Nano and Microsensors for Mammalian Cell Studies
Abstract
:1. Introduction
2. Nano and Micro Cantilever Beam Temperature Sensor for Biological Applications
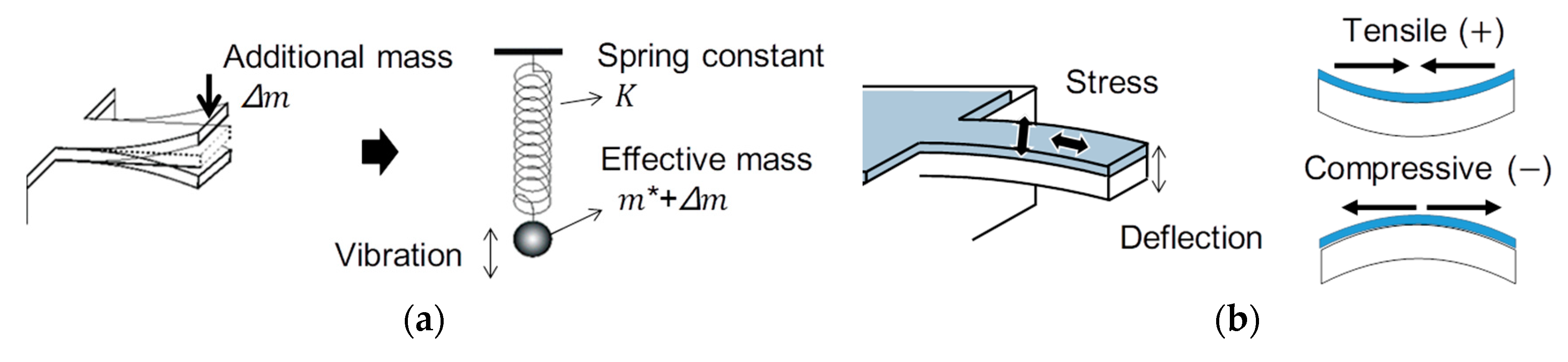
2.1. Principle of Operation of the Cantilever Beam Temperature Sensor
2.2. Bimaterial Cantilever Beam Temperature Sensor
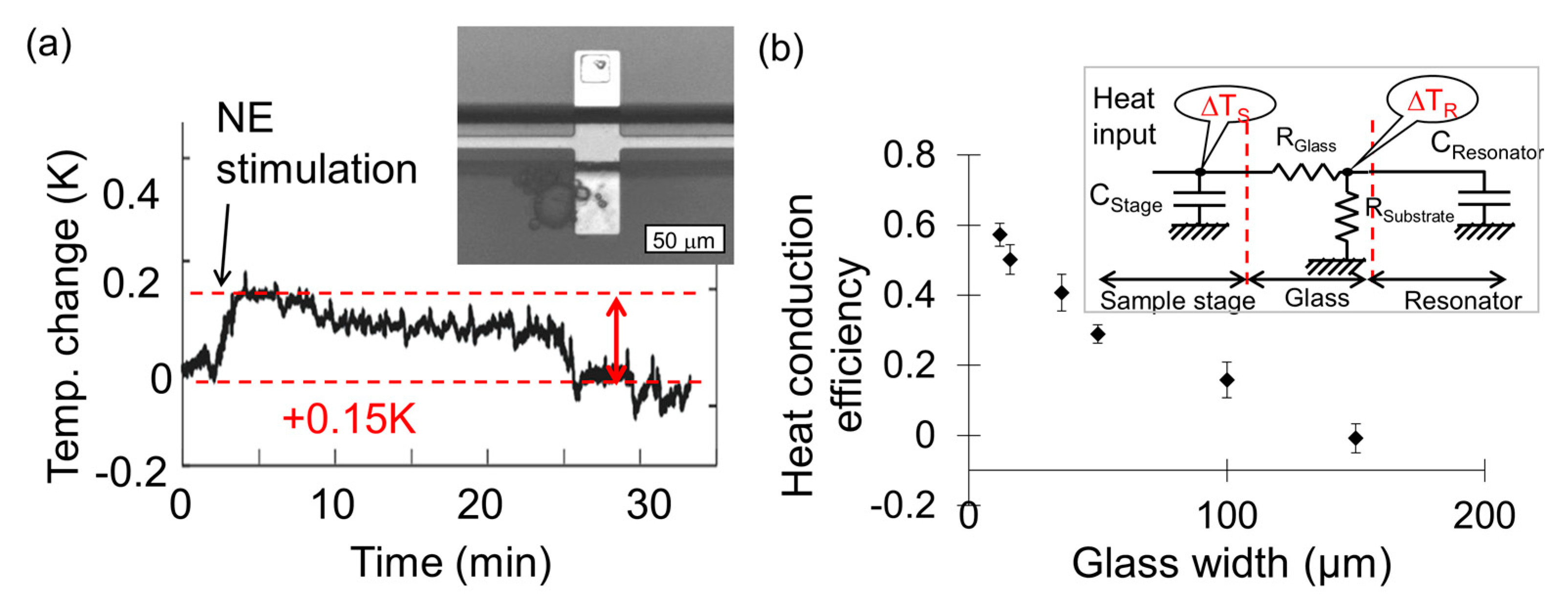
2.3. Resonant Cantilever Beam Temperature Sensor
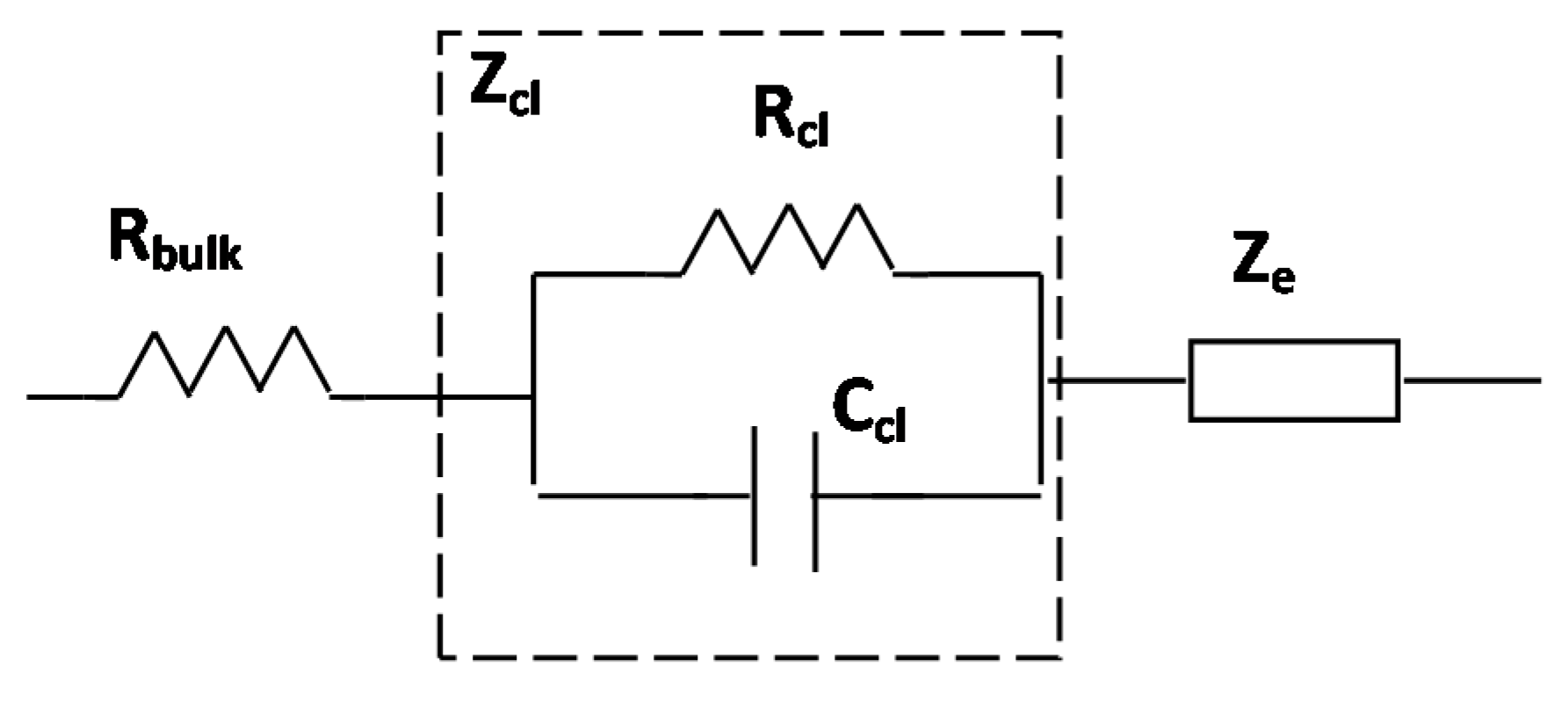
3. Electric Cell-Substrate Impedance Sensing (ECIS) Sensor for Mammalian Cells Applications
3.1. Electric Cell-Substrate Impedance Sensing (ECIS) Technique
3.2. Combination of ECIS Sensor with Quartz Crystal Microbalance (QCM) Resonator for Testing Water Toxicity
3.3. Stretchable ECIS Sensor for Monitoring Cell Proliferation
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Turner, A.; Wilson, G.; Kaube, I. Biosensors: Fundamentals and Applications; Oxford University Press: Oxford, UK, 1987; p. 770. ISBN 0198547242. [Google Scholar]
- Urban, G.A. Micro- and nanobiosensors—State of the art and trends. Meas. Sci. Technol. 2009, 20, 012001. [Google Scholar] [CrossRef]
- Bănică, F.G.; Fogg, A.G. Chemical Sensors and Biosensors: Fundamentals and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2012; pp. 590–595. [Google Scholar]
- Crivianu-Gaita, V.; Thompson, M. Aptamers, antibody scFv, and antibody Fab’ fragments: An overview and comparison of three of the most versatile biosensor biorecognition elements. Biosens. Bioelectron. 2016, 85, 32–45. [Google Scholar] [CrossRef] [PubMed]
- Skrlec, K.; Strukelj, B.; Berlec, A. Non-immunoglobulin scaffolds: A focus on their targets. Trends Biotechnol. 2015, 33, 408–418. [Google Scholar] [CrossRef] [PubMed]
- Jost, C.; Plückthun, A. Engineered proteins with desired specificity: DARPins, other alternative scaffolds and bispecific IgGs. Curr. Opin. Struct. Biol. 2014, 27, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Brient-Litzler, E.; Plückthun, A.; Bedouelle, H. Knowledge-based design of reagentless fluorescent biosensors from a designed ankyrin repeat protein. Protein Eng. Des. Sel. 2010, 23, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Miranda, F.F.; Brient-Litzler, E.; Zidane, N.; Pecorari, F.; Bedouelle, H. Reagentless fluorescent biosensors from artificial families of antigen binding proteins. Biosens. Bioelectron. 2011, 26, 4184–4190. [Google Scholar] [CrossRef] [PubMed]
- Schultz, J.S.; Mansouri, S.; Goldstein, I.J. Affinity sensor: A new technique for developing implantable sensors for glucose and other metabolites. Diabetes Care 1982, 5, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Schultz, J.S.; Sims, G. Affinity sensors for individual metabolites. Biotechnol. Bioeng. Symp. 1979, 9, 65–71. [Google Scholar]
