Immunohistochemical Approach to Study Cylindrospermopsin Distribution in Tilapia (Oreochromis niloticus) under Different Exposure Conditions
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
2.1.1. Acute Dose Assay
| OC | IPC | OI24h | IPI24h | OI5d | IPI5d | |
|---|---|---|---|---|---|---|
| Liver | ||||||
| Hepatocytes | - | - | - | + | + | ++ |
| Pancreatic acini | - | - | + | ++ | + | + |
| Erythrocytes | - | - | - | - | + | ++ |
| Kidney | ||||||
| Tubules | - | - | - | - | + | + |
| Glomeruli | - | - | - | - | + | ++ |
| Erythrocytes | - | - | - | - | - | +++ |
| Intestine | ||||||
| Epithelium | - | - | - | - | - | ++ |
| Gills | ||||||
| Secondary lamellae | - | - | - | - | ++ | + |
| Erythrocytes | - | - | - | - | - | + |
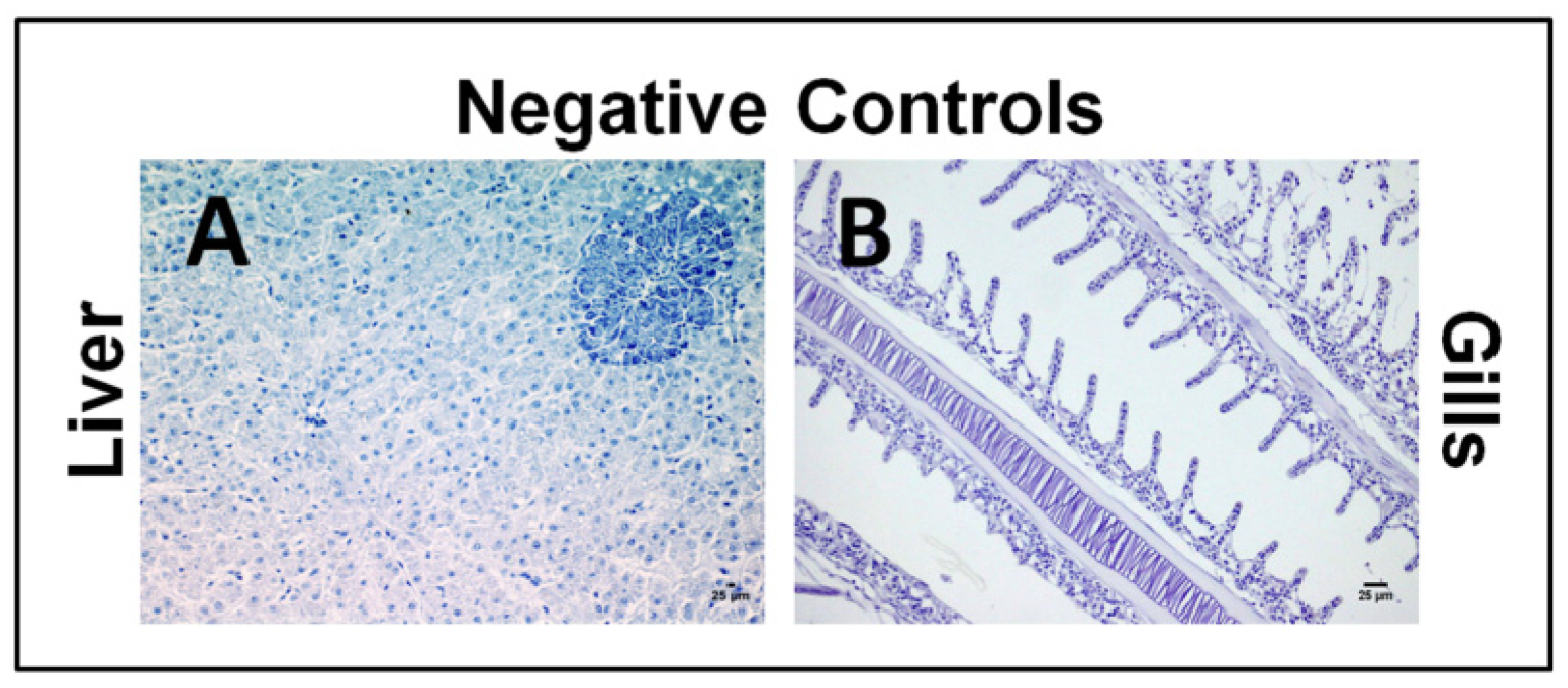


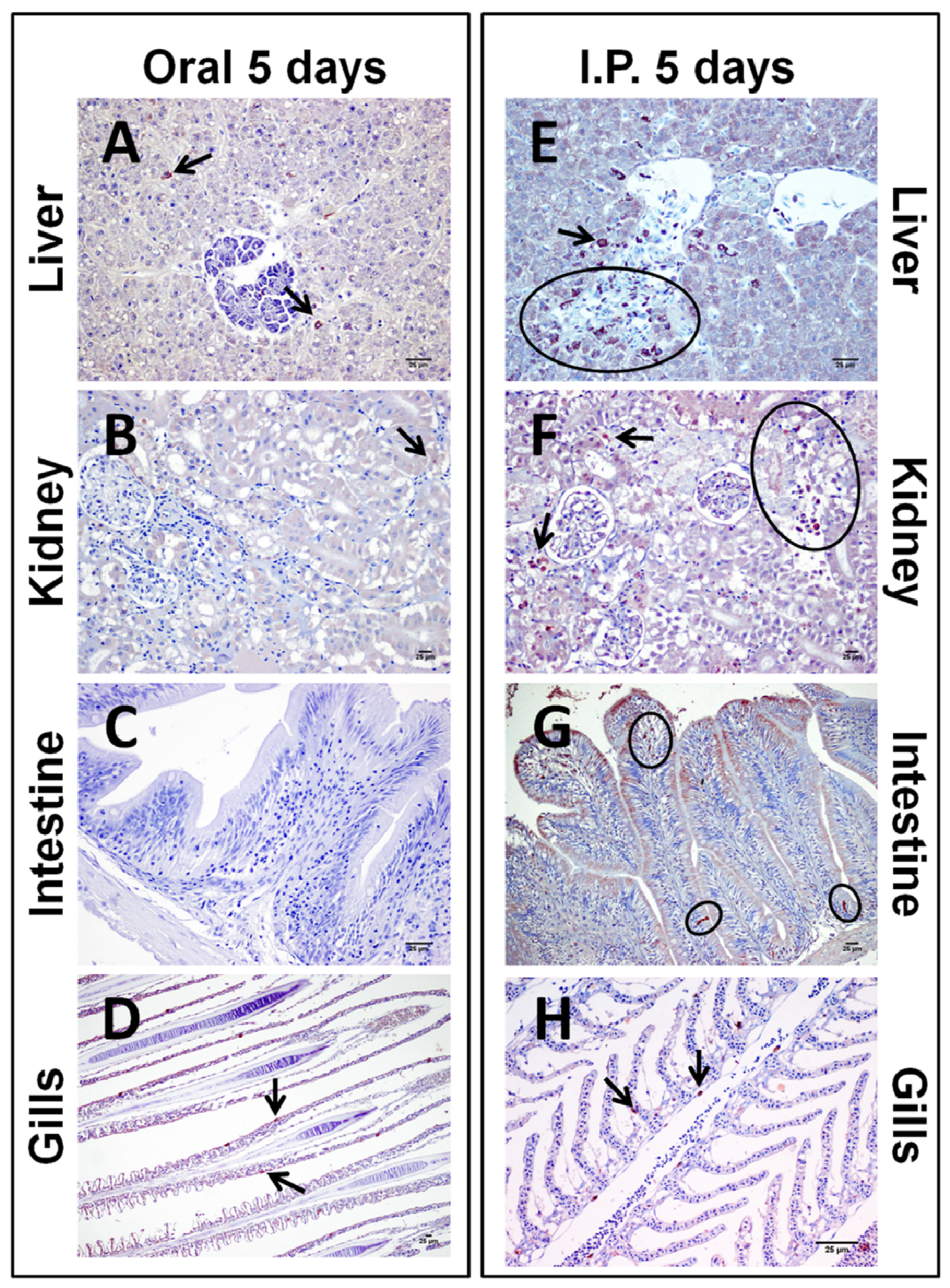
2.1.2. Sub-Chronic Dose Assay
| C7d | 10C7d | 100C7d | C14d | 10C14d | 100C14d | |
|---|---|---|---|---|---|---|
| Liver | ||||||
| Hepatocytes | - | +++ | +++ | - | +++ | +++ |
| Pancreatic acini | - | ++ | +++ | - | - | +++ |
| Erythrocytes | - | - | ++ | - | + | + |
| Kidney | ||||||
| Tubules | - | - | +++ | - | ++ | +++ |
| Glomeruli | - | - | ++ | - | +++ | +++ |
| Erythrocytes | - | - | ++ | - | + | + |
| Intestine | ||||||
| Epithelium | - | + | + | - | +++ | +++ |
| Gills | ||||||
| Secondary lamellae | - | + | ++ | - | ++ | +++ |
| Erythrocytes | - | + | + | - | - | + |

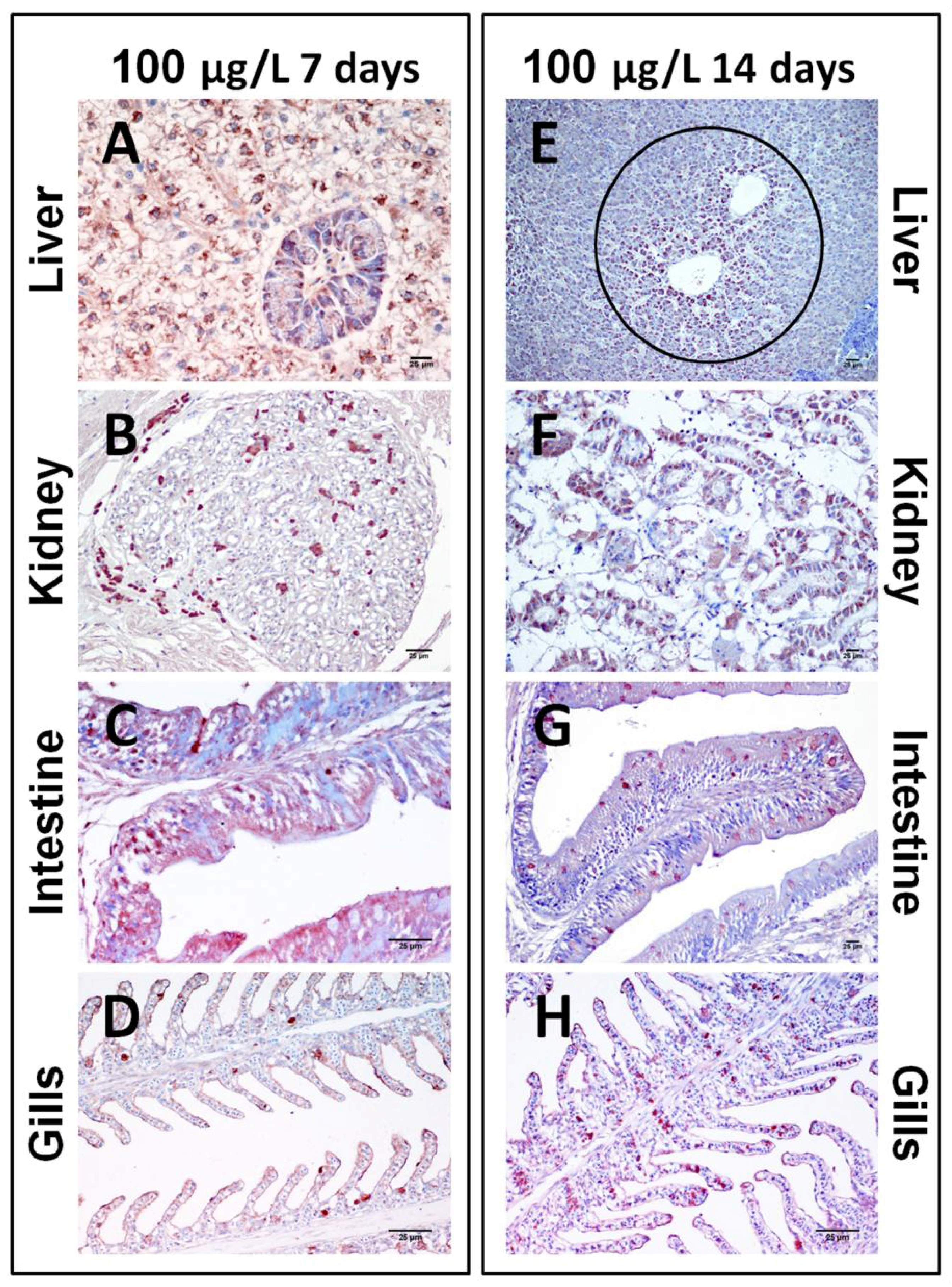
2.2. Discussion
3. Experimental Section
3.1. Chemicals
3.2. Aphanizomenon Ovalisporum Culture and Determination of Cyanobacterial Toxins
3.3. Experimental Setup and Acclimation of Fish
3.4. Experimental Exposure
| 24 h | 5 days | |||||||
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |
| NaCl solution by gavage | + | - | - | - | + | - | - | - |
| NaCl solution by i.p. injection | - | + | - | - | - | + | - | - |
| Pure CYN by gavage | - | - | + | - | - | - | + | - |
| Pure CYN by i.p. injection | - | - | - | + | - | - | - | + |
| 7 days | 14 days | |||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | |
| 10 µg/L of CYN | - | + | - | - | + | - |
| 100 µg/L of CYN | - | - | + | - | - | + |
3.5. Immunization with Cylindrospermopsin (CYN) and Preparation of Polyclonal Antiserum
3.6. Indirect ELISA
3.7. Immunohistochemical Analyses for the Detection of CYN in Tissues
| Antigen retrieval method | Dilutions of anti-CYN rabbit serum † | ||||
|---|---|---|---|---|---|
| 1:10 | 1:50 | 1:100 | 1:200 | ||
| None | - | - | - | - | |
| Tween 20® | - | - | - | - | |
| Protease | - | - | - | - | |
| CB pH 3.2 | oven | + | + | + | + |
| microwave (6 min) | + | + | + | - | |
| microwave (15 min) | - | - | - | - | |
| CB pH 6 | oven | Bs | Bs | +++ | + |
| microwave (6 min) | Bs | ++ | ++ | + | |
| microwave (15 min) | + | + | + | - | |
| CB pH 9 | oven | - | - | - | - |
| microwave (6 min) | - | - | - | - | |
| microwave (15 min) | - | - | - | - | |
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Moreira, C.; Azevedo, J.; Antunes, A.; Vasconcelos, V. Cylindrospermopsin: Occurrence, methods of detection and toxicology. J. Appl. Microbiol. 2012, 114, 605–620. [Google Scholar]
- Shaw, G.; Sukenik, A.; Livne, A.; Chiswell, R.K.; Smith, M.J.; Seawright, A.A.; Norris, R.L.; Eaglesham, G.K.; Moore, M.R. Blooms of the cylindrospermopsin containing cyanobacterium, Aphanizomenon ovalisporum (Forti), in newly constructed lakes, Queensland, Australia. Environ. Toxicol. 1999, 14, 167–177. [Google Scholar] [CrossRef]
- Ohtani, I.; Moore, R.E.; Runnegar, M. Cylindrospermopsin: A potent hepatotoxin from the blue-green alga Cylindrospermopsis raciborskii. Am. Chem. Soc. 1992, 114, 7941–7942. [Google Scholar] [CrossRef]
- Sivonen, K.; Jones, G. Cyanobacterial Toxins. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; Chorus, I., Bartram, M.J., Eds.; E & FN Spon: London, UK, 1999; pp. 41–111. [Google Scholar]
- Chiswell, R.K.; Shaw, G.R.; Eaglesham, G.; Smith, M.J.; Norris, R.L.; Seawright, A.A.; Moore, M.R. Stability of cylindrospermopsin, the toxin from the cyanobacterium, Cylindrospermopsis raciborskii: Effect of pH, temperature, and sunlight on decomposition. Environ. Toxicol. 1999, 14, 155–161. [Google Scholar] [CrossRef]
