Koroki, T.; Saito, M.; Iijima, J.; Naruke, H.; Ozeki, T. Hybrid inorganic-organic crystals composed of octamolybdate isomers and pyridinium surfactant. Chem. Lett. 2010, 39, 1323–1325. [Google Scholar]
- Yamase, T. Photo- and electrochromism of polyoxometalates and related materials. Chem. Rev. 1998, 98, 307–325. [Google Scholar]
- Renneke, R.F.; Pasquali, M.; Hill, C.L. Polyoxometalate systems for the catalytic selective production of nonthermodynamic alkenes from alkanes. Nature of excited-state deactivation processes and control of subsequent thermal processes in polyoxometalate photoredox chemistry. J. Am. Chem. Soc. 1990, 112, 6585–6594. [Google Scholar] [CrossRef]
- Moriguchi, I.; Orishikida, K.; Tokuyama, Y.; Watabe, H.; Kagawa, S.; Teraoka, Y. Photocatalytic property of a decatungstate-containing bilayer system for the conversion of 2-propanol to acetone. Chem. Mater. 2001, 13, 2430–2435. [Google Scholar]
- Hölscher, M.; Englert, U.; Zibrowius, B.; Hölderich, W.F. H3N(CH2)6NH3)4[W18P2O62]·3H2O, a microporous solid from Dawson anions and 1,6-diaminohexane. Angew. Chem. Int. Ed. 1994, 33, 2491–2493. [Google Scholar]
- Gabriel, J.-C.P.; Nagarajan, R.; Natarajan, S.; Cheetham, A.K.; Rao, C.N.R. Hydrothermal synthesis and structure of a mixed valent heteropoly-oxometallate Keggin salt: [PMo4.27W7.73O406−][H3N(CH2)6NH32+]3. J. Solid State Chem. 1997, 129, 257–262. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, J.; Wang, E.; Qin, C.; Li, Y.; Qi, Y.; Wang, X. Two extended structures constructed from sandwich-type polyoxometalates functionalized by organic amines. Dalton Trans. 2008, 463–468. [Google Scholar]
- Paradies, H.H.; Habben, F. Structure of n-hexadeeylpyridinium chloride monohydrate. Acta Crystallogr., Sect. C 1993, 49, 744–747. [Google Scholar] [CrossRef]
- Desiraju, G.R.; Steiner, T. The Weak Hydrogen Bond in Structural Chemistry and Biology; Oxford University Press: New York, NY, USA, 1999. [Google Scholar]
- Suezawa, H.; Yoshida, T.; Umezawa, Y.; Tsuboyama, S.; Nishio, M. CH/π interactions implicated in the crystal structure of transition metal compounds—a database study. Eur. J. Inorg. Chem. 2002, 3148–3155. [Google Scholar]
- Fuchs, J.; Hartl, H.; Schiller, W.; Gerlach, U. Die kristallstruktur des tributylammoniumdekawolframats [(C4H9)3NH]4W10O32. Acta Crystallogr. Sect. B 1976, 32, 740–749. [Google Scholar]
- Clegg, W.; Harrington, R.W. Private communication. University of Newcastle: Newcastle Upon Tyne, UK, 2005. [Google Scholar]
- Long, D.-L.; Kögerler, P.; Parenty, A.D.C.; Fielden, J.; Cronin, L. Discovery of a family of isopolyoxotungstates [H4W19O62]6− encapsulating a {WO6} moiety within a {W18} Dawson-like cluster cage. Angew. Chem. Int. Ed. 2006, 45, 4798–4803. [Google Scholar]
- Slight expansion in the volume of the lattice (~3%) was observed at room temperature, which is common for the compounds with long aliphatic chain. See also ref. 32 and references therein.
- Barsoukov, E.; Macdonald, J.R. Impedance Spectroscopy: Theory, Experiment, and Applications, 2nd ed; Wiley-Interscience: Hoboken, NJ, USA, 2005. [Google Scholar]
- Eder, D.; Kramer, R. Electric impedance spectroscopy of titania: Influence of gas treatment and of surface area. J. Phys. Chem. B 2004, 108, 14823–14829. [Google Scholar]
- Naruke, H.; Kajitani, N.; Konya, T. Insertion-release of guest species and ionic conduction in polyoxometalate solids with a layer-like Anderson structure. J. Solid. State Chem. 2011, 184, 770–777. [Google Scholar]
- Fournier, M. Tetrabutylammonium decatungstate(VI). Inorg. Synth. 1990, 27, 81–83. [Google Scholar]
- PROCESS-AUTO; Rigaku Corporation: Tokyo, Japan, 2002.
- Beurskens, P.T.; Admiraal, G.; Beurskens, G.; Bosman, W.P.; Garcia-Granda, S.; Gould, R.O.; Smits, J.M.M.; Smykalla, C. PATTY; University of Nijmegen: Nijmegen, The Netherlands, 1992. [Google Scholar]
- Beurskens, P.T.; Admiraal, G.; Beurskens, G.; Bosman, W.P.; de Gelder, R.; Israel, R.; Smits, J.M.M. DIRDIF99, University of Nijmegen: Nijmegen, The Netherlands, 1999.
- Sheldrick, G.M. SHELX-97; University of Göttingen: Göttingen, Germany, 1997. [Google Scholar]
- CrystalStructure 3.8, Rigaku/MSC: Woodlands, TX, USA, 2006.
- Pawley, G.S. Unit-cell refinement from powder diffraction scans. J. Appl. Crystallogr. 1981, 14, 357. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ito, T.; Fujimoto, N.; Uchida, S.; Iijima, J.; Naruke, H.; Mizuno, N. Polyoxotungstate-Surfactant Layered Crystal toward Conductive Inorganic-Organic Hybrid. Crystals 2012, 2, 362-373. https://doi.org/10.3390/cryst2020362
Ito T, Fujimoto N, Uchida S, Iijima J, Naruke H, Mizuno N. Polyoxotungstate-Surfactant Layered Crystal toward Conductive Inorganic-Organic Hybrid. Crystals. 2012; 2(2):362-373. https://doi.org/10.3390/cryst2020362
Chicago/Turabian StyleIto, Takeru, Nozomu Fujimoto, Sayaka Uchida, Jun Iijima, Haruo Naruke, and Noritaka Mizuno. 2012. "Polyoxotungstate-Surfactant Layered Crystal toward Conductive Inorganic-Organic Hybrid" Crystals 2, no. 2: 362-373. https://doi.org/10.3390/cryst2020362