- Ballerstadt, R.; Schultz, J.S. A fluorescence affinity hollow fiber sensor for continuous transdermal glucose monitoring. Anal. Chem. 2000, 72, 4185–4192. [Google Scholar] [CrossRef] [PubMed]
- Vollmer, F.; Yang, L. Label-free detection with high-Q microcavities: A review of biosensing mechanisms for integrated devices. Nanophotonics 2012, 1, 267–291. [Google Scholar] [CrossRef] [PubMed]
- Rizzuto, R.; Pinton, P.; Brini, M.; Chiesa, A.; Filippin, L.; Pozzan, T. Mitochondria as biosensors of calcium microdomains. Cell Calcium 1999, 26, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Bragadin, M.; Manente, S.; Piazza, R.; Scutari, G. The mitochondria as biosensors for the monitoring of detergent compounds in solution. Anal. Biochem. 2001, 292, 305–307. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Chaudhury, N.K. Entrapment of biomolecules in sol-gel matrix for applications in biosensors: Problems and future prospects. Biosens. Bioelectron. 2007, 22, 2387–2399. [Google Scholar] [CrossRef] [PubMed]
- Juzgado, A.; Solda, A.; Ostric, A.; Criado, A.; Valenti, G.; Rapino, S.; Conti, G.; Fracasso, G.; Paolucci, F.; Prato, M. Highly sensitive electrochemiluminescence detection of a prostate cancer biomarker. J. Mater. Chem. B 2017, 5, 6681. [Google Scholar] [CrossRef]
- Valenti, G.; Rampazzo, E.; Biavardi, E.; Villani, E.; Fracasso, G.; Marcaccio, M.; Bertani, F.; Ramarli, D.; Dalcanale, E.; Paolucci, F.; et al. An electrochemiluminescence-supramolecular approach to sarcosine detection for early diagnosis of prostate cancer. Faraday Discuss. 2015, 185, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Forster, R.J.; Bertoncello, P.; Keyes, T.E. Electrogenerated chemiluminescence. Ann. Rev. Anal. Chem. 2009, 2, 359–385. [Google Scholar] [CrossRef] [PubMed]
- Valenti, G.; Fiorani, A.; Li, H.; Sojic, N.; Paolucci, F. Essential role of electrode materials in electrochemiluminescence applications. Chem. Electron. Chem. 2016, 3, 1990–1997. [Google Scholar] [CrossRef]
- Donzella, V.; Crea, F. Optical biosensors to analyze novel biomarkers in oncology. J. Biophotonics 2011, 4, 442–452. [Google Scholar] [CrossRef] [PubMed]
- Hiep, H.M.; Endo, T.; Kerman, K.; Chikae, M.; Kim, D.-K.; Yamamura, S.; Takamura, Y.; Tamiya, E. A localized surface plasmon resonance based immunosensor for the detection of casein in milk. Sci. Technol. Adv. Mater. 2007, 8, 331–338. [Google Scholar] [CrossRef] [Green Version]
- Kotanen, C.N.; Gabriel Moussy, F.; Carrara, S.; Guiseppi-Elie, A. Implantable enzyme amperometric biosensors. Biosens. Bioelectron. 2012, 35, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Lambrianou, A.; Demin, S.; Hall, E.A. Protein engineering and electrochemical biosensors. Adv. Biochem Eng. Biotechnol. 2008, 109, 65–96. [Google Scholar] [CrossRef] [PubMed]