- Funari, E.; Testai, E. Human health risk assessment related to cyanotoxins exposure. Crit. Rev. Toxicol. 2008, 38, 97–125. [Google Scholar] [CrossRef]
- Terao, K.; Ohmori, S.; Igarashi, K.; Ohtani, I.; Watanabe, M.F.; Harada, K.I.; Ito, E.; Watanabe, M. Electron microscopic studies on experimental poisoning in mice induced by cylindrospermopsin isolated from blue-green alga Umezakia natans. Toxicon 1994, 32, 833–843. [Google Scholar] [CrossRef]
- Falconer, I.R.; Hardy, S.J.; Humpage, A.R.; Froscio, S.M.; Tozer, G.J.; Hawkins, P.R. Hepatic and renal toxicity of the blue-green alga (cyanobacterium) Cylindrospermopsis raciborskii in male Swiss albino mice. Environ. Toxicol. 1999, 14, 143–150. [Google Scholar]
- Seawright, A.A.; Nolan, C.C.; Shaw, G.R.; Chiswell, R.K.; Norris, R.L.; Moore, M.R.; Smith, M.K. The oral toxicity for mice of the tropical cyanobacterium Cylindrospermopsis raciborskii (Woloszynska). Environ. Toxicol. 1999, 14, 135–142. [Google Scholar]
- Runnegar, M.T.; Kong, S.M.; Zhong, Y.Z.; Lu, S.C. Inhibition of reduced glutathione synthesis by cyanobacterial alkaloid Cylindrospermopsin in cultured rat hepatocytes. Biochem. Pharmacol. 1995, 49, 219–225. [Google Scholar] [CrossRef]
- Humpage, A.R.; Fontaine, F.; Froscio, S.; Burcham, P.; Falconer, I.R. Cylindrospermopsin genotoxicity and cytotoxicity: Role of cytochrome P-450 and oxidative stress. J. Toxicol. Environ. Health A 2005, 68, 739–753. [Google Scholar] [CrossRef]
- Puerto, M.; Jos, A.; Pichardo, S.; Gutiérrez-Praena, D.; Cameán, A.M. Acute effects of pure Cylindrospermopsin on the activity and transcription of antioxidant enzymes in tilapia (Oreochromis niloticus) exposed by gavage. Ecotoxicology 2011, 20, 1852–1860. [Google Scholar] [CrossRef]
- Young, F.M.; Micklem, J.; Humpage, A.R. Effects of the blue-green algal toxin cylindrospermopsin (CYN) on human granulosa cells in vitro. Reprod. Toxicol. 2008, 25, 374–380. [Google Scholar] [CrossRef]
