Epidemiology of Skin Cancer: Role of Some Environmental Factors
Abstract
:1. Introduction
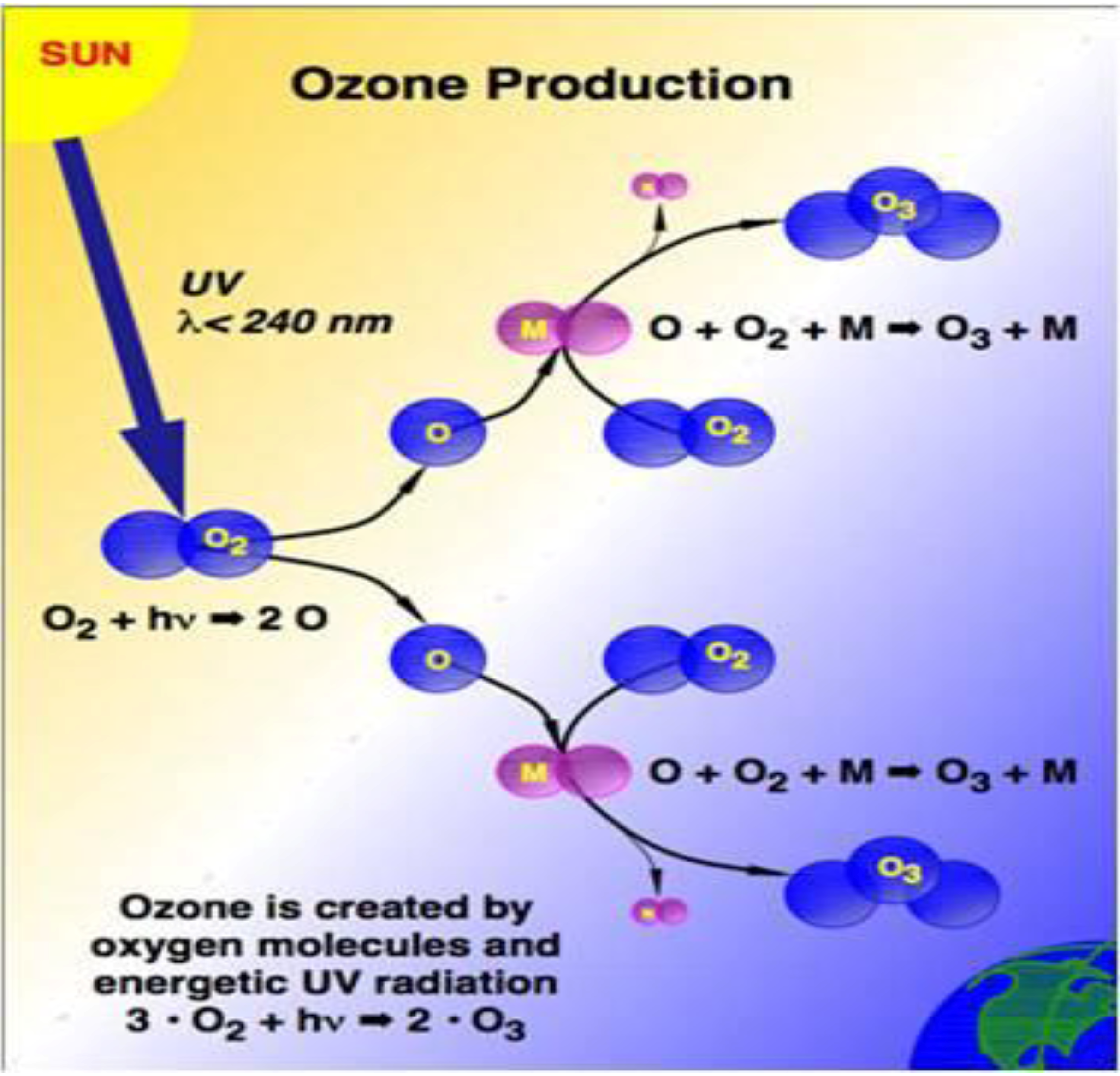
2. Ozone Depletion
3. Relationship between Surface UV Radiation and Air Pollution
4. Effects of the Pollutant Arsenic
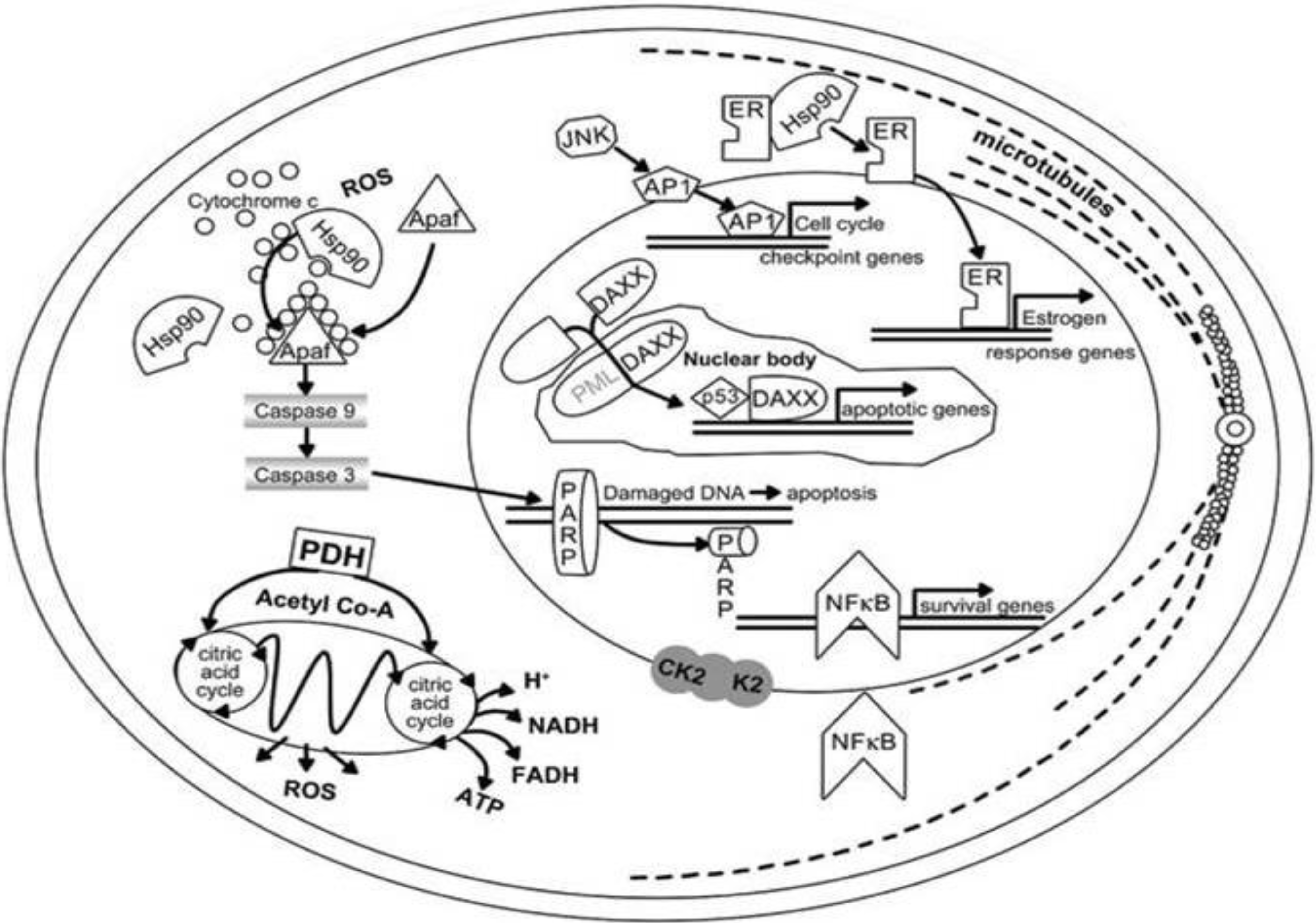
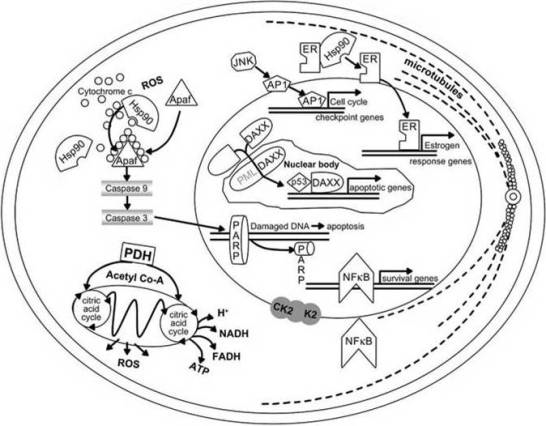
5. Climate Change and Human Skin Cancer
6. Conclusion
Declaration of Conflicts of Interest
References
- Madronich, S.; de Gruijl, F.R. Stratospheric ozone depletion between 1979 and 1992: Implications for biologically active ultraviolet-B radiation and non-melanoma skin cancer incidence. Photochem. Photobiol. 