Fates of Microcystis aeruginosa Cells and Associated Microcystins in Sediment and the Effect of Coagulation Process on Them
Abstract
:1. Introduction
2. Results
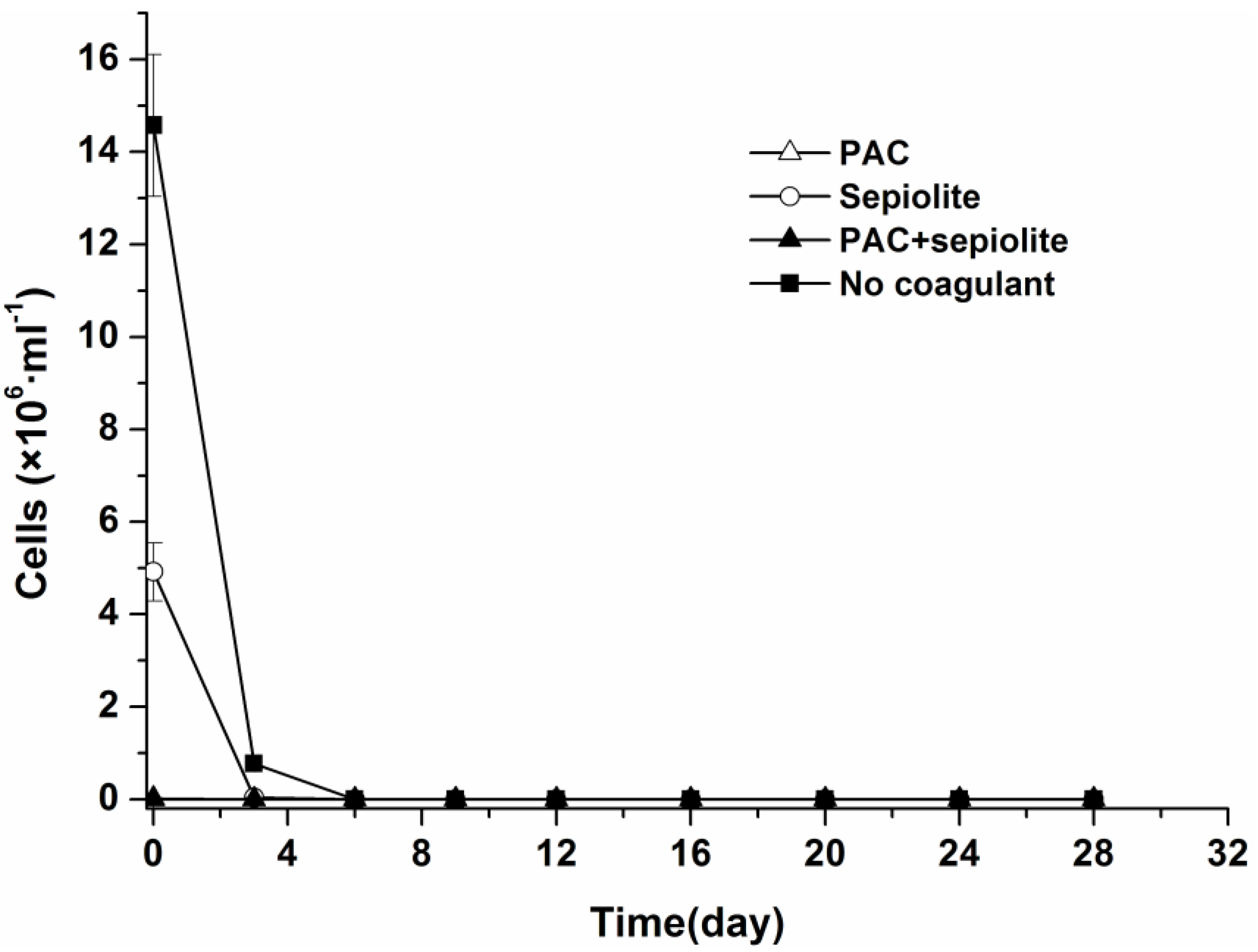
2.1. Natural Settlement and Coagulation Efficiency
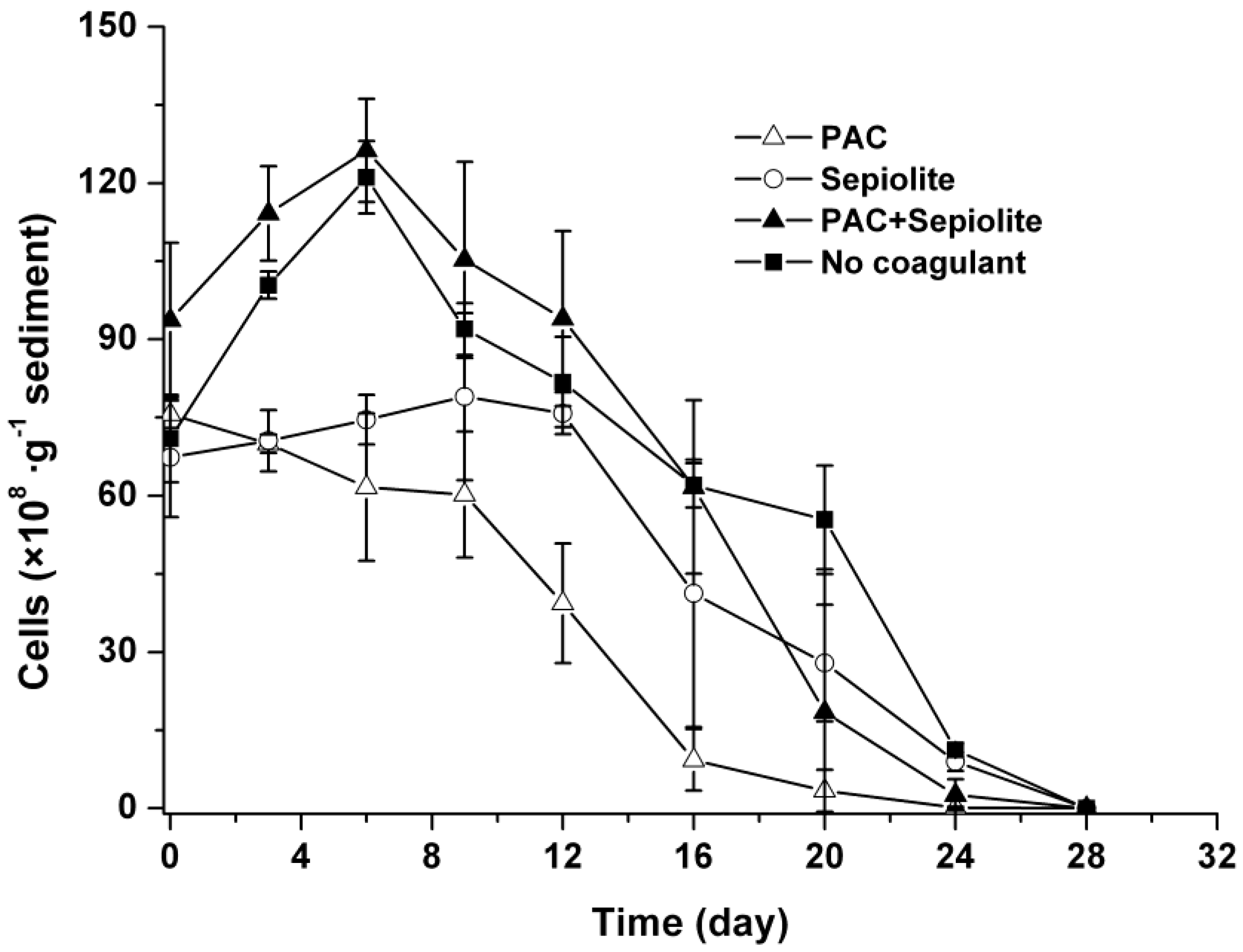
2.2. Dynamics of Microcystis aeruginosa in Sediments

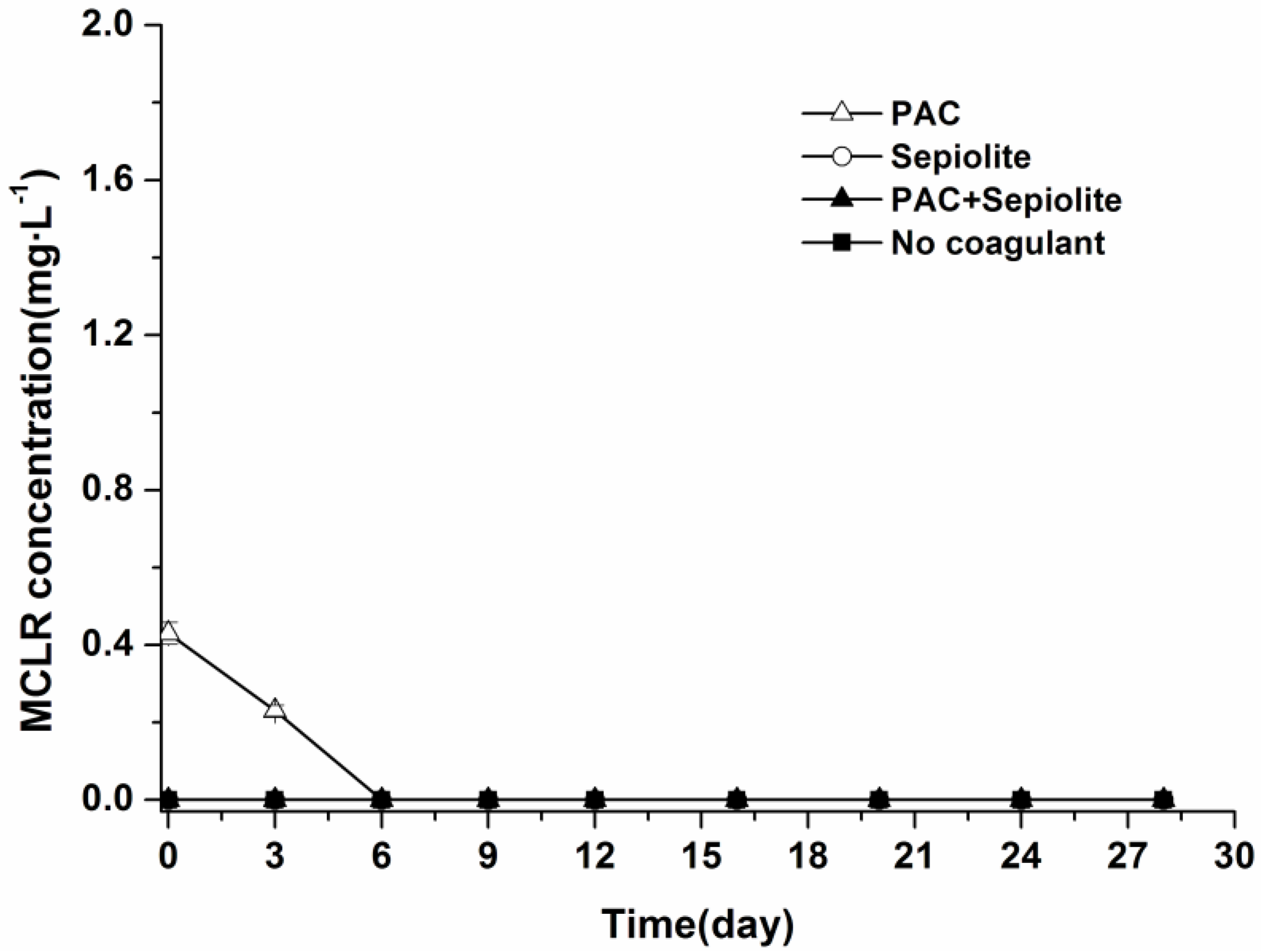
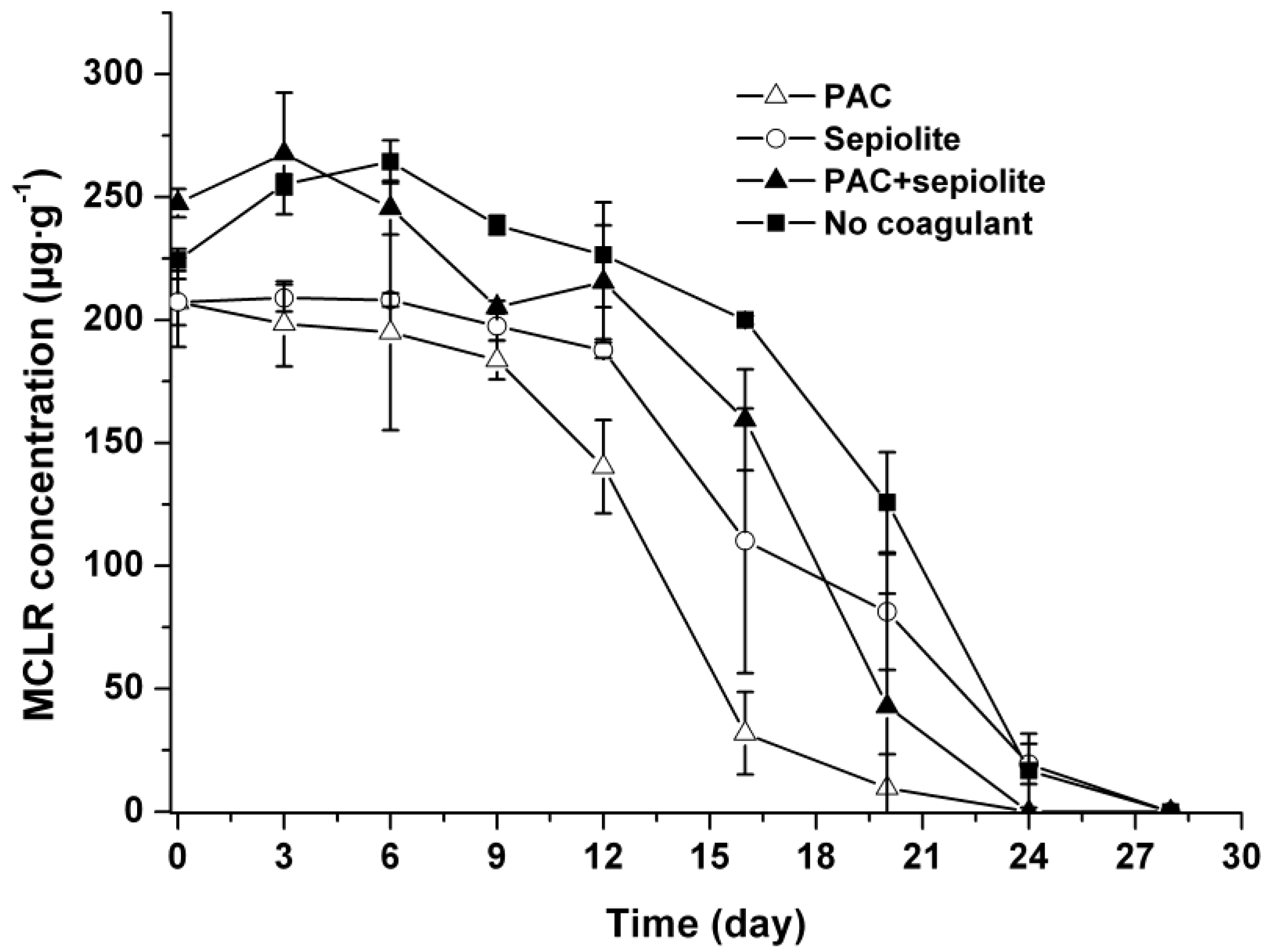
2.3. Microcystin Release and Degradation
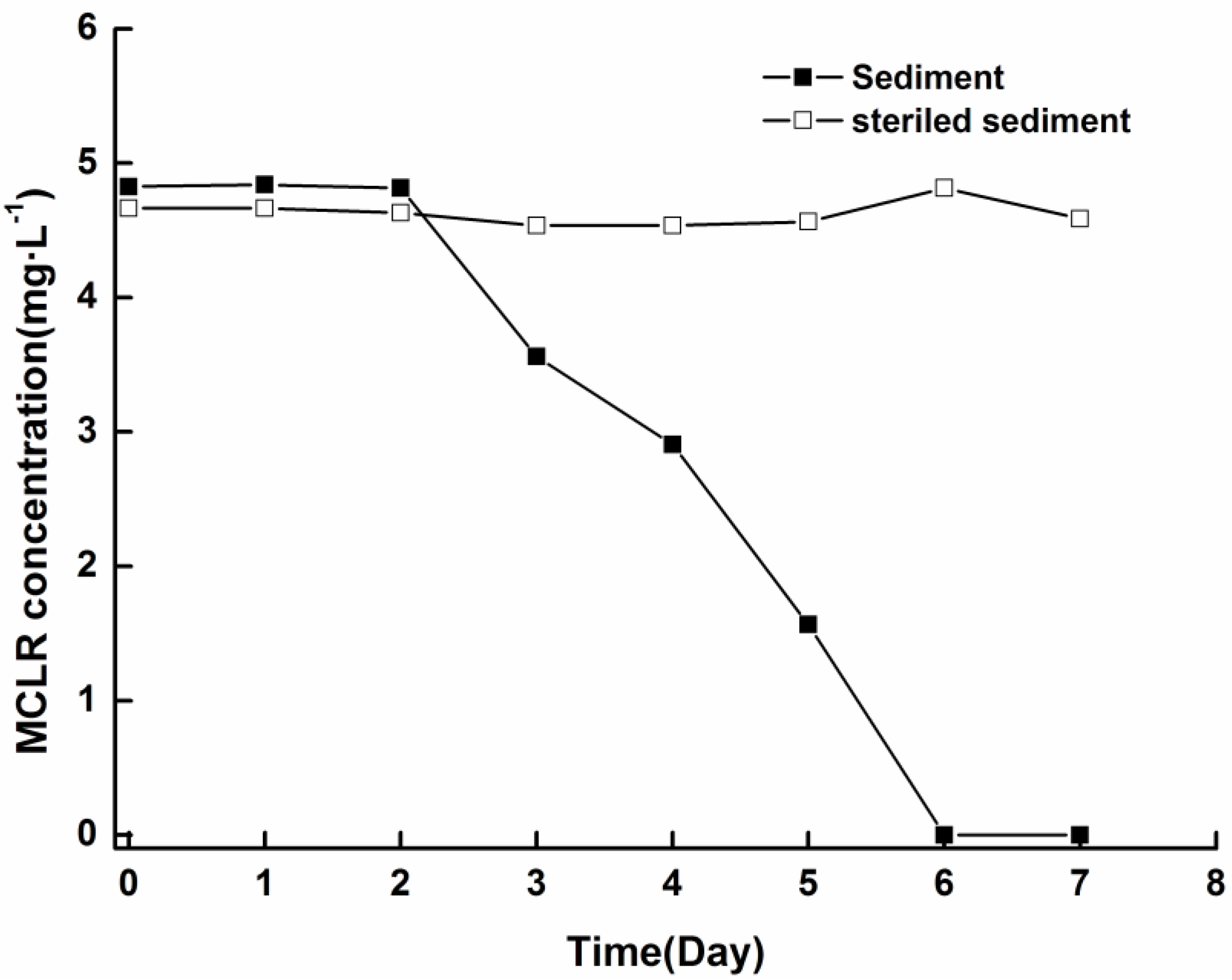
2.4. TN in Water Phase
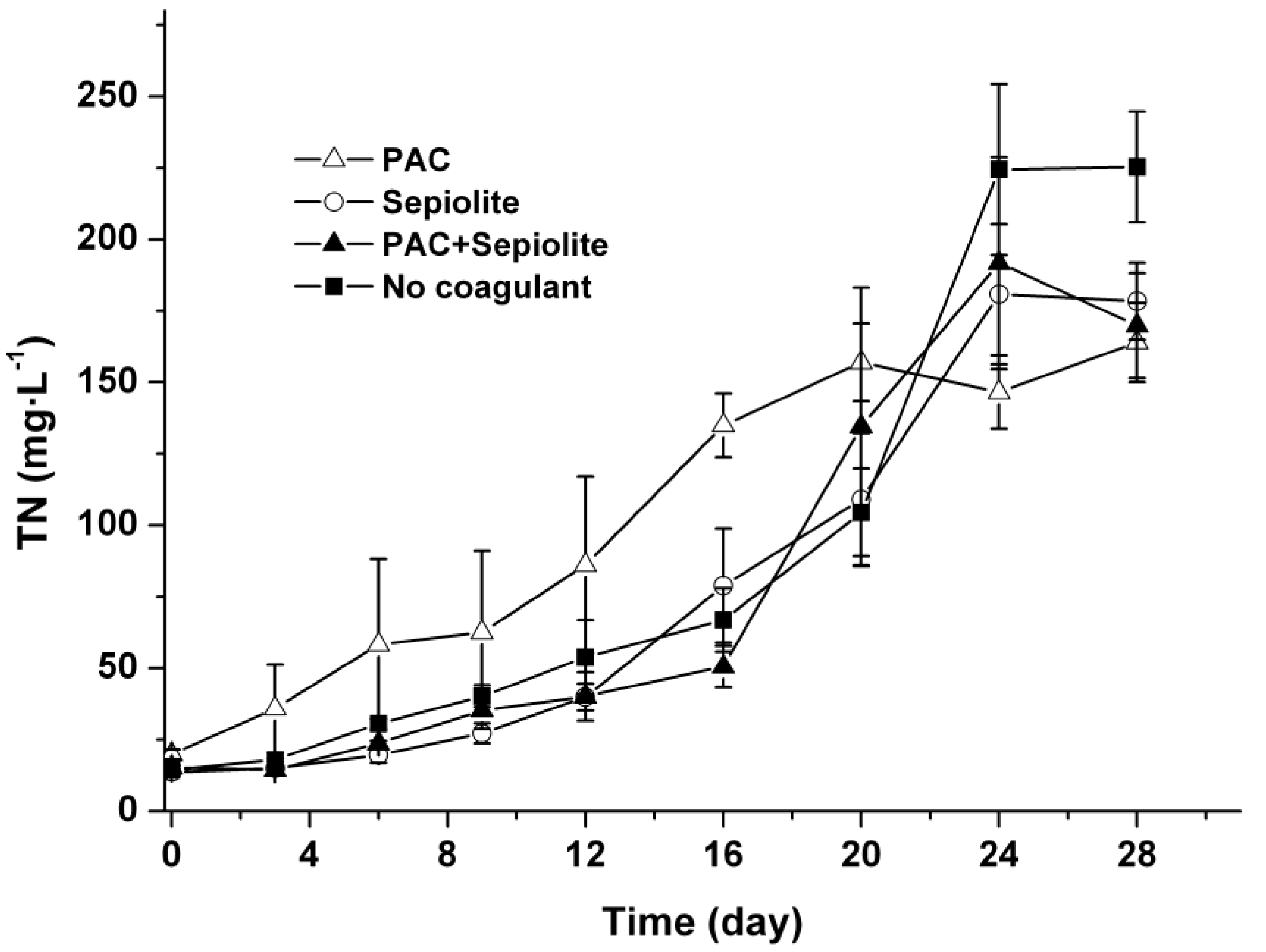
3. Discussion
4. Experimental Section
4.1. Microcystis aeruginosa
4.2. Sediment Samples
4.3. Experimental Design
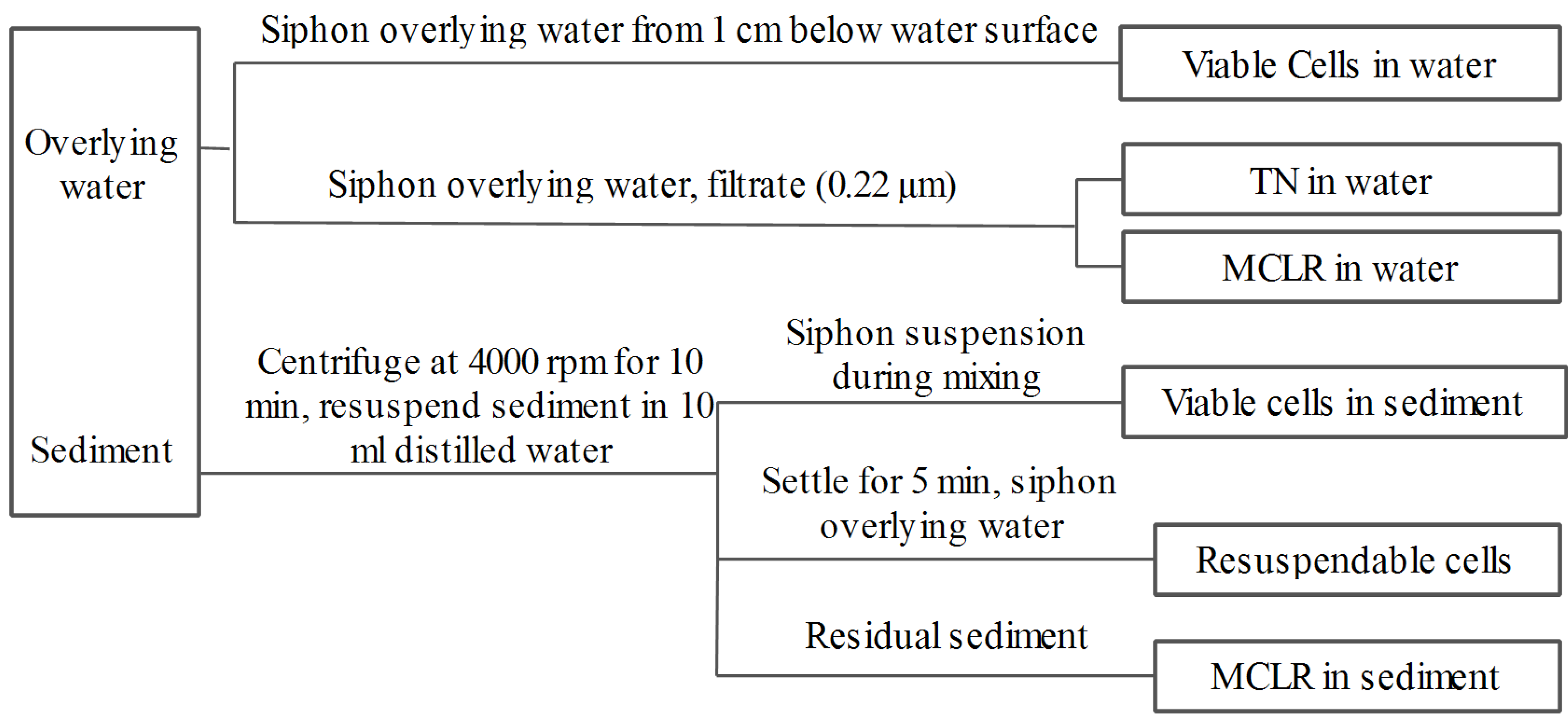
4.4. Biodegradability of Soluble MCs in Sediment
4.5. Viable Cells Assay
4.6. MC Extraction and Analysis
4.7. Statistical Analysis
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Dionysiou, D. Overview: Harmful algal blooms and natural toxins in fresh and marine waters—Exposure, occurrence, detection, toxicity, control, management and policy. Toxicon 2010, 55, 907–908. [Google Scholar] [CrossRef]
- Li, H.; Xing, P.; Chen, M.; Bian, Y.; Wu, Q.L. Short-term bacterial community composition dynamics in response to accumulation and breakdown of Microcystis blooms. Water Res. 2011, 45, 1702–1710. [Google Scholar] [CrossRef]
- Graham, J.L.; Loftin, K.A.; Meyer, M.T.; Ziegler, A.C. Cyanotoxin mixtures and taste-and-odor compounds in cyanobacterial blooms from the Midwestern United States. Environ. Sci. Technol. 2010, 44, 7361–7368. [Google Scholar] [CrossRef]