- Gutiérrez-Praena, D.; Jos., A.; Pichardo, S.; Moreno, I.M.; Cameán, A.M. Presence and bioaccumulation of Microcystin and Cylindrospermopsin in food and the effectiveness of some cooking techniques at decreasing their concentrations: A review. Food Chem. Toxicol. 2013, 53, 139–152. [Google Scholar] [CrossRef]
- White, S.H.; Duivenvoorden, L.J.; Fabbro, L.D.; Eaglesham, G.K. Influence of intracellular toxin concentrations on cylindrospermopsin bioaccumulation in a freshwater gastropod (Melanoides tuberculata). Toxicon 2006, 47, 497–509. [Google Scholar] [CrossRef]
- Berry, J.P.; Lind, O. First evidence of “paralytic shellfish toxins” and cylindrospermopsin in a Mexican freshwater system, Lago Catemaco, and apparent bioaccumulation of the toxins in “tegogolo” snails (Pomacea patula catemacensis). Toxicon 2010, 55, 930–938. [Google Scholar] [CrossRef]
- Anderson, L.; Fabbro, L.D.; Cowden, K. Assessment of Blue-Green Algal Toxins in Barramundi, Red Clay and Mussels from Awoonga Dam; Central Queensland University: Gladstone, Australia, 2003. [Google Scholar]
- Saker, M.L.; Metcalf, J.S.; Codd, G.A.; Vasconcelos, V.M. Accumulation and depuration of the cyanobacterial toxin cylindrospermopsin in the freshwater mussel Anodonta cygnea. Toxicon 2004, 43, 185–194. [Google Scholar] [CrossRef]
- Saker, M.L.; Eaglesham, G.K. The accumulation of cylindrospermopsin from the cyanobacterium Cylindrospermopsis raciborskii in tissues of the Redclaw crayfish Cherax quadricarinatus. Toxicon 1999, 37, 1065–1077. [Google Scholar] [CrossRef]
- White, S.H.; Duivenvoorden, L.J.; Fabbro, L.D.; Eaglesham, G.K. Mortality and toxin bioaccumulation in Bufo marinus following exposure to Cylindrospermopsis raciborskii cell extracts and live cultures. Environ. Pollut. 2007, 147, 158–167. [Google Scholar] [CrossRef]
- Messineo, V.; Melchiorre, S.; Di Corcia, A.; Gallo, P.; Bruno, M. Seasonal succession of Cylindrospermopsis raciborskii and Aphanizomenon Ovalisporum blooms with cylindrospermopsin occurrence in the volcanic Lake Albano, Central Italy. Environ. Toxicol. 2010, 25, 18–27. [Google Scholar]
