Cytokines and Growth Factors Expressed by Human Cutaneous Melanoma
Abstract
:1. Introduction
2. Results
2.1. Cultured Human Melanoma Cell Lines
| Cytokine | ELISA sensitivity
(pg/mL) | Number of cell lines tested | Percentage of cell lines
secreting cytokine |
|---|---|---|---|
| IL-8 | 31 | 16 | 100 |
| IL-6 | 10 | 15 | 87 |
| OPN | 312 | 22 | 82 |
| TNF-α | 16 | 15 | 53 |
| IL-10 | 15 | 17 | 47 |
| GM-CSF | 15 | 14 | 43 |
| IL-1 α | 13 | 15 | 40 |
| IFN -α | 10 | 16 | 19 |
| IL-15 | 4 | 15 | 7 |
| IL-4 | 16 | 17 | 0 |
| IL-13 | 26 | 17 | 0 |
| IL-17 | 31 | 17 | 0 |
| IL-18 | 26 | 17 | 0 |
| IFN-β | 250 | 17 | 0 |
| Growth Factor | ELISA sensitivity (pg/mL) | Number of cell lines tested | Percentage of cell lines secreting growth factor |
|---|---|---|---|
| TGF-β | 31 | 25 | 100 |
| VEGF | 31 | 22 | 95 |
| PDGF-AA | 31 | 21 | 86 |
| PlGF | 16 | 22 | 59 |
| PDGF-AB | 31 | 23 | 52 |
| TGF-α | 16 | 21 | 52 |
| FGF-b | 10 | 22 | 45 |
| IGF-1 | 94 | 21 | 33 |
| EGF | 4 | 22 | 9 |
| PDGF-BB | 31 | 25 | 0 |
2.2. Heterogeneity of Factors
| Patient number | Source of tumor | Date collected | PDGF-AA mean pg/mL (2 SD) | OPN mean pg/mL (2 SD) | PlGF mean pg/mL (2 SD) |
|---|---|---|---|---|---|
| 1 | Lymph node | 14 April 2005 | 0 | 3533 (5801) | 0 |
| Soft tissue | 8 June 2005 | 0 | 2467 (1617) | 95 (139) | |
| Lymph node | 8 June 2005 | 1245 (1414) | 100 (346) | 32 (19) | |
| 2 | Lymph node, Deep groin | 1 September 2006 | 4783 (685) | 60763 (11127) | 4387 (455) |
| Lymph node, Superficial groin | 1 September 2006 | 680 (311) | 467 (115) | 0 |
2.3. Immunohistochemistry Study
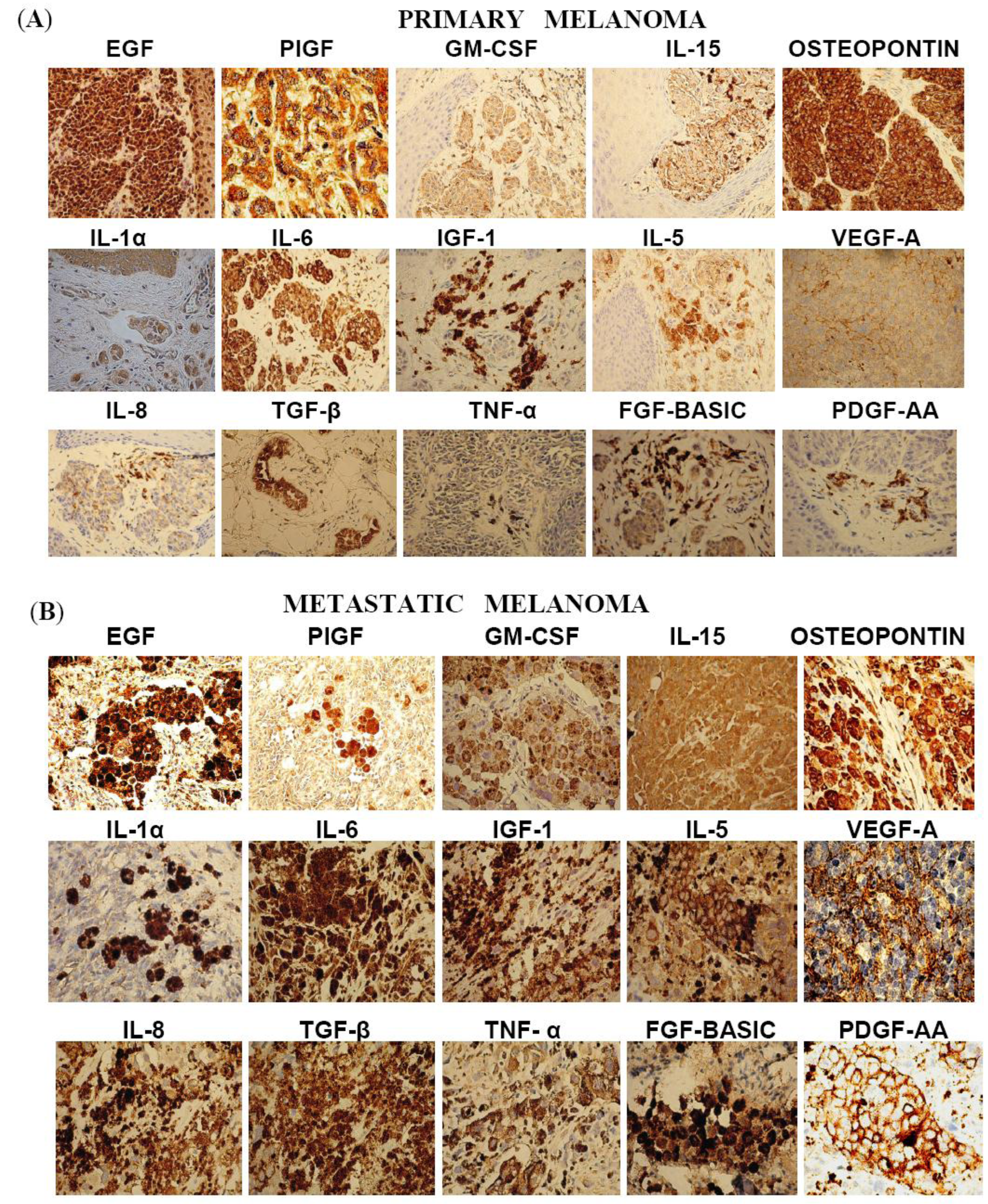
| Cytokines/Growth Factors | Primary Melanoma (n = 12) | Distant Metastasis (n = 19) | Fisher’s Exact Test P-value |
|---|---|---|---|
| EGF | 12 (100%) | 14 (74%) | NS |
| PlGF | 6 (50%) | 7 (37%) | NS |
| GM-CSF | 4 (33%) | 2 (11%) | NS |
| IL-15 | 5 (42%) | 3 (16%) | NS |
| Osteopontin | 12 (100%) | 15 (79%) | NS |
| IL-1α | 8 (66%) | 15 (79%) | NS |
| IL-6 | 7 (58%) | 12 (63%) | NS |
| IGF-1 | 5 (42%) | 8 (42%) | NS |
| IL-5 | 7 (58%) | 14 (74%) | NS |
| VEGF-A | 2 (17%) | 4 (21%) | NS |
| IL-8 | 7 (58%) | 4 (21%) | NS |
| TGF-β | 11 (92%) | 3 (16%) | <0.001 |
| TNF-α | 2 (17%) | 8 (42%) | NS |
| FGF-Basic | 8 (67%) | 9 (47%) | NS |
| PDGF-AA | 1 (8%) | 8 (42%) | NS |
| Cytokines/Growth Factors | Primary Melanoma (n = 12) | Distant Metastasis (n = 19) | Fisher’s Exact Test P-value |
|---|---|---|---|
| EGF | 12 (100%) | 19 (100%) | NS |
| PlGF | 11 (91%) | 18 (94%) | NS |
| GM-CSF | 9 (75%) | 12 (63%) | NS |
| IL-15 | 9 (75%) | 5 (26%) | <0.05 |
| Osteopontin | 9 (75%) | 9 (47%) | NS |
| IL-1α | 8 (66%) | 11 (57%) | NS |