- Renard, M.; Belkadi, L.; Hugo, N.; England, P.; Altschuh, D.; Bedouelle, H. Knowledge-based design of reagentless fluorescent biosensors from recombinant antibodies. J. Mol. Biol. 2002, 318, 429–442. [Google Scholar] [CrossRef]
- Mortellaro, M.; DeHennis, A. Performance characterization of an abiotic and fluorescent-based continuous glucose monitoring system in patients with type 1 diabetes. Biosens. Bioelectron. 2014, 61, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Atay, S.; Pişkin, K.; Yılmaz, F.; Çakır, C.; Yavuz, H.; Denizli, A. Quartz crystal microbalance based biosensors for detecting highly metastatic breast cancer cells via their transferrin receptors. Anal. Methods 2016, 8, 153–161. [Google Scholar] [CrossRef]
- Cavalcanti, A.; Shirinzadeh, B.; Zhang, M.; Kretly, L. C. Nanorobot hardware architecture for medical defense. Sensors 2008, 8, 2932–2958. [Google Scholar] [CrossRef] [PubMed]
- Hierlemann, A.; Brand, O.; Hagleitner, C.; Baltes, H. Microfabrication techniques for chemical/biosensors. Proc. IEEE 2003, 91, 839–863. [Google Scholar] [CrossRef]
- Hierlemann, A.; Baltes, H. CMOS-based chemical microsensors. Analyst 2003, 128, 15–28. [Google Scholar] [CrossRef] [PubMed]
- Quesada-González, D.; Merkoçi, A. Mobile phone-based biosensing: An emerging “diagnostic and communication” technology. Biosens. Bioelectron. 2016, 92, 549–562. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, L.; Wei, D.; Yap, Y.; Chen, F. Moving cancer diagnostics from bench to bedside. Trends Biotechnol. 2007, 25, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Quesada-González, D.; Merkoçi, A. Nanomaterial-based devices for point-of-care diagnostic applications. Chem. Soc. Rev. 2018, 47, 4697–4709. [Google Scholar] [CrossRef] [PubMed]
- Windmiller, J.R.; Wang, J. Wearable electrochemical sensors and biosensors: A review. Electroanalysis 2013, 25, 29–46. [Google Scholar] [CrossRef]
- Ajami, S.; Teimouri, F. Features and application of wearable biosensors in medical care. J. Res. Med. Sci. 2015, 20, 1208–1215. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.A.; Yeo, W.H.; Su, Y.; Hattori, Y.; Lee, W.; Jung, S.Y.; Zhang, Y.; Liu, Z.; Cheng, H.; Falgout, L.; et al. Fractal design concepts for stretchable electronics. Nat. Commun. 2014, 5, 3266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, S.; Zhang, Y.; Cho, J.; Lee, J.; Huang, X.; Jia, L.; Fan, J.A.; Su, Y.; Su, J.; Zhang, H.; et al. Stretchable batteries with self-similar serpentine interconnects and integrated wireless recharging systems. Nat. Commun. 2013, 4, 1543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, L.; Gutbrod, S.R.; Bonifas, A.P.; Su, Y.; Sulkin, M.S.; Lu, N.; Chung, H.J.; Jang, K.I.; Liu, Z.; Ying, M.; et al. 3D multifunctional integumentary membranes for spatiotemporal cardiac measurements and stimulation across the entire epicardium. Nat. Commun. 2014, 5, 3329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, D.H.; Lu, N.; Ma, R.; Kim, Y.S.; Kim, R.H.; Wang, S.; Wu, J.; Won, S.M.; Tao, H.; Islam, A.; et al. Epidermal electronics. Science 2011, 333, 838–843. [Google Scholar] [CrossRef] [PubMed]
