A Review of the Evidence that Ochratoxin A Is an Nrf2 Inhibitor: Implications for Nephrotoxicity and Renal Carcinogenicity
Abstract
:1. Introduction
2. The Nrf2 Pathway
3. OTA: An Inhibitor of Nrf2?
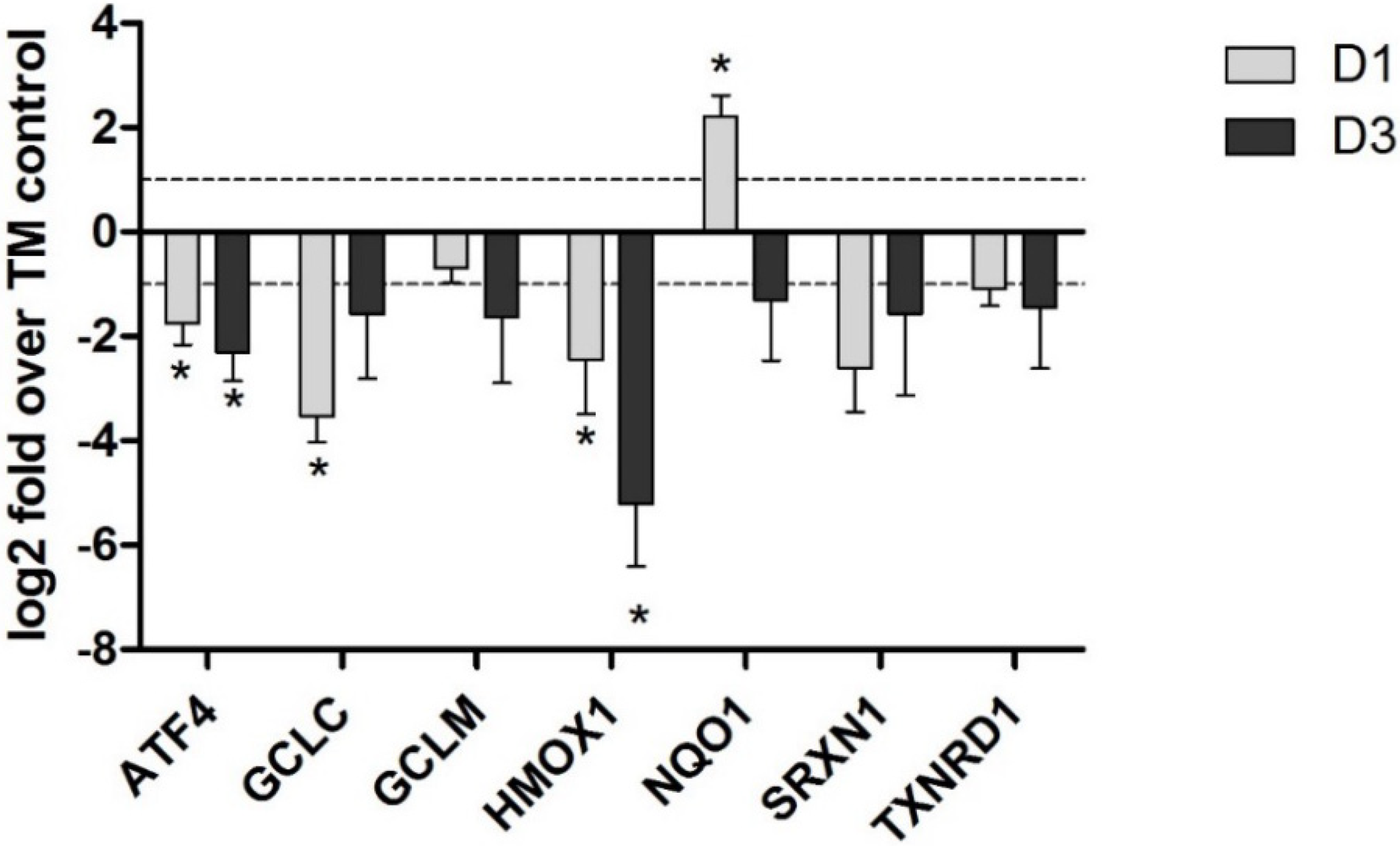
4. Potential Mechanisms of OTA-Induced Nrf2 Inhibition

Acknowledgements
Conflicts of Interest
References
- Clark, H.A.; Snedeker, S.M. Ochratoxin A: Its cancer risk and potential for exposure. J. Toxicol. Environ. Health B Crit. Rev. 2006, 9, 265–296. [Google Scholar] [CrossRef]
- Scientific Cooperation (SCOOP) Task Reports. Reports on tasks for scientific cooperation: Assessment of dietary intake of ochratoxin a by the population of eu member states. Available online: http://ec.europa.eu/food/fs/scoop/3.2.7_en.pdf (accessed on 15 October 2013).