| IL-6 | 8 (66%) | 8 (42%) | NS |
| IGF-1 | 7 (58%) | 1 (5%) | <0.01 |
| IL-5 | 7 (58%) | 9 (47%) | NS |
| VEGF-A | 3 (25%) | 3 (15%) | NS |
| IL-8 | 4 (33%) | 10 (52%) | NS |
| TGF-β | 1 (8%) | 5 (26%) | NS |
| TNF-α | 10 (83%) | 11 (57%) | NS |
| FGF-Basic | 10 (83%) | 8 (42%) | <0.05 |
| PDGF-AA | 6 (50%) | 8 (42%) | NS |
3. Discussion
3.1. Duality of Effects
3.2. Cytokines
3.2.1. Interleukins
3.2.2. Other Cytokines
3.3. The Growth Factors
3.4. Comparison of Factors from Cell Lines and Tissues
3.5. Clinical Considerations
4. Experimental Section
4.1. Establishment and Characterization of Human Melanoma Cell Lines
| Antibody | Clone | Intracellular/Surface | Source |
|---|---|---|---|
| Anti-MART-1 | M2-7C10 | Intracellular | Lab Vision NeoMarkers MS-612P |
| Anti-gp100 | HMB45 | Intracellular | Invitrogen 18-2050 |
| Anti-TRP75 | TA99 | Intracellular | Lab Vision NeoMarkets MS1634P |
| MCSP-APC | EP-1 | Surface | Miltenyi Biotec 130-091-252 |
4.2. Immunohistochemistry Studies
| Antibody | Type of antibody | Catalog # | Vendor |
|---|---|---|---|
| IL-1α | Monoclonal | MAB 200 | R&D Systems, Minneapolis, MN |
| IL-5 | Monoclonal | MAB-605 | R&D Systems, Minneapolis, MN |
| IL-6 | Monoclonal | NCL-IL-6 | Nova Castra, New Castle upon Tyne, UK |
| IL-8 | Polyclonal | AHC-0881 | Biosource, Camarillo, CA |
| IL-15 | Monoclonal | MAB 647 | R&D Systems, Minneapolis, MN |
| GM-CSF | Monoclonal | MAB 215 | R&D Systems, Minneapolis, MN |
| VEGF-A | Monoclonal | MAB-293 | R&D Systems, Minneapolis, MN |
| PDGF-AA | Polyclonal | AF-221NA | R&D Systems, Minneapolis, MN |
| Osteopontin | Monoclonal | NCL-OPNTIN | Nova Castra, New Castle Upon Tyne, UK |
| IGF-1 | Polyclonal | AF-291NA | R&D Systems, Minneapolis, MN |
| PlGF | Monoclonal | P-3863 | Sigma, St Louis, MO |
| TGF-β | Monoclonal | NCL-TGF-B | NovaCastra, New Castle Upon tyne, UK |
| FGF-basic | Polyclonal | AB-233-NA | R&D Systems, Minneapolis, MN |
| EGF | Monoclonal | E-2520 | Sigma, St Louis, MO |
| TGF-alpha | Polyclonal | AF-239-NA | R&D Systems, Minneapolis, MN |
5. Conclusions
Acknowledgements
Abbreviations
| IL | interleukin |
| TNF-α | tumor necrosis factor-alpha |
| IFN | interferon |
| TGF | transforming growth factor |
| VEGF | vascular endothelial growth factor |
| PDGF | platelet derived growth factor |
| PlGF | placenta growth factor |
| FGF | fibroblast growth factor |
| IGF | insulin-like growth factor |
| EGF | epidermal growth factor |
| OPN | osteopontin |
References
- Krasagakis, K.; Garbe, C.; Schrier, P.I.; Orfanos, C.E. Paracrine and autocrine regulation of human melanocyte and melanoma cell growth by transforming growth factor beta in vitro. Anticancer Res. 1994, 14, 2565–2571. [Google Scholar]
