An Ionic Liquid Solution of Chitosan as Organocatalyst
Abstract
:1. Introduction

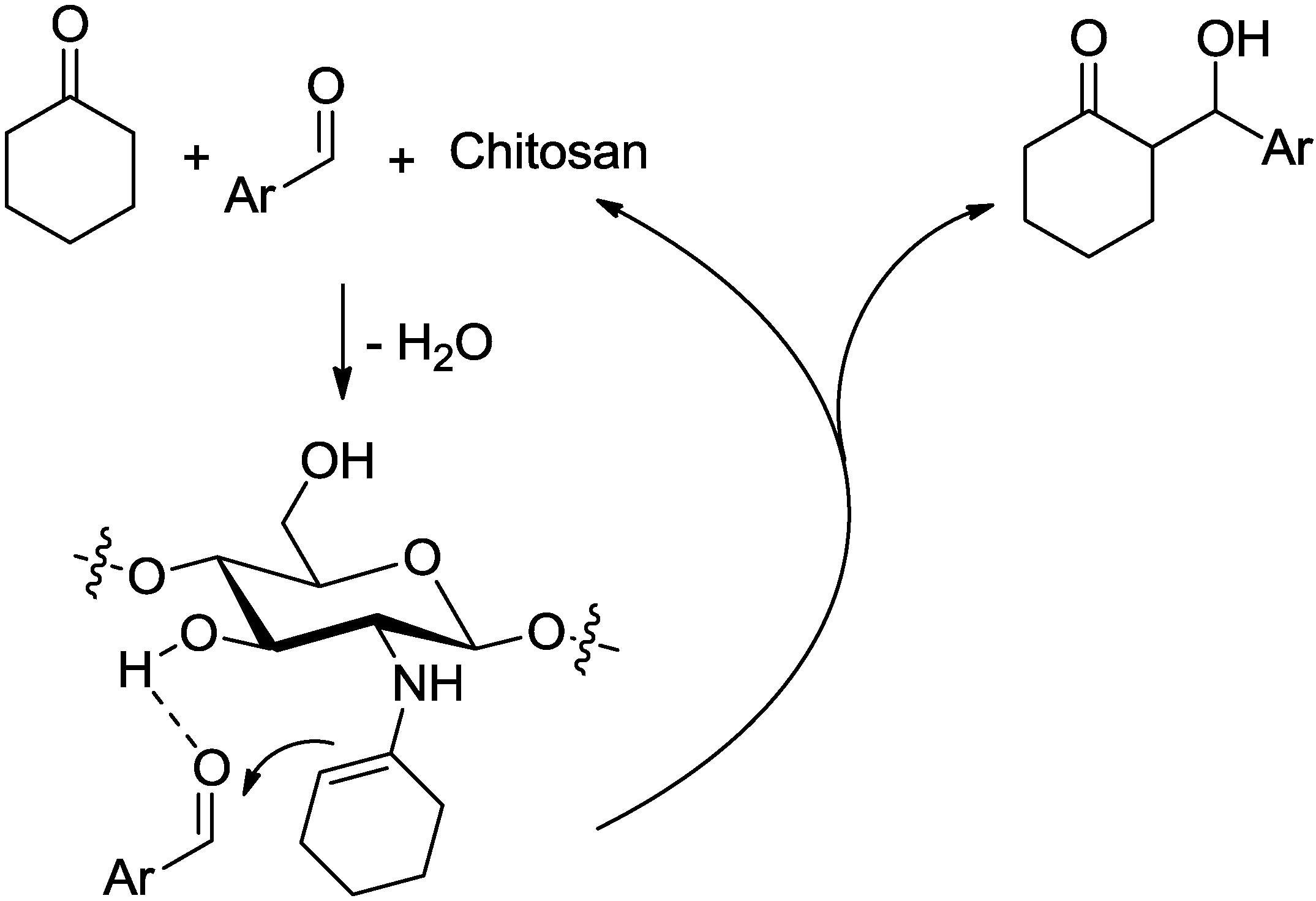
2. Results and Discussion
| Entry | Ionic Liquid | Additive | Yield [%] a | dr [syn/anti] b | ee (syn) [%] c | ee (anti) [%] c |
|---|---|---|---|---|---|---|
| 1 | [BMIM][OAc] | TFA | 71 | 66/34 | 34 | 51 |
| 2 | [BMIM][OAc] | - | - | - | - | - |
| 3 | [EMIM][OAc] | TFA | 46 | 56/44 | 8 | 2 |
| 4 | [EMIM][OAc] | - | - | - | - | - |
| 5 | [BMIM][Cl] | - | 27 | 49/51 | 0 | 14 |
| 6 | [EMIM][Cl] | - | 49 | 51/49 | 24 | 26 |
| 7 | [BMIM][Br] | - | 49 | 38/62 | 13 | 85 |
| 8 | [BMIM][Br] | AcOH | 61 | 47/53 | 4 | 13 |
| 9 | [BMIM][Br] | TFA | - | - | - | - |
| 10 | [EMIM][Br] | - | 44 | 52/48 | 43 | 17 |
| 11 | [EMIM][Br] | AcOH | 25 | 25/75 | 48 | 78 |
| 12 | [EMIM][Br] | TFA | - | - | - | - |
| 13 | [BMIM][BF4] | - | 78 | 65/35 | 7 | 21 |
| 14 | [EMIM][BF4] | - | 12 | 47/53 | 5 | 15 |
| 15 | [BMIM][PF6] | - | 73 | 51/49 | 5 | 23 |
| 16 | [EMIM][PF6] | - | 69 | 59/41 | 4 | 12 |
| 17 | DMSO | - | - | - | - | - |
| 18 | H2O | - | 13 | 97/3 | - | - |
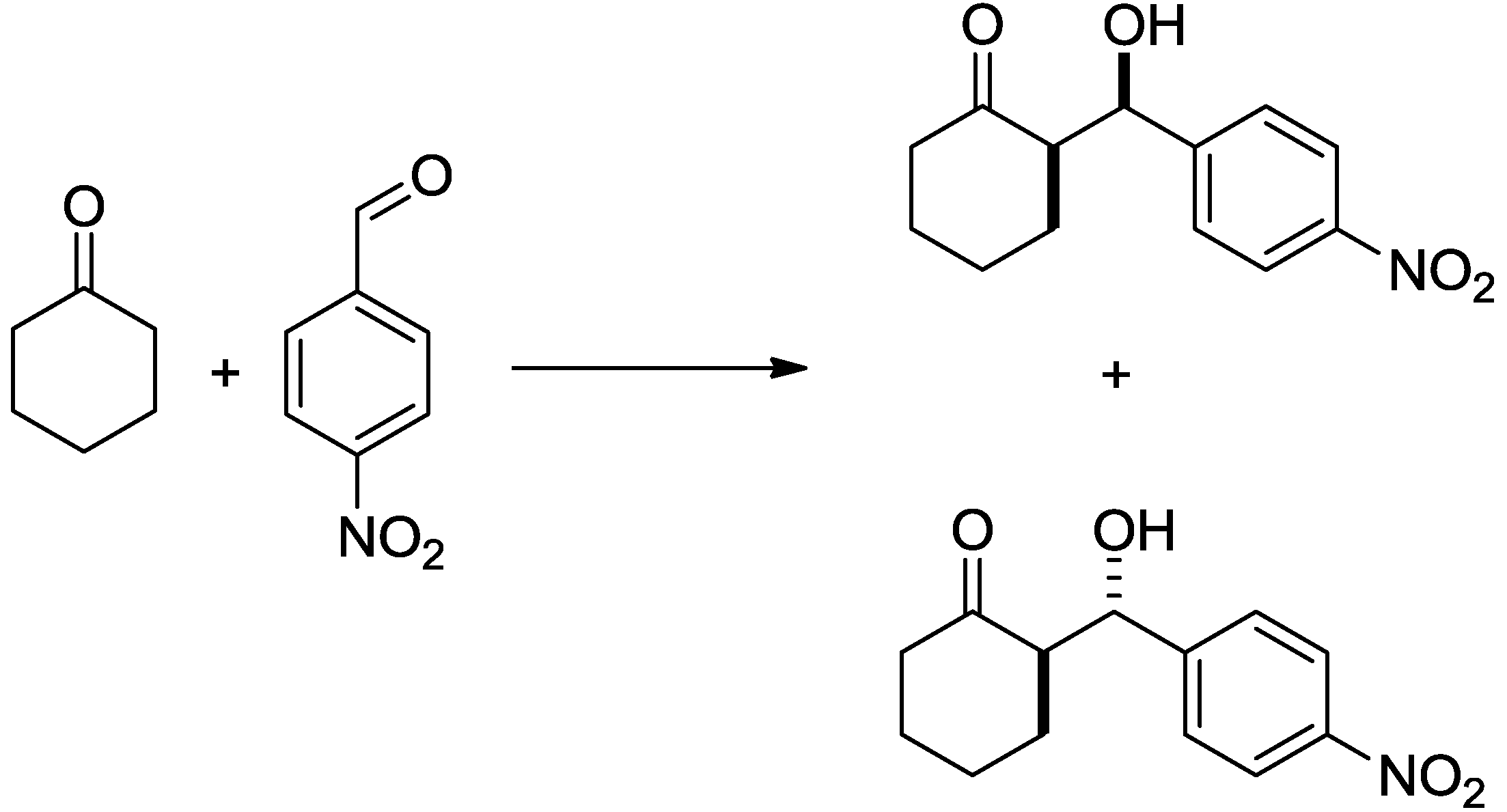
| Entry | Aldehydes | Ketones | Yield [%] a | dr [syn/anti] b | ee (syn) [%] c | ee (anti) [%] c |
|---|---|---|---|---|---|---|
| 1 |  |  | 88 | 94/6 | 30 | 6 |
| 2 |  |  | 13 | 40/60 | - | 38 |
3. Experimental Section
4. Conclusions
Conflicts of Interest
References
- Berkessel, A.; Gröger, H. Asymmetric Organocatalysis; Wiley-VCH: Weinheim, Germany, 2005. [Google Scholar]
- Dalko, P.I. Enantioselective Organocatalysis; Wiley-VCH: Weinheim, Germany, 2007. [Google Scholar]
- Sereda, O.; Tabassum, S.; Wilhelm, R. Lewis acid organocatalysts. Top. Curr. Chem. 2010, 291, 349–394. [Google Scholar] [CrossRef]
- Northrup, A.B.; MacMillan, D.W.C. The first general enantioselective catalytic Diels-Alder reaction with simple α,β-unsaturated ketones. J. Am. Chem. Soc. 2002, 124, 2458–2460. [Google Scholar] [CrossRef]