1994, 59, 541–546. [Google Scholar] [CrossRef]
- De Fabo, E.C. Artic stratospheric ozone depletion and increased UVB radiation: Potential impacts to human health. Int. J. Circumpolar. Health 2005, 64, 509–522. [Google Scholar]
- Schaart, F.M.; Garbe, C.; Orfanos, C.E. Disappearance of the ozone layer and skin cancer: Attempt at risk assessment. Hautarzt 1993, 44, 63–68. [Google Scholar]
- McCarthy, W.H. The Australian experience in sun protection and screening for melanoma. J. Surg. Oncol. 2004, 86, 236–245. [Google Scholar] [CrossRef]
- de Gruijl, F.R. Skin cancer and solar UV radiation. Eur. J. Cancer 1999, 35, 2003–2009. [Google Scholar] [CrossRef]
- Salmon, P.J.; Chan, W.C.; Griffin, J.; McKenzie, R.; Rademaker, M. Extremely high levels of melanoma in Tauranga, New Zealand: Possible causes and comparisons with Australia and the northern hemisphere. Australas J. Dermatol. 2007, 48, 208–216. [Google Scholar] [CrossRef]
- Abarca, J.F.; Casiccia, C.C. Skin cancer and ultraviolet-B radiation under the Antartic ozone hole: Southern Chile, 1987-2000. Photodermatol. Photoimmunol. Photomed. 2002, 18, 294–302. [Google Scholar] [CrossRef]