- Son, D.; Lee, J.; Qiao, S.; Ghaffari, R.; Kim, J.; Lee, J.E.; Song, C.; Kim, S.J.; Lee, D.J.; Jun, S.W.; et al. Multifunctional wearable devices for diagnosis and therapy of movement disorders. Nat. Nanotechnol. 2014, 9, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Rogers, J.A.; Balooch, G. Biomedical materials: A restorative synthetic skin. Nat. Mater. 2016, 15, 828–829. [Google Scholar] [CrossRef] [PubMed]
- Gaio, N.; van Meer, B.; Solano, W.Q.; Bergers, L.; van de Stolpe, A.; Mummery, C.; Sarro, P.M.; Dekker, R. Cytostretch, an organ-on-chip Platform. Micromachines 2016, 7, 120. [Google Scholar] [CrossRef]
- Pakazad, S.K.; Savov, A.; van de Stolpe, A.; Dekker, R. A novel stretchable micro-electrode array (SMEA) design for directional stretching of cells. J. Micromech. Microeng. 2014, 24, 034003. [Google Scholar] [CrossRef]
- Liu, Y.; Jin, Z.; Liu, Y.; Hu, X.; et al. Stretchable electrochemical sensor for real-time monitoring of cells and tissues. Angew. Chem. Int. Ed. Engl. 2016, 55, 4537–4541. [Google Scholar] [CrossRef] [PubMed]
- Bernardeschi, I.; Greco, F.; Ciofani, G.; Marino, A.; Mattoli, V.; Mazzolai, B.; Beccai, L. A soft, stretchable and conductive biointerface for cell mechanobiology. Biomed. Microdevices 2015, 17, 46. [Google Scholar] [CrossRef] [PubMed]
- Raiteri, R.; Grattarola, M.; Butt, H.-J.; Skládal, P. Micromechanical cantilever-based biosensors. Sens. Actuators B 2001, 79, 115–126. [Google Scholar] [CrossRef]
- Lavrik, N.V.; Sepaniak, M.J.; Datskos, P.G. Cantilever transducers as a platform for chemical and biological sensors. Rev. Sci. Instrum. 2004, 75, 2229–2253. [Google Scholar] [CrossRef]
- Lang, H.P.; Hegner, M.; Meyer, E.; Gerber, C. Cantilever array sensors. Mater. Today 2005, 8, 30–36. [Google Scholar] [CrossRef]
- Sushko, M.L.; Harding, J.H.; Shluger, A.L.; McKendry, R.A.; Watari, M. Physics of nanomechanical biosensing on cantilever arrays. Adv. Mater. 2008, 20, 3848–3853. [Google Scholar] [CrossRef]
- Grovera, W.H.; Bryana, A.K.; Diez-Silvac, M.; Sureshb, S.; Higgins, J.M.; Manalis, S.R. Measuring single-cell density. Proc. Natl. Acad. Sci. USA 2011, 108, 10992–10996. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sato, M.K.; Toda, M.; Inomata, N.; Maruyama, H.; Okamatsu-Ogura, Y.; Arai, F.; Ono, T.; Ishijima, A.; Inoue, Y. Temperature changes in brown adipocytes detected with a bimaterial microcantilever. Biophys. J. 2014, 106, 2458–2464. [Google Scholar] [CrossRef] [PubMed]
- Voiculescu, I.; Liu, F.; Ono, T.; Toda, M. Investigation of bimaterial cantilever beam for heat sensing in liquid. Sens. Actuators A 2016, 242, 58–66. [Google Scholar] [CrossRef] [Green Version]