- Berry, J.P.; Jaja-Chimedza, A.; Dávalos-Lind, L.; Lind, O. Apparent bioaccumulation of cylindrospermopsin and paralytic shellfish toxins by finfish in Lake Catemaco (Veracruz, Mexico). Food Addit. Contam. 2012, 2, 314–321. [Google Scholar]
- Kinnear, S. Cylindrospermopsin: A decade of progress on bioaccumulation research. Mar. Drugs 2010, 8, 542–564. [Google Scholar] [CrossRef]
- Norris, R.L.G.; Eaglesham, G.K.; Pierens, G.; Shaw, G.R.; Smith, M.J.; Chiswell, R.K.; Seawright, A.A.; Moore, M.R. Deoxycylindrospermopsin, an analog of cylindrospermopsin from Cylindrospermopsis raciborskii. Environ. Toxicol. 1999, 14, 163–165. [Google Scholar] [CrossRef]
- Seifert, M.; Mcgregor, G.; Eaglesham, G.; Wickramasinghe, W.; Shaw, G. First evidence for the production of cylindrospermopsin and deoxy-cylindrospermopsin by the freshwater benthic cyanobacterium, Lyngbya wollei (Farlow ex Gomont) Speziale and Dyck. Harmful Algae 2007, 6, 73–80. [Google Scholar]
- Hawkins, P.R.; Runnegar, M.T.C.; Jackson, A.R.B.; Falconer, I.R. Severe hepatotoxicity caused by the tropical cyanobacterium (blue-green alga) Cylindrospermopsis raciborskii (Woloszynska) Seenaya and Subba Raju isolated form a domestic supply reservoir. Appl. Environ. Microbiol. 1985, 50, 1292–1295. [Google Scholar]
- Berry, J.P.; Gibbs, P.D.L.; Schmale, M.C.; Saker, M.L. Toxicity of cylindrospermopsin, and other apparent metabolites from Cylindrospermopsis raciborskii and Aphanizomenon ovalisporum, to the zebrafish (Danio rerio) embryo. Toxicon 2009, 53, 289–299. [Google Scholar] [CrossRef]
- Puerto, M.; Jos, A.; Pichardo, S.; Moyano, R.; Blanco, A.; Cameán, A.M. Acute exposure to pure Cylindrospermopsin results in oxidative stress and pathological alterations in tilapia (Oreochromis niloticus). Environ. Toxicol. 2012. [Google Scholar] [CrossRef]
