Overview of Scorpion Species from China and Their Toxins
Abstract
:
1. Scorpion Species from China
| Family | Genera | Species (endemic) | Distribution | References |
|---|---|---|---|---|
| Buthidae | Hottentotta | Hottentotta alticola (Pocock, 1895) | ▲ 1 | [12] |
| Hottentotta songi (Lourenço, Qi and Zhu, 2005) | Xizang (endemic) | [17,24] | ||
| Isometrus | Isometrus maculatus (DeGeer, 1778) | Hainan and Taiwan | [12,30] | |
| Isometrus hainanensis Lourenço, Qi and Zhu, 2005 | Hainan (endemic) | [12,30] | ||
| Isometrus tibetanus Zhu and Lourenço, 2008 | Xizang (endemic) | [37] | ||
| Lychas | Lychas mucronatus (Fabricius, 1798) | Guangxi, Hainan and Yunnan | [30] | |
| Lychas scutilus C. L. Koch, 1845 | Shanghai | [12] | ||
| Buthidae | Mesobuthus | Mesobuthus bolensis Sun, Zhu and Lourenço, 2010 | Xinjiang (endemic) | [17] |
| Mesobuthus caucasicus intermedius (Birula, 1897) | Xinjiang | [12] | ||
| Mesobuthus caucasicus przewalskii (Birula, 1897) | Xinjiang | [12] | ||
| Mesobuthus eupeus mongolcus (Birula, 1911) | Gansu, Inner Mongolia (Neimenggu) and Ningxia | [20] | ||
| Mesobuthus eupeus thersites (C. L. Koch, 1839) | Xinjiang | [20] | ||
| Mesobuthus karshius Sun and Sun, 2011 | Xinjiang (endemic) | [20] | ||
| Mesobuthus longichelus Sun and Zhu, 2010 | Xinjiang (endemic) | [19] | ||
| Mesobuthus martensii martensii (Karsch, 1879) | the south side of 43°N and the north side of the Yangtze River, bordered by the Helan Mountains and the Tengger and Mo Us sand desert in the west and limited by the sea in the east | [39] | ||
| Mesobuthus martensii hainanensis (Birula, 1904) | Hainan (endemic) | [12] | ||
| Orthochirus | Orthochirus scrobiculosus (Grube, 1873) | Northwest | [40] | |
| Razianus | Razianus xinjianganus Lourenço, Sun and Zhu, 2010 | Xinjiang (endemic) | [25] | |
| Chaerilidae | Chaerilus | Chaerilus conchiformus Zhu, Han & Lourenço, 2008 | Xizang (endemic) | [16] |
| Chaerilus dibangvalleycus Bastawade, 2006 | Xizang (endemic) | [34] | ||
| Chaerilus mainlingensis Di and Zhu, 2009 | Xizang (endemic) | [28] | ||
| Chaerilus pictus (Pocock, 1890) | Xizang | [12] | ||
| Chaerilus tessellatus Qi, Zhu and Lourenço, 2005 | Xizang (endemic) | [13,34] | ||
| Chaerilus tricostatus Pocock, 1899 | Xizang | [34] | ||
| Chaerilus tryznai Kovařík, 2000 | Xizang (endemic) | [34] | ||
| Chaerilus wrzecionkoi Kovařík, 2012 | Xizang (endemic) | [26] | ||
| Euscorpiidae | Euscorpiops | Euscorpiops asthenurus (Pocock, 1900) | Xizang | [29] |
| Euscorpiops kamengensis Bastawade, 2006 | Xizang (endemic) | [29] | ||
| Chaerilidae | Chaerilus | Euscorpiops karschi Qi, Zhu and Lourenço, 2005 | Xizang (endemic) | [29] |
| Euscorpiops kubani Kovařík, 2004 | Yunnan | [35] | ||
| Euscorpiops novaki Kovařík, 2005 | Xizang (endemic) | [29] | ||
| Euscorpiops puerensis Di, Wu, Cao, Xiao and Li, 2010 | Yunnan (endemic) | [35] | ||
| Euscorpiops shidian Qi, Zhu and Lourenço, 2005 | Yunnan (endemic) | [35] | ||
| Euscorpiops vachoni Qi, Zhu and Lourenço, 2005 | Yunnan (endemic) | [35] | ||
| Euscorpiops validus Di, Cao, Wu and Li, 2010 | Yunnan (endemic) | [35] | ||
| Euscorpiops xui Sun and Zhu, 2010 | Yunnan (endemic) | [35] | ||
| Euscorpiops yangi Zhu, Zhang and Lourenço, 2007 | Yunnan (endemic) | [35] | ||
| Scorpiops | Scorpiops atomatus Qi, Zhu and Lourenço, 2005 | Xizang (endemic) | [36] | |
| Scorpiops hardwickii (Gervais, 1843) | Xizang | [12,36] | ||
| Scorpiops jendeki Kovařík, 1994 | Yunnan (endemic) | [12,36] | ||
| Scorpiops langxian Qi, Zhu and Lourenço, 2005 | Xizang (endemic) | [36] | ||
| Scorpiops leptochirus Pcock, 1893 | Xizang | [36] | ||
| Scorpiops lhasa Di and Zhu, 2009 | Xizang (endemic) | [27,36] | ||
| Scorpiops luridus Qi, Zhu and Lourenço, 2005 | Xizang (endemic) | [13,36] | ||
| Scorpiops margerisonae Kovařík, 2000 | Xizang (endemic) | [12,36] | ||
| Scorpiops petersii Pocock, 1893 | Xizang | [12,33] | ||
| Scorpiops pococki Qi, Zhu and Lourenço, 2005 | Xizang (endemic) | [13,33] | ||
| Scorpiops tibetanus Hirst, 1911 | Xizang (endemic) | [12,33] | ||
| Hemiscorpiidae | Liocheles | Liocheles australasiae (Fabricius, 1775) | Hainan | [12,30] |
| Tibetiomachus (endemic) | Tibetiomachus himalayensis Lourenço and Qi, 2006 | Xizang (endemic) | [23] | |
| Scorpionidae | Heterometrus | Heterometrus longimanus (Herbst, 1800) | ▲ 2 | [12] |
| Heterometrus tibetanus Lourenço, Qi and Zhu, 2005 | Xizang (endemic) | [24] | ||
