Review of the Inhibition of Biological Activities of Food-Related Selected Toxins by Natural Compounds
Abstract
:1. Introduction
2. Aflatoxin B1 (AFB1)
2.1. Thiol Adducts

2.2. Lysine Adducts
2.3. Chemoprevention of AFB1-Induced Carcinogenesis in Cells
- Rosmarinic acid, a phenolic antioxidant contained in basil, mint, and sage, prevented AFB1-induced carcinogenesis of human hepatoma HepG2 cells [19].
- Cynidin-3-O-β-glucopyranoside, an anthocyanin contained in blackberries, cranberries, oranges, and strawberries inhibited AFB1- and ochratoxin A-induced toxicity in HepG2 and colonic adenocarcinoma (CaCo-2) cells [20].
- Carnosic acid, a phenolic antioxidant present in the rosemary plant, exhibited a dose-dependent protective effect against apoptosis of HepG2 cells [21].
- Leontopodic acid, isolated from the aerial parts of the Leontopodium alpinum plant, showed chemopreventive effects against AFB1- and deoxynivalenol-induced cell damage [22].
2.4. Inhibition of Aflatoxicosis
- Grapefruit juice protected against AFB1–induced liver DNA damage [24].
- Garlic powder protected against AFB1–induced DNA damage in rat liver and colon [25].
- The polysaccharide mannan and yeast reduced AFB1– and ochratoxin–induced DNA damage in rats [9].
- High doses of combinations of indole-3-carbinol and crambene, compounds from cruciferous vegetables, protected against adverse effect of AFB1 [26].
2.5. Reduction of AFB1 in Food
- Treatment with aqueous citric acid degraded 96.7% of AFB1 in maize (corn) with an initial concentration of 93 ng/g [29].
- Citric acid was more effective than lactic acid in reducing AFB1 in extrusion cooked sorghum [30].
- Extrusion cooking of contaminated peanut meal in the presence of calcium chloride, lysine, and methylamine reduced AFB1 from an initial value of 417.7 µg/kg to 66.9 µg/kg [31].
- Intermittent pumping of the volatile soybean aldehyde trans-2-exanal protected stored corn from Aspergillus flavus growth and aflatoxin contamination [34].
- The highest aflatoxin reduction (24.8%) was observed after cooking contaminated rice samples in a rice cooker, but the difference with other home-cooking methods was not statistically significant [35].
2.6. Practical Applications
3. Fumonisins
- Plant essential oils (Cinnamomum zeylanicum, Coriandrum sativum, Melissa officinalis, Mentha piperita, Salvia officinalis, and Thymus vulgaris) inhibited Fusarium mycotoxin production as well as fungal contamination of wheat seeds [46]. Inhibitory effects correlated with antioxidative properties of the oils. The highest inhibition of fungal growth was after 5 days of treatment and inhibition decreased after 22 days. The authors recommend the use of essential oils as natural preservatives for stored cereals.
- Fumigation of corn flour and corn kernels with allyl-, benzyl-, and phenyl isothiocyanates found in garlic resulted in a significant reduction of fumonisin content [47].
- Adsorption of the mycotoxin to a clay-based sorbent resulted in decreased bioavailability [48].
- A mycotoxin binder prevented adverse effects of fumonisin B1 in rats [49].
- An ethanol extract of the plant Aquilegia vulgaris counteracted the oxidative stress and toxicity of fumonisins in rats [52].
- A red ginseng extract also protected rats against AFB1– and fumonisin–induced pre-cancerous lesions [53].
- Several herbal teas and extracts protected against fumonisin B1-induced cancer promotion in rat liver [54].
Practical Applications
4. Ochratoxin A
- Barberis et al. [71] found that food grade antioxidants and antimicrobials controlled the growth of the fungi and ochratoxin A production on peanut kernels.
- Petchkongkaew et al. [72] demonstrated that Bacillus spp. from fermented soybeans can detoxify AFB1 and ochratoxin A.
- Virgili et al. [73] found that native yeast controls the production of ochratoxin production in dry cured ham.
- Kapetanokou et al. [74] observed similar results in beverages.
5. Botulinum Neurotoxins
- Studies by Daifas et al. [87] revealed that a commercial mastic resin and its essential oil in ethanol solution inhibited the growth of proteolytic strains of Clostridium botulinum in media. The anti-botulinal activity was greater when the test substances were applied in the vapor state than in solution. The test substances did not, however, inhibit neurotoxin production in challenge studies with the bacteria in English-style crumpets but the authors suggest that these natural products have the potential to inhibit pathogenic bacteria in bakery products.
- A reduced level of nitrite (75 mg/kg) inhibited the toxigenesis of Clostridium botulinum type B in meat products [88].
- The combined treatment with chlorine and lactic acid inhibited both E. coli O157:H7 and Clostridium sporogenes in spinach packaged in modified atmospheres [89].
- Kaempfenol, kaempferol, and quercetin glycosides isolated from black tea inhibited the neuromuscular inhibitory effects of botulinum neurotoxin A in mouse phrenic nerve–diaphragm preparations [93].
- Ethyl acetate extracts of several teas mixed with botulinum neurotoxin type A also prevented neuromuscular blockade of a mouse phrenic nerve–diaphragm preparation [94] with an order of potency of the extracts of black tea > oolong tea > roasted tea > green tea (no effect).
- Water-soluble fractions of the stinging nettle leaf extract inhibited the protease activity of botulinum neurotoxin type A but not type B [95].
- Chicoric acid isolated from the herbal plant Echinacea is a potent exosite inhibitor of BoNT/A with a synergistic effect when combined with an active site inhibitor [76].
- The natural compound lomofungin inhibited the BoNT serotype A light chain metalloproteinase (LC) by nonclassical inhibition kinetics [96].
Practical Applications
6. Cholera Toxin (CT)
Practical Applications
7. Shiga/Shiga-like Toxins
- Intraperitoneal administration of 1 mg of the green tea catechin epigallocatechin gallate (ECGC) to BALB/c mice completely inhibited the lethal effect of 2 ng of Stx2 [123].
- EGCG and gallocatechin gallate (GCG) also markedly inhibited the extracellular release of Stx2 toxin from E. coli O157:H7 [124]. The mechanism of inhibition seems to involve interference by the catechins of the transfer of periplasmic proteins through the outer membrane of the bacterial cell. The cited findings indicate that tea compounds are potent inhibitors of Stx2. An unanswered question is whether tea compounds and teas can inactivate bacterial toxins present in drinking water and in liquid and solid foods.
- The compound eugenol, which is present in many spices, inhibited verotoxin production in a concentration-dependent manner by E. coli O157:H7 [125].
- The food preservatives potassium sorbate, sodium benzoate, and sodium propionate reduced Shiga toxin activity in E. coli O157:H7 bacteria [126].
- Glycan-encapsulated gold nanoparticles inhibited Stx1 and Stx2 [127]. The authors suggest that tailored glyconanoparticles that mimic the natural display of glycans in lipid rafts could serve as potential therapeutics for the toxins. They also note that a few amino acid changes in emerging Stx2 variants can change receptor specificity.
- In an elegant review, Branson and Turnbull [128] describe mechanistic aspects of the inhibition by multivalent synthetic scaffolds, which include glycopolymers, glycodendrimers, and tailored glycoclusters, that can inhibit the binding of bacterial toxins to specific glycolipids in the cell membrane. The authors conclude that weak interactions of inhibitors can be greatly enhanced through multivalency. The safety and food-compatibility of the synthetic inhibitors need to be established before the inhibitors can be added to food.
- Quiñones et al. [129] describe the development and application of an improved Vero-d2EGFP cell-based fluorescence assay for the detection of Stx2 and inhibitors of toxin activity. Grape seed and grape pomace extracts both provided strong cellular protection against Stx2 inhibition of protein synthesis (Figure 4). The identified anti-toxin compounds can be used to develop food-compatible conditions for toxin inactivation that will benefit microbial food safety, security, and human health.Figure 4. Effect of plant compounds on protein synthesis levels in Stx-treated Vero-d2EGFP cells. Protein synthesis was measured in Vero-d2EGFP cells after a 2-hour co-incubation with plant polyphenolic compounds and Stx2. Cells were co-incubated with no plant compound, 1 mg caffeic acid/mL, 1 mg red wine concentrate/mL, 0.5 mg grape pomace extract/mL, or 0.1 mg grape seed extract/mL. Adapted from [129].Figure 4. Effect of plant compounds on protein synthesis levels in Stx-treated Vero-d2EGFP cells. Protein synthesis was measured in Vero-d2EGFP cells after a 2-hour co-incubation with plant polyphenolic compounds and Stx2. Cells were co-incubated with no plant compound, 1 mg caffeic acid/mL, 1 mg red wine concentrate/mL, 0.5 mg grape pomace extract/mL, or 0.1 mg grape seed extract/mL. Adapted from [129].
- Rasooly et al. [130] discovered that freshly prepared juice from locally purchased Red Delicious apples, but not fresh juice from Golden Delicious apples, inactivated the biological activity of Stx2. However, both Golden Delicious juice and water with added 0.3% polyphenol-rich grape pomace, a byproduct of wine production, also inactivated the Shiga toxin. Additional studies with immunomagnetic beads with specific antibodies against the toxin revealed that only part of the added Stx2 in apple juice appears to be irreversibly bound to apple juice and grape pomace constituents. The authors suggest that food-compatible and safe anti-toxin compounds can be used to inactivate Shiga toxins in apple juice and possibly also in other liquid and solid foods. It would also be of interest to find out whether apple skin, olive, and oregano leaf bactericidal powders [131] would also inhibit Stx2.
- Different grain fractions from pea (Pisum sativum) and faba bean (Vincia faba) inhibited adhesion of enterotoxigenic E. coli cells (ETEC) expressing adhesion and heat-labile LT toxins [132]. Because adhesion is involved in colonization of the host by the pathogens, the authors suggest that some of the fractions have the potential to protect pigs against pathogen-induced diarrhea.
- The probiotic bacteria Lactobacillus plantarum isolated from a fermented milk beverage called Kefir protected Vero cells against the cytotoxicity of Stx2 present in supernatants of E. coli O157:H7 bacteria [133].
- A variety of probiotic bacteria, especially Lactobacilli, inhibited the growth E. coli strains. Whether these in vitro results can be confirmed in vivo merits study [134].