- Toda, M.; Ono, T.; Liu, F.; Voiculescu, I. Evaluation of bimaterial cantilever beam for heat sensing at atmospheric pressure. Rev. Sci. Instrum. 2010, 81, 055104. [Google Scholar] [CrossRef] [PubMed]
- Voiculescu, I.; Toda, M.; Ono, T.; Liu, F. Bimaterial nanocantilever beam calorimeter for atmospheric pressure and liquid applications. In Nanocantilever Beam, Modeling, Fabrication and Applications; Pan Stanford: Singapore, 2016; pp. 411–429. ISBN 9789814613231. [Google Scholar]
- Inomata, N.; Toda, M.; Sato, M.; Ishijima, A.; Ono, T. Pico calorimeter for detection of heat produced in an individual brown fat cell. Appl. Phys. Lett. 2012, 100, 154104. [Google Scholar] [CrossRef]
- Inomata, N.; Toda, M.; Ono, T. Highly sensitive thermometer using a vacuum-packed Si resonator in a microfluidic chip for the thermal measurement of single cells. Lab Chip 2016, 16, 3597–3603. [Google Scholar] [CrossRef] [PubMed]
- Barnes, J.R.; Stephenson, R.J.; Woodburn, C.N.; O’Shea, S.J.; Welland, M.E.; Rayment, T.; Gimzewski, J.K.; Gerber, C. A femtojoule calorimeter using micromechanical sensors. Rev. Sci. Instrum. 1994, 65, 3793–3798. [Google Scholar] [CrossRef]
- Barnes, J.R.; Stephenson, R.J.; Welland, M.E.; Gerber, Ch.; Gimzewski, J.K. Photothermal spectroscopy with femtojoule sensitivity using a micromechanical device. Nature 1994, 372, 79–81. [Google Scholar] [CrossRef]
- Nelson, I.C.; Banerjee, D.; Rogers, W.J.; Mannan, M.S. Detection of explosives using heated microcantilever sensors. In Micro (MEMS) and Nanotechnologies for Space Applications, Proceedings of the Defense and Security Symposium, Orlando, FL, USA, 17–21 April 2006; George, T., Cheng, Z.-Y., Eds.; SPIE: Bellingham, WA, USA, 2006. [Google Scholar]
- Oden, P.I.; Datskos, P.G.; Thundat, T.; Warmack, R.J. Uncooled thermal imaging using a piezoresistive microcantilever. Appl. Phys. Lett. 1996, 69, 3277–3279. [Google Scholar] [CrossRef]
- Datskos, P. G.; Rajic, S.; Senesac, L. R.; Datskou, I. Fabrication of quantum well microcantilever photon detectors. Ultramicroscopy 2001, 86, 191–206. [Google Scholar] [CrossRef]
- Datskos, P.G.; Oden, P.I.; Thundat, T.; Wachter, E.A.; Warmack, R.J.; Hunter, S.R. Remote infrared radiation detection using piezoresistive microcantilevers. Appl. Phys. Lett. 1996, 69, 2986–2988. [Google Scholar] [CrossRef]
- Perazzo, T.; Mao, M.; Kwon, O.; Majumdar, A.; Varesi, J.B.; Norton, P. Infrared vision using uncooled micro-optomechanical camera. Appl. Phys. Lett. 1999, 74, 3567. [Google Scholar] [CrossRef]
- Datskos, P.G.; Rajic, S.; Datskou, I. Photoinduced and thermal stress in silicon microcantilevers. Appl. Phys. Lett. 1998, 73, 2319–2321. [Google Scholar] [CrossRef]