| Heterometrus petersii (Thorell, 1876) | ▲ 3 | [12] | ||
| 5 | 12 (1) | 53 (33) |
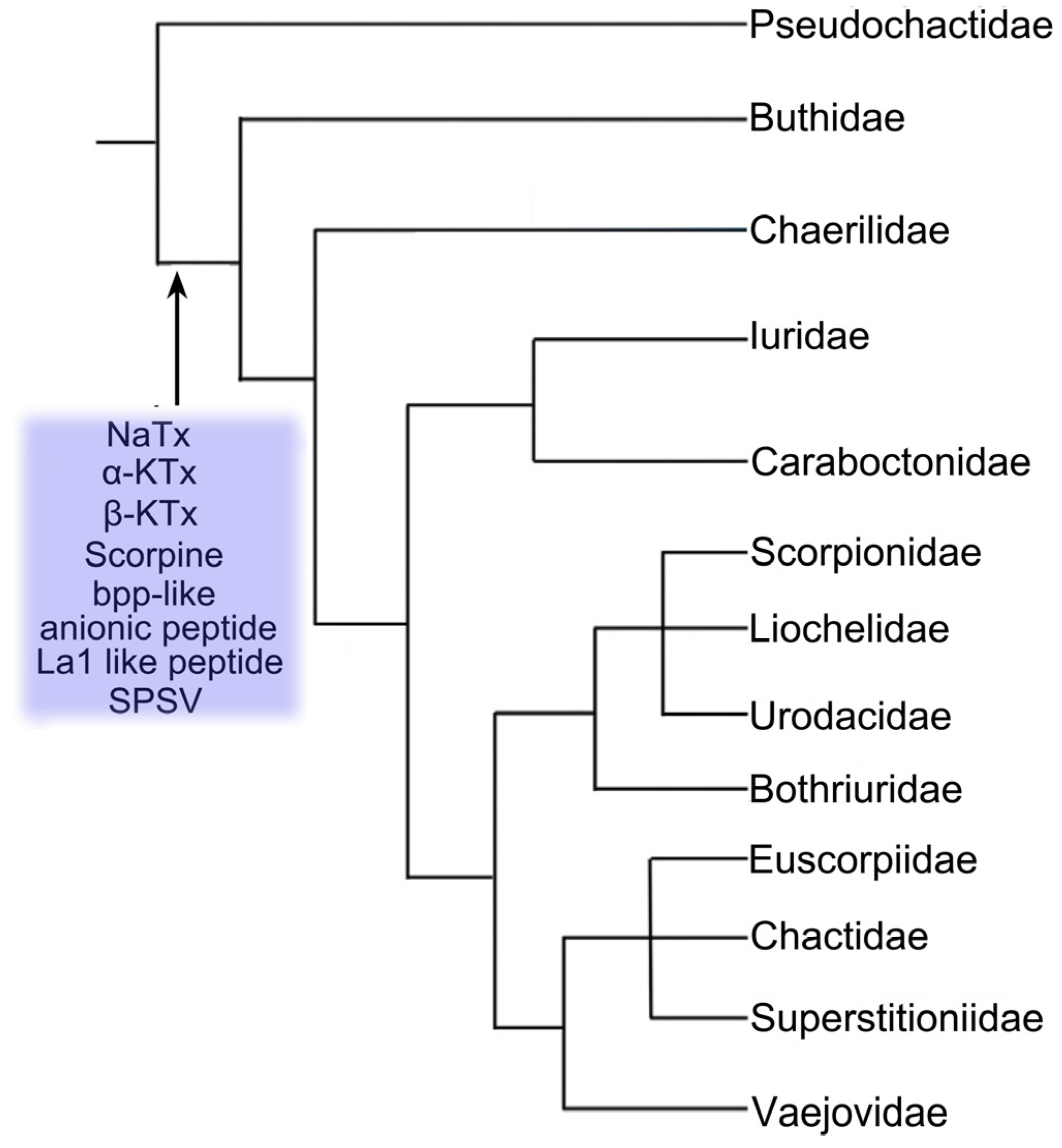
2. Toxins from Chinese Scorpion Species
| Family | Species | Transcriptomic analysis | Proteomic analysis | References |
|---|---|---|---|---|
| Buthidae | Lychas mucronatus | + | [48,73] | |
| Isometrus maculates | [48] | |||
| Buthus martensii (=M. martensii) | + | [49] | ||
| Hottentotta judaicus | + | [77] | ||
| Tityus discrepans | + | + | [43,78] | |
| Tityus stigmurus | + | + | [79,80] | |
| Tityus cambridgei | + | [81] | ||
| Tityus costatus | + | [82] | ||
| Tityus pachyurus | + | [83] | ||
| Androctonus crassicauda | + | + | [84] | |
| Androctonus mauretanicus mauretanicus | + | [85] | ||
| Scorpionidae | Heterometrus petersii | + | + | [45] |
| Heterometrus longimanus | + | [86] | ||
| Pandinus cavimanus | + | + | [87] | |
| Urodacus yaschenkoi | + | + | [88] | |
| Euscorpiidae | Scorpiops jendeki | + | [72] | |
| Scorpiops margerisonae | + | [48] | ||
| Hemiscorpiidae | Liocheles australasiae | + | [89] | |
| (Liochelidae) | Opisthacanthus cayaporum | + | + | [90,91] |
| Caraboctonidae | Hadrurus gertschi | + | [47] | |
| Chaerilidae | Chaerilus tricostatus | + | [75] | |
| Chaerilus tryznai | + | [75] |
3. Diverse Functions of Toxins from Chinese Scorpion Species
3.1. Na+ Channel Modulators
3.2. K+ Channel Blockers
3.3. Antimicrobial Peptides
3.4. Protease Inhibitors
4. Conclusions
Funding
Conflicts of Interest
References
- Shultz, J.W. A phylogenetic analysis of the arachnid orders based on morphological characters. Zool. J. Linn. Soc. 2007, 150, 221–265. [Google Scholar] [CrossRef]
- Chippaux, J.P.; Goyffon, M. Epidemiology of scorpionism: A global appraisal. Acta Trop. 2008, 107, 71–79. [Google Scholar] [CrossRef]
- Rodriguez de la Vega, R.C.; Schwartz, E.F.; Possani, L.D. Mining on scorpion venom biodiversity. Toxicon 2010, 56, 1155–1161. [Google Scholar] [CrossRef]
- Lourenço, W.R.; Pham, D.S. First record of a cave species of Euscorpiops Vachon from Viet Nam (Scorpiones, Euscorpiidae, Scorpiopinae). C. R. Biol. 2013, 336, 370–374. [Google Scholar] [CrossRef]
- Miller, A.L.; Makowsky, R.A.; Formanowicz, D.R.; Prendini, L.; Cox, C.L. Cryptic genetic diversity and complex phylogeography of the boreal North American scorpion, Paruroctonus boreus (Vaejovidae). Mol. Phylogenet. Evol. Mol. Phylogenet. Evol. 2014, 71, 298–307. [Google Scholar] [CrossRef]
- Graham, M.R.; Jaeger, J.R.; Prendini, L.; Riddle, B.R. Phylogeography of Beck’s Desert Scorpion, Paruroctonus becki, reveals Pliocene diversification in the Eastern California Shear Zone and postglacial expansion in the Great Basin Desert. Mol. Phylogenet. Evol. 2013, 69, 502–513. [Google Scholar] [CrossRef]
- Soleglad, M.E.; Fet, V. Further observations on scorpion genera Hadrurus and Hoffmannihadrurus (Scorpiones, Caraboctonidae). Zookeys 2010, 59, 1–14. [Google Scholar]
- Lourenço, W.R.; Ythier, E. The remarkable scorpion diversity in the Ecuadorian Andes and description of a new species of Tityus C. L. Koch, 1836 (Scorpiones, Buthidae). Zookeys 2013, 307, 1–13. [Google Scholar] [CrossRef]
- Smith, J.J.; Vetter, I.; Lewis, R.J.; Peigneur, S.; Tytgat, J.; Lam, A.; Gallant, E.M.; Beard, N.A.; Alewood, P.F.; Dulhunty, A.F. Multiple actions of phi–LITX–Lw1a on ryanodine receptors reveal a functional link between scorpion DDH and ICK toxins. Proc. Natl. Acad. Sci. USA 2013, 110, 8906–8911. [Google Scholar] [CrossRef]
- Feng, J.; Hu, Y.T.; Yi, H.; Yin, S.J.; Han, S.; Hu, J.; Chen, Z.Y.; Yang, W.S.; Cao, Z.J.; de Waard, M.; et al. Two conserved arginine residues from the SK3 potassium channel outer vestibule control selectivity of recognition by scorpion toxins. J. Biol. Chem. 2013, 288, 12544–12553. [Google Scholar] [CrossRef]
- Mamelak, A.N.; Rosenfeld, S.; Bucholz, R.; Raubitschek, A.; Nabors, L.B.; Fiveash, J.B.; Shen, S.; Khazaeli, M.B.; Colcher, D.; Liu, A.; et al. Phase I single–dose study of intracavitary–administered iodine–131–TM–601 in adults with recurrent high–grade glioma. J. Clin. Oncol. 2006, 24, 3644–3650. [Google Scholar] [CrossRef]
- Zhu, M.S.; Qi, J.X.; Song, D.X. A checklist of scorpions from China (Arachnida: Scorpiones). Acta Arachnol. Sin. 2004, 13, 111–118. [Google Scholar]
- Qi, J.X.; Zhu, M.S.; Lourenço, W.R. Eight new species of the genera Scorpiops Peters, Euscorpiops Vachon, and Chaerilus Simon (Scorpiones: Euscorpiidae, Chaerilidae) from Tibet and Yunnan, China. Euscorpius 2005, 32, 1–40. [Google Scholar]