- Lazar-Molnar, E.; Hegyesi, H.; Toth, S.; Falus, A. Autocrine and paracrine regulation by cytokines and growth factors in melanoma. Cytokines 2000, 12, 547–554. [Google Scholar] [CrossRef]
- Richmond, A. The pathogenic role of growth factors in melanoma. Semin. Dermatol. 1991, 10, 246–255. [Google Scholar]
- Luan, J.; Shattuk-Brandt, R.; Haghnegahdar, H.; Owen, J.D.; Strieter, R.; Burdick, M.; Nirodi, C.; Beauchamp, D.; Johnson, K.N.; Richmond, A. Mechanism and biological significance of constitutive expression of MGSA/GRO chemokines in malignant melanoma tumor progression. J. Leukoc. Biol. 1997, 62, 588–597. [Google Scholar]
- Bracke, M.C.; van Roy, F.M.; Mareel, M.M. The E-cadherin/catenin complex in invasion and metastasis. Curr. Top. Microbiol. Immunol. 1996, 213, 123–161. [Google Scholar] [CrossRef]
- Selzer, E.; Schlagbauer-Wadl, H.; Okamoto, I.; Pehamberger, H.; Pötter, R.; Jansen, B. Expression of Bcl-2 family members in human melanocytes, in melanoma metastases and in melanoma cell lines. Melanoma Res. 1998, 8, 197–203. [Google Scholar] [CrossRef]
- Grover, R.; Wilson, G.D. Bcl-2 expression in malignant melanoma and its prognostic significance. Eur. J. Surg. Oncol. 1996, 22, 347–349. [Google Scholar] [CrossRef]
- Cruse, J.M.; Lewis, R.E.; Wang, H. Cytokines and chemokines. In Immunology Guidebook; Cruse, J., Lewis, R., Wang, H., Eds.; Elsevier Science & Technology Books: Amsterdam, The Netherlands, 2004; pp. 339–392. [Google Scholar]
- Rodeck, U. Growth factor independence and growth regulatory pathways in human melanoma development. Cancer Metastasis Rev. 1993, 12, 219–226. [Google Scholar] [CrossRef]
- Krasagakis, K.; Garbe, C.; Orfanos, C.E. Cytokines in human melanoma cells: synthesis, autocrine stimulation and regulatory functions-an overview. Melanoma Res. 1993, 3, 425–433. [Google Scholar] [CrossRef]
- Gray-Schopfer, V.; Wellbrock, C.; Marias, R. Melanoma biology and new targeted therapy. Nature 2007, 445, 851–867. [Google Scholar] [CrossRef]
- Kitadai, Y.; Haruma, K.; Sumii, K.; Yamamoto, S.; Ue, T.; Yokozaki, H.; Yasui, W.; Ohmoto, Y.; Kajiyama, G.; Fidler, I.J.; et al. Expression of interleukin-8 correlates with vascularity of human gastric carcinoma. Am. J. Path. 1998, 152, 93–100. [Google Scholar]
- Leong, S.R.; Lowman, H.B.; Liu, J.; Shire, S.; Deforge, L.E.; Gillece-Castro, B.L.; McDowell, R.; Hébert, C.A. IL-8 single chain homodimers and heterodimers: interactions with chemokine receptors CXCR-1, CXCR-2 and DARC. Protein Sci. 1997, 6, 609–617. [Google Scholar]