- Gutiérrez-Praena, D.; Jos, A.; Pichardo, S.; Cameán, A.M. Oxidative stress responses in tilapia (Oreochromis niloticus) exposed to a single dose of pure Cylindrospermopsin under laboratory conditions: Influence of exposure route and time of sacrifice. Aquat. Toxicol. 2011, 105, 100–106. [Google Scholar] [CrossRef]
- Gutiérrez-Praena, D.; Jos, A.; Pichardo, S.; Moyano, R.; Blanco, A.; Monterde, J.G.; Cameán, A.M. Time-dependent histopathological changes induced in tilapia (Oreochromis niloticus) after acute exposure to pure Cylindrospermopsin by oral and intraperitoneal route. Ecotoxicol. Environ. Saf. 2012, 76, 102–113. [Google Scholar] [CrossRef]
- Gutiérrez-Praena, D.; Puerto, M.; Prieto, A.I.; Jos, A.; Pichardo, S.; Vasconcelos, V.; Cameán, A.M. Protective role of dietary N-Acetylcysteine on the oxidative stress induced by Cylindrospermopsin in tilapia (Oreochromis niloticus). Environ. Toxicol. Chem. 2012, 31, 1548–1555. [Google Scholar] [CrossRef]
- Yoshida, T.; Makita, Y.; Tsutsumi, T.; Nagata, S.; Tashiro, F.; Yoshida, F.; Sekijima, M.; Tamura, S.I.; Harada, T.; Maita, K.; et al. Immunohistochemical localization of Microcystin-LR in the liver of mice: A study on the pathogenesis of Microcystin-LR-induced hepatotoxicity. Toxicol. Pathol. 1998, 26, 411–418. [Google Scholar]
- Guzmán, R.E.; Solter, P.F. Characterization of sublethal microcystin-LR exposure in mice. Vet. Pathol. 2002, 39, 17–26. [Google Scholar] [CrossRef]
- Fischer, W.J.; Hitzfeld, B.C.; Tencalla, F.; Eriksson, J.E.; Mikhailov, A.; Dietrich, D.R. Microcystin-LR toxicodynamics, induced pathology, and immunohistochemical localization in livers of blue-green algae exposed Rainbow trout (Oncorhynchus mykiss). Toxicol. Sci. 2000, 54, 365–373. [Google Scholar] [CrossRef]
- Lance, E.; Josso, C.; Dietrich, D.; Ernst, B.; Paty, C.; Senger, F.; Bormans, M.; Gérard, C. Histopathology and Microcystin distribution in Lymnaea stagnalis (Gastropoda) following toxic cyanobacterial or dissolved microcystin-LR exposure. Aquat. Toxicol. 2010, 98, 211–220. [Google Scholar] [CrossRef]
