Electroactive Bisiminopyridine Ligands: Synthesis and Complexation Studies
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis and Characterization


| bond | length (Å) | bond | length (Å) | bond | length (Å) |
|---|---|---|---|---|---|
| C11–C12 | 1.330(6) | C14–S4 | 1.761(5) | N1–Zn1 | 2.052(5) |
| C11–S1 | 1.774(4) | C15–C16 | 1.345(6) | N2–Zn1 | 2.351(3) |
| C12–S2 | 1.720(4) | C15–S6 | 1.743(4) | Cl1–Zn1 | 2.2345(13) |
| C13–C14 | 1.340(5) | C15–S3 | 1.769(4) | Zn1–Cl1 i | 2.2345(13) |
| C13–S2 | 1.756(4) | C16–S5 | 1.742(4) | Zn1–N2 i | 2.351(3) |
| C13–S1 | 1.759(4) | C18–S5 | 1.794(5) | H10–Cl1 i | 3.1536(15) |
| C14–S3 | 1.758(4) | N1–C1i | 1.342(5) | Cl1–Cl1 i | 3.8584(19) |
| angle | value (°) | angle | value (°) | angle | value (°) |
| N1–Zn1–Cl1 | 120.30(4) | N1–Zn1–N2 i | 74.31(9) | Cl1–Zn1–N2 | 97.74(9) |
| N1–Zn1–Cl1 i | 120.30(4) | Cl1–Zn1–N2 i | 97.94(9) | Cl1 i–Zn1–N2 | 97.94(9) |
| Cl1–Zn1–Cl1 i | 119.40(9) | N1–Zn1–N2 | 74.31(9) | N2 i–Zn1–N2 | 148.63(18) |
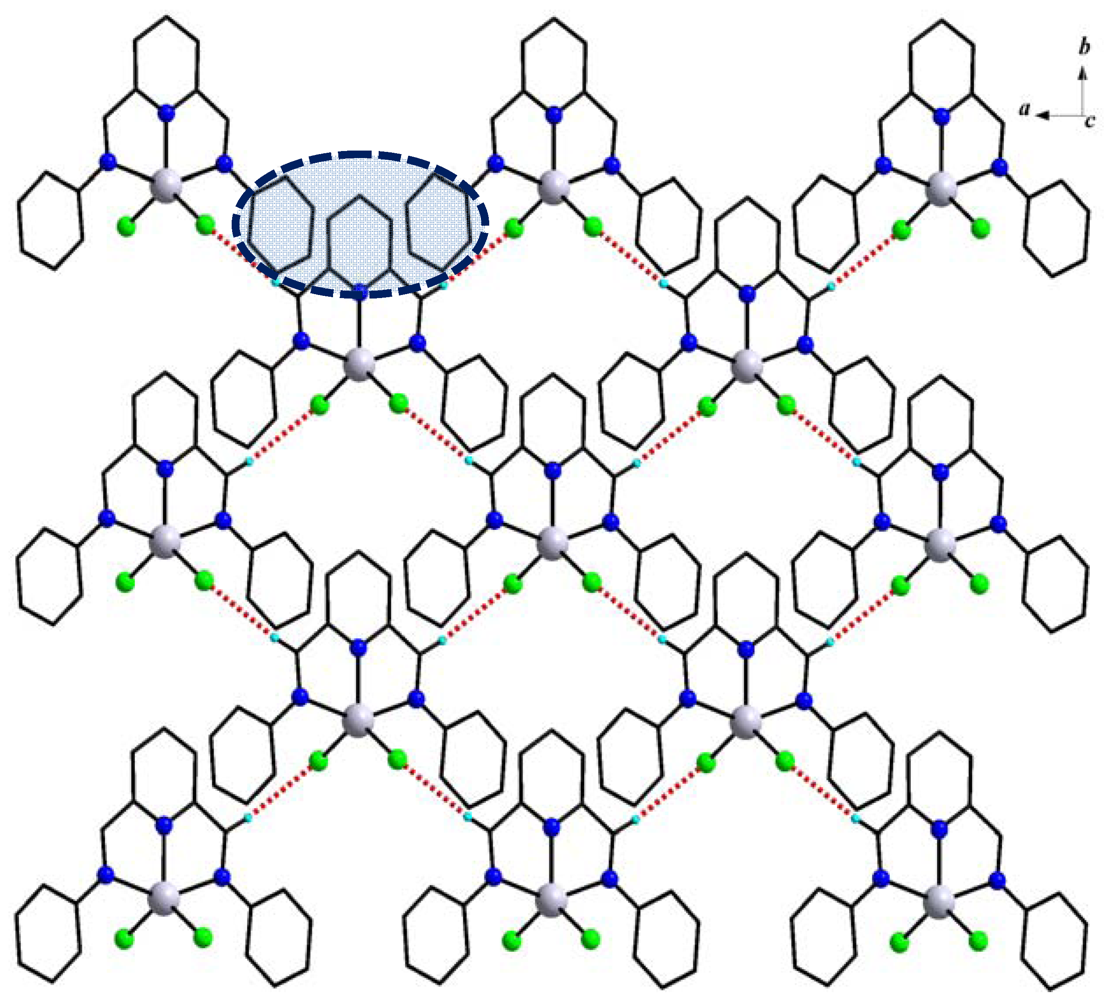
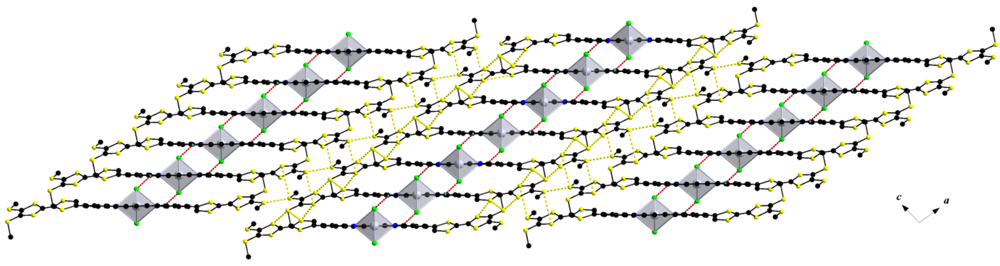
2.2. UV-Vis Spectroscopy
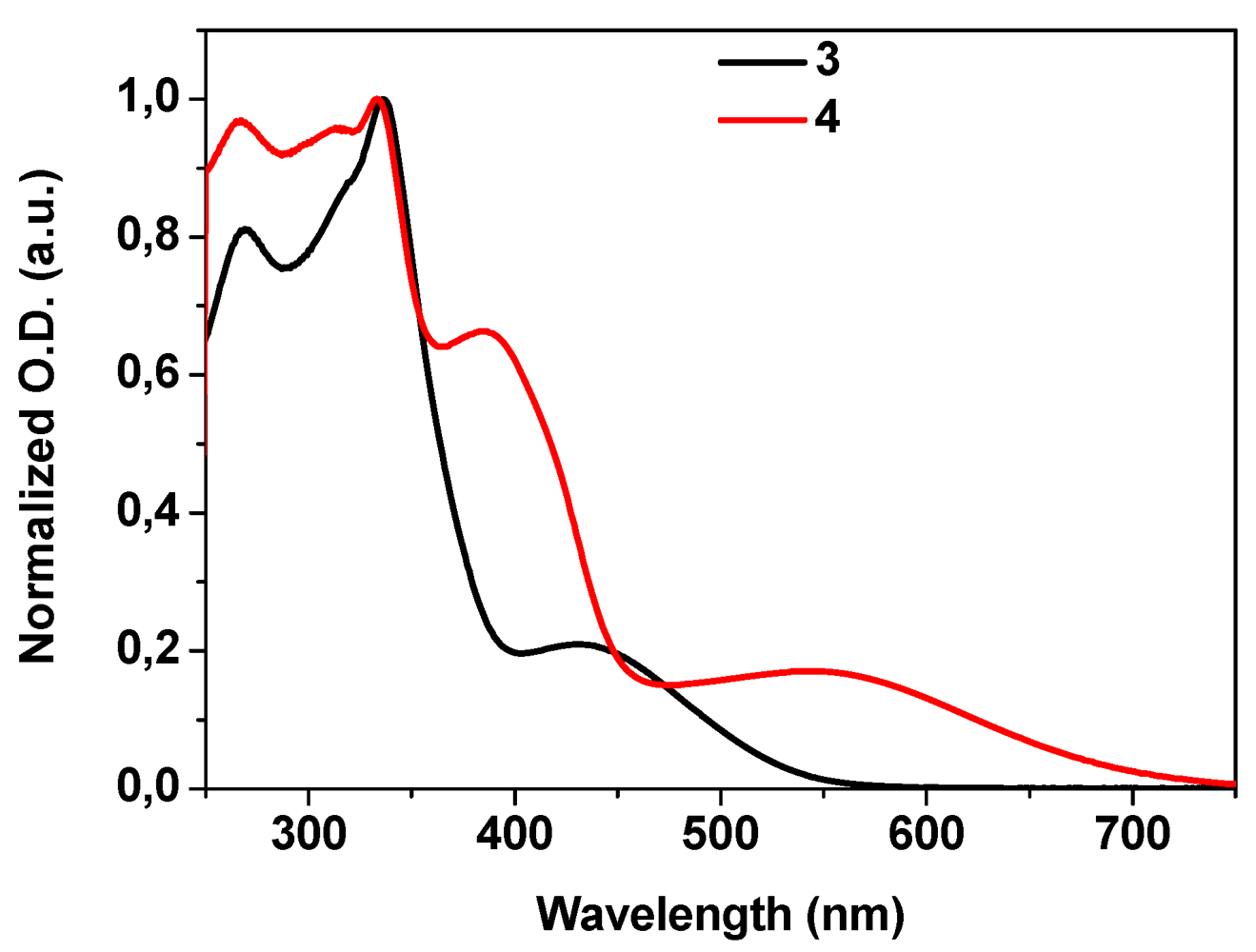
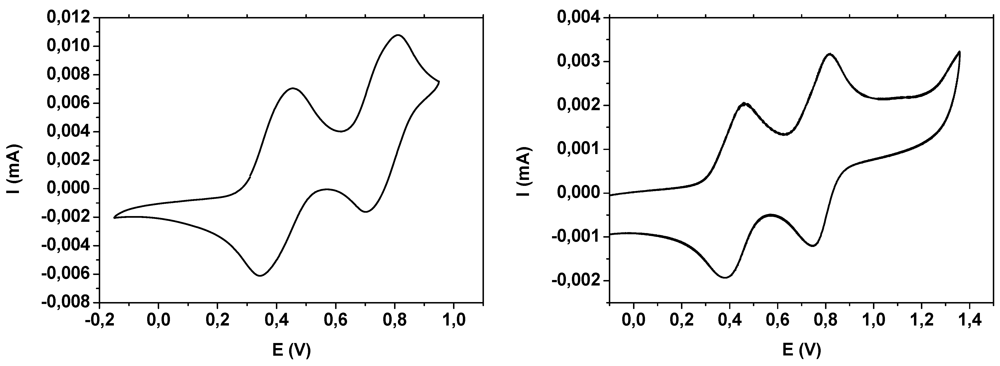
2.3. Electrochemistry
3. Experimental Section
3.1. X-Ray Structure Determinations
| complex | 4 |
|---|---|
| empirical formula | C35H27Cl2N3S12Zn |
| fw | 1010.59 |
| T (K) | 293(2) |
| wavelength (Å) | 0.71073 |
| crystal system | monoclinic |
| space group | C2/c |
| unit cell dimensions: | |
| a (Å) | 11.8152(16) |
| b (Å) | 10.100(2) |
| c (Å) | 35.579(6) |
| α(deg) | 90 |
| β(deg) | 98.111(13) |
| γ(deg) | 90 |
| V (Å3) | 4202.2(12) |
| Z | 4 |
| Dc (g cm−3) | 1.597 |
| abs coeff (mm−1) | 1.340 |
| θ range for data collection (deg) | 1.16–27.54 |
