Cellulases in Ionic Liquids—The Long Term Stability of Aspergillus sp. Cellulase
Abstract
:1. Introduction
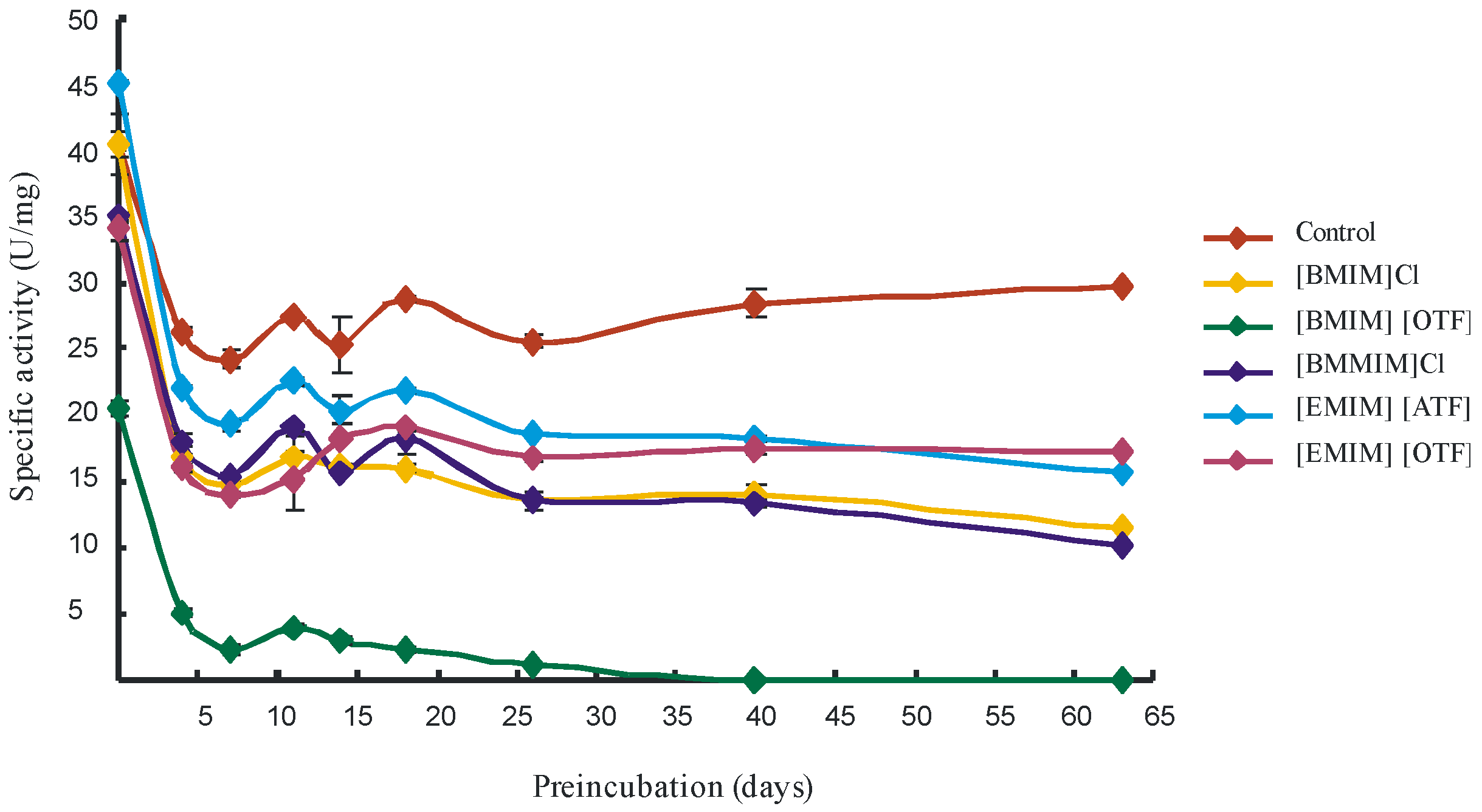
2. Results and Discussion

3. Experimental Section
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Heinze, T.; Schwikal, K.; Barthel, S. Ionic liquids as reaction medium in cellulose functionalization. Macromol. Biosci. 2005, 5, 520–525. [Google Scholar] [CrossRef]
- Swatloski, R.P.; Spear, S.K.; Holbrey, J.D.; Rogers, R.D. Dissolution of cellulose [correction of cellose] with ionic liquids. J. Am. Chem. Soc. 2002, 124, 4974–4975. [Google Scholar]
- Pottkämper, J.; Barthen, P.; Ilmberger, N.; Schwaneberg, U.; Schenk, A.; Schulte, M.; Ignatiev, N.; Streit, W.R. Applying metagenomics for the identification of bacterial cellulases that are stable in ionic liquids. Green Chem. 2009, 11, 957–965. [Google Scholar] [CrossRef]
- Turner, M.B.; Spear, S.K.; Huddleston, J.G.; Holbrey, J.D.; Rogers, R.D. Ionic liquid salt-induced inactivation and unfolding of cellulase from Trichoderma reesei. Green Chem. 2003, 5, 443–447. [Google Scholar] [CrossRef]
- Ilmberger, N.; Meske, D.; Juergensen, J.; Schulte, M.; Barthen, P.; Rabausch, U.; Angelov, A.; Mientus, M.; Liebl, W.; Schmitz, R.A.; et al. Metagenomic cellulases highly tolerant towards the presence of ionic liquids-linking thermostability and halotolerance. Appl. Microbiol. Biotechnol. 2012, 95, 135–146. [Google Scholar] [CrossRef]
- Liang, C.; Xue, Y.; Fioroni, M.; Rodriguez-Ropero, F.; Zhou, C.; Schwaneberg, U.; Ma, Y. Cloning and characterization of a thermostable and halo-tolerant endoglucanase from Thermoanaerobacter tengcongensis MB4. Appl. Microbiol. Biotechnol. 2011, 89, 315–325. [Google Scholar] [CrossRef]