- Niedobitek, G.; Kremmer, E.; Herbst, H.; Whitehead, L.; Dawson, C.W.; Niedobitek, E.; von Ostau, C.; Rooney, N.; Grässer, F.A.; Young, L.S. Immunohistochemical detection of the Epstein-Barr Virus-encoded latent membrane protein 2A in Hodkin’s disease and infectious mononucleosis. Blood 1997, 90, 1664–1672. [Google Scholar]
- Risalde, M.A.; Molina, V.; Sánchez-Cordón, P.J.; Pedrera, M.; Romero-Palomo, F.; Bautista, M.J.; Moreno, A.; Gómez-Villamandos, J.C. Comparison of pathologic changes and viral antigen distribution in tissues of calves with and without pre-existing bovine viral diarrhea virus infection following challenge with bovine Herpesvirus-1. Am. J. Vet. Res. 2013, 74, 598–610. [Google Scholar] [CrossRef]
- Oosterwijk, E.; Ruiter, M.D.; Wakka, J.C.; Huiskens-van der Meij, J.W.; Jonas, U.; Fleuren, G.J.; Zwartendijk, J.; Hoedemaeker, P.; Warnaar, S.O. Immunohistochemical analysis of monoclonal antibodies to renal antigens. Application in the diagnosis of renal cell carcinoma. J. Am. Pathol. 1986, 123, 301–309. [Google Scholar]
- Guzmán-Guillén, R.; Prieto, A.I.; Moreno, I.; Vasconcelos, V.M.; Moyano, R.; Blanco, A.; Cameán, A.M. Cyanobacterium producing Cylindrospermopsin cause histopathological changes at environmentally relevant concentrations in sub-chronically exposed tilapia (Oreochromis niloticus). Environ. Toxicol. 2013. [Google Scholar] [CrossRef]
- Ito, E.; Kondo, F.; Harada, K.H. First report of the distribution of orally administered Microcystin-LR in mouse tissue using an immunostaining method. Toxicon 2000, 38, 37–48. [Google Scholar] [CrossRef]
- Humpage, A.R.; Falconer, I.R. Oral toxicity of the cyanobacterial toxin cylindrospermopsin in male Swiss albino mice: Determination of no observed adverse effect level for deriving a drinking water guideline value. Environ. Toxicol. 2003, 18, 94–103. [Google Scholar] [CrossRef]
- Norris, R.L.G.; Seawright, A.A.; Shaw, G.R.; Smith, M.J.; Chiswell, R.K.; Moore, M.R. Distribution of 14C Cylindrospermopsin in vivo in the Mouse. Environ. Toxicol. 2001, 16, 498–505. [Google Scholar] [CrossRef]