Practical Applications
8. Staphylococcus Enterotoxins
- Intraperitoneal administration of a green tea extract and of the tea catechin ECGC to BALB/c mice bound to and inhibited the staphylococcal enterotoxin B (SEB) [138]. The inhibition of the heat-resistant enterotoxin was both dose and time dependent. ECGC also inhibited Staphylococcal superantigens-induced activation of T cells both in vitro and in vivo. Because these antigens aggravate atopic dermatitis, the authors suggest that catechins may be useful in the treatment of this human disease.
- Ether extracts of the herb Helichrysum italicum inhibited the production of enterotoxins (A–D) by S. aureus strains in culture media, suggesting that the extract interfered with the production of the enterotoxins [139].
- Lactobacillus starter cultures inhibited both growth of S. aureus and enterotoxin production in sausages during fermentation [140]. The authors suggest that intestinal Lactobacillus strains could be used as a starter culture to produced microbiologically safe meat products.
- Microbial growth and SEA production rates of S. aureus in the presence of undissociated lactic acid can be used as indicators of bacterial growth and SEA formation during initial stages of cheese production [141].
- An ethanol extract from the bulb of the Eleutherine americana plant inhibited both S. aureus strains and enterotoxin A–D production in broth and cooked pork [144]. The extract at 2 mg/mL delayed production of toxins A and C for 8 and 4 h, respectively, whereas toxin B was not detected in the pork after 48 h. The authors suggest that the ability of the extract to inhibit lipase and protease enzymes and to delay enterotoxin production in food indicates that it could be a novel additive against S. aureus in food.
- The 12-carbon fatty acid monoether dodecylglycerol (DDG) was more effective than glycerol monolaurate (GML) in inhibiting S. aureus growth in vitro [145]. By contrast, GML was more effective than DDG in reducing mortality, suppressing TNF-α, S. aureus growth and exotoxin production, and mortality in a rabbit model. The authors suggest that GML has the potential to be an effective anti-staphylococcal topical anti-infective candidate.
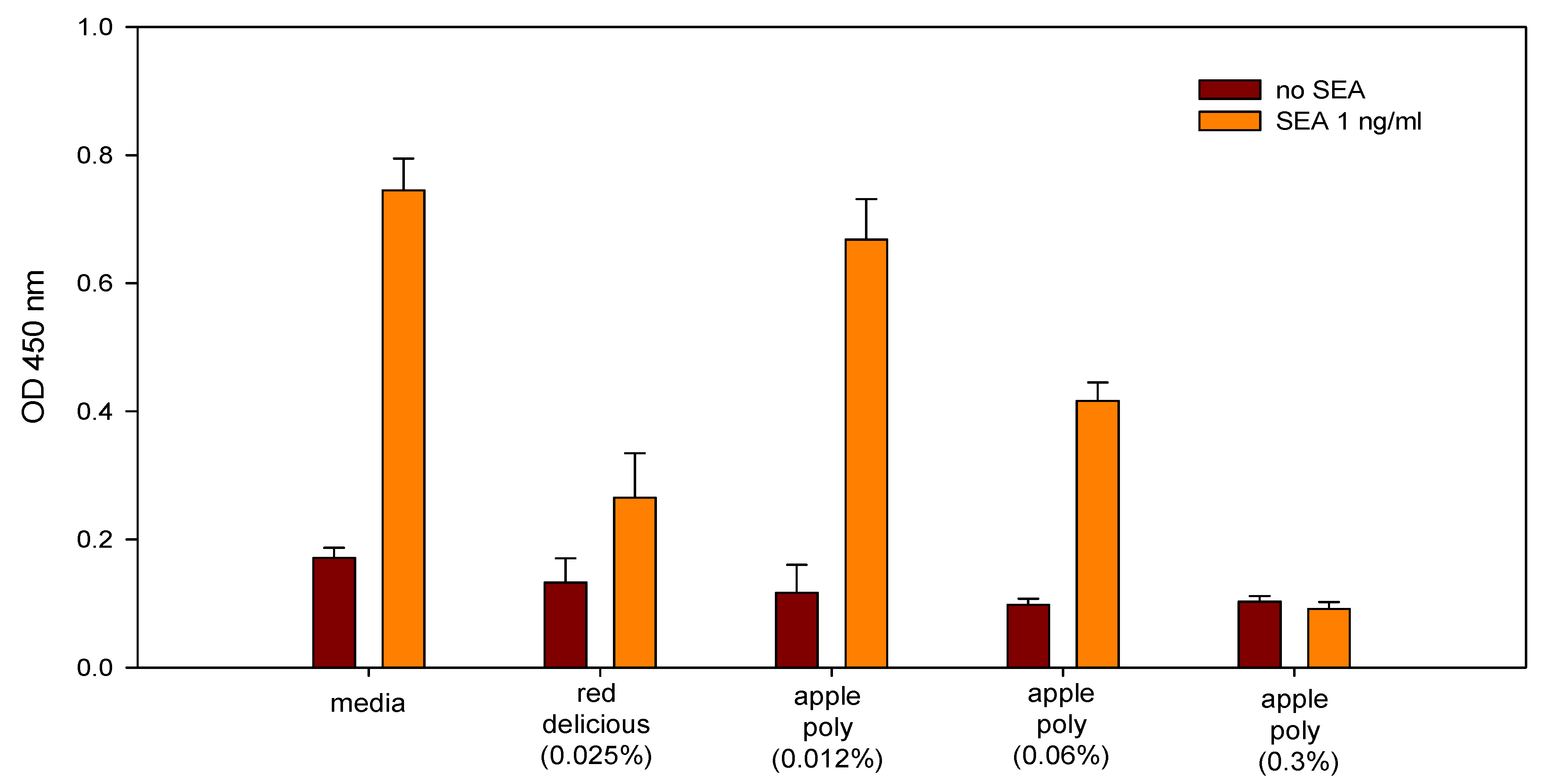
- Dilutions of freshly prepared apple juices and a commercial apple polyphenol preparation (Apple Poly®) inhibited the biological activity of SEA in a spleen cell assay (Figure 5) [146]. Studies with antibody-coated immunomagnetic beads bearing specific antibodies against the toxin showed that SEA added to apple juice seems to be largely bound to the juice constituents. Figure 6 depicts a possible mechanistic scheme for the inhibition.Figure 5. Comparison of inhibition of SEA by Red Delicious apple juice and apple polyphenols (Apple Poly). Splenocytes and SEA (1 ng/mL) were incubated for 48 h with Red Delicious juice or decreasing concentrations (0.3%, 0.06% and 0.012% w/v in PBS) of Apple Poly. The level of newly synthesized DNA was then determined, by measuring optical density at 450 nm. Adapted from [146].Figure 5. Comparison of inhibition of SEA by Red Delicious apple juice and apple polyphenols (Apple Poly). Splenocytes and SEA (1 ng/mL) were incubated for 48 h with Red Delicious juice or decreasing concentrations (0.3%, 0.06% and 0.012% w/v in PBS) of Apple Poly. The level of newly synthesized DNA was then determined, by measuring optical density at 450 nm. Adapted from [146].Figure 6. Schematic representation of cellular events that lead to the inhibition of SEA induced cell proliferation by apple juice. The individual steps in this scheme involve (A) the formation of a bridge between antigen presenting cells (APC) and T cells that results in the induction of T-cell proliferation; and (B) the inhibition of T-cell proliferation by added pure apple juice that disrupt the connection between APC and T cells. The net beneficial result of these events is the prevention of release and the consequent adverse effects induced by cytokines. Abbreviations: MHC, major histocompatibility complex; TCR, T-cell receptor. Adapted from [146].Figure 6. Schematic representation of cellular events that lead to the inhibition of SEA induced cell proliferation by apple juice. The individual steps in this scheme involve (A) the formation of a bridge between antigen presenting cells (APC) and T cells that results in the induction of T-cell proliferation; and (B) the inhibition of T-cell proliferation by added pure apple juice that disrupt the connection between APC and T cells. The net beneficial result of these events is the prevention of release and the consequent adverse effects induced by cytokines. Abbreviations: MHC, major histocompatibility complex; TCR, T-cell receptor. Adapted from [146].