- Di, Z.Y.; Zhu, M.S. Redescription of Scorpiops margerisonae Kovařík, 2000, with the first record of its female, from China (Scorpiones: Euscorpiidae: Scorpiopinae). Euscorpius 2010, 104, 1–9. [Google Scholar]
- Zhu, M.S.; Zhang, L.; Lourenço, W.R. One new species of scorpion belonging to the genus Euscorpiops Vachon, 1980 from South China (Scorpiones: Euscorpiidae, Scorpiopinae). Zootaxa 2007, 1582, 19–25. [Google Scholar]
- Zhu, M.S.; Han, G.X.; Lourenço, W.R. The chaerilid scorpions of China (Scorpiones: Chaerilidae). Zootaxa 2008, 1943, 37–52. [Google Scholar]
- Sun, D.; Zhu, M.S.; Lourenço, W.R. A new species of Mesobuthus (Scorpiones: Buthidae) from Xinjiang, China, with notes on Mesobuthus songi. J. Arachnol. 2010, 38, 35–43. [Google Scholar] [CrossRef]
- Sun, D.; Zhu, M.S. One new species of scorpion belonging to the genus Euscorpiops Vachon, 1980 from Yunnan, China (Scorpiones: Euscorpiidae, Scorpiopinae). Zootaxa 2010, 2399, 61–68. [Google Scholar]
- Sun, D.; Zhu, M.S. A new species of the genus Mesobuthus Vachon, 1950 (Scorpiones, Buthidae) from Xinjiang, China. ZooKeys 2010, 37, 1–12. [Google Scholar]
- Sun, D.; Sun, Z.N. Notes on the genus Mesobuthus (Scorpiones: Buthidae) in China, with description of a new species. J. Arachnol. 2011, 39, 59–75. [Google Scholar] [CrossRef]
- Qi, J.X.; Zhu, M.S.; Lourenço, W.R. Redescription of Mesobuthus martensii martensii (Karsch, 1879)(Scorpiones: Buthidae) from China. Revista ibérica de Aracnol. 2004, 10, 137–144. [Google Scholar]
- Lourenço, W.R.; Qi, J.X.; Zhu, M.S. Description of a new species of Isometrus Ehrenberg 1828 (Scorpiones, Buthidae) from the Island of Hainan. China. Boletín SEA 2005, 36, 57–63. [Google Scholar]
- Lourenço, W.R.; Qi, J.X. Mountain scorpions: A new genus and species from Tibet (China). Comptes Rendus Biol. 2006, 329, 289–295. [Google Scholar] [CrossRef]
- Lourenço, W.R.; Qi, J.X.; Zhu, M.S. Description of two new species of scorpions from China (Tibet) belonging to the genera Mesobuthus Vachon (Buthidae) and Heterometrus Ehrenberg (Scorpionidae). Zootaxa 2005, 985, 1–16. [Google Scholar]
- Lourenço, W.R.; Sun, D.; Zhu, M.S. Razianus xinjianganus sp. nov.: A New Record Genus and New Species of (Scorpiones, Buthidae) from China. J. Hebei Univ. (Nat. Sci. Ed.) 2010, 30, 307–312. [Google Scholar]
- Kovařík, F. Five new species of Chaerilus Simon, 1877 from China, Indonesia, Malaysia, Philippines, Thailand, and Vietnam (Scorpiones: Chaerilidae). Euscorpius 2012, 149, 1–14. [Google Scholar]
- Di, Z.Y.; Zhu, M.S. One new species of the Genus Scorpiops Peters, 1861 (Scorpiones: Euscorpiidae, Scorpiopinae) from Xizang, China. Zootaxa 2009, 2030, 39–48. [Google Scholar]
- Di, Z.Y.; Zhu, M.S. A new species of Chaerilus Simon, 1877 (Scorpiones, Chaerilidae) from China. Acta Arachnol. 2009, 58, 97–102. [Google Scholar] [CrossRef]
- Di, Z.Y.; Wu, Y.L.; Cao, Z.J.; Xiao, H.; Li, W.X. A catalogue of the genus Euscorpiops Vachon, 1980 (Scorpiones: Euscorpiidae, Scorpiopinae) from China, with description of a new species. Zootaxa 2010, 2477, 49–61. [Google Scholar]
- Di, Z.Y.; Cao, Z.J.; Wu, Y.L.; Zhu, L.; Liu, H.; Li, W.X. The scorpions of Hainan Island, China (Arachnida: Scorpiones). Euscorpius 2013, 153, 1–22. [Google Scholar]
- Di, Z.Y.; Cao, Z.J.; Wu, Y.L.; Li, W.X. A new species of the genus Euscorpiops Vachon, 1980 (Scorpiones: Euscorpiidae, Scorpiopinae) from Yunnan, China. Zootaxa 2010, 2361, 13–22. [Google Scholar]
- Di, Z.Y.; Zhu, M.S. The male of Euscorpiops karschi (Scorpiones: Euscorpiidae, Scorpiopinae) from China (Xizang). Arthr. Sel. 2009, 18, 9–16. [Google Scholar]
- Di, Z.Y.; Xu, X.B.; Cao, Z.J.; Wu, Y.L.; Li, W.X. Notes on the scorpions (Arachnida, Scorpiones) from Xizang with the redescription of Scorpiops jendeki Kovařík, 2000 (Scorpiones, Euscorpiidae) from Yunnan (China). ZooKeys 2013, 301, 51–99. [Google Scholar] [CrossRef]
- Di, Z.Y.; Wu, Y.L.; Cao, Z.J.; Fan, L.Q.; Li, W.X. The genus Chaerilus Simon, 1877 (Scorpiones: Chaerilidae) in China, with a description of the female C. tricostatus Pocock, 1899. Arthr. Sel. 2009, 18, 131–138. [Google Scholar]
- Di, Z.Y.; He, Y.W.; Wu, Y.L.; Cao, Z.J.; Liu, H.; Jiang, D.H.; Li, W.X. The scorpions of Yunnan (China): Updated identification key, new record and redescriptions of Euscorpiops kubani and E. shidian (Arachnida, Scorpiones). ZooKeys 2011, 82, 1–33. [Google Scholar]