- Hajos, Z.G.; Parrish, D.R. Asymmetric synthesis of bicyclic intermediates of natural product chemistry. J. Org. Chem. 1974, 39, 1615–1621. [Google Scholar] [CrossRef]
- Eder, U.; Sauer, G.; Wiechert, R. New type of asymmetric cyclization to optically active steroid CD partial structures. Angew. Chem. Int. Ed. Engl. 1971, 10, 496–497. [Google Scholar] [CrossRef]
- List, B.; Lerner, R.A.; Barbas, C.F. Proline-catalyzed direct asymmetric Aldol reactions. J. Am. Chem. Soc. 2000, 122, 2395–2396. [Google Scholar] [CrossRef]
- Gröger, H.; Wilken, J. The application of l-proline as an enzyme mimic and further new asymmetric syntheses using small organic molecules as chiral catalysis. Angew. Chem. Int. Ed. 2001, 40, 529–532. [Google Scholar] [CrossRef]
- Sakthivel, K.; Notz, W.; Bui, T.; Barbas, C.F. Amino acid Catalyzed Direct Asymmetric Aldol Reactions: A Bioorganic Approach to Catalytic Asymmetric Carbon-Carbon Bond-Forming reactions. J. Am. Chem. Soc. 2001, 123, 5260–5267. [Google Scholar] [CrossRef]
- Xu, L.-W.; Lu, Y. Primary amino acids: Privileged catalysts in enantioselective organocatalysis. Org. Biomol. Chem. 2008, 6, 2047–2053. [Google Scholar] [CrossRef]
- Gioa, C.; Ricci, A.; Bernardi, L.; Bourahla, K.; Tanchoux, N.; Robitzer, M.; Quignard, F. chitosan aerogel Beads as a Heterogeneous Organocatalyst for the asymmetric Aldol Reaction in the presence of water: An assessment of the effect of additives. Eur. J. Org. Chem. 2013, 588–594. [Google Scholar]
- Ricci, A.; Bernardi, L.; Gioia, C.; Vierucci, S.; Robitzer, M.; Quignard, F. Chitosan aerogel: A recyclable, heterogeneous organocatalyst for the asymmetric direct aldol reaction in water. Chem. Commun. 2010, 6288–6290. [Google Scholar]
- Reddy, K.R.; Rajgopal, K.; Maheswari, C.U.; Kantam, M.L. Chitosan hydrogel: A green and recyclable biopolymer catalyst for aldol and Knoevenagel reactions. New J. Chem. 2006, 30, 1549–1552. [Google Scholar] [CrossRef]