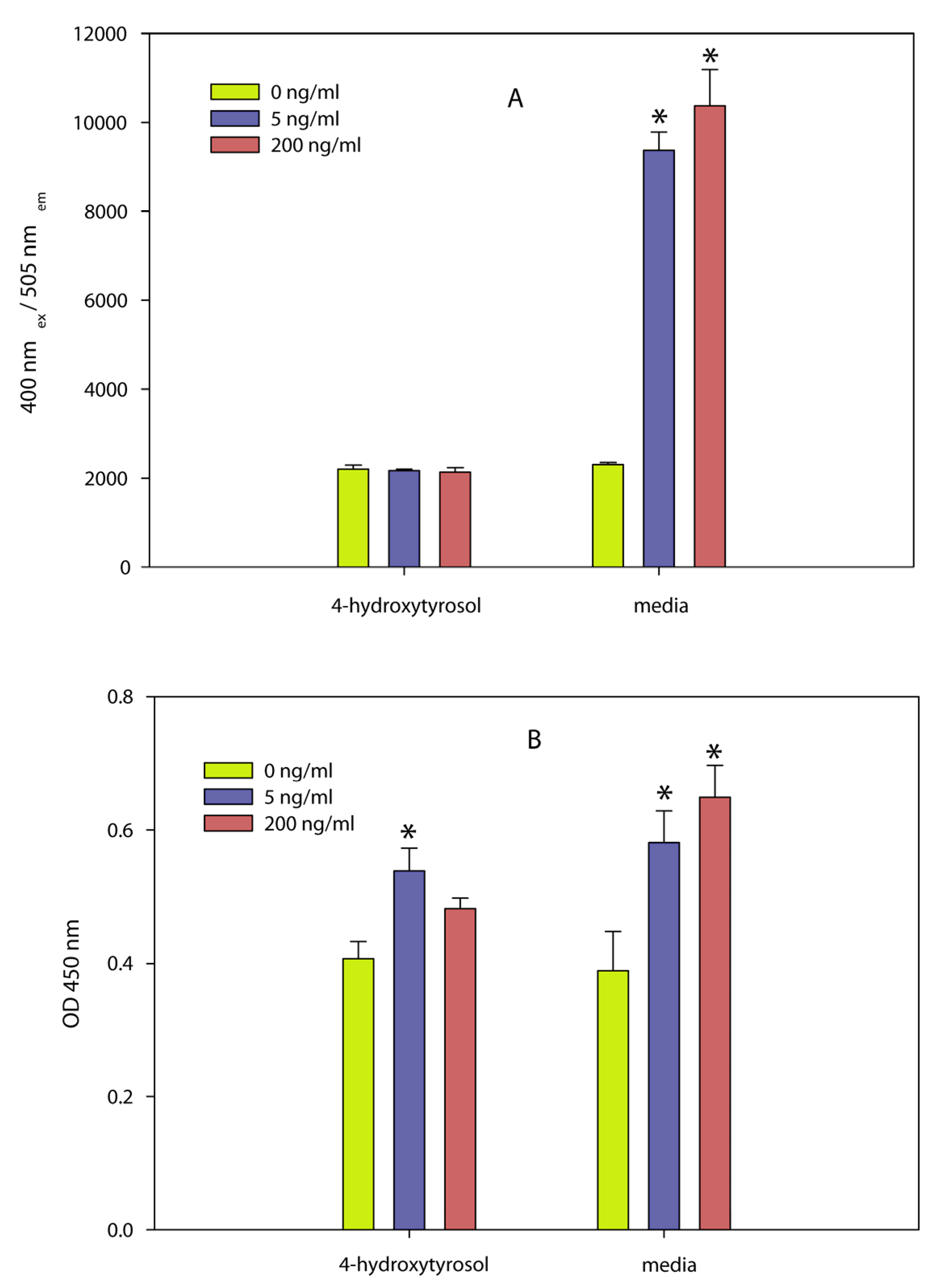
- A dilution series of the olive compound 4-hydroxytyrosol and a commercial olive powder containing approximately 6% 4-hydroxytyrosol and 6% of other phenolic compounds inactivated the pathogen [147]. Two independent assays, (5-bromo-2-deoxyuridine (BrdU) incorporation into newly synthesized DNA, and glycyl-phenylalanyl-aminofluorocoumarin proteolysis) showed that the olive compound also inactivated the biological activity of SEA at concentrations that were not toxic to spleen cells used in the assay. Efforts to determine the inhibition of the toxin by the olive powder were not successful because the powder was cytotoxic to the spleen cells at concentrations that are effective against the bacteria. The results (Figure 7) show that the olive compound can be used to inactivate both the bacteria and the toxin produced by the bacteria and that the use of cell assays to determine inhibition can only be done with concentrations of the inhibitor that are not toxic to cells.Figure 7. Effect of 4-hydroxytyrosol on splenocyte proliferation determined by two independent methods. Different concentrations of the toxin (0, 5, and 200 ng/mL) were exposed to 4-hydroxytyrosol or the control (media) and were then incubated for 48 h with splenocyte cells followed by determining (A) GF-AFC cleavage by live cell protease (a measure of cellular activity) or (B) BrdU incorporation into newly synthesized DNA (a measure of cellular proliferation). Conditions: (A) GF-AFC substrate in intact cells is cleaved by live cell protease releasing the fluorescent AFC, which is quantified at an excitation wavelength of 355 nm and an emission wavelength of 523 nm. (B) BrdU-labeled DNA was determined spectrophotometrically at absorbances of 620 nm and 450 nm. Both assays show that 4-hydroxytyrosol inhibited the biological activity of SEA. Adapted from [147].Figure 7. Effect of 4-hydroxytyrosol on splenocyte proliferation determined by two independent methods. Different concentrations of the toxin (0, 5, and 200 ng/mL) were exposed to 4-hydroxytyrosol or the control (media) and were then incubated for 48 h with splenocyte cells followed by determining (A) GF-AFC cleavage by live cell protease (a measure of cellular activity) or (B) BrdU incorporation into newly synthesized DNA (a measure of cellular proliferation). Conditions: (A) GF-AFC substrate in intact cells is cleaved by live cell protease releasing the fluorescent AFC, which is quantified at an excitation wavelength of 355 nm and an emission wavelength of 523 nm. (B) BrdU-labeled DNA was determined spectrophotometrically at absorbances of 620 nm and 450 nm. Both assays show that 4-hydroxytyrosol inhibited the biological activity of SEA. Adapted from [147].
- The Chinese herbal extract anisodamine inhibited the S. aureus toxin in human blood mononuclear cells [148].
- Hemoglobin inhibits the production of S. aureus exotoxins in a cell assay [149].
- Several phenolic antioxidants showed antimicrobial activity against several S. aureus strains [150] .
- Human monoclonal antibodies against SEB possess high affinity and toxin neutralization qualities essential for any therapeutic agent [151] .
- Several synthetic peptides inhibited the emetic and superantigenic activities of SEA in house musk shrews [152].
- Apple and olive powders and oregano leaves exhibited exceptionally high activity at nanogram levels against S. aureus [131].
Practical Applications
9. Ricin

Practical Applications
10. α-Chaconine
Practical Applications
| Toxin | Adverse effects | Inhibitors |
|---|---|---|
| Fungal | ||
| Aflatoxin B1 | mutagen; carcinogen | apple juice, caffeic, carnosic, cinnamic, citric, ferulic, lactic, leontopodic, rosmarinic, and vanillic acids, crambene, cysteine, cyanidinglucopyranoside, extrusion cooking, fisetin, garlic powder, glutathione, grapefruit juice, lactic acid, leontopodic acid, N-acetylcysteine, rosmarinic acid, yeast |
| Fumonisins | carcinogen, neurotoxin | clay-based sorbent, essential oils, ginseng, herbal teas, isothiocyanates, Aquilegia extract |
| Ochratoxin A | cytotoxin, nephrotoxin | antioxidants, Bacilli, yeast |
| Bacterial | ||
| Botulinum neurotoxin | neurotoxin, flaccid paralysis, botulism | chicoric acid, lactic acid, lomofungin, mastic essential oil, mastic resin, theaflavin, thearubigin, kaempferol, quercetin, teas, stinging nettle leaf extract |
| Cholera toxin | cholera disease, diarrhea | capsaicin, catechins, cysteine, epicatechin, glutathione, fenugreek seeds, galactose, quinazolines, rice bran, sialyloligosaccharides, skim milk, chilli pepper extract |
| Shiga toxins | diarrhea, hemolytic uremic syndrome, kidney, spleen, and thymus necrosis | bean fractions, apple juice, epigallocatechin, eugenol, fermented milk, glycan, glycodendrides, glycopolymers, grape seed extract, grape pomace extract, Lactobacillus, pea fractions, probiotic bacteria, yeast |
| Staphylococcus enterotoxin | atopic dermatitis, gastritis, mastitis, superantigen | anisodamine, apple juice, apple extract, dodecylglycerol, Eleutherine extract, glycerol monolaurate, green tea, Helichrysum extract, hemoglobin, hydroxytyrosol, kefir, olive powder, oregano leaves, sour milk |
| Plant | ||
| Ricin | cytotoxin | anti-ricin antibodies, reconstituted milk, ricin hydrogel |
| α-Chaconine | teratogen | folic acid, glucose-6-posphate, methotrexate, NADP |
11. Conclusions
- Determine whether natural compounds can concurrently reduce both pathogens and toxins produced by the pathogens.
- Define additive and/or synergistic effects of mixtures of natural toxin inhibitors.
- Compare efficacy of natural inhibitors against toxins in different foods, including fruit and vegetable juices, milk and cheeses, cereal grains, and meat and poultry products.
- Develop anti-toxin films and coatings to protect foods against contamination by toxins [171].
- Determine whether anti-toxin effects of natural compounds and extracts in vitro can be duplicated in vivo, especially in humans.
- Determine the biological significance of low levels of residual AFB1 and ricin, which seem to stimulate cell growth.
- Explore the use of molecular biology anti-sense RNA methods to suppress genes that govern the biosynthesis of plant and microbial toxins.