- Gabellini, C.; Trisciuoglio, D.; Desideri, M.; Candiloro, A.; Ragazzoni, Y.; Orlandi, A.; Zupi, G.; Del Bufalo, D. Functional activity of CXCL8 receptors, CXCR1 and CXCR2, on human malignant melanoma progression. Eur. J. Cancer 2009, 45, 2618–2627. [Google Scholar] [CrossRef]
- Varney, M.L.; Johansson, S.L.; Singh, R.K. Distinct expression of CXCL8 and its receptors CXCR1 and CXCR2 and their association with vessel density and aggressiveness in malignant melanoma. Am. J. Clin. Pathol. 2006, 125, 209–216. [Google Scholar]
- Singh, S.; Sadanandam, A.; Nannuru, KC.; Varney, M.L.; Mayer-Ezell, R.; Bond, R.; Singh, R.K. Small-molecule antagonists for CXCR2 and CXCR1 inhibit human melanoma growth by decreasing tumor cell proliferation, survival and angiogenesis. Clin. Cancer Res. 2009, 15, 2380–2386. [Google Scholar] [CrossRef]
- Akira, S.; Taga, T.; Kishimoto, T. Interleukin-6 in biology and medicine. Adv.Immunol. 1993, 54, 1–78. [Google Scholar] [CrossRef]
- Tartour, E.; Dorval, T.; Mosseri, V.; Deneux, L.; Mathiot, C.; Brailly, H.; Montero, F.; Joyeux, I.; Pouillart, P.; Fridman, W.H. Serum interleukin 6 and C-reactive protein levels correlate with resistance to IL-2 therapy and poor survival in melanoma patients. Br. J. Cancer 1994, 69, 911–913. [Google Scholar] [CrossRef]
- Pestka, S.; Krause, C.D.; Sarkar, D.; Walter, M.R.; Shi, Y.; Fisher, P.B. Interleukin-10 and related cytokines and receptors. Ann. Rev. Immunol. 2004, 22, 929–979. [Google Scholar] [CrossRef]
- Dummer, W.; Becker, J.C.; Schwaaf, A.; Leverkus, M.; Moll, T.; Bröcker, E.B. Elevated serum level of interleukin-10 in patients with metastatic malignant melanoma. Melanoma Res. 1995, 5, 67–68. [Google Scholar] [CrossRef]
- Le Feuvre, R.A.; Brough, D.; Iwakura, Y.; Takeda, K.; Rothwell, N.J. Priming of macrophages with lip-poly saccharide potentiates P2X7-mediated cell death via caspase-1 dependant mechanism independently of cytokine production. J. Biol. Chem. 2002, 277, 3210–3718. [Google Scholar]
- Tyler, D.S.; Francis, G.M.; Fredrick, M.; Tran, A.H.; Ordóñez, N.G.; Smith, J.L.; Eton, O.; Ross, M.; Grimm, E.A. Interleukin-1 production in tumor cells of human melanoma surgical specimens. J. Interferon Cytokine Res. 1995, 15, 331–340. [Google Scholar] [CrossRef]
- Fehniger, T.A.; Caligiuri, M.A. Interleukin-15: biology and relevance to human disease. Blood 2001, 97, 14–32. [Google Scholar] [CrossRef]
- Carson, W.E.; Giri, J.G.; Lindemann, M.J.; Linett, M.L.; Ahdieh, M.; Paxton, R.; Anderson, D.; Eisenmann, J.; Grabstein, K.; Caligiuri, M.A. Interleukin (IL) 15 is a novel cytokine that activates natural killer cells via components of the IL-2 receptor. J. Exp. Med. 1994, 180, 1395–1403. [Google Scholar] [CrossRef]