- Di, Z.Y.; He, Y.W.; Cao, Z.J.; Wu, Y.L.; Li, W.X. The first record of the family Euscorpiidae (Arachnida: Scorpiones) from Central China, with a key of Chinese species of the genus Scorpiops. Euscorpius 2011, 118, 1–9. [Google Scholar]
- Lourenco, W.R.; Zhu, M.S. A new species of the genus Isometrus Ehrenberg 1828 from China (Scorpiones, Buthidae). Acta Zootaxon. Sin. 2008, 33, 264–271. [Google Scholar]
- Shi, C.M.; Ji, Y.J.; Liu, L.; Wang, L.; Zhang, D.X. Impact of climate changes from Middle Miocene onwards on evolutionary diversification in Eurasia: Insights from the mesobuthid scorpions. Mol. Ecol. 2013, 22, 1700–1716. [Google Scholar] [CrossRef]
- Shi, C.M.; Huang, Z.S.; Wang, L.; He, L.J.; Hua, Y.P.; Leng, L.; Zhang, D.X. Geographical distribution of two species of Mesobuthus (Scorpiones, Buthidae) in China: Insights from systematic field surveys and predictive models. J. Arachnol. 2007, 35, 215–226. [Google Scholar] [CrossRef]
- Shi, C.M.; Zhang, D.X. A review of the systematic research on buthid scorpions (Scorpiones: Buthidae). Acta Zootaxon. Sin. 2005, 30, 470–477. [Google Scholar]
- Zhu, S.Y.; Peigneur, S.; Gao, B.; Luo, L.; Jin, D.; Zhao, Y.; Tytgat, J. Molecular diversity and functional evolution of scorpion potassium channel toxins. Mol. Cell Proteom. 2011, 10, M11 0002832. [Google Scholar] [CrossRef]
- Cao, Z.J.; Luo, F.; Wu, Y.L.; Mao, X.; Li, W.X. Genetic mechanisms of scorpion venom peptide diversification. Toxicon 2006, 47, 348–355. [Google Scholar] [CrossRef]
- Batista, C.V.; D’Suze, G.; Gomez–Lagunas, F.; Zamudio, F.Z.; Encarnacion, S.; Sevcik, C.; Possani, L.D. Proteomic analysis of Tityus discrepans scorpion venom and amino acid sequence of novel toxins. Proteomics 2006, 6, 3718–3727. [Google Scholar] [CrossRef]
- Abdel–Rahman, M.A.; Quintero–Hernandez, V.; Possani, L.D. Venom proteomic and venomous glands transcriptomic analysis of the Egyptian scorpion Scorpio maurus palmatus (Arachnida: Scorpionidae). Toxicon 2013, 74C, 193–207. [Google Scholar]
- Ma, Y.B.; Zhao, Y.; Zhao, R.; Zhang, W.P.; He, Y.W.; Wu, Y.L.; Cao, Z.J.; Guo, L.; Li, W.X. Molecular diversity of toxic components from the scorpion Heterometrus petersii venom revealed by proteomic and transcriptome analysis. Proteomics 2010, 10, 2471–2485. [Google Scholar] [CrossRef]
- Abdel–Rahman, M.A.; Omran, M.A.; Abdel–Nabi, I.M.; Ueda, H.; McVean, A. Intraspecific variation in the Egyptian scorpion Scorpio maurus palmatus venom collected from different biotopes. Toxicon 2009, 53, 349–359. [Google Scholar] [CrossRef]
- Schwartz, E.F.; Diego-Garcia, E.; Rodriguez de la Vega, R.C.; Possani, L.D. Transcriptome analysis of the venom gland of the Mexican scorpion Hadrurus gertschi (Arachnida: Scorpiones). BMC Genomics 2007, 8, 119. [Google Scholar] [CrossRef]
- Ma, Y.B.; He, Y.W.; Zhao, R.M.; Wu, Y.L.; Li, W.X.; Cao, Z.J. Extreme diversity of scorpion venom peptides and proteins revealed by transcriptomic analysis: Implication for proteome evolution of scorpion venom arsenal. J. Proteomics 2012, 75, 1563–1576. [Google Scholar] [CrossRef]
- Zhu, S.Y.; Li, W.X.; Zeng, X.C.; Jiang, D.H.; Mao, X.; Liu, H. Molecular cloning and sequencing of two ‘short chain’ and two ‘long chain’ K(+) channel–blocking peptides from the Chinese scorpion Buthus martensii Karsch. FEBS Lett. 1999, 457, 509–514. [Google Scholar] [CrossRef]
- Goudet, C.; Chi, C.W.; Tytgat, J. An overview of toxins and genes from the venom of the Asian scorpion Buthus martensi Karsch. Toxicon 2002, 40, 1239–1258. [Google Scholar] [CrossRef]
- Zeng, X.C.; Li, W.X.; Zhu, S.Y.; Peng, F.; Jiang, D.H.; Yang, F.H.; Wu, K.L. Cloning and characterization of the cDNA sequences of two venom peptides from Chinese scorpion Buthus martensii Karsch (BmK). Toxicon 2000, 38, 893–899. [Google Scholar] [CrossRef]
- Dai, H.; Yin, S.J.; Li, T.; Cao, Z.J.; Ji, Y.H.; Wu, Y.L.; Li, W.X. Recombinant expression, purification, and characterization of scorpion toxin BmalphaTX14. Protein Expr. Purif. 2012, 82, 325–331. [Google Scholar] [CrossRef]
- Wang, K.; Yin, S.J.; Lu, M.; Yi, H.; Dai, C.; Xu, X.J.; Cao, Z.J.; Wu, Y.L.; Li, W.X. Functional analysis of the alpha–neurotoxin, BmalphaTX14, derived from the Chinese scorpion, Buthus martensii Karsch. Biotechnol. Lett. 2006, 28, 1767–1672. [Google Scholar] [CrossRef]
- Zeng, X.C.; Li, W.X.; Zhu, S.Y.; Peng, F.; Zhu, Z.H.; Liu, H.; Mao, X. Molecular cloning and sequence analysis of cDNAs encoding a beta–toxin–like peptide and two MkTx I homologues from scorpion Buthus martensii Karsch. Toxicon 2001, 39, 225–232. [Google Scholar] [CrossRef]