- Jones, R.R. Ozone depletion and its effects on human populations. Br. J. Dermatol. 1992, 127, 2–6. [Google Scholar] [CrossRef]
- Leiter, U.; Garbe, C. Epidemiology of melanoma and nonmelanoma skin cancer--the role of sunlight. Adv. Exp. Med. Biol. 2008, 624, 89–103. [Google Scholar] [CrossRef]
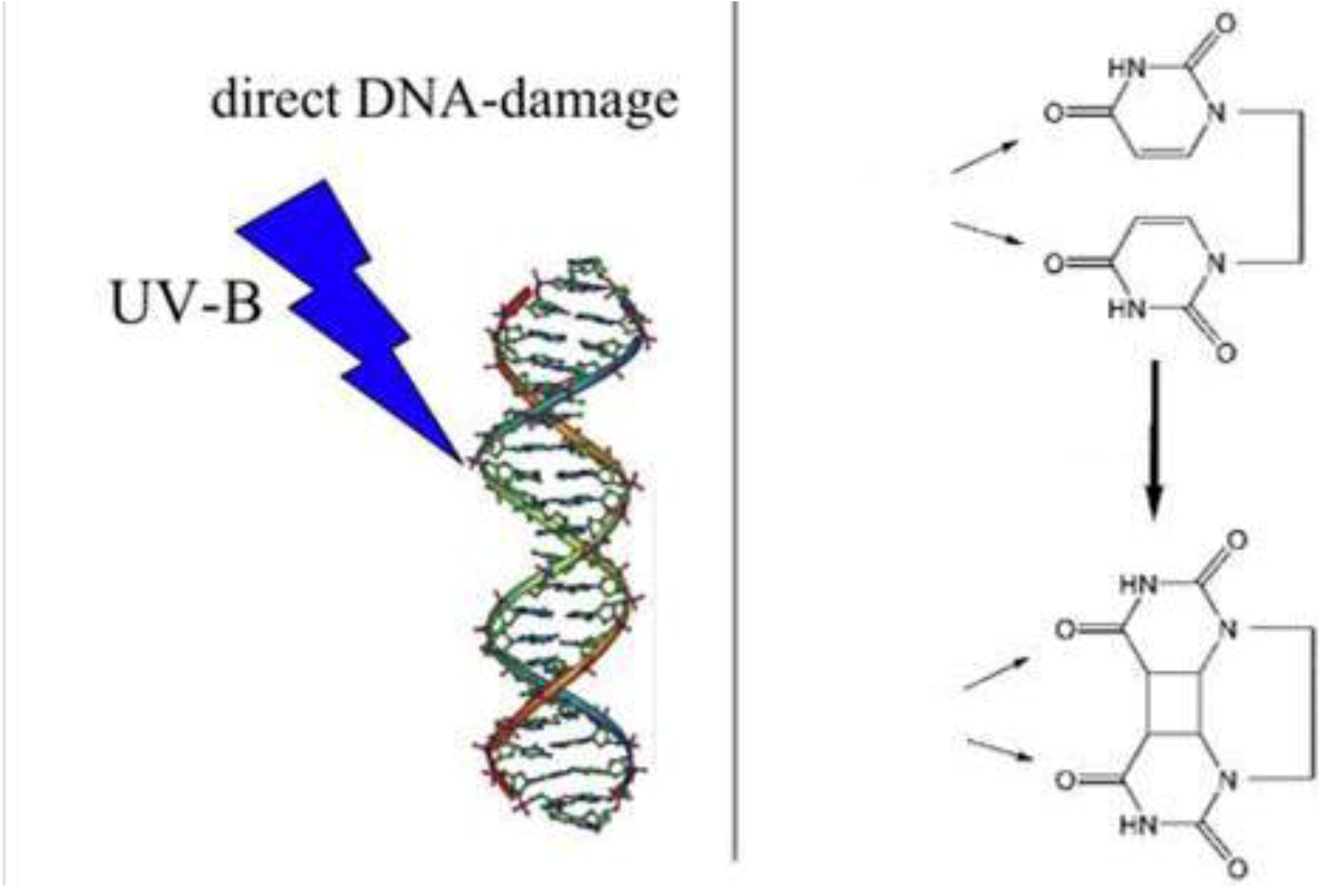
- Gerriet 41. The direct DNA damage that sometimes occurs after DNA absorbs a UVB photon. Feel free to copy it. Available online: http://upload.wikimedia.org (accessed on 24 November 2010).