- Huarte, E.; Fisher, J.; Turk, M.J. ; Mellinger, D.; Foster, C.; Wolf, B.; Meehan, K.R.; Fadul, C.E.; Ernstoff, M.S. Ex vivo expansion of tumor specific lymphocytes with IL-15 and IL-21 for adoptive immunotherapy in melanoma. Cancer Lett. 2009, 285, 80–88. [Google Scholar] [CrossRef]
- Doucet, C.; Meazza, R.; Pottin-Clemenceau, C.; Scudeletti, M.; Brouty-Boye, D.; Ferrini, S.; Alileche, A.; Taoufik, Y.; Jasmin, C.; Azzarone, B.; Indiveri, F. Role of interleukin (IL)-2 and IL-15 in the tumour progression of a melanoma cell line MELP, derived from an IL-2 progressor patient. Melanoma Res. 1997, 7 (Suppl. 2), S7–S17. [Google Scholar]
- Murugaiyan, G.; Saha, B. Protumor vs antitumor functions of IL-17. J. Immunol. 2009, 183, 4169–4175. [Google Scholar] [CrossRef]
- Von Euw, E.; Chodon, T.; Attar, N.; Jalil, J.; Koya, R.C.; Comin-Anduix, B.; Ribas, A. CTLA4 blockade increases Th17 cells in patients with metastatic melanoma. J. Transl. Med. 2009, 7, 35–47. [Google Scholar] [CrossRef]
- Park, H.; Byun, D.; Kim, T.S.; Kim, Y.I.; Kang, J.S.; Hahm, E.S.; Kim, S.H.; Lee, W.J.; Song, H.K.; Yoon, D.Y.; et al. nhanced IL-18 expression in common skin tumors. Immunol. Lett. 2001, 79, 215–219. [Google Scholar] [CrossRef]
- Hacker, E.; Muller, K.; Whiteman, D.C.; Pavey, S.; Hayward, N.; Walker, G. Reduced expression of IL-18 is a marker of ultraviolet radiation-induced melanomas. Int. J. Cancer 2008, 123, 227–231. [Google Scholar] [CrossRef] [Green Version]
- Johnson, N.I.; Gunasekharan, V.K.; Ravindranath, A.; O'Connell, C.; Johnston, P.G.; El-Tanani, M.K. Osteopontin as a target for cancer therapy. Frontiers Biosc. 2008, 13, 4361–4372. [Google Scholar]
- Baldwin, A.S. Control of oncogenesis and cancer therapy resistance by transcription factor NF-κB. J. Clin. Invest. 2001, 107, 241–246. [Google Scholar] [CrossRef]
- Hayes, A.J.; Neuhaus, S.J.; Clark, M.A.; Thomas, J.M. Isolated limb perfusion with melphalan and tumor necrosis factor α for advanced melanoma and soft-tissue sarcoma. Ann. Surg. Oncol. 2007, 14, 230–238. [Google Scholar]
- Manna, S.K.; Mukhopadhyay, A.; Aggarwal, B.B. IFN-alpha suppresses activation of nuclear transcription factors NF-Kappa and activation protein 1 and protentiates TNF-induced apoptosis. J. Immunol. 2000, 165, 4927–4934. [Google Scholar]
- Moschos, S.J.; Lesinski, G.B.; Carson, W.E., III; Kirkwood, J.M. The type1 interferon system with emphasis on its role in malignancies. In Cytokines in the Genesis and Treatment of Cancer; Caligiuri, M.A., Lotze, M.T., Eds.; Human Press: Totowa, NJ, USA, 2007; pp. 339–372. [Google Scholar]