- Dorr, F.A.; Pinto, E.; Soares, R.M.; Feliciano de Oliveira e Azevedo, S.M. Microcystins in South American aquatic ecosystems: Occurrence, toxicity and toxicological assays. Toxicon 2010, 56, 1247–1256. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Guidelines for Drinking-Water Quality. In Addendum to Volume 2: Health Criteria and Other Supporting Information; WHO: Geneva, Switzerland, 1998. [Google Scholar]
- Ihle, T.; Jähnichen, S.; Benndorf, J. Wax and wane of microcystis (Cyanophyceae) and microcystins in lake sediments: A case study in Quitzdorf Reservoir (Germany). J. Phycol. 2005, 41, 479–488. [Google Scholar] [CrossRef]
- Welker, M.; Šejnohová, L.; Némethová, D.; von Döhren, H.; Jarkovský, J.; Maršálek, B. Seasonal shifts in chemotype composition of Microcystis sp. Communities in the pelagial and the sediment of a shallow reservoir. Limnol. Oceanogr. 2007, 52, 609–619. [Google Scholar] [CrossRef]
- Kankaanpää, H.T.; Sjövall, O.; Huttunen, M.; Olin, M.; Karlsson, K.; Hyvärinen, K.; Sneitz, L.; Härkönen, J.; Sipiä, V.O.; Meriluoto, J.A.O. Production and sedimentation of peptide toxins nodularin-R and microcystin-LR in the northern Baltic Sea. Environ. Pollut. 2009, 157, 1301–1309. [Google Scholar] [CrossRef]
- Cirés, S.; Wörmer, L.; Carrasco, D.; Quesada, A. Sedimentation patterns of toxin-producing Microcystis morphospecies in freshwater reservoirs. Toxins 2013, 5, 939–957. [Google Scholar] [CrossRef]
