2.2.1. Structures of α'-(ET)4(tcpd)·THF (1) and α'-(ET)4(tcpd)·2H2O (2)
Compounds
1 and
2 are isostructural and crystallize in the monoclinic space group C2/c. Crystal and refinement data are shown in
Table 4. The only difference is the presence of a disordered THF molecule in the anionic layer in compound
1 and two water molecules in compound
2. Both compounds present cationic ET layers alternating along the
c axis with layers containing the tcpd
2− anions and the solvent molecules (
Figure 1).
The ET molecules show the so-called α' packing in which the organic donors form chains where all the ET molecular planes are parallel [
53]. The difference with other parallel packings is that now the long axes of the ET molecules are not parallel but form a twist angle (ω, in the range 32–34° in compounds
1 and
2) with the neighboring molecules in the chain (
Figure 1). Besides the twisting, there is a displacement of the center of adjacent ET molecules, noted as D in
Figure 1. Note that the so-called δ and δ' phases are very similar but in these phases the overlap between adjacent ET molecules in the chains follows the alternating sequence ...-RA-twisted-... (RA = ring-over-atom) and ...-RB-twisted-... (RB = ring-over-bond), respectively, whereas in the α' phase the overlap is always twisted [
53].
Table 4.
Crystal data for compounds α'-ET4(tcpd)(THF) (1) and α'-ET4(tcpd)(H2O)2 (2).
Table 4.
Crystal data for compounds α'-ET4(tcpd)(THF) (1) and α'-ET4(tcpd)(H2O)2 (2).
| Compound | 1 | 2 |
|---|
| Formula | C54H40N6OS32 | C50H36N6O2S32 |
| M | 1814.84 | 1778.76 |
| Cryst. Syst. | Monoclinic | Monoclinic |
| Space group | C2/c | C2/c |
| a (Å) | 15.7423(13) | 15.5152 (13) |
| b (Å) | 13.2947(11) | 13.2644(10) |
| c (Å) | 33.810(3) | 33.673(2) |
| β (°) | 96.701(6) | 97.443(6) |
| V (Å3) | 7027.7(10) | 6871.5(9) |
| Z | 4 | 4 |
| T (K) | 293(2) | 170(2) |
| ρcalc (g·cm−3) | 1.708 | 1.719 |
| μ (cm−1) | 9.406 | 10.36 |
| F(000) | 3672 | 3624 |
| Refl. Collec. | 40764 | 16,092 |
| Refl. Uniq. | 5029 | 3379 |
| Rint | 0.0588 | 0.0807 |
| Refl. Obs I > 2σ(I) | 2614 | 2051 |
| Nv | 568 | 274 |
| a R1 | 0.0790 | 0.1562 |
| b wR2 | 0.2542 | 0.4738 |
| c GooF | 0.963 | 1.730 |
| ∆ρmax (eÅ−3) | 0.690 | +2.271 |
| ∆ρmin (eÅ−3) | −0.409 | −1.359 |