- Zlender, V.; Breljak, D.; Ljubojevic, M.; Flajs, D.; Balen, D.; Brzica, H.; Domijan, A.M.; Peraica, M.; Fuchs, R.; Anzai, N.; et al. Low doses of ochratoxin a upregulate the protein expression of organic anion transporters oat1, oat2, oat3 and oat5 in rat kidney cortex. Toxicol. Appl. Pharmacol. 2009, 239, 284–296. [Google Scholar] [CrossRef]
- Anzai, N.; Jutabha, P.; Endou, H. Molecular mechanism of ochratoxin a transport in the kidney. Toxins 2010, 2, 1381–1398. [Google Scholar]
- Jennings, P.; Weiland, C.; Limonciel, A.; Bloch, K.M.; Radford, R.; Aschauer, L.; McMorrow, T.; Wilmes, A.; Pfaller, W.; Ahr, H.J.; et al. Transcriptomic alterations induced by ochratoxin A in rat and human renal proximal tubular in vitro models and comparison to a rat in vivo model. Arch. Toxicol. 2012, 86, 571–589. [Google Scholar] [CrossRef]
- Boorman, G.A.; McDonald, M.R.; Imoto, S.; Persing, R. Renal lesions induced by ochratoxin a exposure in the f344 rat. Toxicol. Pathol. 1992, 20, 236–245. [Google Scholar] [CrossRef]
- Boorman, G.A. Toxicology and carcinogenesis studies of ochratoxin a in f344/n rats (gavage studies). Natl. Toxicol. Progr. Tech. Rep. Ser. 1989, 5, 1–142. [Google Scholar]
- Pfohl-Leszkowicz, A.; Manderville, R.A. An update on direct genotoxicity as a molecular mechanism of ochratoxin a carcinogenicity. Chem. Res. Toxicol. 2012, 25, 252–262. [Google Scholar] [CrossRef]
- Rached, E.; Pfeiffer, E.; Dekant, W.; Mally, A. Ochratoxin A: Apoptosis and aberrant exit from mitosis due to perturbation of microtubule dynamics? Toxicol. Sci. 2006, 92, 78–86. [Google Scholar] [CrossRef]
- Czakai, K.; Muller, K.; Mosesso, P.; Pepe, G.; Schulze, M.; Gohla, A.; Patnaik, D.; Dekant, W.; Higgins, J.M.; Mally, A. Perturbation of mitosis through inhibition of histone acetyltransferases: The key to ochratoxin a toxicity and carcinogenicity? Toxicol. Sci. 2011, 122, 317–329. [Google Scholar] [CrossRef]
- Jennings, P.; Limonciel, A.; Felice, L.; Leonard, M.O. An overview of transcriptional regulation in response to toxicological insult. Arch. Toxicol. 2013, 87, 49–72. [Google Scholar] [CrossRef]
- Itoh, K.; Chiba, T.; Takahashi, S.; Ishii, T.; Igarashi, K.; Katoh, Y.; Oyake, T.; Hayashi, N.; Satoh, K.; Hatayama, I.; et al. An Nrf2/small maf heterodimer mediates the induction of phase ii detoxifying enzyme genes through antioxidant response elements. Biochem. Biophys. Res. Commun. 1997, 236, 313–322. [Google Scholar] [CrossRef]
- Baird, L.; Dinkova-Kostova, A.T. The cytoprotective role of the keap1-Nrf2 pathway. Arch. Toxicol. 2011, 85, 241–272. [Google Scholar] [CrossRef]
- Yates, M.S.; Tran, Q.T.; Dolan, P.M.; Osburn, W.O.; Shin, S.; McCulloch, C.C.; Silkworth, J.B.; Taguchi, K.; Yamamoto, M.; Williams, C.R.; et al. Genetic versus chemoprotective activation of Nrf2 signaling: Overlapping yet distinct gene expression profiles between keap1 knockout and triterpenoid-treated mice. Carcinogenesis 2009, 30, 1024–1031. [Google Scholar] [CrossRef]