- Lai, J.; Perazzo, T.; Shi, Z.; Majumdar, A. Optimization and performance of high-resolution micro-optomechanical thermal sensors. Sens. Actuators A 1997, 58, 113–119. [Google Scholar] [CrossRef]
- Senesac, L.R.; Corbeil, J.L.; Rajic, S.; Lavrik, N.V.; Datskos, P.G. IR imaging using uncooled microcantilever detectors. Ultramicroscopy 2003, 97, 451–458. [Google Scholar] [CrossRef]
- Varesi, J.; Lai, J.; Perazzo, T.; Shi, Z.; Majumdar, A. Photothermal measurements at picowatt resolution using uncooled micro-optomechanical sensors. Appl. Phys. Lett. 1997, 71, 306–308. [Google Scholar] [CrossRef]
- Arakawa, E.T.; Lavrik, N.V.; Rajic, S.; Datskos, P.G. Detection and differentiation of biological species using microcalorimetric spectroscopy. Ultramicroscopy 2003, 97, 459–465. [Google Scholar] [CrossRef]
- Arakawa, E.T.; Lavrik, N.V.; Datskos, P.G. Detection of anthrax simulants with microcalorimetric spectroscopy: Bacillus subtilis and Bacillus cereus spores. Appl. Opt. 2003, 42, 1757–1762. [Google Scholar] [CrossRef] [PubMed]
- Wig, A.; Arakawa, E.T.; Passian, A.; Ferrell, T.L.; Thundat, T. Photothermal spectroscopy of bacillus cereus and bacillus anthracis from 2.5 µm to 14.5 µm with microcantilevers. Sens. Actuators B 2006, 114, 206–211. [Google Scholar] [CrossRef]
- Peizman, E.R.; Zaidman, N.A.; Maniak, P.J.; O’Grady, S.M. Agonist Binding to β-Adrenergic receptors on human airway epithelial cells inhibits migration and wound repair. Am. J. Physiol. Cell Physiol. 2015, 309, C847–C855. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie, T.N.; Mujumdar, N.; Banerjee, S.; Sangwan, V.; Sarver, A.; Vickers, S.; Subramanian, S.; Saluja, A.K. Triptolide induces the expression of miR-142-3p: A negative regulator of heat shock protein 70 and pancreatic cancer cell proliferation. Mol. Cancer Ther. 2013, 12, 1266–1275. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.S.; Yang, L.; Zhang, G.; Zhao, H.; Selim, M.; McCullough, L.; Kulik, M.; Sanchez, T. Critical role of sphingosine-1-phosphate receptor-2 in the disruption of cerebrovascular integrity in experimental stroke. Nat. Commun. 2015, 6, 7893. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shinde, A.V.; Motiani, R.K.; Zhang, X.; Abdullaev, I.F.; Adam, A.P.; González-Cobos, J.C.; Zhang, W.; Matrougui, K.; Vincent, P.A.; Trebak, M. STIM1 controls endothelial barrier function independently of Orai1 and Ca2+ entry. Sci. Signal 2013, 6, ra18. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, J.I.; Dodelet-Devillers, A.; Kebir, H.; Ifergan, I.; Fabre, P.J.; Terouz, S.; Sabbagh, M.; Wosik, K.; Bourbonnière, L.; Bernard, M.; et al. The Hedgehog pathway promotes blood-brain barrier integrity and CNS immune quiescence. Science 2011, 334, 1727–1731. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Sangwan, V.; McGinn, O.; Chugh, R.; Dudeja, V.; Vickers, S.; Saluja, A. Triptolide-induced cell death in pancreatic cancer is mediated by O-GlcNAc modification of transcription factor Sp1. J. Biol. Chem. 2013, 288, 33927–33938. [Google Scholar] [CrossRef] [PubMed]