- Wörmer, L.; Cirés, S.; Quesada, A. Importance of natural sedimentation in the fate of microcystins. Chemosphere 2011, 82, 1141–1146. [Google Scholar] [CrossRef]
- Brunberg, A.-K.; Blomqvist, P. Benthic overwintering of Microcystis colonies under different environmental conditions. J. Plankton Res. 2002, 24, 1247–1252. [Google Scholar] [CrossRef]
- Tsujimura, S.; Tsukada, H.; Nakahara, H.; Nakajima, T.; Nishino, M. Seasonal variations of Microcystis populations in sediments of Lake Biwa, Japan. Hydrobiologia 2000, 434, 183–192. [Google Scholar] [CrossRef]
- Oberholster, P.J.; Botha, A.M.; Cloete, T.E. Use of molecular markers as indicators for winter zooplankton grazing on toxic benthic cyanobacteria colonies in an urban Colorado lake. Harmful Algae 2006, 5, 705–716. [Google Scholar] [CrossRef]
- Mohamed, Z.A.; Al-Shehri, A.M. Grazing on Microcystis aeruginosa and degradation of microcystins by the heterotrophic flagellate Diphylleia rotans. Ecotox. Environ. Safe. 2013, 96, 48–52. [Google Scholar] [CrossRef]
- Schöne, K.; Jähnichen, S.; Ihle, T.; Ludwig, F.; Benndorf, J. Arriving in better shape: Benthic Microcystis as inoculum for pelagic growth. Harmful Algae 2010, 9, 494–503. [Google Scholar] [CrossRef]
- Kim, S.G.; Joung, S.H.; Ahn, C.Y.; Ko, S.R.; Boo, S.M.; Oh, H.M. Annual variation of Microcystis genotypes and their potential toxicity in water and sediment from a eutrophic reservoir. FEMS Microbiol. Ecol. 2010, 74, 93–102. [Google Scholar] [CrossRef]