- Okawa, H.; Motohashi, H.; Kobayashi, A.; Aburatani, H.; Kensler, T.W.; Yamamoto, M. Hepatocyte-specific deletion of the keap1 gene activates Nrf2 and confers potent resistance against acute drug toxicity. Biochem. Biophys. Res. Commun. 2006, 339, 79–88. [Google Scholar]
- Reichard, J.F.; Motz, G.T.; Puga, A. Heme oxygenase-1 induction by Nrf2 requires inactivation of the transcriptional repressor bach1. Nucleic Acids Res. 2007, 35, 7074–7086. [Google Scholar] [CrossRef]
- Yueh, M.F.; Tukey, R.H. Nrf2-keap1 signaling pathway regulates human ugt1a1 expression in vitro and in transgenic ugt1 mice. J. Biol. Chem. 2007, 282, 8749–8758. [Google Scholar] [CrossRef]
- Harvey, C.J.; Thimmulappa, R.K.; Singh, A.; Blake, D.J.; Ling, G.; Wakabayashi, N.; Fujii, J.; Myers, A.; Biswal, S. Nrf2-regulated glutathione recycling independent of biosynthesis is critical for cell survival during oxidative stress. Free Radic. Biol. Med. 2009, 46, 443–453. [Google Scholar] [CrossRef]
- Jung, K.A.; Choi, B.H.; Nam, C.W.; Song, M.; Kim, S.T.; Lee, J.Y.; Kwak, M.K. Identification of aldo-keto reductases as Nrf2-target marker genes in human cells. Toxicol. Lett. 2013, 218, 39–49. [Google Scholar] [CrossRef]
- Limonciel, A.; Wilmes, A.; Aschauer, L.; Radford, R.; Bloch, K.M.; McMorrow, T.; Pfaller, W.; van Delft, J.H.; Slattery, C.; Ryan, M.P.; et al. Oxidative stress induced by potassium bromate exposure results in altered tight junction protein expression in renal proximal tubule cells. Arch. Toxicol. 2012, 86, 1741–1751. [Google Scholar] [CrossRef]
- Wilmes, A.; Crean, D.; Aydin, S.; Pfaller, W.; Jennings, P.; Leonard, M.O. Identification and dissection of the Nrf2 mediated oxidative stress pathway in human renal proximal tubule toxicity. Toxicol. in Vitro 2011, 25, 613–622. [Google Scholar] [CrossRef]
- Wilmes, A.; Limonciel, A.; Aschauer, L.; Moenks, K.; Bielow, C.; Leonard, M.O.; Hamon, J.; Carpi, D.; Ruzek, S.; Handler, A.; et al. Application of integrated transcriptomic, proteomic and metabolomic profiling for the delineation of mechanisms of drug induced cell stress. J. Proteomics 2013, 79, 180–194. [Google Scholar] [CrossRef]
- Forti, E.; Bulgheroni, A.; Cetin, Y.; Hartung, T.; Jennings, P.; Pfaller, W.; Prieto, P. Characterisation of cadmium chloride induced molecular and functional alterations in airway epithelial cells. Cell. Phys. Biochem. 2010, 25, 159–168. [Google Scholar] [CrossRef]
- Forti, E.; Salovaara, S.; Cetin, Y.; Bulgheroni, A.; Tessadri, R.; Jennings, P.; Pfaller, W.; Prieto, P. In vitro evaluation of the toxicity induced by nickel soluble and particulate forms in human airway epithelial cells. Toxicol. in Vitro 2011, 25, 454–461. [Google Scholar] [CrossRef]