- Zhu, S.Y.; Li, W.X.; Zeng, X.C.; Liu, H.; Jiang, D.H.; Mao, X. Nine novel precursors of Buthus martensii scorpion alpha–toxin homologues. Toxicon 2000, 38, 1653–1661. [Google Scholar] [CrossRef]
- Peng, F.; Zeng, X.C.; He, X.H.; Pu, J.; Li, W.X.; Zhu, Z.H.; Liu, H. Molecular cloning and functional expression of a gene encoding an antiarrhythmia peptide derived from the scorpion toxin. Eur. J. Biochem. 2002, 269, 4468–4475. [Google Scholar] [CrossRef]
- Zeng, X.C.; Zhu, Z.H.; Li, W.X.; Zhu, S.Y.; Peng, F.; Mao, X.; Liu, H. Molecular cloning and genomic organization of a K(+) channel toxin from the Chinese scorpion Buthus martensii Karsch. Toxicon 2001, 39, 407–410. [Google Scholar] [CrossRef]
- Zhu, S.Y.; Li, W.X.; Zeng, X.C. Precursor nucleotide sequence and genomic organization of BmTXKS1, a new scorpion toxin–like peptide from Buthus martensii Karsch. Toxicon 2001, 39, 1291–1296. [Google Scholar] [CrossRef]
- Zeng, X.C.; Peng, F.; Luo, F.; Zhu, S.Y.; Liu, H.; Li, W.X. Molecular cloning and characterization of four scorpion K(+)–toxin–like peptides: A new subfamily of venom peptides (alpha–KTx14) and genomic analysis of a member. Biochimie 2001, 83, 883–889. [Google Scholar] [CrossRef]
- Zhu, S.Y.; Li, W.X.; Cao, Z.J. A naturally occurring non–coding fusion transcript derived from scorpion venom gland: Implication for the regulation of scorpion toxin gene expression. FEBS Lett. 2001, 508, 241–244. [Google Scholar] [CrossRef]
- Sheng, J.Q.; Xu, X.L.; Cao, Z.J.; Liu, W.H.; Wu, Y.L.; Zhu, S.Y.; Zeng, X.C.; Jang, D.H.; Mao, X.; Liu, H.; et al. Molecular cloning, genomic organization and functional characterization of a new short–chain potassium channel toxin–like peptide BmTxKS4 from Buthus martensii Karsch(BmK). J. Biochem. Mol. Toxicol. 2004, 18, 187–195. [Google Scholar] [CrossRef]
- Zeng, X.; Luo, F.; Li, W.X. Molecular dissection of venom from Chinese scorpion Mesobuthus martensii: Identification and characterization of four novel disulfide–bridged venom peptides. Peptides 2006, 27, 1745–1754. [Google Scholar] [CrossRef]
- Mao, X.; Cao, Z.J.; Yin, S.J.; Ma, Y.B.; Wu, Y.L.; Li, W.X. Cloning and characterization of BmK86, a novel K+ –channel blocker from scorpion venom. Biochem. Biophys. Res. Commun. 2007, 360, 728–734. [Google Scholar] [CrossRef]
- Zeng, X.C.; Li, W.X.; Zhu, S.Y.; Peng, F.; Zhu, Z.H.; Wu, K.L.; Yiang, F.H. Cloning and characterization of a cDNA sequence encoding the precursor of a chlorotoxin–like peptide from the Chinese scorpion Buthus martensii Karsch. Toxicon 2000, 38, 1009–1014. [Google Scholar] [CrossRef]
- Cao, Z.J.; Xie, Y.; Dai, C.; Zhu, S.Y.; Yin, S.J.; Wu, Y.L.; Li, W.X. Cloning and characterization of a novel calcium channel toxin–like gene BmCa1 from Chinese scorpion Mesobuthus martensii Karsch. Peptides 2006, 27, 1235–1240. [Google Scholar] [CrossRef]
- Zeng, X.C.; Li, W.X.; Peng, F.; Zhu, Z.H. Cloning and characterization of a novel cDNA sequence encoding the precursor of a novel venom peptide (BmKbpp) related to a bradykinin–potentiating peptide from Chinese scorpion Buthus martensii Karsch. IUBMB Life 2000, 49, 207–210. [Google Scholar]
- Zeng, X.C.; Wang, S.X.; Zhu, Y.; Zhu, S.Y.; Li, W.X. Identification and functional characterization of novel scorpion venom peptides with no disulfide bridge from Buthus martensii Karsch. Peptides 2004, 25, 143–150. [Google Scholar]
- Zeng, X.C.; Li, W.X.; Wang, S.X.; Zhu, S.Y.; Luo, F. Precursor of a novel scorpion venom peptide (BmKn1) with no disulfide bridge from Buthus martensii Karsch. IUBMB Life 2001, 51, 117–120. [Google Scholar]
- Zeng, X.C.; Wang, S.X.; Li, W.X. Identification of BmKAPi, a novel type of scorpion venom peptide with peculiar disulfide bridge pattern from Buthus martensii Karsch. Toxicon 2002, 40, 1719–1722. [Google Scholar] [CrossRef]
- Zhu, S.Y.; Li, W.X.; Jiang, D.H.; Zeng, X.C. Evidence for the existence of insect defensin–like peptide in scorpion venom. IUBMB Life 2000, 50, 57–61. [Google Scholar] [CrossRef]
- Zhu, S.Y.; Li, W.X. Precursors of three unique cysteine–rich peptides from the scorpion Buthus martensii Karsch. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2002, 131, 749–756. [Google Scholar] [CrossRef]
- Ma, Y.B.; Zhao, R.M.; He, Y.W.; Li, S.R.; Liu, J.; Wu, Y.L.; Cao, Z.J.; Li, W.X. Transcriptome analysis of the venom gland of the scorpion Scorpiops jendeki: Implication for the evolution of the scorpion venom arsenal. BMC Genomics 2009, 10, 290. [Google Scholar] [CrossRef]