| reflections collected | 4760 |
| indep reflns | 3371 |
| completeness (%) | 98.5 |
| data/restraints/param | 4760/0/243 |
| GOF on F 2 | 1.197 |
| final R indices [I > 2σ(I)] | R1 = 0.0597, wR2 = 0.1051 |
| R indices (all data) | R1 = 0.0968, wR2 = 0.1227 |
| largest diff. peak and hole (e Å−3) | 0.394 and −0.416 |
3.2. General
3.3. Synthesis of (1-(6,7-Dimethyldithio-tétrathiafulvalene)-4-nitro-benzene) (1)
3.4. Synthesis of (4-(6,7-Dimethyldithio-tétrathiafulvalene)-aniline) (2)
3.5. (2,6-Bis(4-(4',5'-bis(Methylthio)-[2,2'-bi(1,3-dithiolylidene)]-4-yl)aniline)-iminopyridine) (3)
3.6. (2,6-Bis(4-(4',5'-bis(methylthio)-[2,2'-bi(1,3-dithiolylidene)]-4-yl)aniline)-iminopyridine) Zinc Chloride (4)
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Williams, J.M.; Ferraro, J.R.; Thorn, R.J.; Carlson, K.D.; Geiser, U.; Wang, H.H.; Kini, A.M.; Whangbo, M.-H. Organic Superconductors (Including Fullerenes), Synthesis, Structure, Properties and Theory; Prentice Hall: Upper Saddle River, NJ, USA, 1992. [Google Scholar]
- Bryce, M.R.; Murphy, L.C. Organic metals. Nature 1984, 309, 119–126. [Google Scholar]
- Segura, J.L.; Martin, N. New concepts in tetrathiafulvalene chemistry. Angew. Chem. Int. Ed. 2001, 40, 1372–1409. [Google Scholar] [CrossRef]
- Coronado, E.; Galan-Marcos, J.R.; Gomez-Garcia, C.J.; Laukhin, V. Coexistence of ferromagnetism and metallic conductivity in a molecule-based layered compound. Nature 2000, 408, 447–449. [Google Scholar]
- Coronado, E.; Day, P. Magnetic molecular conductors. Chem. Rev. 2004, 104, 5419–5448. [Google Scholar] [CrossRef]
- Lorcy, D.; Bellec, N.; Fourmigué, M.; Avarvari, N. Tetrathiafulvalene-based group XV ligands: Synthesis, coordination chemistry and radical cation salts. Coord. Chem. Rev. 2009, 253, 1398–1438. [Google Scholar] [Green Version]