- Crean, D.; Felice, L.; Taylor, C.T.; Rabb, H.; Jennings, P.; Leonard, M.O. Glucose reintroduction triggers the activation of nrf2 during experimental ischemia reperfusion. Mol. Cell. Biochem. 2012, 366, 231–238. [Google Scholar] [CrossRef]
- Liu, F.; Ichihara, S.; Valentine, W.M.; Itoh, K.; Yamamoto, M.; Sheik Mohideen, S.; Kitoh, J.; Ichihara, G. Increased susceptibility of nrf2-null mice to 1-bromopropane-induced hepatotoxicity. Toxicol. Sci. 2010, 115, 596–606. [Google Scholar] [CrossRef]
- Copple, I.M.; Goldring, C.E.; Kitteringham, N.R.; Park, B.K. The nrf2-keap1 defence pathway: Role in protection against drug-induced toxicity. Toxicology 2008, 246, 24–33. [Google Scholar] [CrossRef]
- Aleksunes, L.M.; Goedken, M.J.; Rockwell, C.E.; Thomale, J.; Manautou, J.E.; Klaassen, C.D. Transcriptional regulation of renal cytoprotective genes by nrf2 and its potential use as a therapeutic target to mitigate cisplatin-induced nephrotoxicity. J. Pharmacol. Exp. Ther. 2010, 335, 2–12. [Google Scholar] [CrossRef]
- Enomoto, A.; Itoh, K.; Nagayoshi, E.; Haruta, J.; Kimura, T.; O’Connor, T.; Harada, T.; Yamamoto, M. High sensitivity of nrf2 knockout mice to acetaminophen hepatotoxicity associated with decreased expression of are-regulated drug metabolizing enzymes and antioxidant genes. Toxicol. Sci. 2001, 59, 169–177. [Google Scholar] [CrossRef]
- Sorrenti, V.; Di Giacomo, C.; Acquaviva, R.; Barbagallo, I.; Bognanno, M.; Galvano, F. Toxicity of ochratoxin a and its modulation by antioxidants: A review. Toxins 2013, 5, 1742–1766. [Google Scholar] [CrossRef]
- Schaaf, G.J.; Nijmeijer, S.M.; Maas, R.F.; Roestenberg, P.; de Groene, E.M.; Fink-Gremmels, J. The role of oxidative stress in the ochratoxin a-mediated toxicity in proximal tubular cells. Biochim. Biophys. Acta 2002, 1588, 149–158. [Google Scholar] [CrossRef]
- Marin-Kuan, M.; Nestler, S.; Verguet, C.; Bezencon, C.; Piguet, D.; Mansourian, R.; Holzwarth, J.; Grigorov, M.; Delatour, T.; Mantle, P.; et al. A toxicogenomics approach to identify new plausible epigenetic mechanisms of ochratoxin a carcinogenicity in rat. Toxicol. Sci. 2006, 89, 120–134. [Google Scholar]
- Cavin, C.; Delatour, T.; Marin-Kuan, M.; Holzhauser, D.; Higgins, L.; Bezencon, C.; Guignard, G.; Junod, S.; Richoz-Payot, J.; Gremaud, E.; et al. Reduction in antioxidant defenses may contribute to ochratoxin a toxicity and carcinogenicity. Toxicol. Sci. 2007, 96, 30–39. [Google Scholar]
- Kumar, R.; Ansari, K.M.; Chaudhari, B.P.; Dhawan, A.; Dwivedi, P.D.; Jain, S.K.; Das, M. Topical application of ochratoxin a causes DNA damage and tumor initiation in mouse skin. PloS ONE 2012, 7, e47280. [Google Scholar]