- Kourtidis, A.; Ngok, S.P.; Pulimeno, P.; Feathers, R.W.; Carpio, L.R.; Baker, T.R.; Carr, J.M.; Yan, I.K.; Borges, S.; Perez, E.A.; et al. Distinct E-cadherin-based complexes regulate cell behavior through miRNA processing or Src and p120 catenin activity. Nat. Cell Biol. 2015, 17, 1145–1157. [Google Scholar] [CrossRef] [PubMed]
- Goc, A.; Al-Azayzih, A.; Abdalla, M.; Al-Husein, B.; Kavuri, S.; Lee, J.; Moses, K.; Somanath, P.R. P21 activated kinase-1 (Pak1) promotes prostate tumor growth and microinvasion via inhibition of transforming growth factor β expression and enhanced matrix metalloproteinase 9 secretion. J. Biol. Chem. 2013, 288, 3025–3035. [Google Scholar] [CrossRef] [PubMed]
- Migliorini, A.; Angelotti, M.L.; Mulay, S.R.; Kulkarni, O.O.; Demleitner, J.; Dietrich, A.; Sagrinati, C.; Ballerini, L.; Peired, A.; Shankland, S. J.; et al. The antiviral cytokines IFN-α and IFN-β modulate parietal epithelial cells and promote podocyte loss: Implications for IFN toxicity, viral glomerulonephritis, and glomerular regeneration. Am. J. Pathol. 2013, 183, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.G. Electric Cell-Substrate Impedance Sensing and Cancer Metastasis; Springer: Dordrecht, The Netherlands, 2012. [Google Scholar]
- Liu, F.; Nordin, A.N.; Li, F.; Voiculescu, I. A lab-on-chip cell-based biosensor for label-free sensing of water toxicants. Lab Chip 2014, 14, 1270–1280. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Li, F.; Nordin, A.N.; Voiculescu, I. A novel cell-based hybrid acoustic wave biosensor with impedimetric sensing capabilities. Sensors 2013, 13, 3039–3055. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, W.; Li, F.; Voiculescu, I. Stretchable impedance sensor for mammalian cell proliferation measurements. Lab Chip 2017, 17, 2054–2066. [Google Scholar] [CrossRef] [PubMed]
- Toda, M.; Inomata, N.; Ono, T.; Voiculescu, I. Cantilever beam temperature sensors for biological applications. IEEJ Trans. Electr. Electron. Eng. 2017, 12, 153–160. [Google Scholar] [CrossRef]
- Giaever, I.; Keese, C.R. Micromotion of mammalian cells measured electrically. Proc. Natl. Acad. Sci. USA 1991, 88, 7896–7900. [Google Scholar] [CrossRef] [PubMed]
- Mitra, P.; Keese, C.R.; Giaever, I. Electric measurements can be used to monitor the attachment and spreading of cells in tissue culture. Biotechniques 1991, 11, 504–510. [Google Scholar] [PubMed]
- Lo, C.M.; Keese, C.R.; Giaever, I. Monitoring motion of confluent cells in tissue culture. Exp. Cell Res. 1993, 204, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Keese, C.R.; Giaever, I. A Biosensor that monitors cell morphology with electrical fields. IEEE Eng. Med. Biol. 1994, 13, 402–408. [Google Scholar] [CrossRef]