- An, J.L.; Wang, Y.S.; Li, X.; Sun, Y.; Shen, S.H. Relationship between surface UV radiation and air pollution in Bejiing. Huan Jing Ke Xue 2008, 29, 1053–1058. [Google Scholar]
- Barnard, W.F.; Saxena, V.K.; Wenny, B.N.; DeLuisi, J.J. Daily surface UV exposure and its relationship to surface pollutant measurements. J. Air Waste Manag. Assoc. 2003, 53, 237–245. [Google Scholar] [CrossRef]
- Sabburg, J.; Parisi, A.; Wong, J. Ozone, cloud, solar and UV-B levels at a low pollution, Southern Hemisphere, sub-tropical site for winter/spring 1995. Australas Phys. Eng. Sci. Med. 1997, 20, 198–202. [Google Scholar]
- Burke, K.E.; Wei, H. Synergistic damage by UVA radiation and pollutants. Toxicol. Ind. Health. 2009, 25, 219–224. [Google Scholar] [CrossRef]
- Tokunaga. Risk assessment of arsenic for people who live in the area of arsenic-contaminated undergroundwater in Bangladesh. Kokuritsu Iyakuhin Shokuhin Eisei Kenkyusho Hokoku 2007, 125, 1–16. [Google Scholar]
- Rossman, T.G.; Uddin, A.N.; Burns, F.J. Evidence that arsenite acts as a carcinogen in skin cancer. Toxicol. App. Pharmacol. 2004, 198, 394–404. [Google Scholar] [CrossRef]
- Abernathy, C.O.; Thomas, D.J.; Calderon, R.L. Health effects and risk assessment of arsenic. Am. Soc. Nutr. Sci. J. Nutr. 2003, 133, 1536–1538. [Google Scholar]
- Kitchin, K.T. Recent advances in arsenic carcinogenesis: Modes of action, animal model system methylated arsenic metabolites. Toxicol. Appl. Pharmacol. 2001, 172, 249–261. [Google Scholar] [CrossRef]
- Tchounwou, P.B.; Wilson, B.; Ishaque, A. Important considerations in the development of public health advisories for arsenic and arsenic-containing compounds in drinking water. Rev. Environ. Health 1999, 14, 211–229. [Google Scholar]
- National Research Council, Arsenic in Drinking Water; National Academy Press: Washington, DC, USA, 1999.
- National Research Council, Arsenic in Drinking Water; National Academy Press: Washington, DC, USA, 2001.
- Nordstrom, D.K. Worldwide occurrences of arsenic in ground water. Science 2002, 21, 2143–2145. [Google Scholar] [CrossRef]
- Smedley, P.L.; Kinniburgh, D.G. A review of the source, behavior and distribution of arsenic in natural waters. Appl. Geochem. 2002, 17, 517–568. [Google Scholar] [CrossRef]
- Florea, A.M.; Yamoah, E.N.; Dopp, E. Intracellular calcium disturbances induced by arsenic and its Methylated derivatives in relation to genomic damage and apoptosis induction. Rev. Environ. Health Perspect. 2005, 113, 659–664. [Google Scholar] [CrossRef]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Some drinking-water disinfectants and contaminants, including arsenic. IARC Monogr. Eval. Carcinog. Risks Hum. 2004, 84, 269–477.
- Chouhan, S.; Flora, S.J. Arsenic and fluoride: two major ground water pollutants. Indian J Exp Biol. 2010, 48, 666–678. [Google Scholar]
- Yu, H.S.; Liao, W.T.; Chai, C.Y. Arsenic carcinogenesis in the skin. J. Biomed. Sci. 2006, 13, 657–666. [Google Scholar] [CrossRef]
- Akao, Y.; Yamada, H.; Nakagawa, Y. Arsenic induced apoptosis in malignant cells in vitro. Leuk Lymphoma. 2000, 37, 53–63. [Google Scholar] [CrossRef]
- Cai, X.; Yu, Y.; Huang, Y.; Zhang, L.; Jia, P.M.; Zhao, Q. Arsenic tioxide-induced mitotic arrest and apoptosis in acute promyelocytic leukemia cells. Leukemia 2003, 17, 1333–1337. [Google Scholar] [CrossRef]