Acknowledgment
References
- Cousin, M.A.; Riley, R.T.; Pestka, J.J. Foodborne Mycotoxins: Chemistry, Biology, Ecology, and Toxicology. In Foodborne Pathogens-Microbiology and Molecular Biology; Fratamico, P.M., Bhunia, A.K., Smith, J.L., Eds.; Caister Academic Press: Norfolk, UK, 2005; pp. 163–226. [Google Scholar]
- Friedman, M. The Chemistry and Biochemistry of the Sulfhydryl Group in Amino Acids, Peptides, and Proteins; Pergamon Press: Oxford, UK, 1973; p. 499. [Google Scholar]
- Friedman, M.; Wehr, C.M.; Schade, J.E.; MacGregor, J.T. Inactivation of aflatoxin B1 mutagenicity by thiols. Food Chem. Toxicol. 1982, 20, 887–892. [Google Scholar]
- Friedman, M. Sulfhydryl Groups and Food Safety. In Nutritional and Toxicological Aspects of Food Safety (Advances in Experimental Medicine and Biology); Friedman, M., Ed.; Plenum Press: New York, NY, USA, 1984; Volume 177, pp. 31–63. [Google Scholar]
- De Flora, S.; Izzotti, A.; D'Agostini, F.; Cesarone, C.F. Antioxidant activity and other mechanisms of thiols involved in chemoprevention of mutation and cancer. Am. J. Med. 1991, 91, 122S–130S. [Google Scholar] [CrossRef]
- De Flora, S.; Bennicelli, C.; Serra, D.; Izzotti, A.; Cesarone, C.F. Role of Glutathione and N-Acetylcysteine as Inhibitors of Mutagenesis and Carcinogenesis. In Absorption and Utilization of Amino Acids; Friedman, M., Ed.; CRC Press: Boca Raton, FL, USA, 1989; Volume 3, pp. 19–54. [Google Scholar]
- Shetty, T.K.; Francis, A.R.; Bhattacharya, R.K. Modifying role of dietary factors on the mutagenicity of aflatoxin B1: In vitro effect of sulphur-containing amino acids. Mutat. Res. Genet. Toxicol. 1989, 222, 403–407. [Google Scholar] [CrossRef]
- Guengerich, F.P.; Johnson, W.W.; Shimada, T.; Ueng, Y.-F.; Yamazaki, H.; Langouët, S. Activation and detoxication of aflatoxin B1. Mutat. Res. Fund. Mol. Mech. Mutagen. 1998, 402, 121–128. [Google Scholar] [CrossRef]
- Madrigal-Santillán, E.; Morales-González, J.A.; Vargas-Mendoza, N.; Reyes-Ramírez, P.; Cruz-Jaime, S.; Sumaya-Martínez, T.; Pérez-Pastén, R.; Madrigal-Bujaidar, E. Antigenotoxic studies of different substances to reduce the DNA damage induced by aflatoxin B1 and ochratoxin A. Toxins 2010, 2, 738–757. [Google Scholar]
- Valencia-Quintana, R.; Sánchez-Alarcón, J.; Tenorio, M.G.; Deng, Y.; Waliszewski, S.M.; Valera, M.Á. Preventive strategies aimed at reducing the health risks of Aflatoxin B1. Toxicol. Environ. Health Sci. 2012, 4, 71–79. [Google Scholar]
- Cavalcante, A.A.M.; Rübensam, G.; Erdtmann, B.; Brendel, M.; Henriques, J.A.P. Cashew (Anacardium occidentale) apple juice lowers mutagenicity of aflatoxin B1 in S. typhimurium TA102. Genet. Mol. Biol. 2005, 28, 328–333. [Google Scholar] [CrossRef]
- Stevens, K.L.; Wilson, R.E.; Friedman, M. Inactivation of a tetrachloroimide mutagen from simulated processing water. J. Agric. Food Chem. 1995, 43, 2424–2427. [Google Scholar] [CrossRef]
- Friedman, M.; Levin, C.E. Review of methods for the reduction of dietary content and toxicity of acrylamide. J. Agric. Food Chem. 2008, 56, 6113–6140. [Google Scholar] [CrossRef]
- Rayburn, J.R.; Friedman, M. l-Cysteine, N-acetyl-l-cysteine, and glutathione protect Xenopus laevis embryos against acrylamide-induced malformations and mortality in the Frog Embryo Teratogenesis Assay. J. Agric. Food Chem. 2010, 58, 11172–11178. [Google Scholar]
- Friedman, M. Dietary Significance of Processing-Induced Lysinoalanine in Food. In Process-Induced Food Toxicants: Occurrence, Formation, Mitigation, and Health Risks; Stadler, R.H., Lineback, D.R., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009; pp. 475–508. [Google Scholar]
- Guengerich, F.P.; Arneson, K.O.; Williams, K.M.; Deng, Z.; Harris, T.M. Reaction of aflatoxin B1 oxidation products with lysine. Chem. Res. Toxicol. 2002, 15, 780–792. [Google Scholar]
- McCoy, L.F.; Scholl, P.F.; Sutcliffe, A.E.; Kieszak, S.M.; Powers, C.D.; Rogers, H.S.; Gong, Y.Y.; Groopman, J.D.; Wild, C.P.; Schleicher, R.L. Human aflatoxin albumin adducts quantitatively compared by ELISA, HPLC with fluorescence detection, and HPLC with isotope dilution mass spectrometry. Cancer Epidemiol. Biomarkers Prev. 2008, 17, 1653–1657. [Google Scholar] [CrossRef]
- Qian, G.; Tang, L.; Wang, F.; Guo, X.; Massey, M.E.; Williams, J.H.; Phillips, T.D.; Wang, J.-S. Physiologically based toxicokinetics of serum aflatoxin B1-lysine adduct in F344 rats. Toxicology 2013, 303, 147–151. [Google Scholar] [CrossRef]
- Renzulli, C.; Galvano, F.; Pierdomenico, L.; Speroni, E.; Guerra, M.C. Effects of rosmarinic acid against aflatoxin B1 and ochratoxin-A-induced cell damage in a human hepatoma cell line (Hep G2). J. Appl. Toxicol. 2004, 24, 289–296. [Google Scholar] [CrossRef]
- Guerra, M.C.; Galvano, F.; Bonsi, L.; Speroni, E.; Costa, S.; Renzulli, C.; Cervellati, R. Cyanidin-3-O-β-glucopyranoside, a natural fee-radical scavenger against aflatoxin B1-and ochratoxin A- induced cell damage in a human hepatoma cell line (Hep G2) and a human colonic adenocarcinoma cell line (CaCo-2). Br. J. Nutr. 2005, 94, 211–220. [Google Scholar] [CrossRef]
- Costa, S.; Utan, A.; Speroni, E.; Cervellati, R.; Piva, G.; Prandini, A.; Guerra, M.C. Carnosic acid from rosemary extracts: A potential chemoprotective agent against aflatoxin B1. An in vitro study. J. Appl. Toxicol. 2007, 27, 152–159. [Google Scholar] [CrossRef]
- Costa, S.; Schwaiger, S.; Cervellati, R.; Stuppner, H.; Speroni, E.; Guerra, M.C. In vitro evaluation of the chemoprotective action mechanisms of leontopodic acid against aflatoxin B1 and deoxynivalenol-induced cell damage. J. Appl. Toxicol. 2009, 29, 7–14. [Google Scholar] [CrossRef]
- Baptista, A.S.; Abdalla, A.L.; Aguiar, C.L.; Baptista, A.A.S.; Micheluchi, D.; Zampronio, A.C.; Pires, D.S.; Glória, E.M.; Calori-Domingues, M.A.; Walder, J.M.M.; et al. Utilization of diets amended with yeast and amino acids for the control of aflatoxicosis. World J. Microbiol. Biotechnol. 2008, 24, 2547–2554. [Google Scholar]
- Miyata, M.; Takano, H.; Guo, L.Q.; Kiyoshi, N.; Yamazoe, Y. Grapefruit juice intake does not enhance but rather protects against aflatoxin B1-induced liver DNA damage through a reduction in hepatic CYP3A activity. Carcinogenesis 2004, 25, 203–209. [Google Scholar]
- Singh, V.; Belloir, C.; Siess, M.-H.; Le Bon, A.-M. Inhibition of carcinogen-induced DNA damage in rat liver and colon by garlic powders with varying alliin content. Nutr. Cancer 2006, 55, 178–184. [Google Scholar]
- Wallig, M.A.; Heinz-Taheny, K.M.; Epps, D.L.; Gossman, T. Synergy among phytochemicals within crucifers: Does it translate into chemoprotection? J. Nutr. 2005, 135, 2972S–2977S. [Google Scholar]
- Dalvi, R.R. An overview of aflatoxicosis of poultry: Its characteristics, prevention and reduction. Vet. Res. Commun. 1986, 10, 429–443. [Google Scholar] [CrossRef]
- Rawal, S.; Kim, J.E.; Coulombe, R. Aflatoxin B1 in poultry: Toxicology, metabolism and prevention. Res. Vet. Sci. 2010, 89, 325–331. [Google Scholar] [CrossRef]
- Méndez-Albores, A.; Arámbula-Villa, G.; Loarca-Piña, M.G.F.; Castaño-Tostado, E.; Moreno-Martínez, E. Safety and efficacy evaluation of aqueous citric acid to degrade B-aflatoxins in maize. Food Chem. Toxicol. 2005, 43, 233–238. [Google Scholar]
- Méndez-Albores, A.; Martínez-Bustos, F.; Gaytán-Martínez, M.; Moreno-Martínez, E. Effect of lactic and citric acid on the stability of B-aflatoxins in extrusion-cooked sorghum. Lett. Appl. Microbiol. 2008, 47, 1–7. [Google Scholar]
- Saalia, F.K.; Phillips, R.D. Reduction of aflatoxins in peanut meal by extrusion cooking in the presence of nucleophiles. Lebensm.-Wiss. u.-Technol. 2011, 44, 1511–1516. [Google Scholar]
- Nesci, A.; Gsponer, N.; Etcheverry, M. Natural maize phenolic acids for control of aflatoxigenic fungi on maize. J. Food Sci. 2007, 72, M180–M185. [Google Scholar] [CrossRef]
- Samapundo, S.; De Meulenaer, B.; Osei-Nimoh, D.; Lamboni, Y.; Debevere, J.; Devlieghere, F. Can phenolic compounds be used for the protection of corn from fungal invasion and mycotoxin contamination during storage? Food Microbiol. 2007, 24, 465–473. [Google Scholar] [CrossRef]
- De Lucca, A.J.; Carter-Wientjes, C.H.; Boué, S.; Bhatnagar, D. Volatile trans-2-hexenal, a soybean aldehyde, inhibits Aspergillus flavus growth and aflatoxin production in corn. J. Food Sci. 2011, 76, M381–M386. [Google Scholar] [CrossRef]
- Mohamadi Sani, A.; Gholampour Azizi, E.; Ataye Salehi, E.; Rahimi, K. Reduction of aflatoxin in rice by different cooking methods. Toxicol. Ind. Health 2012. Published online Sept. 27. [Google Scholar] [CrossRef]