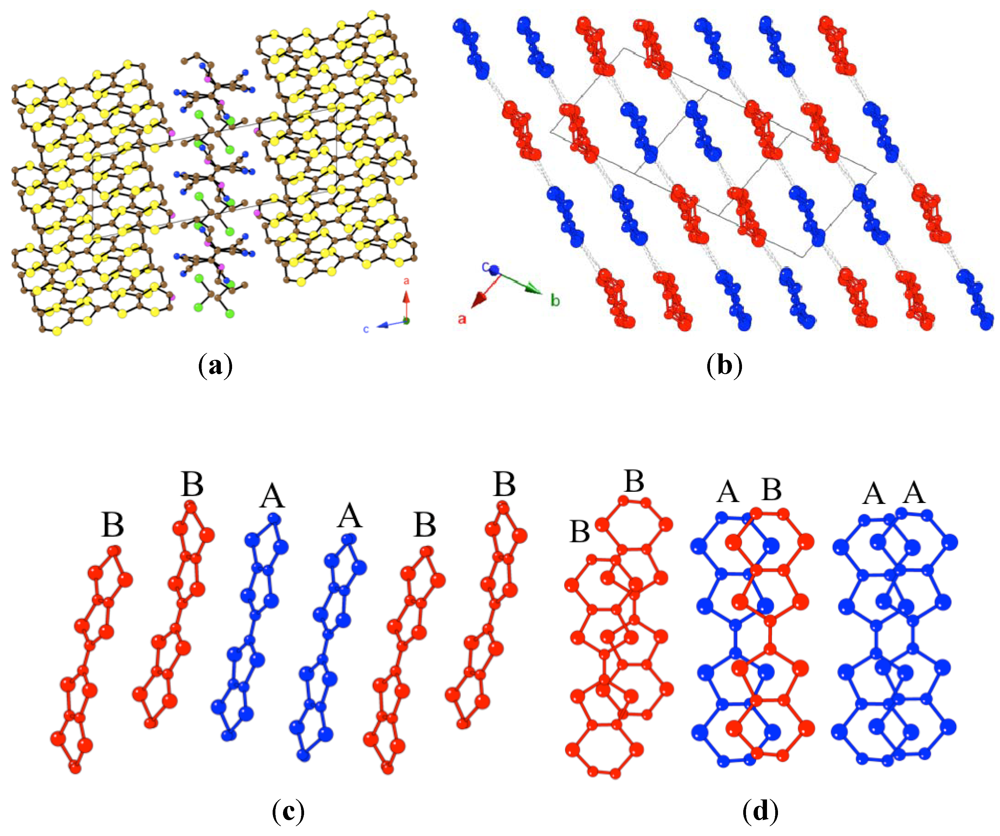
Figure 1.
(a) View of the alternating cationic and anionic layers in compound 1 along the c axis (similar structure for compound 2); (b) View of the ET layer. Dotted lines indicate the S···S contacts shorter than the sum of the van der Waals radii; (c) Side view of the ET chains; (d) View of the overlap between neighboring ET molecules in the chain.
Figure 1.
(a) View of the alternating cationic and anionic layers in compound 1 along the c axis (similar structure for compound 2); (b) View of the ET layer. Dotted lines indicate the S···S contacts shorter than the sum of the van der Waals radii; (c) Side view of the ET chains; (d) View of the overlap between neighboring ET molecules in the chain.
Since the chains are formed by two crystallographically independent ET molecules (noted as A and B) following the sequence ...–AABB–..., there are three different intra-chain overlaps: A–A, A–B and B‑B, all with similar twisting angles (ω) and displacements (
D): ω
AA = 34.0°,
DAA = 1.39 Å, ω
AB = 33.2°,
DAB = 1.38 Å, ω
BB = 32.4° and
DBB = 1.41 Å, in compound
1 and ω
AA = 34.0°,
DAA = 1.39 Å, ω
AB = 32.8°,
DAB = 1.32 Å, ω
BB = 32.0° and
DBB = 1.28 Å in compound
2 (
Figure 1).
The two terminal ethylene groups of the A-type ET molecule in compound 1 present a boat conformation and are staggered whereas those on the B-type ET molecule present a half-chair conformation and are also staggered. In compound 2 the A-type molecule presents a boat conformation in one of the ethylene groups and a half chair in the other whereas the B-type molecules presents a staggered half chair conformation in both ethylene groups.
As observed in other α' phases [
53], there are many intermolecular S···S contacts shorter than the sum of the van der Waals radii in both compounds (
Table 5). Although the structure of compound
2 is not very precise, the analysis of the bond lengths in the central TTF skeleton of the ET molecules in both compounds [
54] suggests an homogeneous charge distribution where both ET molecules bear similar charges, close to +0.5, giving an overall charge of +2 for the four ET molecules, in agreement with the −2 charge of the anion and the stoichiometry of the salts (4:1).
Table 5.