- Johnston, B.R.; Jacoby, J.M. Cyanobacterial toxicity and migration in a mesotrophic lake in western Washington, USA. Hydrobiologia 2003, 495, 79–91. [Google Scholar] [CrossRef]
- Rinta-Kanto, J.M.; Saxton, M.A.; DeBruyn, J.M.; Smith, J.L.; Marvin, C.H.; Krieger, K.A.; Sayler, G.S.; Boyer, G.L.; Wilhelm, S.W. The diversity and distribution of toxigenic Microcystis spp. in present day and archived pelagic and sediment samples from Lake Erie. Harmful Algae 2009, 8, 385–394. [Google Scholar] [CrossRef]
- Rohrlack, T.; Hyenstrand, P. Fate of intracellular microcystins in the cyanobacterium Microcystis aeruginosa (Chroococcales, Cyanophyceae). Phycologia 2007, 46, 277–283. [Google Scholar] [CrossRef]
- Tsuji, K.; Masui, H.; Uemura, H.; Mori, Y.; Harada, K.-I. Analysis of microcystins in sediments using MMPB method. Toxicon 2001, 39, 687–692. [Google Scholar] [CrossRef]
- Chen, W.; Song, L.; Peng, L.; Wan, N.; Zhang, X.; Gan, N. Reduction in microcystin concentrations in large and shallow lakes: Water and sediment-interface contributions. Water Res. 2008, 42, 763–773. [Google Scholar] [CrossRef]
- Mohamed, Z.A.; El-Sharouny, H.M.; Ali, W.S. Microcystin concentrations in the Nile river sediments and removal of microcystin-LR by sediments during batch experiments. Arch. Environ. Contam. Toxicol. 2007, 52, 489–495. [Google Scholar] [CrossRef]
- Hitzfeld, B.C.; Hoger, S.J.; Dietrich, D.R. Cyanobacterial toxins: Removal during drinking water treatment, and human risk assessment. Environ. Health Perspect. 2000, 108, 113–122. [Google Scholar]