- Rossi, C.R.; Russano, F.; Mocellin, S.; Chiarion-Sileni, V.; Foletto, M.; Pilati, P.; Campana, L.G.; Zanon, A.; Picchi, G.F.; Lise, M.; Nitti, D. TNF-based isolated limb perfusion followed by consolidation biotherapy with systemic low-dose interferon alpha 2b in patients with in-transit melanoma metastases: a pilot trial. Ann. Surg. Oncol. 2008, 15, 1218–1223. [Google Scholar] [CrossRef]
- Huang, A.Y.; Golumbek, P.; Ahmadzadeh, M.; Jaffee, E.; Pardoll, D.; Levitsky, H. Role of bone marrow-derived cells in presenting MHC class-1 restricted tumor antigens. Science 1994, 264, 961–965. [Google Scholar]
- Javelaud, D.; Alexaki, V.; Mauviel, A. Transforming growth factor-β in cutaneous melanoma. Pigment Cell Melanoma Res. 2008, 21, 123–132. [Google Scholar] [CrossRef]
- Grunstein, J.; Masbad, J.J.; Hickley, R.; Giordano, F.; Johnson, R.S. Isoforms of vascular epithelial factor act in coordinated fashion to recruit and expand tumor vasculature. Mol. Cell Biol. 2000, 20, 7282–7291. [Google Scholar] [CrossRef]
- Heldin, C.H.; Eriksson, U.; Ostman, A. New members of the platelet-derived growth factor family of mitogens. Arch. Biochem. Biophys. 2002, 398, 284–290. [Google Scholar] [CrossRef]
- Mitsiades, C.S.; Mitsiades, N.S.; McMullan, C.J.; Poulaki, V.; Shringarpure, R.; Akiyama, M.; Hideshima, T.; Chauhan, D.; Joseph, M.; Libermann, T.A.; et al. Inhibition of the insulin-like growth-1 tyrosine kinase activity as a therapeutic strategy for multiple myeloma, other hematologic malignancies and solid tumors. Cancer Cell 2004, 5, 221–230. [Google Scholar] [CrossRef]
- Hilmi, C.; Larribere, L.; Giuliano, S.; Bille, K.; Ortonne, J.; Ballotti, R.; Bertolotto, C. IGF1 promotes resistance to apoptosis in melanoma cells through an increased expression of BCL2, BCL-X(L) and survivin. J. Invest. Dermatol. 2008, 128, 1499–1505. [Google Scholar] [CrossRef]
- Marcellini, M.; De Luca, N.; Riccioni, T.; Ciucci, A.; Orecchia, A.; Lacal, P.M.; Ruffini, F.; Pesce, M.; Cianfaarani, F.; Zambruno, G.; et al. Increased melanoma growth and metastasis spreading in mice overexpressing placenta growth factor. Am. J. Path. 2006, 169, 643–654. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. The hallmark of cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef]
- Souza, R.F. Molecular and biologic basis of upper gastrointestinal malignancy-esophageal carcinoma. Sur. Oncol. Clin. N. Am. 2002, 11, 257–272. [Google Scholar] [CrossRef]
- Dow, J.K.; de Vere-White, R.W. Fibroblast growth factor 2, its structure and property paracrine function, tumor angiogenesis and prostate-related mitogenic functions. Urology 2000, 55, 800–806. [Google Scholar] [CrossRef]
- Folkman, J. Angiogenesis in cancer, vascular, rheumatoid and other diseases. Nat. Med. 1995, 1, 27–31. [Google Scholar] [CrossRef]