- Iwama, K.; Nakajo, S.; Aiuchi, T.; Nakaya, K. Apoptosis induced by arsenic trioxide in leukemia U937 cells is dependent on activation of p38, inactivation of ERK and the Ca2+-dependent production of superoxide. Int. J. Cancer 2001, 92, 518–526. [Google Scholar] [CrossRef]
- Miller, W.H., Jr.; Schipper, H.M.; Lee, J.S.; Singer, J.; Waxman, S. Mechanism of action of arsenic trioxide. Cancer Res. 2002, 62, 3893–3903. [Google Scholar]
- Guo, H.R.; Yu, H.S.; Hu, H.; Monson, R.R. Arsenic in drinking water and Skin cancers: Cell-type specificity (R.O.C.). Cancer Causes Contr. 2001, 12, 909–916. [Google Scholar] [CrossRef]
- Murata, J.; Abe, R.; Shimizu, H. Increased soluble Fas ligand levels in patients with Stevens-Johnson syndrome and toxic epidermal necrolysis preceding skin detachment. J. Allergy Clin. Immunol. 2008, 122, 992–1000. [Google Scholar] [CrossRef]
- Tay, C.H.; Seah, S.C. Arsenic poisoning from anti-asmatic herbal preparations. Med. J. Aus. 1975, 2, 424–428. [Google Scholar]
- Waalkes, M.P.; Liu, J.; Diwan, B.A. Transplacental arsenic carcinogenesis in mice. Toxicol. Appl. Pharmacol. 2007, 222, 271–280. [Google Scholar] [CrossRef]
- Kitchin, K.T.; Ahmad, S. Oxidative stress as a possible mode of action for arsenic carcinogenesis. Toxicol. Lett. 2003, 137, 3–13. [Google Scholar] [CrossRef]
- IPCC. IPCC Fourth Assessment Report: Climate Change 2007. The Physical Science Basis. Working Group I Assessment Report. Available online: http://www.ipcc.ch/ipccreports/ar4-wg1.htm (accessed on 24 November 2010).
- Van der Leun, J.C.; Piacentini, R.D.; de Gruijl, F.R. Climate change and human skin cancer. Photochem. Photobiol. Sci. 2008, 7, 730–733. [Google Scholar] [CrossRef]
- Norval, M.; Cullen, A.P.; de Gruijo, F.R.; Longstreth, J.; Takizawa, Y.; Lucas, R.M.; Noona, F.P.; Van der Leun, J.C. The effevts on human health from stratospheric ozone depletion and its interactions with climate change. Photochem. Photobiol. Sci. 2007, 6, 232–251. [Google Scholar] [CrossRef]
- Bain, J.; Rusch, H.; Kline, B. The effect of temperature upon ultraviolet carcinogenesis with wavelengths 2800–3400 A. Cancer Res. 1943, 3, 610–612. [Google Scholar]
- Freeman, R.G.; Knox, J.M. Influence of temperature on ultraviolet injury. Arch. Dermatol. 1964, 89, 858–864. [Google Scholar] [CrossRef]
- Diffey, B.L.; Gibson, C.J.; Haylock, R.; McKinlay, A.F. Outdoor ultraviolet exposure of children and adolescents. Br. J. Dermatol. 1996, 134, 1030–1034. [Google Scholar] [CrossRef]
- Hill, D.; Boulter, J. Sun protection behaviour: Determinants and trends. Cancer Forum 1996, 20, 204–210. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fabbrocini, G.; Triassi, M.; Mauriello, M.C.; Torre, G.; Annunziata, M.C.; De Vita, V.; Pastore, F.; D’Arco, V.; Monfrecola, G. Epidemiology of Skin Cancer: Role of Some Environmental Factors. Cancers 2010, 2, 1980-1989. https://doi.org/10.3390/cancers2041980
Fabbrocini G, Triassi M, Mauriello MC, Torre G, Annunziata MC, De Vita V, Pastore F, D’Arco V, Monfrecola G. Epidemiology of Skin Cancer: Role of Some Environmental Factors. Cancers. 2010; 2(4):1980-1989. https://doi.org/10.3390/cancers2041980
Chicago/Turabian StyleFabbrocini, Gabriella, Maria Triassi, Maria Chiara Mauriello, Guglielma Torre, Maria Carmela Annunziata, Valerio De Vita, Francesco Pastore, Vincenza D’Arco, and Giuseppe Monfrecola. 2010. "Epidemiology of Skin Cancer: Role of Some Environmental Factors" Cancers 2, no. 4: 1980-1989. https://doi.org/10.3390/cancers2041980