- Kühbeck, D.; Saidulu, G.; Reddy, K.R.; Diaz, D.D. Critical assessment of the efficiency of chitosan biohydrogel beads as recyclable and heterogeneous organocatalyst for C–C bond formation. Green Chem. 2012, 14, 378–392. [Google Scholar] [CrossRef]
- Dekamin, M.G.; Azimoshan, M.; Ramezani, L. Chitosan: A highly efficient renewable and recoverable bio-polymer catalyst for the expeditious synthesis of α-amino nitriles and imines under mild conditions. Green Chem. 2013, 15, 811–820. [Google Scholar] [CrossRef]
- Chtchigrovsky, M.; Primo, A.; Gonzalez, P.; Molvinger, K.; Robitzer, M.; Quignard, F.; Taran, F. Functionalized chitosan as a green, recyclable, biopolymer-supported catalyst for the [3 + 2] huisgen cycloaddition. Angew. Chem. Int. Ed. 2009, 48, 5916–5920. [Google Scholar] [CrossRef]
- Pinkert, A.; Marsh, K.N.; Pang, S.S.; Staiger, M.P. Ionic liquids and Their Interaction with cellulose. Chem. Rev. 2009, 109, 6712–6728. [Google Scholar] [CrossRef]
- Barber, P.S.; Griggs, C.S.; Bonner, J.R.; Rogers, R.D. Electrospinning of chitin nanofibers directly from an ionic liquid extract of shrimp shells. Green Chem. 2013, 15, 601–607. [Google Scholar] [CrossRef]
- Wasserscheid, P.; Welton, T. Ionic Liquids in Synthesis, 2nd ed.; Wiley-VCH: Weinheim, Germay, 2008; Volume 1–2. [Google Scholar]
- Khan, S.S.; Shah, J.; Liebscher, J. Ionic-liquid tagged prolines as recycable organocatalysts for enantioselective α-aminoxylations of carbonyl compounds. Tetrahedron 2011, 67, 1812–1820. [Google Scholar] [CrossRef]
- Winkel, A.; Reddy, P.V.G.; Wilhelm, R. Recent advances in the synthesis and application of chiral ionic liquids. Synthesis 2008, 999–1016. [Google Scholar]
- Jurčík, V.; Wilhelm, R. The preparation of new enantiopure imidazolinium salts and their evaluation as catalysts and shift reagents. Tetrahedron 2006, 17, 801–810. [Google Scholar] [CrossRef]