Intermolecular S···S distances (in Å) shorter than the sum of the van der Waals radii (3.6 Å) in compounds 1 and 2.
Table 5.
Intermolecular S···S distances (in Å) shorter than the sum of the van der Waals radii (3.6 Å) in compounds 1 and 2.
| Compound 1 | Compound 2 |
|---|
| Atoms | Distance | Atoms | Distance | Atoms | Distance |
|---|
| S1···S10 | 3.524(3) | S1A-S3B | 3.59(1) | S6A-S1B | 3.43(1) |
| S2A···S9 | 3.477(7) | S1A-S6B | 3.48(1) | S6A-S5B | 3.42(1) |
| S2A···S12 | 3.426(7) | S3A-S1B | 3.59(1) | S7A-S4B | 3.32(1) |
| S4···S10 | 3.527(3) | S4A-S7B | 3.55(1) | S7A-S8B | 3.38(1) |
| S6···S16 | 3.468(3) | S5A-S6B | 3.57(1) | S8A-S7B | 3.50(1) |
| S7···S16 | 3.534(3) | | | | |
| S8···S14 | 3.438(3) | | | | |
| S8···S15 | 3.429(3) | | | | |
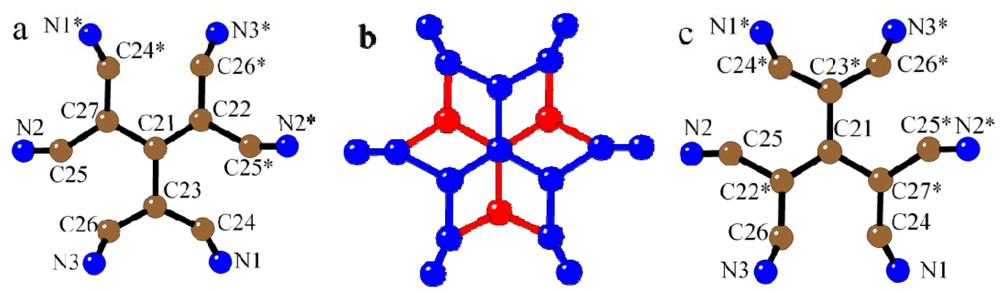
The anionic layer (in the
ab plane) is formed by tcpd
2− anions that appear disordered since they are located on an inversion center on the central carbon atom (C21). This inversion center generates two superimposed anions, as clearly shown in
Figure 2 and already observed in a Fe(II) chain with tcpd
2− [
55]. The solvent molecules (disordered THF in
1 and two water molecules in
2) are located between the tcpd
2− anions.
Figure 2.
View of the statistical disorder of the tcpd2− anion in compound 1 (similar in 2). Symmetry operation: * = −x, −y, −z.
Figure 2.
View of the statistical disorder of the tcpd2− anion in compound 1 (similar in 2). Symmetry operation: * = −x, −y, −z.
2.2.2. Structures of θ-(ET)2(tcnoetOH) (3) and β''-(ET)2(tcnoetOH) (4)
Compounds
3 and
4 crystallize in the triclinic space group P-1 but show different ET packings. Crystal and refinement data are shown in
Table 6. According to the asymmetric unit the chemical formula of compound
3 should be θ-(ET)
8(tcnoetOH)
4 (see description below), but the chemical formula θ-(ET)
2(tcnoetOH) was preferred to facilitate its crystal structure description. This compound presents layers of ET molecules parallel to the
ac plane alternating with layers of the tcnoetOH
− anions (
Figure 3). The ET molecules are packed in parallel chains although the molecules of one chain are twisted with respect to those of adjacent chains forming the so-called α and θ packings [
56]. Note that although compound
3 could be considered as an α phase (since there is only one different cationic layer), following Mori’s suggestion to unify both notations [
56], we should call compound
3 as a θ
80+80 multiple θ phase, since there are two different chains in the layers and each chain contains a repeating unit formed by eight eclipsed molecules with no dislocation (see
Figure 3).