- Sun, Z.; Chen, T.; Thorgeirsson, S.S.; Zhan, Q.; Chen, J.; Park, J.H.; Lu, P.; Hsia, C.C.; Wang, N.; Xu, L.; et al. Dramatic reduction of liver cancer incidence in young adults: 28 year follow-up of etiological interventions in an endemic area of China. Carcinogenesis 2013. published online Jan. 14. [Google Scholar] [CrossRef]
- Chulze, S.N. Strategies to reduce mycotoxin levels in maize during storage: A review. Food Addit. Contam. 2010, 27, 651–657. [Google Scholar] [CrossRef]
- Jha, A.; Shah, K.; Verma, R.J. Aflatoxin-induced biochemical changes in liver of mice and its mitigation by black tea extract. Acta Poloniae Pharm. 2012, 69, 851–857. [Google Scholar]
- Rasooly, R.; Hernlem, B.; Friedman, M. Low levels of aflatoxin B1, ricin, and milk enhance recombinant protein production in mammalian cells. PLoS One 2013. submitted for publication.. [Google Scholar]
- Müller, S.; Dekant, W.; Mally, A. Fumonisin B1 and the kidney: Modes of action for renal tumor formation by fumonisin B1 in rodents. Food Chem. Toxicol. 2012, 50, 3833–3846. [Google Scholar] [CrossRef]
- Persson, E.C.; Sewram, V.; Evans, A.A.; Thomas London, W.; Volkwyn, Y.; Shen, Y.-J.; Van Zyl, J.A.; Chen, G.; Lin, W.; Shephard, G.S.; et al. Fumonisin B1 and risk of hepatocellular carcinoma in two Chinese cohorts. Food Chem. Toxicol. 2012, 50, 679–683. [Google Scholar] [CrossRef]
- Domijan, A.M. Fumonisin B1: A neurotoxic mycotoxin. Arh. Hig. Rada Toksikol. 2012, 63, 531–544. [Google Scholar] [CrossRef]
- Magan, N.; Aldred, D. Post-harvest control strategies: Minimizing mycotoxins in the food chain. Int. J. Food Microbiol. 2007, 119, 131–139. [Google Scholar] [CrossRef] [Green Version]
- Kimanya, M.E.; De Meulenaer, B.; Roberfroid, D.; Lachat, C.; Kolsteren, P. Fumonisin exposure through maize in complementary foods is inversely associated with linear growth of infants in Tanzania. Mol. Nutr. Food Res. 2010, 54, 1659–1667. [Google Scholar] [CrossRef]
- Kimanya, M.E.; De Meulenaer, B.; Van Camp, J.; Baert, K.; Kolsteren, P. Strategies to reduce exposure of fumonisins from complementary foods in rural Tanzania. Matern. Child Nutr. 2012, 8, 503–511. [Google Scholar]
- Sumalan, R.-M.; Alexa, E.; Poiana, M.-A. Assessment of inhibitory potential of essential oils on natural mycoflora and Fusarium mycotoxins production in wheat. Chem. Cent. J. 2013, 7. [Google Scholar] [CrossRef]
- Azaiez, I.; Meca, G.; Manyes, L.; Luciano, F.B.; Fernández-Franzón, M. Study of the chemical reduction of the fumonisins toxicity using allyl, benzyl and phenyl isothiocyanate in model solution and in food products. Toxicon 2013, 63, 137–146. [Google Scholar] [CrossRef]
- Brown, K.A.; Mays, T.; Romoser, A.; Marroquin-Cardona, A.; Mitchell, N.J.; Elmore, S.E.; Phillips, T.D. Modified hydra bioassay to evaluate the toxicity of multiple mycotoxins and predict the detoxification efficacy of a clay-based sorbent. J. Appl. Toxicol. 2012. [Google Scholar] [CrossRef]
- Qiang, Z.; Truong, M.; Meynen, K.; Murphy, P.A.; Hendrich, S. Efficacy of a mycotoxin binder against dietary fumonisin, deoxynivalenol, and zearalenone in rats. J. Agric. Food Chem. 2011, 59, 7527–7533. [Google Scholar]
- Voss, K.A.; Riley, R.T.; Jackson, L.S.; Jablonski, J.E.; Bianchini, A.; Bullerman, L.B.; Hanna, M.A.; Ryu, D. Extrusion cooking with glucose supplementation of fumonisin-contaminated corn grits protects against nephrotoxicity and disrupted sphingolipid metabolism in rats. Mol. Nutr. Food Res. 2011, 55, S312–S320. [Google Scholar] [CrossRef]
- Voss, K.A.; Riley, R.T.; Moore, N.D.; Burns, T.D. Alkaline cooking (nixtamalisation) and the reduction in the in vivo toxicity of fumonisin-contaminated corn in a rat feeding bioassay. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2012. [Google Scholar] [CrossRef]
- Hassan, A.M.; Mohamed, S.R.; El-Nekeety, A.A.; Hassan, N.S.; Abdel-Wahhab, M.A. Aquilegia vulgaris L. extract counteracts oxidative stress and cytotoxicity of fumonisin in rats. Toxicon 2010, 56, 8–18. [Google Scholar]
- Abdel-Wahhab, M.A.; Hassan, N.S.; El-Kady, A.A.; Khadrawy, Y.A.; El-Nekeety, A.A.; Mohamed, S.R.; Sharaf, H.A.; Mannaa, F.A. Red ginseng extract protects against aflatoxin B1 and fumonisins-induced hepatic pre-cancerous lesions in rats. Food Chem. Toxicol. 2010, 48, 733–742. [Google Scholar] [CrossRef]
- Marnewick, J.L.; van der Westhuizen, F.H.; Joubert, E.; Swanevelder, S.; Swart, P.; Gelderblom, W.C. Chemoprotective properties of rooibos (Aspalathus linearis), honeybush (Cyclopia intermedia) herbal and green and black (Camellia sinensis) teas against cancer promotion induced by fumonisin B1 in rat liver. Food Chem. Toxicol. 2009, 47, 220–229. [Google Scholar]
- Friedman, M.; Henika, P.R.; Mandrell, R.E. Bactericidal activities of plant essential oils and some of their isolated constituents against Campylobacter jejuni, Escherichia coli, Listeria monocytogenes, and Salmonella enterica. J. Food Prot. 2002, 65, 1545–1560. [Google Scholar]
- Juneja, V.K.; Yadav, A.S.; Hwang, C.-A.; Sheen, S.; Mukhopadhyay, S.; Friedman, M. Kinetics of thermal destruction of Salmonella in ground chicken containing trans-cinnamaldehyde and carvacrol. J. Food Prot. 2012, 75, 289–296. [Google Scholar]
- Moore-Neibel, K.; Gerber, C.; Patel, J.; Friedman, M.; Jaroni, D.; Ravishankar, S. Antimicrobial activity of oregano oil against antibiotic-resistant Salmonella enterica on organic leafy greens at varying exposure times and storage temperatures. Food Microbiol. 2013, 34, 123–129. [Google Scholar]
- Friedman, M.; Levin, C.E. Composition of jimson weed (Datura stramonium) seeds. J. Agric. Food Chem. 1989, 37, 998–1005. [Google Scholar]
- Crawford, L.; Friedman, M. The effects of low levels of dietary toxic weed seeds (jimson weed, Datura stramonium and sicklepod, Cassia obtusifolia) on the relative size of rat liver and levels and function of cytochrome P-450. Toxicol. Lett. 1990, 54, 175–181. [Google Scholar] [CrossRef]
- Dugan, G.M.; Gumbmann, M.R.; Friedman, M. Toxicological evaluation of jimson weed (Datura stramonium) seed. Food Chem. Toxicol. 1989, 27, 501–510. [Google Scholar] [CrossRef]
- Höhler, D. Ochratoxin A in food and feed: Occurrence, legislation and mode of action. Z. Ernaehrungswiss. 1998, 37, 2–12. [Google Scholar] [CrossRef]
- Cairns-Fuller, V.; Aldred, D.; Magan, N. Water, temperature and gas composition interactions affect growth and ochratoxin A production by isolates of Penicillium verrucosum on wheat grain. J. Appl. Microbiol. 2005, 99, 1215–1221. [Google Scholar] [CrossRef]
- Chulze, S.N.; Magnoli, C.E.; Dalcero, A.M. Occurrence of ochratoxin A in wine and ochratoxigenic mycoflora in grapes and dried vine fruits in South America. Int. J. Food Microbiol. 2006, 111 Suppl. 1, S5–S9. [Google Scholar]
- Lasram, S.; Belli, N.; Chebil, S.; Nahla, Z.; Ahmed, M.; Sanchis, V.; Ghorbel, A. Occurrence of ochratoxigenic fungi and ochratoxin A in grapes from a Tunisian vineyard. Int. J. Food Microbiol. 2007, 114, 376–379. [Google Scholar] [CrossRef]
- EL Khoury, A.; Rizk, T.; Lteif, R.; Azouri, H.; Delia, M.-L.; Lebrihi, A. Fungal contamination and Aflatoxin B1 and Ochratoxin A in Lebanese wine-grapes and musts. Food Chem. Toxicol. 2008, 46, 2244–2250. [Google Scholar] [CrossRef]
- Kamp, H.G.; Eisenbrand, G.; Schlatter, J.; Würth, K.; Janzowski, C. Ochratoxin A: Induction of (oxidative) DNA damage, cytotoxicity and apoptosis in mammalian cell lines and primary cells. Toxicology 2005, 206, 413–425. [Google Scholar] [CrossRef]
- Bouslimi, A.; Bouaziz, C.; Ayed-Boussema, I.; Hassen, W.; Bacha, H. Individual and combined effects of ochratoxin A and citrinin on viability and DNA fragmentation in cultured Vero cells and on chromosome aberrations in mice bone marrow cells. Toxicology 2008, 251, 1–7. [Google Scholar] [CrossRef]
- Chopra, M.; Link, P.; Michels, C.; Schrenk, D. Characterization of ochratoxin A-induced apoptosis in primary rat hepatocytes. Cell Biol. Toxicol. 2010, 26, 239–254. [Google Scholar] [CrossRef]
- Pfohl-Leszkowicz, A.; Manderville, R.A. Ochratoxin A: An overview on toxicity and carcinogenicity in animals and humans. Mol. Nutr. Food Res. 2007, 51, 61–99. [Google Scholar] [CrossRef]
- Napoletano, M.; Pennino, D.; Izzo, G.; de Maria, S.; Ottaviano, R.; Ricciardi, M.; Mancini, R.; Schiattarella, A.; Farina, E.; Metafora, S.; et al. Ochratoxin A induces craniofacial malformation in mice acting on Dlx5 gene expression. Front. Biosci. Elite Ed. 2010, 2, 133–142. [Google Scholar]