- Avarvari, N.; Martin, D.; Fourmigué, M. Structural and electrochemical study of metal carbonyl complexes with chelating bis- and tetrakis(diphenylphosphino)tetrathiafulvalenes. J. Organomet. Chem. 2002, 643/644, 292–300. [Google Scholar]
- Pellon, P.; Gachot, G.; Le Bris, J.; Marchin, S.; Carlier, R.; Lorcy, D. Complexing ability of the versatile, redox-active, 3-[3-(Diphenylphosphino)propylthio]-3',4,4'-trimethyl-tetrathiafulvalene Ligand. Inorg. Chem. 2003, 42, 2056–2060. [Google Scholar] [CrossRef]
- Avarvari, N.; Fourmigué, M. First radical cation salt of a tetrathiafulvalene-based phosphine metal complex. Chem. Commun. 2004, 2, 1300–1301. [Google Scholar] [CrossRef]
- Réthoré, C.; Fourmigué, M.; Avarvari, N. Tetrathiafulvalene based phosphino-oxazolines: A new family of redox active chiral ligands. Chem. Commun. 2004, 34, 1384–1385. [Google Scholar]
- Devic, T.; Batail, P.; Fourmigué, M.; Avarvari, N. Unexpected reactivity of PdCl2 and PtCl2 complexes of the unsaturated diphosphine o-Me2TTF(PPh2)2 towards chloride abstraction with thallium triflate. Inorg. Chem. 2004, 43, 3136–3141. [Google Scholar]
- Gouverd, C.; Biaso, F.; Cataldo, L.; Berclaz, T.; Geoffroy, M.; Levillain, E.; Avarvari, N.; Fourmigué, M.; Sauvage, F.X.; Wartelle, C. Tetrathiafulvalene–phosphine-based iron and ruthenium carbonyl complexes: Electrochemical and EPR studies. Phys. Chem. Chem. Phys. 2005, 7, 85–93. [Google Scholar]
- Kobayashi, A.; Fujiwara, E.; Kobayashi, H. Single-component molecular metals with extended-TTF dithiolateligands. Chem. Rev. 2004, 104, 5243–5264. [Google Scholar] [CrossRef]