Table 6.
Crystal data for compounds θ-ET2(tcnoetOH) (3) and β''-ET2(tcnoetOH) (4).
Table 6.
Crystal data for compounds θ-ET2(tcnoetOH) (3) and β''-ET2(tcnoetOH) (4).
| Compounds | 3 | 4 |
|---|
| Formula | C116H84N16O8S64 | C29H21N4O2S16 |
| M | 3881.83 | 970.46 |
| Cryst. Syst. | Triclinic | Triclinic |
| Space group | P-1 | P-1 |
| a (Å) | 17.3403(13) | 10.2496(7) |
| b (Å) | 20.1481(15) | 11.2314(10) |
| c (Å) | 23.9412(18) | 18.0649(11) |
| α (°) | 84.225(6) | 96.525(6) |
| β (°) | 69.596(7) | 90.593(5) |
| γ (°) | 76.254(6) | 115.397(7) |
| V (Å3) | 7613.6(10) | 1,862.4(2) |
| Z | 2 | 2 |
| T (K) | 293(2) | 170(2) |
| ρcalc (g·cm−3) | 1.693 | 1.731 |
| μ (cm−1) | 9.46 | 9.67 |
| F(000) | 3960 | 990 |
| Refl. Collec. | 66,331 | 19,184 |
| Refl. Uniq. | 42,071 | 11,234 |
| Rint | 0.1655 | 0.0361 |
| Refl. Obs
I > 2σ(I) | 4381 | 6269 |
| Nv | 1257 | 461 |
| a R1 | 0.1015 | 0.0487 |
| b wR2 | 0.1793 | 0.1171 |
| c GooF | 0.758 | 0.888 |
| ∆ρmax (eÅ−3) | +0.673 | +1.107 |
| ∆ρmin (eÅ−3) | −0.503 | −0.677 |
Figure 3.
(a) View of the alternating cationic and anionic layers in compound 3 along the b axis; (b) View of the ET layer. Dotted lines indicate the S···S contacts shorter than the sum of the van der Waals radii; (c) Side view of the ET chains; (d) View of the overlap between neighboring ET molecules in the chain.
Figure 3.
(a) View of the alternating cationic and anionic layers in compound 3 along the b axis; (b) View of the ET layer. Dotted lines indicate the S···S contacts shorter than the sum of the van der Waals radii; (c) Side view of the ET chains; (d) View of the overlap between neighboring ET molecules in the chain.
There are eight independent ET molecules (noted as A–H). Molecules A, B, C and H form one of the chains following the sequence ...CBAHHABC...and the remaining ET molecules (D, E, F and G) form the other chain with the sequence ...GFEDDEFG... (
Figure 3). All the ethylene groups of the ET molecules have half chair conformations and are eclipsed except for A and F molecules where they are staggered.
As it is usual in other θ phases, there are many inter-chains S···S contacts shorter than the sum of the van der Waals radii (3.60 Å) (
Table 7). Furthermore, there are also three short cation-anion S···N contacts shorter than the sum of the van der Walls radii (3.35 Å) implying ET molecules of B, C and E-type (
Table 7). The analysis of the charge of each ET molecule shows an inhomogeneous charge distribution with approximate charges of 0.9, 0.3, 0.7, 0.4, 0.3, 0.4, 0.6 and 0.2 for molecules A‑H, respectively, giving a total charge of
ca. +4 for the eight ET molecules, in agreement with the stoichiometry of compound
3 and with the presence of four tcnoetOH
− anions in the anionic layer per eight ET molecules [
54].
Table 7.
Intermolecular S···S and S···N distances (in Å) shorter than the sum of the van der Waals radii (3.60 and 3.35 Å, respectively) in compound 3.
Table 7.