- Barberis, C.L.; Astoreca, A.L.; Dalcero, A.M.; Magnoli, C.E. Food-grade antioxidants and antimicrobials to control growth and ochratoxin a production by Aspergillus section Nigri on peanut kernels. J. Food Prot. 2010, 73, 1493–1501. [Google Scholar]
- Petchkongkaew, A.; Taillandier, P.; Gasaluck, P.; Lebrihi, A. Isolation of Bacillus spp. from Thai fermented soybean (Thua-nao): Screening for aflatoxin B1 and ochratoxin A detoxification. J. Appl. Microbiol. 2008, 104, 1495–1502. [Google Scholar] [CrossRef]
- Virgili, R.; Simoncini, N.; Toscani, T.; Leggieri, M.C.; Formenti, S.; Battilani, P. Biocontrol of Penicillium nordicum growth and ochratoxin A production by native yeasts of dry cured ham. Toxins 2012, 4, 68–82. [Google Scholar] [CrossRef]
- Kapetanakou, A.E.; Kollias, J.N.; Drosinos, E.H.; Skandamis, P.N. Inhibition of A. carbonarius growth and reduction of ochratoxin A by bacteria and yeast composites of technological importance in culture media and beverages. Int. J. Food Microbiol. 2012, 152, 91–99. [Google Scholar] [CrossRef]
- Pirazzini, M.; Bordin, F.; Rossetto, O.; Shone, C.C.; Binz, T.; Montecucco, C. The thioredoxin reductase-thioredoxin system is involved in the entry of tetanus and botulinum neurotoxins in the cytosol of nerve terminals. FEBS Lett. 2013, 587, 150–155. [Google Scholar] [CrossRef]
- Šilhár, P.; Capková, K.; Salzameda, N.T.; Barbieri, J.T.; Hixon And, M.S.; Janda, K.D. Botulinum neurotoxin a protease: Discovery of natural product exosite inhibitors. J. Am. Chem. Soc. 2010, 132, 2868–2869. [Google Scholar]
- Chalk, C.; Benstead, T.J.; Keezer, M. Medical treatment for botulism. Cochrane Database Syst. Rev. 2011, 3. [Google Scholar] [CrossRef] [Green Version]
- Hill, S.E.; Iqbal, R.; Cadiz, C.L.; Le, J. Foodborne botulism treated with heptavalent botulism antitoxin. Ann. Pharmacother. 2013, 47. [Google Scholar] [CrossRef]
- Juliao, P.C.; Maslanka, S.; Dykes, J.; Gaul, L.; Bagdure, S.; Granzow-Kibiger, L.; Salehi, E.; Zink, D.; Neligan, R.P.; Barton-Behravesh, C.; et al. National outbreak of type a foodborne botulism associated with a widely distributed commercially canned hot dog chili sauce. Clin. Infect. Dis. 2013, 56, 376–382. [Google Scholar] [CrossRef]
- Date, K.; Fagan, R.; Crossland, S.; Maceachern, D.; Pyper, B.; Bokanyi, R.; Houze, Y.; Andress, E.; Tauxe, R. Three outbreaks of foodborne botulism caused by unsafe home canning of vegetables-ohio and Washington, 2008 and 2009. J. Food Prot. 2011, 74, 2090–2096. [Google Scholar]
- Daifas, D.P.; Smith, J.P.; Tarte, I.; Blanchfield, B.; Austin, J.W. Effect of ethanol vapor on growth and toxin production by Clostridium botulinum in a high moisture bakery product. J. Food Saf. 2000, 20, 111–125. [Google Scholar] [CrossRef]
- Daifas, D.P.; Smith, J.P.; Blanchfield, B.; Austin, J.W. Challenge studies with Clostridium botulinum in high moisture bakery products packaged under modified atmospheres. Food Aust. 1999, 51, 507–511. [Google Scholar]
- Daifas, D.P.; Smith, J.P.; Blanchfield, B.; Austin, J.W. Growth and toxin production by Clostridium botulinum in English-style crumpets packaged under modified atmospheres. J. Food Prot. 1999, 62, 349–355. [Google Scholar]
- Malakar, P.K.; Barker, G.C.; Peck, M.W. Quantitative risk assessment for hazards that arise from non-proteolytic Clostridium botulinum in minimally processed chilled dairy-based foods. Food Microbiol. 2011, 28, 321–330. [Google Scholar] [CrossRef]
- Newell, C.R.; Ma, L.; Doyle, M. Botulism challenge studies of a modified atmosphere package for fresh mussels: Inoculated pack studies. J. Food Prot. 2012, 75, 1157–1166. [Google Scholar] [CrossRef]
- Sevenier, V.; Delannoy, S.; André, S.; Fach, P.; Remize, F. Prevalence of Clostridium botulinum and thermophilic heat-resistant spores in raw carrots and green beans used in French canning industry. Int. J. Food Microbiol. 2012, 155, 263–268. [Google Scholar] [CrossRef]
- Daifas, D.P.; Smith, J.P.; Blanchfield, B.; Sanders, G.; Austin, J.W.; Koukoutisis, J. Effects of mastic resin and its essential oil on the growth of proteolytic Clostridium botulinum. Int. J. Food Microbiol. 2004, 94, 313–322. [Google Scholar] [CrossRef]
- Keto-Timonen, R.; Lindström, M.; Puolanne, E.; Niemistö, M.; Korkeala, H. Inhibition of toxigenesis of group II (nonproteolytic) Clostridium botulinum type B in meat products by using a reduced level of nitrite. J. Food Prot. 2012, 75, 1346–1349. [Google Scholar] [CrossRef]
- Brown, A.L.; Brooks, J.C.; Karunasena, E.; Echeverry, A.; Laury, A.; Brashears, M.M. Inhibition of Escherichia coli O157:H7 and Clostridium sporogenes in spinach packaged in modified atmospheres after treatment combined with chlorine and lactic acid bacteria. J. Food Sci. 2011, 76, M427–M432. [Google Scholar] [CrossRef]
- Sawamura, S.-I.; Sakane, I.; Satoh, E.; Ishii, T.; Shimizu, Y.; Nishimura, M. Black tea extract reduces the oral toxicity of botulinum neurotoxin applied to food. Nippon Shokuhin Kagaku Kogaku Kaishi 2002, 49, 808–812. [Google Scholar] [CrossRef]
- Satoh, E.; Ishii, T.; Shimizu, Y.; Sawamura, S.; Nishimura, M. A mechanism of the thearubigin fraction of black tea (Camellia sinensis) extract protecting against the effect of tetanus toxin. J. Toxicol. Sci. 2002, 27, 441–447. [Google Scholar] [CrossRef]
- Satoh, E.; Ishii, T.; Shimizu, Y.; Sawamura, S.-I.; Nishimura, M. Black tea extract, thearubigin fraction, counteract the effects of botulinum neurotoxins in mice. Br. J. Pharmacol. 2001, 132, 797–798. [Google Scholar] [CrossRef]
- Sawamura, S.-I.; Sakane, I.; Satoh, E.; Ishii, T.; Shimizu, Y.; Nishimura, M.; Umehara, K. Isolation and determination of an antidote for botulinum neurotoxin from black tea extract. Folia Pharmacol. Jpn. 2002, 120, 116P–118P. [Google Scholar]
- Satoh, E. Ethyl acetate extract from black tea prevents neuromuscular blockade by botulinum neurotoxin type A in vitro. Int. J. Food Sci. Nutr. 2005, 56, 543–550. [Google Scholar] [CrossRef]
- Gul, N.; Ahmed, S.A.; Smith, L.A. Inhibition of the protease activity of the light chain of type A botulinum neurotoxin by aqueous extract from stinging nettle (Urtica dioica) leaf. Basic Clin. Pharmacol. Toxicol. 2004, 95, 215–219. [Google Scholar] [CrossRef]
- Eubanks, L.M.; Šilhár, P.; Salzameda, N.T.; Zakhari, J.S.; Xiaochuan, F.; Barbieri, J.T.; Shoemaker, C.B.; Hixon, M.S.; Janda, K.D. Identification of a natural product antagonist against the botulinum neurotoxin light chain protease. ACS Med. Chem. Lett. 2010, 1, 268–272. [Google Scholar] [CrossRef]
- Friedman, M. Overview of antibacterial, antitoxin, antiviral, and antifungal activities of tea flavonoids and teas. Mol. Nutr. Food Res. 2007, 51, 116–134. [Google Scholar] [CrossRef]
- Friedman, M.; Kim, S.-Y.; Lee, S.-J.; Han, G.-P.; Han, J.-S.; Lee, K.-R.; Kozukue, N. Distribution of catechins, theaflavins, caffeine, and theobromine in 77 teas consumed in the United States. J. Food Sci. 2005, 70, C550–C559. [Google Scholar]
- Friedman, M.; Levin, C.E.; Choi, S.-H.; Kozukue, E.; Kozukue, N. HPLC analysis of catechins, theaflavins, and alkaloids in commercial teas and green tea dietary supplements: Comparison of water and 80% ethanol/water extracts. J. Food Sci. 2006, 71, C328–C337. [Google Scholar] [CrossRef]
- Juneja, V.K.; Bari, M.L.; Inatsu, Y.; Kawamoto, S.; Friedman, M. Control of Clostridium perfringens spores by green tea leaf extracts during cooling of cooked ground beef, chicken, and pork. J. Food Prot. 2007, 70, 1429–1433. [Google Scholar]
- Juneja, V.K.; Friedman, M. Carvacrol, cinnamaldehyde, oregano oil, and thymol inhibit Clostridium perfringens spore germination and outgrowth in ground turkey during chilling. J. Food Prot. 2007, 70, 218–222. [Google Scholar]
- Shukla, H.D.; Sharma, S.K. Clostridium botulinum: A bug with beauty and weapon. Crit. Rev. Microbiol. 2005, 31, 11–18. [Google Scholar] [CrossRef]
- Rasmussen, L.; White, E.L.; Pathak, A.; Ayala, J.C.; Wang, H.; Wu, J.-H.; Benitez, J.A.; Silva, A.J. A high-throughput screening assay for inhibitors of bacterial motility identifies a novel inhibitor of the Na+-driven flagellar motor and virulence gene expression in Vibrio cholerae. Antimicrob. Agents Chemother. 2011, 55, 4134–4143. [Google Scholar] [CrossRef]
- Toda, M.; Okubo, S.; Ikigai, H.; Suzuki, T.; Suzuki, Y.; Shimamura, T. The protective activity of tea against infection by Vibrio cholerae O1. J. Appl. Bacteriol. 1991, 70, 109–112. [Google Scholar] [CrossRef]