- Zhao, R.M.; Ma, Y.B.; He, Y.W.; Di, Z.Y.; Wu, Y.L.; Cao, Z.J.; Li, W.X. Comparative venom gland transcriptome analysis of the scorpion Lychas mucronatus reveals intraspecific toxic gene diversity and new venomous components. BMC Genomics 2010, 11, 452. [Google Scholar] [CrossRef]
- Oliveira, F.N.; Mortari, M.R.; Carneiro, F.P.; Guerrero-Vargas, J.A.; Santos, D.M.; Pimenta, A.M.; Schwartz, E.F. Another record of significant regional variation in toxicity of Tityus serrulatus venom in Brazil: A step towards understanding the possible role of sodium channel modulators. Toxicon 2013, 73, 33–46. [Google Scholar] [CrossRef]
- He, Y.W.; Zhao, R.M.; di, Z.Y.; Li, Z.J.; Xu, X.B.; Hong, W.; Wu, Y.L.; Zhao, H.B.; Li, W.X.; Cao, Z.J. Molecular diversity of Chaerilidae venom peptides reveals the dynamic evolution of scorpion venom components from Buthidae to non–Buthidae. J. Proteomics 2013, 89, 1–14. [Google Scholar]
- Cao, Z.J.; Yu, Y.; Wu, Y.L.; Hao, P.; Di, Z.Y.; He, Y.W.; Chen, Z.Y.; Yang, W.S.; Shen, Z.Y.; He, X.H.; et al. The genome of Mesobuthus martensii reveals a unique adaptation model of arthropods. Nat Commun. 2013, 4, 2602. [Google Scholar]
- Morgenstern, D.; Rohde, B.H.; King, G.F.; Tal, T.; Sher, D.; Zlotkin, E. The tale of a resting gland: Transcriptome of a replete venom gland from the scorpion Hottentotta judaicus. Toxicon 2011, 57, 695–703. [Google Scholar] [CrossRef]
- D’Suze, G.; Schwartz, E.F.; Garcia–Gomez, B.I.; Sevcik, C.; Possani, L.D. Molecular cloning and nucleotide sequence analysis of genes from a cDNA library of the scorpion Tityus discrepans. Biochimie 2009, 91, 1010–1019. [Google Scholar] [CrossRef]
- Almeida, D.D.; Scortecci, K.C.; Kobashi, L.S.; Agnez-Lima, L.F.; Medeiros, S.R.; Silva, A.A., Jr.; Junqueira-de-Azevedo Ide, L.; Fernandes-Pedrosa Mde, F. Profiling the resting venom gland of the scorpion Tityus stigmurus through a transcriptomic survey. BMC Genomics 2012, 13, 362. [Google Scholar] [CrossRef]
- Batista, C.V.; Roman-Gonzalez, S.A.; Salas-Castillo, S.P.; Zamudio, F.Z.; Gomez-Lagunas, F.; Possani, L.D. Proteomic analysis of the venom from the scorpion Tityus stigmurus: Biochemical and physiological comparison with other Tityus species. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2007, 146, 147–157. [Google Scholar] [CrossRef]
- Batista, C.V.; del Pozo, L.; Zamudio, F.Z.; Contreras, S.; Becerril, B.; Wanke, E.; Possani, L.D. Proteomics of the venom from the Amazonian scorpion Tityus cambridgei and the role of prolines on mass spectrometry analysis of toxins. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2004, 803, 55–66. [Google Scholar]
- Diego–Garcia, E.; Batista, C.V.; Garcia–Gomez, B.I.; Lucas, S.; Candido, D.M.; Gomez–Lagunas, F.; Possani, L.D. The Brazilian scorpion Tityus costatus Karsch: Genes, peptides and function. Toxicon 2005, 45, 273–283. [Google Scholar] [CrossRef]
- Barona, J.; Batista, C.V.; Zamudio, F.Z.; Gomez–Lagunas, F.; Wanke, E.; Otero, R.; Possani, L.D. Proteomic analysis of the venom and characterization of toxins specific for Na+ – and K+ –channels from the Colombian scorpion Tityus pachyurus. Biochim. Biophys. Acta 2006, 1764, 76–84. [Google Scholar] [CrossRef]
- Caliskan, F.; Garcia, B.I.; Coronas, F.I.; Batista, C.V.; Zamudio, F.Z.; Possani, L.D. Characterization of venom components from the scorpion Androctonus crassicauda of Turkey: Peptides and genes. Toxicon 2006, 48, 12–22. [Google Scholar] [CrossRef]
- Oukkache, N.; Rosso, J.P.; Alami, M.; Ghalim, N.; Saile, R.; Hassar, M.; Bougis, P.E.; Martin-Eauclaire, M.F. New analysis of the toxic compounds from the Androctonus mauretanicus mauretanicus scorpion venom. Toxicon 2008, 51, 835–852. [Google Scholar] [CrossRef]
- Bringans, S.; Eriksen, S.; Kendrick, T.; Gopalakrishnakone, P.; Livk, A.; Lock, R.; Lipscombe, R. Proteomic analysis of the venom of Heterometrus longimanus (Asian black scorpion). Proteomics 2008, 8, 1081–1096. [Google Scholar] [CrossRef]
- Diego-Garcia, E.; Peigneur, S.; Clynen, E.; Marien, T.; Czech, L.; Schoofs, L.; Tytgat, J. Molecular diversity of the telson and venom components from Pandinus cavimanus (Scorpionidae Latreille 1802): Transcriptome, venomics and function. Proteomics 2012, 12, 313–328. [Google Scholar] [CrossRef]