- Anderson, D.M. Approaches to monitoring, control and management of harmful algal blooms (HABs). Ocean. Coastal Manag. 2009, 52, 342–347. [Google Scholar] [CrossRef]
- Zamyadi, A.; Dorner, S.; Sauve, S.; Ellis, D.; Bolduc, A.; Bastien, C.; Prevost, M. Species-dependence of cyanobacteria removal efficiency by different drinking water treatment processes. Water Res. 2013, 47, 2689–2700. [Google Scholar] [CrossRef]
- Sun, F.; Pei, H.-Y.; Hu, W.-R.; Li, X.-Q.; Ma, C.-X.; Pei, R.-T. The cell damage of Microcystis aeruginosa in PACl coagulation and floc storage processes. Sep. Purif. Technol. 2013, 115, 123–128. [Google Scholar] [CrossRef]
- Hall, T.; Hart, J.; Croll, B.; Gregory, R. Laboratory-scale investigations of algal toxin removal by water treatment. J. Chart. Inst. Water. Environ. Manag. 2000, 14, 143–149. [Google Scholar] [CrossRef]
- Drikas, M.; Chow, C.W.K.; House, J.; Burch, M.D. Using coagulation, flocculation, and settling to remove toxic cyanobacteria. J. Am. Water Work Assoc. 2001, 93, 100–111. [Google Scholar]
- Jurczak, T.; Tarczynska, M.; Izydorczyk, K.; Mankiewicz, J.; Zalewski, M.; Meriluoto, J. Elimination of microcystins by water treatment processes—Examples from Rulejow Reservoir, Poland. Water Res. 2005, 39, 2394–2406. [Google Scholar] [CrossRef]
- Henderson, R.; Parsons, S.A.; Jefferson, B. The impact of algal properties and pre-oxidation on solid-liquid separation of algae. Water Res. 2008, 42, 1827–1845. [Google Scholar] [CrossRef]