- Toda, M.; Okubo, S.; Ikigai, H.; Suzuki, T.; Suzuki, Y.; Hara, Y.; Shimamura, T. The protective activity of tea catechins against experimental infection by Vibrio cholerae O1. Microbiol. Immunol. 1992, 36, 999–1001. [Google Scholar]
- Morinaga, N.; Iwamaru, Y.; Yahiro, K.; Tagashira, M.; Moss, J.; Noda, M. Differential activities of plant polyphenols on the binding and internalization of cholera toxin in vero cells. J. Biol. Chem. 2005, 280, 23303–23309. [Google Scholar]
- Toda, M.; Shimamura, T. Catechin - Multifunctional biodefense agent. J. Showa Med. Assoc. 1997, 57, 175–189. [Google Scholar]
- Shimamura, T.; Watanabe, S.; Sasaki, S. Inhibition of cholera toxin production by thiols in Vibrio cholerae. Infect. Immun. 1986, 53, 700–701. [Google Scholar]
- Friedman, M.; Brandon, D.L. Nutritional and health benefits of soy proteins. J. Agric. Food Chem. 2001, 49, 1069–1086. [Google Scholar] [CrossRef]
- Friedman, M. Application of the S-pyridylethylation reaction to the elucidation of the structures and functions of proteins. J. Protein Chem. 2001, 20, 431–453. [Google Scholar] [CrossRef]
- Becker, P.M.; Widjaja-Greefkes, H.C.A.; Van Wikselaar, P.G. Inhibition of binding of the AB5-type enterotoxins LT-I and cholera toxin to ganglioside GM1 by galactose-rich dietary components. Foodborne Pathog. Dis. 2010, 7, 225–233. [Google Scholar]
- Sinclair, H.R.; Kemp, F.; Slegte, J.D.; Gibson, G.R.; Rastall, R.A. Carbohydrate-based anti-adhesive inhibition of Vibrio cholerae toxin binding to GM1-OS immobilized into artificial planar lipid membranes. Carbohydr. Res. 2009, 344, 1968–1974. [Google Scholar] [CrossRef]
- Chatterjee, S.; Asakura, M.; Chowdhury, N.; Neogi, S.B.; Sugimoto, N.; Haldar, S.; Awasthi, S.P.; Hinenoya, A.; Aoki, S.; Yamasaki, S. Capsaicin, a potential inhibitor of cholera toxin production in Vibrio cholerae. FEMS Microbiol. Lett. 2010, 306, 54–60. [Google Scholar] [CrossRef]
- Kozukue, N.; Han, J.S.; Kozukue, E.; Lee, S.J.; Kim, J.A.; Lee, K.R.; Levin, C.E.; Friedman, M. Analysis of eight capsaicinoids in peppers and pepper-containing foods by high-performance liquid chromatography and liquid chromatography-mass spectrometry. J. Agric. Food Chem. 2005, 53, 9172–9181. [Google Scholar]
- Choi, S.H.; Suh, B.S.; Kozukue, E.; Kozukue, N.; Levin, C.E.; Friedman, M. Analysis of the contents of pungent compounds in fresh Korean red peppers and in pepper-containing foods. J. Agric. Food Chem. 2006, 54, 9024–9031. [Google Scholar] [CrossRef]
- Yamasaki, S.; Asakura, M.; Neogi, S.B.; Hinenoya, A.; Iwaoka, E.; Aoki, S. Inhibition of virulence potential of Vibrio cholerae by natural compounds. Indian J. Med. Res. 2011, 133, 232–239. [Google Scholar]
- Velázquez, C.; Correa-Basurto, J.; Garcia-Hernandez, N.; Barbosa, E.; Tesoro-Cruz, E.; Calzada, S.; Calzada, F. Anti-diarrheal activity of (-)-epicatechin from Chiranthodendron pentadactylon Larreat: Experimental and computational studies. J. Ethnopharmacol. 2012, 143, 716–719. [Google Scholar] [CrossRef]
- Choi, S.P.; Kim, S.P.; Nam, S.H.; Friedman, M. Antitumor effects of dietary black and brown rice brans in tumor-bearing mice: Relationship to composition. Mol. Nutr. Food Res. 2013, 57, 390–400. [Google Scholar]
- Kondo, S.; Teongtip, R.; Srichana, D.; Itharat, A. Antimicrobial activity of rice bran extracts for diarrheal disease. J. Med. Assoc. Thai. 2011, 94 Suppl 7, S117–S121. [Google Scholar]
- Holmgren, J.; Bourgeois, L.; Carlin, N.; Clements, J.; Gustafsson, B.; Lundgren, A.; Nygren, E.; Tobias, J.; Walker, R.; Svennerholm, A.M. Development and preclinical evaluation of safety and immunogenicity of an oral ETEC vaccine containing inactivated E. coli bacteria overexpressing colonization factors CFA/I, CS3, CS5 and CS6 combined with a hybrid LT/CT B subunit antigen, administered alone and together with dmLT adjuvant. Vaccine 2013. [Google Scholar] [CrossRef]
- Rasooly, R.; Do, P.M. Shiga toxin Stx2 is heat-stable and not inactivated by pasteurization. Int. J. Food Microbiol. 2010, 136, 290–294. [Google Scholar] [CrossRef]
- Rasooly, R.; Do, P.M.; Griffey, S.M.; Vilches-Moure, J.G.; Friedman, M. Ingested Shiga toxin 2 (Stx2) causes histopathological changes in kidney, spleen and thymus tissues and mortality in mice. J. Agric. Food Chem. 2010, 58, 9281–9286. [Google Scholar]
- Okubo, S.; Sasaki, T.; Hara, Y.; Mori, F.; Shimamura, T. [Bactericidal and anti-toxin activities of catechin on enterohemorrhagic Escherichia coli]. Kansenshogaku Zasshi 1998, 72, 211–217. [Google Scholar]
- Sugita-Konishi, Y.; Hara-Kudo, Y.; Amano, F.; Okubo, T.; Aoi, N.; Iwaki, M.; Kumagai, S. Epigallocatechin gallate and gallocatechin gallate in green tea catechins inhibit extracellular release of Vero toxin from enterohemorrhagic Escherichia coli O157:H7. Biochim. Biophys. Acta 1999, 1472, 42–50. [Google Scholar]
- Takemasa, N.; Ohnishi, S.; Tsuji, M.; Shikata, T.; Yokoigawa, K. Screening and analysis of spices with ability to suppress verocytotoxin production by Escherichia coli O157. J. Food Sci. 2009, 74. [Google Scholar] [CrossRef]
- Subils, T.; Aquili, V.; Ebner, G.; Balagué, C. Effect of preservatives on Shiga toxigenic phages and Shiga toxin of Escherichia coli O157:H7. J. Food Prot. 2012, 75, 959–965. [Google Scholar] [CrossRef]
- Kulkarni, A.A.; Fuller, C.; Korman, H.; Weiss, A.A.; Iyer, S.S. Glycan encapsulated gold nanoparticles selectively inhibit Shiga toxins 1 and 2. Bioconjug. Chem. 2010, 21, 1486–1493. [Google Scholar]
- Branson, T.R.; Turnbull, W.B. Bacterial toxin inhibitors based on multivalent scaffolds. Chem. Soc. Rev. 2012. [Google Scholar] [CrossRef]
- Quiñones, B.; Massey, S.; Friedman, M.; Swimley, M.S.; Teter, K. Novel cell-based method to detect Shiga toxin 2 from Escherichia coli O157:H7 and inhibitors of toxin activity. Appl. Environ. Microbiol. 2009, 75, 1410–1416. [Google Scholar] [CrossRef]
- Rasooly, R.; Do, P.M.; Levin, C.E.; Friedman, M. Inhibition of Shiga toxin 2 (Stx2) in apple juices and its resistance to pasteurization. J. Food Sci. 2010, 75, M296–M301. [Google Scholar]
- Friedman, M.; Henika, P.R.; Levin, C.E. Bactericidal activities of health-promoting, food-derived powders against the foodborne pathogens Escherichia coli, Listeria monocytogenes, Salmonella enterica, and Staphylococcus aureus. J. Food Sci. 2013, 78, M270–M275. [Google Scholar] [CrossRef]
- Becker, P.M.; van der Meulen, J.; Jansman, A.J.M.; van Wikselaar, P.G. In vitro inhibition of ETEC K88 adhesion by pea hulls and of LT enterotoxin binding by faba bean hulls. J. Anim. Physiol. Anim. Nutr. (Berl.) 2012, 96, 1121–1126. [Google Scholar]
- Kakisu, E.; Abraham, A.G.; Farinati, C.T.; Ibarra, C.; De Antoni, G.L. Lactobacillus plantarum isolated from kefir protects Vero cells from cytotoxicity by type-II Shiga toxin from Escherichia coli O157:H7. J. Dairy Res. 2013, 80, 64–71. [Google Scholar] [CrossRef]
- Mogna, L.; Del Piano, M.; Deidda, F.; Nicola, S.; Soattini, L.; Debiaggi, R.; Sforza, F.; Strozzi, G.; Mogna, G. Assessment of the in vitro inhibitory activity of specific probiotic bacteria against different Escherichia coli strains. J. Clin. Gastroenterol. 2012, 46, S29–S32. [Google Scholar] [CrossRef]
- Du, W.-X.; Olsen, C.W.; Avena-Bustillos, R.J.; McHugh, T.H.; Levin, C.E.; Friedman, M. Effects of allspice, cinnamon, and clove bud essential oils in edible apple films on physical properties and antimicrobial activities. J. Food Sci. 2009, 74, M372–M378. [Google Scholar] [CrossRef]
- Du, W.-X.; Olsen, C.W.; Avena-Bustillos, R.J.; McHugh, T.H.; Levin, C.E.; Mandrell, R.; Friedman, M. Antibacterial effects of allspice, garlic, and oregano essential oils in tomato films determined by overlay and vapor-phase methods. J. Food Sci. 2009, 74, M390–M397. [Google Scholar] [CrossRef]
- Rasooly, R.; Friedman, M. Plant Compounds Inhibit Staphylococcus aureus Bacteria and the Toxicity of Staphylococcus Enterotoxin A (SEA) Associated with Atopic Dermatitis. In Atopic Dermatitis-Disease Etiology and Clinical Management; Esparza-Gordillo, J., Dekio, I., Eds.; InTech-Open Access Publishers: Rijeka, Croatia, 2012; pp. 387–404. [Google Scholar]
- Hisano, M.; Yamaguchi, K.; Inoue, Y.; Ikeda, Y.; Iijima, M.; Adachi, M.; Shimamura, T. Inhibitory effect of catechin against the superantigen staphylococcal enterotoxin B (SEB). Arch. Dermatol. Res. 2003, 295, 183–189. [Google Scholar]