- Winkel, A.; Wilhelm, R. New chiral ionic liquids Based on Imidazolinium salts. Tetrahedron 2009, 20, 2344–2350. [Google Scholar] [CrossRef]
- Winkel, A.; Wilhelm, R. New chiral ionic liquids based on enantiopure sulphate and sulfonate anions for chiral recognition. Eur. J. Org. Chem. 2010, 5817–5824. [Google Scholar] [CrossRef]
- Hollóczki, O.; Gerhard, D.; Massone, K.; Szarvas, L.; Németh, B.; Veszprémi, T.; Nyulászi, L. Carbenes in ionic liquids. New. J. Chem. 2010, 34, 3004–3009. [Google Scholar] [CrossRef]
- Hollóczki, O.; Nyulászi, L. Neutral species from “non-protic” N-heterocyclic ionic liquids. Org. Biomol. Chem. 2011, 9, 2634–2640. [Google Scholar] [CrossRef]
- Kelemen, Z.; Hollóczki, O.; Nagy, J.; Nyulászi, L. An organocatalytic ionic liquid. Org. Biomol. Chem. 2011, 9, 5362–5364. [Google Scholar] [CrossRef]
- Xie, B.-H.; Li, W.; Liu, Y.; Li, H.-H.; Guan, Z.; He, Y.-H. The enzymeticasymmetric aldol reaction using acidic protease from Aspergillus usamii. Tetrahedron 2012, 68, 3160–3164. [Google Scholar] [CrossRef]
- Gauchot, V.; Schmitzer, A.R. Asymmetric Aldol Reaction catalyszed by the anion of an ionic liquid. J. Org. Chem. 2012, 77, 4917–4923. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Heckel, T.; Konieczna, D.D.; Wilhelm, R. An Ionic Liquid Solution of Chitosan as Organocatalyst. Catalysts 2013, 3, 914-921. https://doi.org/10.3390/catal3040914
Heckel T, Konieczna DD, Wilhelm R. An Ionic Liquid Solution of Chitosan as Organocatalyst. Catalysts. 2013; 3(4):914-921. https://doi.org/10.3390/catal3040914
Chicago/Turabian StyleHeckel, Tatjana, Dagny Dagmara Konieczna, and René Wilhelm. 2013. "An Ionic Liquid Solution of Chitosan as Organocatalyst" Catalysts 3, no. 4: 914-921. https://doi.org/10.3390/catal3040914
APA StyleHeckel, T., Konieczna, D. D., & Wilhelm, R. (2013). An Ionic Liquid Solution of Chitosan as Organocatalyst. Catalysts, 3(4), 914-921. https://doi.org/10.3390/catal3040914