- Pan, G.; Yang, B.; Wang, D.; Chen, H.; Tian, B.-H.; Zhang, M.-L.; Yuan, X.-Z.; Chen, J. In-lake algal bloom removal and submerged vegetation restoration using modified local soils. Ecol. Eng. 2011, 37, 302–308. [Google Scholar] [CrossRef]
- Lürling, M.; Oosterhout, F.V. Controlling eutrophication by combined bloom precipitation and sediment phosphorus inactivation. Water Res. 2013, 47, 6527–6537. [Google Scholar] [CrossRef]
- Pilgrim, K.M.; Brezonik, P.L. Evaluation of the potential adverse effects of lake inflow treatment with alum. Lake Reserv. Manag. 2005, 21, 77–87. [Google Scholar]
- MacDiarmid, C.W.; Gardner, R.C. Overexpression of the Saccharomyces cerevisiaemagnesium transport system confers resistance to aluminum ion. J. Biol. Chem. 1998, 273, 1727–1732. [Google Scholar] [CrossRef]
- Ståhl-Delbanco, A.; Hansson, L.-A. Effects of bioturbation on recruitment of algal cells from the “seed bank” of lake sediments. Limnol. Oceanogr. 2002, 47, 1836–1843. [Google Scholar] [CrossRef]
- Ståhl-Delbanco, A.; Hansson, L.-A.; Gyllström, M. Recruitment of resting stages may induce blooms of Microcystis at low n:P ratios. J. Plankton Res. 2003, 25, 1099–1106. [Google Scholar] [CrossRef]
- Yamamoto, Y. Contribution of bioturbation by the red swamp crayfish Procambarus clarkii to the recruitment of bloom-forming cyanobacteria from sediment. J. Limnol. 2010, 69, 102–111. [Google Scholar] [CrossRef]
- Han, J.; Jeon, B.-S.; Futatsugi, N.; Park, H.-D. The effect of alum coagulation for in-lake treatment of toxic Microcystis and other cyanobacteria related organisms in microcosm experiments. Ecotox. Environ. Safe. 2013, 96, 17–23. [Google Scholar] [CrossRef]