- Massue, J.; Bellec, N.; Chopin, S.; Levillain, E.; Roisnel, T.; Clérac, R.; Lorcy, D. Electroactiveligands: The first metal complexes of tetrathiafulvenyl−acetylacetonate. Inorg. Chem. 2005, 44, 8740–8748. [Google Scholar]
- Setifi, F.; Ouahab, L.; Golhen, S.; Yoshida, Y.; Saito, G. First radical cation salt of paramagnetic transition metal complex containing TTF as ligand, [CuII(hfac)2(TTF-py)2](PF6)·2CH2Cl2 (hfac = Hexafluoroacetylacetonate and TTF-py = 4-(2-Tetrathiafulvalenyl-ethenyl)pyridine). Inorg. Chem. 2003, 42, 1791–1793. [Google Scholar]
- Liu, S.-X.; Dolder, S.; Franz, P.; Neels, A.; Stoeckli-Evans, H.; Decurtins, S. Structural studies of transition metal complexes with 4,5-Bis(2-pyridylmethylsulfanyl)-4',5'-ethylenedithiotetrathiafulvalene: Probing their potential for the construction of multifunctional molecular assemblies. Inorg. Chem. 2003, 42, 4801–4803. [Google Scholar]
- Devic, T.; Avarvari, N.; Batail, P. A series of redox active, tetrathiafulvalene-based amidopyridines and bipyridinesligands: Syntheses, crystal structures, a radical cation salt and group 10 transition-metal complexes. Chem. Eur. J. 2004, 10, 3697–3707. [Google Scholar] [CrossRef]
- Devic, T.; Rondeau, D.; Şahin, Y.; Levillain, E.; Clérac, R.; Batail, P.; Avarvari, N. Copper(I/II) complexes of a bis(tetrathiafulvalene)-2,2'-bipyridine: Synthesis, characterization, magnetic and electrochemical properties. Dalton Trans. 2006, 10, 1331–1337. [Google Scholar]
- Hervé, K.; Liu, S.-X.; Cador, O.; Golhen, S.; le Gal, Y.; Bousseksou, A.; Stoeckli-Evans, H.; Decurtins, S.; Ouahab, L. Synthesis of a BEDT-TTF bipyridine organic donor and the first FeII coordination complex with a redox-active ligand. Eur. J. Inorg. Chem. 2006, 2006, 3498–3502. [Google Scholar]