Intermolecular S···S and S···N distances (in Å) shorter than the sum of the van der Waals radii (3.60 and 3.35 Å, respectively) in compound 3.
| Atoms | Distance | Atoms | Distance | Atoms | Distance |
|---|
| S3A-S8G | 3.490(6) | S3B-S5G | 3.552(6) | S3C-S5F | 3.559(6) |
| S5A-S6G | 3.582(6) | S5B-S2G | 3.575(6) | S6C-S5E | 3.548(6) |
| S5A-S2F | 3.553(7) | S7B-S5G | 3.526(6) | S7C-S5F | 3.442(6) |
| S6A-S5D | 3.547(8) | S8B-S3E | 3.531(7) | S8C-S7F | 3.592(8) |
| S7A-S8G | 3.551(6) | S8B-S7E | 3.462(8) | S2E-S5H | 3.596(6) |
| S8A-S6D | 3.525(6) | S4D-S6H | 3.527(7) | S7B-N32 | 3.30(1) |
| S8A-S7D | 3.484(8) | S8F-S3H | 3.494(6) | S7C-N34 | 3.17(1) |
| S8E-S6H | 3.536(6) | S6E-S8H | 3.520(6) | S8E-N14 | 3.28(1) |
Compound
4 also presents a layered structure (
Figure 4) with anionic and cationic layers alternating along the
c axis. The cationic layer is formed by two independent ET molecules (A and B) packed in parallel chains with all the molecules parallel and tilted with respect to the chain direction, giving rise to the so-called β'' phase (
Figure 4) [
57]. The chains present a repeating unit formed by four ET molecules following the sequence ...AABB... and present three dislocations along this repeating unit, giving rise to a β''
43 phase according to Mori’s notation [
57].
Figure 4.
(a) View of the alternating cationic and anionic layers in compound 4 along the c axis; (b) View of the ET layer. Dotted lines indicate the S···S contacts shorter than the sum of the van der Waals radii; (c) Side view of the ET chains; (d) View of the overlap between neighboring ET molecules in the chain.
Figure 4.
(a) View of the alternating cationic and anionic layers in compound 4 along the c axis; (b) View of the ET layer. Dotted lines indicate the S···S contacts shorter than the sum of the van der Waals radii; (c) Side view of the ET chains; (d) View of the overlap between neighboring ET molecules in the chain.
The ethylene groups of the A-type ET molecules present a half-chair and a boat conformation whereas the B-type molecules present a staggered half chair conformation. As in other β'' phases, there are many inter-chain S···S contacts shorter than the sum of the van der Waals radii (
Table 8).
Table 8.
Intermolecular S···S distances (in Å) shorter than the sum of the van der Waals radii (3.6 Å) in compound 4.
Table 8.
Intermolecular S···S distances (in Å) shorter than the sum of the van der Waals radii (3.6 Å) in compound 4.
| atoms | distance | atoms | distance |
|---|
| S1A-S7B | 3.502(1) | S7A-S1B | 3.506(2) |
| S4A-S6A | 3.494(1) | S7A-S4B | 3.501(1) |
| S5A-S7B | 3.529(1) | S7A-S5B | 3.354(2) |
| S6A-S8A | 3.357(1) | S7A-S8B | 3.573(1) |
An estimation of the charge of the two ET molecules show a checkerboard-type charge distribution (
Figure 4b) since the A-type ET molecule presents an approximate charge of +1/4 whereas the B-type molecule bears a charge of
ca. +3/4, giving an overall charge of +1 per two ET molecules, in agreement with the single negative charge of the tcnoetOH
− anion and the stoichiometry of the salt [
54].
2.2.3. Structure of β''-(ET)2(tcnoprOH)·(CHCl2CH2Cl)0.5 (5)
Compound
5 crystallizes in the triclinic space group P-1. Crystal and refinement data are shown in
Table 9.
Table 9.
Crystal data for compound β''-ET2(tcnoprOH)(CHCl2CH2Cl)0.5 (5).
Table 9.