- Kumar, R.; Yoneda, J.; Bucana, C.D.; Fidler, I.J. Regulation of distinct steps of angiogenesis by different angiogenic molecules. Int. J. Oncol. 1998, 12, 749–757. [Google Scholar]
- Nicholson, R.I.; Gee, J.M.; Harper, M.E. EGFR and cancer prognosis. Eur. J. Cancer 2001, 37 (Suppl. 4), S9–S15. [Google Scholar] [CrossRef]
- Rakosy, Z.; Vizkeleti, L.; Ecsedi, S.; Voko, Z.; Begany, A.; Barok, M.; Krekk, Z.; Gallai, M.; Szentirmay, Z.; Adany, R.; Balazs, M. EGFR gene copy number alterations in primary cutaneous malignant melanoma are associated with poor prognosis. Int. J. Cancer 2007, 121, 1729–1737. [Google Scholar] [CrossRef]
- Schicher, N.; Paulitschke, V.; Swoboda, A.; Kunstfeld, R.; Loewe, R.; Pilarski, P.; Pehamberger, H.; Hoeller, C. Erlotinib and bevacizumab have synergistic activity against melanoma. Clin. Cancer Res. 2009, 15, 3495–3502. [Google Scholar] [CrossRef]
- Smits, A.; Funa, K.; Vassbotn, F.S.; Beausang-Linder, M.; af Ekenstam, F.; Heldin, C.H.; Westermark, B.; Nistér, M. Expression of platelet-derived growth factor and its receptor in proliferative disorders of fibroblastic origin. Am. J. Path. 1992, 140, 639–648. [Google Scholar]
- Ohlsson, R.; Falck, P.; Hellstrom, M.; Lindahl, P.; Boström, H.; Franklin, G.; Ahrlund-Richter, L.; Pollard, J.; Soriano, P.; Betsholtz, C. PDGFB regulates the development of the labyrinthine layer of the mouse fetal placenta. Dev. Biol. 1999, 212, 124–136. [Google Scholar] [CrossRef]
- Serafini, P.; Carbley, R.; Noonan, K.A.; Tan, G.; Bronte, V.; Borrello, I. High-dose granulocyte-macrophage colony- stimulating factor-producing vaccines impair the immune response through the recruitment of myeloid suppressor cells. Cancer Res. 2004, 64, 6337–6343. [Google Scholar] [CrossRef]
- Filipazzi, P.; Valenti, R.; Huber, V.; Pilla, L.; Canese, P.; Iero, M.; Castelli, C.; Mariani, L.; Parmiani, G.; Rivoltini, L. Identification of a new subset of myeloid suppressor cells in the peripheral blood of melanoma patients with modulation by granulocyte-macrophage colony-stimulating factor-based antitumor vaccine. J. Clin. Oncol. 2007, 25, 2546–2553. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Elias, E.G.; Hasskamp, J.H.; Sharma, B.K. Cytokines and Growth Factors Expressed by Human Cutaneous Melanoma. Cancers 2010, 2, 794-808. https://doi.org/10.3390/cancers2020794
Elias EG, Hasskamp JH, Sharma BK. Cytokines and Growth Factors Expressed by Human Cutaneous Melanoma. Cancers. 2010; 2(2):794-808. https://doi.org/10.3390/cancers2020794
Chicago/Turabian StyleElias, Elias G., Joanne H. Hasskamp, and Bhuvnesh K. Sharma. 2010. "Cytokines and Growth Factors Expressed by Human Cutaneous Melanoma" Cancers 2, no. 2: 794-808. https://doi.org/10.3390/cancers2020794