- Chahma, M.; Hassan, N.; Alberola, A.; Stoeckli-Evans, H.; Pilkington, M. Preparation and coordination complex of the first imine-bridged tetrathiafulvalene–pyridine donor ligand. Inorg. Chem. 2007, 46, 3807–3809. [Google Scholar] [CrossRef]
- Qin, J.; Qian, C.-X.; Zhou, N.; Zhu, R.-M.; Li, Y.-Z.; Zuo, J.-L.; You, X.-Z. Metal complexes based on tetrathiafulvalene-fused π-extended schiff base ligands—Syntheses, characterization, and properties. Eur. J. Inorg. Chem. 2012, 234–245. [Google Scholar]
- Wu, J.-C.; Liu, S.-X.; Keene, T.D.; Neels, A.; Mereacre, V.; Powell, A.K.; Decurtins, S. Coordination chemistry of a π-extended, rigid and redox-active tetrathiafulvalene-fused schiff-base ligand. Inorg. Chem. 2008, 47, 3452–3459. [Google Scholar] [CrossRef]
- Britovsek, G.J.P.; Gibson, V.C.; Kimberley, B.S.; Maddox, P.J.; McTavish, S.J.; Solan, G.A.; White, A.J.P.; Williams, D.J. Novel olefin polymerization catalysts based on iron and cobalt. Chem. Commun. 1998, 849–850. [Google Scholar]
- Small, B.L.; Brookhart, M.; Bennett, A.M.A. Highly active iron and cobalt catalysts for the polymerization of ethylene. J. Am. Chem. Soc. 1998, 120, 4049–4050. [Google Scholar]
- Iyoda, M.; Hara, K.; Kuwatani, Y.; Nagase, S. Helical tetrathiafulvalene oligomers. Synthesis and properties of Bi-, Ter-, and quatertetrathiafulvalenes. Org. Lett. 2000, 2, 2217–2220. [Google Scholar]
- Lamère, J.F.; Malfant, I.; Sournia-Saquet, A.; Lacroix, P.G. Quadric non linear optical response in partially charged donor-substituted tetrathiafulvalene: From a computational investigation to a rational synthetic feasibility. Chem. Mater. 2007, 19, 805–815. [Google Scholar] [CrossRef]