- Beckham, G.T.; Dai, Z.; Matthews, J.F.; Momany, M.; Payne, C.M.; Adney, W.S.; Baker, S.E.; Himmel, M.E. Harnessing glycosylation to improve cellulase activity. Curr. Opin. Biotechnol. 2012, 23, 338–345. [Google Scholar] [CrossRef]
- Hong, J.; Tamaki, H.; Akiba, S.; Yamamoto, K.; Kumagai, H. Cloning of a gene encoding a highly stable endo-beta-1,4-glucanase from Aspergillus niger and its expression in yeast. J. Biosci. Bioeng. 2001, 92, 434–441. [Google Scholar]
- Steen, P.V.D.; Rudd, P.M.; Dwek, R.A.; Opdenakker, G. Concepts and principles of O-linked glycosylation. Crit. Rev. Biochem. Mol. Biol. 1998, 33, 151–208. [Google Scholar] [CrossRef]
- Zheng, J.; Xu, Z.; Wang, Y.; Hong, K.; Liu, P.; Zhu, W. Cyclic tripeptides from the halotolerant fungus Aspergillus sclerotium PT06–1. J. Nat. Prod. 2010, 73, 1133–1137. [Google Scholar] [CrossRef]
- Ilmberger, N.; Streit, W.R. Screening for cellulase-encoding clones in metagenomic libraries. Methods Mol. Biol. 2010, 668, 177–188. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ilmberger, N.; Pottkämper, J.; Streit, W.R. Cellulases in Ionic Liquids—The Long Term Stability of Aspergillus sp. Cellulase. Catalysts 2013, 3, 584-587. https://doi.org/10.3390/catal3020584
Ilmberger N, Pottkämper J, Streit WR. Cellulases in Ionic Liquids—The Long Term Stability of Aspergillus sp. Cellulase. Catalysts. 2013; 3(2):584-587. https://doi.org/10.3390/catal3020584
Chicago/Turabian StyleIlmberger, Nele, Julia Pottkämper, and Wolfgang R. Streit. 2013. "Cellulases in Ionic Liquids—The Long Term Stability of Aspergillus sp. Cellulase" Catalysts 3, no. 2: 584-587. https://doi.org/10.3390/catal3020584