- Luna–Ramirez, K.; Quintero–Hernandez, V.; Vargas–Jaimes, L.; Batista, C.V.; Winkel, K.D.; Possani, L.D. Characterization of the venom from the Australian scorpion Urodacus yaschenkoi: Molecular mass analysis of components, cDNA sequences and peptides with antimicrobial activity. Toxicon 2013, 63, 44–54. [Google Scholar] [CrossRef]
- Miyashita, M.; Otsuki, J.; Hanai, Y.; Nakagawa, Y.; Miyagawa, H. Characterization of peptide components in the venom of the scorpion Liocheles australasiae (Hemiscorpiidae). Toxicon 2007, 50, 428–437. [Google Scholar] [CrossRef]
- Silva, E.C.; Camargos, T.S.; Maranhao, A.Q.; Silva–Pereira, I.; Silva, L.P.; Possani, L.D.; Schwartz, E.F. Cloning and characterization of cDNA sequences encoding for new venom peptides of the Brazilian scorpion Opisthacanthus cayaporum. Toxicon 2009, 54, 252–261. [Google Scholar] [CrossRef]
- Schwartz, E.F.; Camargos, T.S.; Zamudio, F.Z.; Silva, L.P.; Bloch, C.J.; Caixeta, F.; Schwartz, C.A.; Possani, L.D. Mass spectrometry analysis, amino acid sequence and biological activity of venom components from the Brazilian scorpion Opisthacanthus cayaporum. Toxicon 2008, 51, 1499–1508. [Google Scholar] [CrossRef]
- Rodriguez de la Vega, R.C.; Possani, L.D. Overview of scorpion toxins specific for Na+ channels and related peptides: Biodiversity, structure–function relationships and evolution. Toxicon 2005, 46, 831–844. [Google Scholar] [CrossRef]
- Chen, J.; Tan, Z.Y.; Zhao, R.; Feng, X.H.; Shi, J.; Ji, Y.H. The modulation effects of BmK I, an alpha–like scorpion neurotoxin, on voltage–gated Na(+) currents in rat dorsal root ganglion neurons. Neurosci. Lett. 2005, 390, 66–71. [Google Scholar] [CrossRef]
- Chai, Z.F.; Zhu, M.M.; Bai, Z.T.; Liu, T.; Tan, M.; Pang, X.Y.; Ji, Y.H. Chinese–scorpion (Buthus martensi Karsch) toxin BmK alphaIV, a novel modulator of sodium channels: From genomic organization to functional analysis. Biochem. J. 2006, 399, 445–453. [Google Scholar] [CrossRef]
- Zhu, S.Y.; Peigneur, S.; Gao, B.; Lu, X.X.; Cao, C.Y.; Tytgat, J. Evolutionary diversification of Mesobuthus alpha–scorpion toxins affecting sodium channels. Mol. Cell Proteomics 2012, 11, M111012054. [Google Scholar] [CrossRef]
- Mouhat, S.; Andreotti, N.; Jouirou, B.; Sabatier, J.M. Animal toxins acting on voltage–gated potassium channels. Curr. Pharm. Des. 2008, 14, 2503–2518. [Google Scholar] [CrossRef]
- Ji, Y.H.; Wang, W.X.; Ye, J.G.; He, L.L.; Li, Y.J.; Yan, Y.P.; Zhou, Z. Martentoxin, a novel K+–channel–blocking peptide: Purification, cDNA and genomic cloning, and electrophysiological and pharmacological characterization. J. Neurochem. 2003, 84, 325–335. [Google Scholar] [CrossRef]
- Shi, J.; He, H.Q.; Zhao, R.; Duan, Y.H.; Chen, J.; Chen, Y.; Yang, J.; Zhang, J.W.; Shu, X.Q.; Zheng, P.; Ji, Y.H. Inhibition of martentoxin on neuronal BK channel subtype (alpha + beta4): Implications for a novel interaction model. Biophys. J. 2008, 94, 3706–3713. [Google Scholar] [CrossRef]
- Yi, H.; Cao, Z.J.; Yin, S.J.; Dai, C.; Wu, Y.L.; Li, W.X. Interaction simulation of hERG K+ channel with its specific BeKm–1 peptide: Insights into the selectivity of molecular recognition. J. Proteome Res. 2007, 6, 611–620. [Google Scholar] [CrossRef]
- Yi, H.; Qiu, S.; Cao, Z.J.; Wu, Y.L.; Li, W.X. Molecular basis of inhibitory peptide maurotoxin recognizing Kv1.2 channel explored by ZDOCK and molecular dynamic simulations. Proteins 2008, 70, 844–854. [Google Scholar]
- Gan, G.L.; Yi, H.; Chen, M.R.; Sun, L.; Li, W.X.; Wu, Y.L.; Ding, J.P. Structural basis for toxin resistance of beta4–associated calcium–activated potassium (BK) channels. J. Biol. Chem. 2008, 283, 24177–24184. [Google Scholar]
- Qiu, S.; Yi, H.; Liu, H.; Cao, Z.J.; Wu, Y.L.; Li, W.X. Molecular Information of charybdotoxin blockade in the large conductance calcium–activated potassium channel. J. Chem. Inf. Model. 2009, 49, 1831–1838. [Google Scholar] [CrossRef]
- Chen, Z.Y.; Hu, Y.T.; Yang, W.S.; He, Y.W.; Feng, J.; Wang, B.; Zhao, R.M.; Ding, J.P.; Cao, Z.J.; Li, W.X.; et al. Hg1, novel peptide inhibitor specific for Kv1.3 channels from first scorpion Kunitz–type potassium channel toxin family. J. Biol. Chem. 2012, 287, 13813–13821. [Google Scholar] [CrossRef]
- Han, S.; Yin, S.; Yi, H.; Mouhat, S.; Qiu, S.; Cao, Z.; Sabatier, J.M.; Wu, Y.; Li, W. Protein–protein recognition control by modulating electrostatic interactions. J. Proteome Res. 2010, 9, 3118–3125. [Google Scholar] [CrossRef]