- Addison, A.W.; Rao, T.N.; Reedijk, J.; van Rijn, J.; Verschoor, G.C. Synthesis, structure, and spectroscopic properties of copper(II) compounds containing nitrogen-sulphur donor ligands; The crystal and molecular structure of aqua[1,7-bis(N-methylbenzimidazol-2'-yl)-2,6-dithiaheptane]copper(II) perchlorate. J. Chem. Soc. Dalton Trans. 1984, 1349–1356. [Google Scholar]
- Andreu, R.; Malfant, I.; Lacroix, P.G.; Cassoux, P. New tetrathiafulvalene-π-spacer-acceptor derivatives: Synthesis, crystal structure, optical and electrochemical properties. Eur. J. Org. Chem. 2000, 2000, 737–741. [Google Scholar]
- Riobé, F.; Grosshans, P.; Sidorenkova, H.; Geoffroy, M.; Avarvari, N. Mono- and Bis(tetrathiafulvalene)-1,3,5-triazines as covalently linked donor–acceptor systems: Structural, spectroscopic, and theoretical investigations. Chem. Eur. J. 2009, 15, 380–387. [Google Scholar] [CrossRef] [Green Version]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nita, G.; Branzea, D.; Pop, F.; El-Ghayoury, A.; Avarvari, N. Electroactive Bisiminopyridine Ligands: Synthesis and Complexation Studies. Crystals 2012, 2, 338-348. https://doi.org/10.3390/cryst2020338
Nita G, Branzea D, Pop F, El-Ghayoury A, Avarvari N. Electroactive Bisiminopyridine Ligands: Synthesis and Complexation Studies. Crystals. 2012; 2(2):338-348. https://doi.org/10.3390/cryst2020338
Chicago/Turabian StyleNita, Gabriela, Diana Branzea, Flavia Pop, Abdelkrim El-Ghayoury, and Narcis Avarvari. 2012. "Electroactive Bisiminopyridine Ligands: Synthesis and Complexation Studies" Crystals 2, no. 2: 338-348. https://doi.org/10.3390/cryst2020338
APA StyleNita, G., Branzea, D., Pop, F., El-Ghayoury, A., & Avarvari, N. (2012). Electroactive Bisiminopyridine Ligands: Synthesis and Complexation Studies. Crystals, 2(2), 338-348. https://doi.org/10.3390/cryst2020338