- Guzmán-Guillén, R.; Prieto, A.I.; Moreno, I.M.; González, G.; Soria-Díaz, M.E.; Vasconcelos, V.; Cameán, A.M. Development and optimization of a method for the determination of Cylindrospermopsin from strains of Aphanizomenon cultures: Intra-laboratory assessment of its accuracy by using validation standards. Talanta 2012, 100, 356–363. [Google Scholar]
- Hancock, D.C.; O’Reilly, N.J. Production of Polyclonal Antibodies in Rabbits. Methods Mol. Biol. 2005, 295, 27–40. [Google Scholar]
- Elliot, C.T.; Redshaw, C.H.; George, S.E.; Campbell, K. First development and characterization of polyclonal and monoclonal antibodies to the emerging fresh water toxin Cylindrospermopsin. Harmful Algae 2013, 24, 10–19. [Google Scholar] [CrossRef]
- Straus, W. Cleavage of heme from horseradish peroxidase by methanol with inhibition of enzyme activity. J. Histochem. Cytochem. 1974, 22, 908–911. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Guzmán-Guillén, R.; Gutiérrez-Praena, D.; De los Ángeles Risalde, M.; Moyano, R.; Prieto, A.I.; Pichardo, S.; Jos, Á.; Vasconcelos, V.; Cameán, A.M. Immunohistochemical Approach to Study Cylindrospermopsin Distribution in Tilapia (Oreochromis niloticus) under Different Exposure Conditions. Toxins 2014, 6, 283-303. https://doi.org/10.3390/toxins6010283
Guzmán-Guillén R, Gutiérrez-Praena D, De los Ángeles Risalde M, Moyano R, Prieto AI, Pichardo S, Jos Á, Vasconcelos V, Cameán AM. Immunohistochemical Approach to Study Cylindrospermopsin Distribution in Tilapia (Oreochromis niloticus) under Different Exposure Conditions. Toxins. 2014; 6(1):283-303. https://doi.org/10.3390/toxins6010283
Chicago/Turabian StyleGuzmán-Guillén, Remedios, Daniel Gutiérrez-Praena, María De los Ángeles Risalde, Rosario Moyano, Ana Isabel Prieto, Silvia Pichardo, Ángeles Jos, Vitor Vasconcelos, and Ana María Cameán. 2014. "Immunohistochemical Approach to Study Cylindrospermopsin Distribution in Tilapia (Oreochromis niloticus) under Different Exposure Conditions" Toxins 6, no. 1: 283-303. https://doi.org/10.3390/toxins6010283