- Ayliffe, H.E.; Frazier, A.B.; Rabbitt, R.D. Electric impedance spectroscopy using microchannels with integrated metal electrodes. J. Microelectromech. Syst. 1999, 8, 50–57. [Google Scholar] [CrossRef]
- Han, A.; Yang, L.; Frazier, A.B. Quantification of the heterogeneity in breast cancer cell lines using whole-cell impedance spectroscopy. Clin. Cancer Res. 2007, 13, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, F.; Nordin, A.N.; Tarbell, J.; Voiculescu, I. Toxicity studies using mammalian cells and impedance spectroscopy method. Sens. Biosens. Res. 2015, 3, 112–121. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, W.; Nordin, A.N.; Li, F.; Voiculescu, I. The influence of the electrode dimension on the detection sensitivity of electric cell–substrate impedance sensing (ECIS) and its mathematical modeling. Sens. Actuators B 2017, 247, 780–790. [Google Scholar] [CrossRef]
- Wegener, J.; Keese, C.R.; Giaever, I. Electric cell-substrate impedance sensing (ECIS) as a noninvasive means to monitor the kinetics of cell spreading to artificial surfaces. Exp. Cell Res. 2000, 259, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Lo, C.M.; Keese, C.R.; Giaever, I. Cell-substrate contact: Another factor may influence transepithelial electrical resistance of cell layers cultured on permeable filters. Exp. Cell Res. 1999, 250, 576–580. [Google Scholar] [CrossRef] [PubMed]
- Lo, C.M.; Keese, C.R.; Giaever, I. Impedance analysis of MDCK cells measured by electric cell-substrate impedance sensing. Biophys. J. 1995, 69, 2800–2807. [Google Scholar] [CrossRef] [Green Version]
- Szulcek, R.; Bogaard, H.J.; van Nieuw Amerongen, G.P. Electric cell-substrate impedance sensing for the quantification of endothelial proliferation, barrier function, and motility. J. Vis. Exp. 2014, 85, 51300. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.; Lachance, B.; Sunahara, G.; Luong, J.H. Assessment of cytotoxicity using electric cell-substrate impedance sensing: Concentration and time response function approach. Anal. Chem. 2002, 74, 5748–5753. [Google Scholar] [CrossRef] [PubMed]








© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Voiculescu, I.; Toda, M.; Inomata, N.; Ono, T.; Li, F. Nano and Microsensors for Mammalian Cell Studies. Micromachines 2018, 9, 439. https://doi.org/10.3390/mi9090439
Voiculescu I, Toda M, Inomata N, Ono T, Li F. Nano and Microsensors for Mammalian Cell Studies. Micromachines. 2018; 9(9):439. https://doi.org/10.3390/mi9090439
Chicago/Turabian StyleVoiculescu, Ioana, Masaya Toda, Naoki Inomata, Takahito Ono, and Fang Li. 2018. "Nano and Microsensors for Mammalian Cell Studies" Micromachines 9, no. 9: 439. https://doi.org/10.3390/mi9090439