- Han, S.; Yi, H.; Yin, S.J.; Chen, Z.Y.; Liu, H.; Cao, Z.J.; Wu, Y.L.; Li, W.X. Structural basis of a potent peptide inhibitor designed for Kv1.3 channel, a therapeutic target of autoimmune disease. J. Biol. Chem. 2008, 283, 19058–19065. [Google Scholar]
- Yin, S.J.; Jiang, L.; Yi, H.; Han, S.; Yang, D.W.; Liu, M.L.; Liu, H.; Cao, Z.J.; Wu, Y.L.; Li, W.X. Different residues in channel turret determining the selectivity of ADWX–1 inhibitor peptide between Kv1.1 and Kv1.3 channels. J. Proteome Res. 2008, 7, 4890–4897. [Google Scholar] [CrossRef]
- Cociancich, S.; Goyffon, M.; Bontems, F.; Bulet, P.; Bouet, F.; Menez, A.; Hoffmann, J. Purification and characterization of a scorpion defensin, a 4 kDa antibacterial peptide presenting structural similarities with insect defensins and scorpion toxins. Biochem. Biophys. Res. Commun. 1993, 194, 17–22. [Google Scholar] [CrossRef]
- Dai, L.; Yasuda, A.; Naoki, H.; Corzo, G.; Andriantsiferana, M.; Nakajima, T. IsCT, a novel cytotoxic linear peptide from scorpion Opisthacanthus madagascariensis. Biochem. Biophys. Res. Commun. 2001, 286, 820–825. [Google Scholar] [CrossRef]
- Dai, C.; Ma, Y.B.; Zhao, Z.H.; Zhao, R.M.; Wang, Q.; Wu, Y.L.; Cao, Z.J.; Li, W.X. Mucroporin, the first cationic host defense peptide from the venom of Lychas mucronatus. Antimicrob. Agents Chemother. 2008, 52, 3967–3972. [Google Scholar] [CrossRef]
- Zhao, Z.H.; Ma, Y.B.; Dai, C.; Zhao, R.M.; Li, S.R.; Wu, Y.L.; Cao, Z.J.; Li, W.X. Imcroporin, a new cationic antimicrobial peptide from the venom of the scorpion Isometrus maculates. Antimicrob. Agents Chemother. 2009, 53, 3472–3477. [Google Scholar] [CrossRef]
- Cao, L.Y.; Dai, C.; Li, Z.J.; Fan, Z.; Song, Y.; Wu, Y.L.; Cao, Z.J.; Li, W.X. Antibacterial activity and mechanism of a scorpion venom peptide derivative in vitro and in vivo. PLoS One 2012, 7, e40135. [Google Scholar]
- Fan, Z.; Cao, L.Y.; He, Y.W.; Hu, J.; Di, Z.Y.; Wu, Y.L.; Li, W.X.; Cao, Z.J. Ctriporin, a new anti–methicillin–resistant Staphylococcus aureus peptide from the venom of the scorpion Chaerilus tricostatus. Antimicrob. Agents Chemother. 2011, 55, 5220–5229. [Google Scholar] [CrossRef]
- Yuan, W.Y.; Cao, L.Y.; Ma, Y.B.; Mao, P.Y.; Wang, W.P.; Zhao, R.M.; Wu, Y.L.; Cao, Z.J.; Li, W.X. Cloning and functional characterization of a new antimicrobial peptide gene StCT1 from the venom of the scorpion Scorpiops tibetanus. Peptides 2010, 31, 22–26. [Google Scholar] [CrossRef]
- Cao, L.Y.; Li, Z.J.; Zhang, R.M.; Wu, Y.L.; Li, W.X.; Cao, Z.J. StCT2, a new antibacterial peptide characterized from the venom of the scorpion Scorpiops tibetanus. Peptides 2012, 36, 213–220. [Google Scholar] [CrossRef]
- Zhao, R.M.; Dai, H.; Qiu, S.; Li, T.; He, Y.W.; Ma, Y.B.; Chen, Z.Y.; Wu, Y.L.; Li, W.X.; Cao, Z.J. SdPI, the first functionally characterized Kunitz–type trypsin inhibitor from scorpion venom. PLoS One 2011, 6, e27548. [Google Scholar]
- Chen, Z.Y.; Wang, B.; Hu, J.; Yang, W.S.; Cao, Z.J.; Zhuo, R.M.; Li, W.X.; Wu, Y.L. SjAPI, the first functionally characterized Ascaris–type protease inhibitor from animal venoms. PLoS One 2013, 8, e57529. [Google Scholar]
- Chen, Z.Y.; Cao, Z.; Li, W.; Wu, Y. Cloning and characterization of a novel Kunitz–type inhibitor from scorpion with unique cysteine framework. Toxicon 2013, 72, 5–10. [Google Scholar] [CrossRef]
- Craik, C.S.; Page, M.J.; Madison, E.L. Proteases as therapeutics. Biochem. J. 2011, 435, 1–16. [Google Scholar] [CrossRef]
- Wang, G.J.; Gao, C.F.; Wei, D.; Wang, C.; Ding, S.Q. Acute pancreatitis: Etiology and common pathogenesis. World J. Gastroenterol. 2009, 15, 1427–1430. [Google Scholar] [CrossRef]
- Hansen, K.K.; Oikonomopoulou, K.; Li, Y.; Hollenberg, M.D. Proteinases, proteinase–activated receptors (PARs) and the pathophysiology of cancer and diseases of the cardiovascular, musculoskeletal, nervous and gastrointestinal systems. Naunyn Schmiedebergs Arch. Pharmacol. 2008, 377, 377–392. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cao, Z.; Di, Z.; Wu, Y.; Li, W. Overview of Scorpion Species from China and Their Toxins. Toxins 2014, 6, 796-815. https://doi.org/10.3390/toxins6030796
Cao Z, Di Z, Wu Y, Li W. Overview of Scorpion Species from China and Their Toxins. Toxins. 2014; 6(3):796-815. https://doi.org/10.3390/toxins6030796
Chicago/Turabian StyleCao, Zhijian, Zhiyong Di, Yingliang Wu, and Wenxin Li. 2014. "Overview of Scorpion Species from China and Their Toxins" Toxins 6, no. 3: 796-815. https://doi.org/10.3390/toxins6030796