- Ramyaa, P.; Krishnaswamy, R.; Padma, V.V. Quercetin modulates ota-induced oxidative stress and redox signalling in hepg2 cells—Up regulation of nrf2 expression and down regulation of nf-kappab and cox-2. Biochim. Biophys. Acta 2013, 1840, 681–692. [Google Scholar]
- Boesch-Saadatmandi, C.; Wagner, A.E.; Graeser, A.C.; Hundhausen, C.; Wolffram, S.; Rimbach, G. Ochratoxin a impairs Nrf2-dependent gene expression in porcine kidney tubulus cells. J. Anim. Physiol. Anim. Nutr. 2009, 93, 547–554. [Google Scholar] [CrossRef]
- Boesch-Saadatmandi, C.; Loboda, A.; Jozkowicz, A.; Huebbe, P.; Blank, R.; Wolffram, S.; Dulak, J.; Rimbach, G. Effect of ochratoxin a on redox-regulated transcription factors, antioxidant enzymes and glutathione-s-transferase in cultured kidney tubulus cells. Food Chem. Toxicol. 2008, 46, 2665–2671. [Google Scholar] [CrossRef]
- Cavin, C.; Holzhaeuser, D.; Scharf, G.; Constable, A.; Huber, W.W.; Schilter, B. Cafestol and kahweol, two coffee specific diterpenes with anticarcinogenic activity. Food Chem. Toxicol. 2002, 40, 1155–1163. [Google Scholar] [CrossRef]
- Stachurska, A.; Ciesla, M.; Kozakowska, M.; Wolffram, S.; Boesch-Saadatmandi, C.; Rimbach, G.; Jozkowicz, A.; Dulak, J.; Loboda, A. Cross-talk between micrornas, nuclear factor e2-related factor 2, and heme oxygenase-1 in ochratoxin a-induced toxic effects in renal proximal tubular epithelial cells. Mol. Nutr. Food Res. 2013, 57, 504–515. [Google Scholar]
- Dopp, E.; Muller, J.; Hahnel, C.; Schiffmann, D. Induction of genotoxic effects and modulation of the intracellular calcium level in syrian hamster embryo (she) fibroblasts caused by ochratoxin A. Food Chem. Toxicol. 1999, 37, 713–721. [Google Scholar] [CrossRef]
- Kansanen, E.; Kuosmanen, S.M.; Leinonen, H.; Levonen, A.L. The Keap1-Nrf2 pathway: Mechanisms of activation and dysregulation in cancer. Redox Biol. 2013, 1, 45–49. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Limonciel, A.; Jennings, P. A Review of the Evidence that Ochratoxin A Is an Nrf2 Inhibitor: Implications for Nephrotoxicity and Renal Carcinogenicity. Toxins 2014, 6, 371-379. https://doi.org/10.3390/toxins6010371
Limonciel A, Jennings P. A Review of the Evidence that Ochratoxin A Is an Nrf2 Inhibitor: Implications for Nephrotoxicity and Renal Carcinogenicity. Toxins. 2014; 6(1):371-379. https://doi.org/10.3390/toxins6010371
Chicago/Turabian StyleLimonciel, Alice, and Paul Jennings. 2014. "A Review of the Evidence that Ochratoxin A Is an Nrf2 Inhibitor: Implications for Nephrotoxicity and Renal Carcinogenicity" Toxins 6, no. 1: 371-379. https://doi.org/10.3390/toxins6010371
APA StyleLimonciel, A., & Jennings, P. (2014). A Review of the Evidence that Ochratoxin A Is an Nrf2 Inhibitor: Implications for Nephrotoxicity and Renal Carcinogenicity. Toxins, 6(1), 371-379. https://doi.org/10.3390/toxins6010371