- Edwards, C.; Graham, D.; Fowler, N.; Lawton, L.A. Biodegradation of microcystins and nodularin in freshwaters. Chemosphere 2008, 73, 1315–1321. [Google Scholar] [CrossRef]
- Catherine, Q.; Susanna, W.; Isidora, E.-S.; Mark, H.; Aurélie, V.; Jean-François, H. A review of current knowledge on toxic benthic freshwater cyanobacteria—Ecology, toxin production and risk management. Water Res. 2013, 47, 5464–5479. [Google Scholar] [CrossRef]
- Ou, D.Y.; Song, L.R.; Gan, N.Q.; Chen, W. Effects of microcystins on and toxin degradation by Poterioochromonas sp. Environ. Toxicol. 2005, 20, 373–380. [Google Scholar] [CrossRef]
- Kim, B.R.; Nakan, S.; Kim, B.H.; Han, M.S. Grazing and growth of the heterotrophic flagellate Diphylleia rotans on the cyanobacterium Microcystis aeruginosa. Aquat. Microb. Ecol. 2006, 45, 163–170. [Google Scholar] [CrossRef]
- Giaramida, L.; Manage, P.M.; Edwards, C.; Singh, B.K.; Lawton, L.A. Bacterial communities’ response to microcystins exposure and nutrient availability: Linking degradation capacity to community structure. Int. Biodeterior. Biodegrad. 2013, 84, 111–117. [Google Scholar] [CrossRef]
- Chen, X.; Yang, X.; Yang, L.; Xiao, B.; Wu, X.; Wang, J.; Wan, H. An effective pathway for the removal of microcystin LR via anoxic biodegradation in lake sediments. Water Res. 2010, 44, 1884–1892. [Google Scholar] [CrossRef]