Crystal data for compound β''-ET2(tcnoprOH)(CHCl2CH2Cl)0.5 (5).
| Compound | 5 |
|---|
| Formula | C31H24.5Cl1.5N4O2S16 |
| M | 1051.18 |
| Cryst Syst | Triclinic |
| Space group | P-1 |
| a (Å) | 7.6688(3) |
| b (Å) | 12.6432(4) |
| c (Å) | 22.6204(5) |
| α (°) | 98.430(2) |
| β (°) | 98.463(2) |
| γ (°) | 106.246(3) |
| V (Å3) | 2041.60(11) |
| Z | 2 |
| T (K) | 120(2) |
| ρcalc (g·cm−3) | 1.71 |
| μ (cm−1) | 9.84 |
| F(000) | 1072 |
| Refl. Collec. | 27108 |
| Refl. Uniq. | 10518 |
| Rint | 0.0491 |
| Refl. Obs I > 2σ(I) | 8669 |
| Nv | 524 |
| a R1 | 0.0358 |
| b wR2 | 0.1033 |
| c GooF | 1.049 |
| ∆ρmax (eÅ−3) | 0.743 |
| ∆ρmin (eÅ−3) | −0.566 |
Compound
5 presents an alternating layered structure with the ET molecules forming a β'' packing (see above) (
Figure 5). As observed in compound
4, the chains are formed by two independent ET molecules (A and B) and present a repeating unit formed by four ET molecules following the sequence ...AABB... although now there are no dislocations along this repeating unit, giving rise to a β''
40 phase according to Mori’s notation (
Figure 5) [
57].
Figure 5.
(a) View of the alternating cationic and anionic layers in compound 5 along the c axis; (b) View of the ET layer. Dotted lines indicate the S···S contacts shorter than the sum of the van der Waals radii; (c) Side view of the ET chains; (d) View of the overlap between neighboring ET molecules in the chain. The disorder on the ET molecule, the tcnoprOH− anion and the solvent has been removed for clarity.
Figure 5.
(a) View of the alternating cationic and anionic layers in compound 5 along the c axis; (b) View of the ET layer. Dotted lines indicate the S···S contacts shorter than the sum of the van der Waals radii; (c) Side view of the ET chains; (d) View of the overlap between neighboring ET molecules in the chain. The disorder on the ET molecule, the tcnoprOH− anion and the solvent has been removed for clarity.
The ethylene groups of the ET molecules present an eclipsed half-chair conformation although we have observed a partial disorder in the ethylene group C9–C10 of one ET molecule corresponding to two positions with occupancies factors of 0.28 and 0.72, labelled as C9A/C9B and C10A/C10B, respectively. An additional small disorder has also been detected on the –CH
2CH
2OH group of the tcnoprOH
− anion leading to two statistical positions for carbon C29 and C30, labelled as C29A/C29B and C30A/C30B with occupancies factors of 0.89 and 0.11 for A and B positions, respectively. As is common in the β'' phases, compound
5 presents several S···S contacts shorter than the sum of the van der Waals radii (
Table 10).
Table 10.
Intermolecular S···S distances (in Å) shorter than the sum of the van der Waals radii (3.60 Å) in compound 5.
Table 10.
Intermolecular S···S distances (in Å) shorter than the sum of the van der Waals radii (3.60 Å) in compound 5.
| atoms | distance | atoms | distance |
|---|
| S3-S16 | 3.446(1) | S6-S9 | 3.441(1) |
| S4-S15 | 3.404(1) | S6-S13 | 3.424(1) |
| S5-S14 | 3.376(1) | S7-S16 | 3.429(1) |
| S5-S10 | 3.396(1) | S8-S15 | 3.370(1) |
The analysis of the bond lengths in compound
5 shows an homogeneous charge distribution with both ET molecules (A and B) bearing a charge close to +0.5, in agreement with the stoichiometry (2:1) and the anionic charge (−1) [
54].
The anionic layers, parallel to the ac plane, contain tcnoprOH− anions and half a disordered CHCl2CH2Cl molecule.