- Nostro, A.; Cannatelli, M.A.; Musolino, A.D.; Procopio, F.; Alonzo, V. Helichrysum italicum extract interferes with the production of enterotoxins by Staphylococcus aureus. Lett. Appl. Microbiol. 2002, 35, 181–184. [Google Scholar] [CrossRef]
- Sameshima, T.; Magome, C.; Takeshita, K.; Arihara, K.; Itoh, M.; Kondo, Y. Effect of intestinal Lactobacillus starter cultures on the behaviour of Staphylococcus aureus in fermented sausage. Int. J. Food Microbiol. 1998, 41, 1–7. [Google Scholar] [CrossRef]
- Lindblad, M.; Lindqvist, R. The effect of undissociated lactic acid on Staphylococcus aureus growth and enterotoxin A production. Int. J. Food Microbiol. 2013, 162, 159–166. [Google Scholar] [CrossRef]
- Fluer, F.S.; Kuznetsova, G.G.; Batishcheva, S.; Matushevskaia, V.N.; Donskaia, G.A. Influence of the food products, enriched with pectin, on properties of potentially patogenic representatives of the microflora of the large intenstine. Voprosy pitaniia 2006, 75, 46–49. [Google Scholar]
- Aoyama, K.; Takahashi, C.; Yamauchi, Y.; Sakai, F.; Igarashi, H.; Yanahira, S.; Konishi, H. Examination of Staphylococcus aureus survival and growth during cheese-making process. J. Food Hyg. Soc. Jpn. 2008, 49, 116–123. [Google Scholar] [CrossRef]
- Ifesan, B.O.T.; Voravuthlkunchai, S.P. Effect of Eleutherine americana merr. Extract on enzymatic activity and enterotoxin production of Staphylococcus aureus in broth and cooked pork. Foodborne Pathog. Dis. 2009, 6, 699–704. [Google Scholar] [CrossRef]
- Lin, Y.-C.; Schlievert, P.M.; Anderson, M.J.; Fair, C.L.; Schaefers, M.M.; Muthyala, R.; Peterson, M.L. Glycerol monolaurate and dodecylglycerol effects on Staphylococcus aureus and toxic shock syndrome toxin-1 in vitro and in vivo. PLoS One 2009, 4, e7499. [Google Scholar] [CrossRef]
- Rasooly, R.; Do, P.M.; Friedman, M. Inhibition of biological activity of staphylococcal enterotoxin A (SEA) by apple juice and apple polyphenols. J. Agric. Food Chem. 2010, 58, 5421–5426. [Google Scholar] [CrossRef]
- Friedman, M.; Rasooly, R.; Do, P.M.; Henika, P.R. The olive compound hydroxytyrosol inactivates Staphylococcus aureus bacteria and Staphylococcal enterotoxin A (SEA). J. Food Sci. 2011, 76, M558–M563. [Google Scholar] [CrossRef]
- Nakagawa, S.; Kushiya, K.; Taneike, I.; Imanishi, K.; Uchiyama, T.; Yamamoto, T. Specific inhibitory action of anisodamine against a staphylococcal superantigenic toxin, toxic shock syndrome toxin 1 (TSST-1), leading to down-regulation of cytokine production and blocking of TSST-1 toxicity in mice. Clin. Diagn. Lab. Immunol. 2005, 12, 399–408. [Google Scholar]
- Schlievert, P.M.; Case, L.C.; Nemeth, K.A.; Davis, C.C.; Sun, Y.; Qin, W.; Wang, F.; Brosnahan, A.J.; Mleziva, J.A.; Peterson, M.L.; et al. α and β chains of hemoglobin inhibit production of Staphylococcus aureus exotoxins. Biochemistry 2007, 46, 14349–14358. [Google Scholar] [CrossRef]
- Rúa, J.; Fernández-Álvarez, L.; Gutiérrez-Larraínzar, M.; Del Valle, P.; De Arriaga, D.; García-Armesto, M.R. Screening of phenolic antioxidants for their inhibitory activity against foodborne Staphylococcus aureus strains. Foodborne Pathog. Dis. 2010, 7, 695–705. [Google Scholar] [CrossRef]
- Larkin, E.A.; Stiles, B.G.; Ulrich, R.G. Inhibition of toxic shock by human monoclonal antibodies against staphylococcal enterotoxin B. PLoS One 2010, 5, e13253. [Google Scholar] [CrossRef]
- Maina, E.K.; Hu, D.-L.; Asano, K.; Nakane, A. Inhibition of emetic and superantigenic activities of staphylococcal enterotoxin A by synthetic peptides. Peptides 2012, 38, 1–7. [Google Scholar] [CrossRef]
- Lord, M.J.; Jolliffe, N.A.; Marsden, C.J.; Pateman, C.S.; Smith, D.C.; Spooner, R.A.; Watson, P.D.; Roberts, L.M. Ricin. Mechanism of cytotoxicity. Toxicol. Rev. 2003, 22, 53–64. [Google Scholar] [CrossRef]
- Jasheway, K.; Pruet, J.; Anslyn, E.V.; Robertus, J.D. Structure-based design of ricin inhibitors. Toxins 2011, 3, 1233–1248. [Google Scholar] [CrossRef]
- Jandhyala, D.M.; Thorpe, C.M.; Magun, B. Ricin and Shiga Toxins: Effects on Host Cell Signal Transduction. In Ricin and Shiga Toxins; Mantis, N., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; Volume 357, pp. 41–65. [Google Scholar]
- Barbieri, L.; Ciani, M.; Girbés, T.; Liu, W.-Y.; Van Damme, E.J.M.; Peumans, W.J.; Stirpe, F. Enzymatic activity of toxic and non-toxic type 2 ribosome-inactivating proteins. FEBS Lett. 2004, 563, 219–222. [Google Scholar] [CrossRef]
- Deeks, E.D.; Cook, J.P.; Day, P.J.; Smith, D.C.; Roberts, L.M.; Lord, J.M. The low lysine content of ricin A chain reduces the risk of proteolytic degradation after translocation from the endoplasmic reticulum to the cytosol. Biochemistry 2002, 41, 3405–3413. [Google Scholar]
- Rasooly, R.; He, X.; Friedman, M. Milk inhibits the biological activity of ricin. J. Biol. Chem. 2012, 287, 27924–27929. [Google Scholar] [CrossRef]
- Rasooly, R.; He, X. Sensitive bioassay for detection of biologically active ricin in food. J. Food Prot. 2012, 75, 951–954. [Google Scholar] [CrossRef]
- Bai, Y.; Watt, B.; Wahome, P.G.; Mantis, N.J.; Robertus, J.D. Identification of new classes of ricin toxin inhibitors by virtual screening. Toxicon 2010, 56, 526–534. [Google Scholar] [CrossRef]
- Chen, Z.-K.; Lin, L.-W.; Weng, X.-H.; Xue, E.-S.; Lin, Y.-H. Interstitial chemotherapy with ricin-loaded thermosensitive hydrogel in pancreatic cancer xenograft. Hepatobiliary Pancreatic Dis. Int. 2009, 8, 418–423. [Google Scholar]
- Fernandes, K.V.; Deus-de-Oliveira, N.; Godoy, M.G.; Guimarães, Z.A.; Nascimento, V.V.; Melo, E.J.; Freire, D.M.; Dansa-Petretski, M.; Machado, O.L. Simultaneous allergen inactivation and detoxification of castor bean cake by treatment with calcium compounds. Braz. J. Med. Biol. Res. 2012, 45, 1002–1010. [Google Scholar] [CrossRef]
- Jackson, L.S.; Zhang, Z.; Tolleson, W.H. Thermal stability of ricin in orange and apple juices. J. Food Sci. 2010, 75, T65–T71. [Google Scholar] [CrossRef]
- Ruiz Olivares, A.; Carrillo-González, R.; González-Chávez, M.D.C.A.; Soto Hernández, R.M. Potential of castor bean (Ricinus communis L.) for phytoremediation of mine tailings and oil production. J. Environ. Manage. 2013, 114, 316–323. [Google Scholar] [CrossRef]
- Friedman, M. Potato glycoalkaloids and metabolites: Roles in the plant and in the diet. J. Agric. Food Chem. 2006, 54, 8655–8681. [Google Scholar] [CrossRef]
- Friedman, M.; Rayburn, J.R.; Bantle, J.A. Developmental toxicology of potato alkaloids in the frog embryo teratogenesis assay-Xenopus (FETAX). Food Chem. Toxicol. 1991, 29, 537–547. [Google Scholar] [CrossRef]
- Rayburn, J.R.; Bantle, J.A.; Qualls, C.W., Jr.; Friedman, M. Protective effects of glucose-6-phosphate and NADP against α-chaconine-induced developmental toxicity in Xenopus embryos. Food Chem. Toxicol. 1995, 33, 1021–1025. [Google Scholar] [CrossRef]
- Friedman, M.; Burns, C.F.; Butchko, C.A.; Blankemeyer, J.T. Folic acid protects against potato glycoalkaloid α-chaconine-induced disruption of frog embryo cell membranes and developmental toxicity. J. Agric. Food Chem. 1997, 45, 3991–3994. [Google Scholar]
- McWilliams, M.L.; Blankemeyer, J.T.; Friedman, M. The folic acid analogue methotrexate protects frog embryo cell membranes against damage by the potato glycoalkaloid α-chaconine. Food Chem. Toxicol. 2000, 38, 853–859. [Google Scholar] [CrossRef]
- Williams, P.J.; Bulmer, J.N.; Innes, B.A.; Pipkin, F.B. Possible roles for folic acid in the regulation of trophoblast invasion and placental development in normal early human pregnancy. Biol. Reprod. 2011, 84, 1148–1153. [Google Scholar] [CrossRef]
- Ravishankar, S.; Jaroni, D.; Zhu, L.; Olsen, C.W.; McHugh, T.H.; Friedman, M. Inactivation of Listeria monocytogenes on ham and bologna using pectin-based apple, carrot, and hibiscus edible films containing carvacrol and cinnamaldehyde. J. Food Sci. 2012, 77, M377–M382. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Friedman, M.; Rasooly, R. Review of the Inhibition of Biological Activities of Food-Related Selected Toxins by Natural Compounds. Toxins 2013, 5, 743-775. https://doi.org/10.3390/toxins5040743
Friedman M, Rasooly R. Review of the Inhibition of Biological Activities of Food-Related Selected Toxins by Natural Compounds. Toxins. 2013; 5(4):743-775. https://doi.org/10.3390/toxins5040743
Chicago/Turabian StyleFriedman, Mendel, and Reuven Rasooly. 2013. "Review of the Inhibition of Biological Activities of Food-Related Selected Toxins by Natural Compounds" Toxins 5, no. 4: 743-775. https://doi.org/10.3390/toxins5040743