- Zou, H.; Pan, G.; Chen, H.; Yuan, X. Removal of cyanobacterial blooms in Taihu lake using local soils II. Effective removal of Microcystis aeruginosa using local soils and sediments modified by chitosan. Environ. Pollut. 2006, 141, 201–205. [Google Scholar] [CrossRef]
- Mirón, A.S.; Garcı́a, M.C.C.; Gómez, A.C.; Camacho, F.G.A.; Grima, E.M.; Chisti, Y. Shear stress tolerance and biochemical characterization of Phaeodactylum tricornutum in quasi steady-state continuous culture in outdoor photobioreactors. Biochem. Eng. J. 2003, 16, 287–297. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chen, X.; Xiang, H.; Hu, Y.; Zhang, Y.; Ouyang, L.; Gao, M. Fates of Microcystis aeruginosa Cells and Associated Microcystins in Sediment and the Effect of Coagulation Process on Them. Toxins 2014, 6, 152-167. https://doi.org/10.3390/toxins6010152
Chen X, Xiang H, Hu Y, Zhang Y, Ouyang L, Gao M. Fates of Microcystis aeruginosa Cells and Associated Microcystins in Sediment and the Effect of Coagulation Process on Them. Toxins. 2014; 6(1):152-167. https://doi.org/10.3390/toxins6010152
Chicago/Turabian StyleChen, Xiaoguo, Huiyi Xiang, Yue Hu, Yang Zhang, Liao Ouyang, and Meiying Gao. 2014. "Fates of Microcystis aeruginosa Cells and Associated Microcystins in Sediment and the Effect of Coagulation Process on Them" Toxins 6, no. 1: 152-167. https://doi.org/10.3390/toxins6010152
APA StyleChen, X., Xiang, H., Hu, Y., Zhang, Y., Ouyang, L., & Gao, M. (2014). Fates of Microcystis aeruginosa Cells and Associated Microcystins in Sediment and the Effect of Coagulation Process on Them. Toxins, 6(1), 152-167. https://doi.org/10.3390/toxins6010152



