Off Label Use of Botulinum Toxin in Children under Two Years of Age: A Systematic Review
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
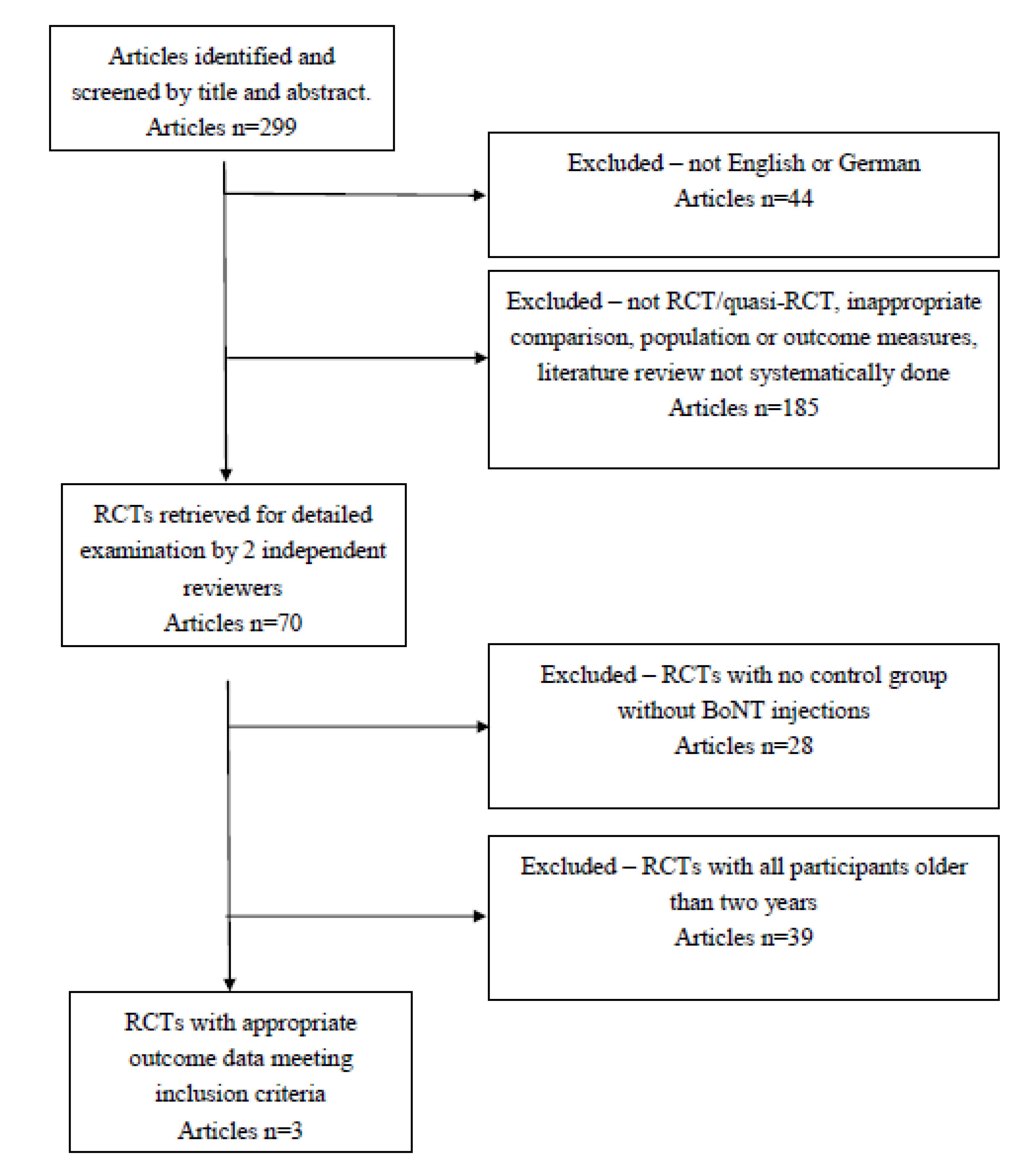
2.1.1. Study Characteristics
2.1.2. Methodological Quality
| Study | Olesch et al. | Tedroff et al. | Graham et al. |
|---|---|---|---|
| 1. Specification of eligibility criteria | 1 | 1 | 1 |
| 2. Random allocation | 1 | 1 | 1 |
| 3. Concealed allocation | 1 | 1 | 1 |
| 4. Prognostic similarity at baseline | 1 | 0 | 0 |
| 5. participant blinding | 0 | 0 | 0 |
| 6. therapist blinding | 0 | 0 | 0 |
| 7. assessor blinding | 0 | 0 | 1 |
| 8. >85% follow up of at least one key outcome | 1 | 1 | 1 |
| 9. intention to treat analysis | 1 | 1 | 1 |
| 10. between-group statistical comparison for at least one key outcome | 1 | 1 | 1 |
| 11. Point estimates and measures of variability for at least one key outcome | 1 | 1 | 1 |
| Total score | 8 | 7 | 8 |
| % of maximum score | 72.7 | 63.6 | 72.7 |
2.1.3. Comparison of Interventions
| Study | Dosage | Drug | Injection site | Muscle identification | Time to reinjection | Anesthesia |
|---|---|---|---|---|---|---|
| Olesch et al. | 0.5 U/kg to 2 U/kg body weight | Ona-BoNT-A (Botox®, Allergan) | M. biceps brachii (100%), M. pronator teres (91%), M. flex. carpi uln. (55%), M. flex. carpi rad. (18%), M. flex. dig. prof. (73%), M. flex. dig. sup. (73%) M. flex. poll. long. (45%) M. adductor pollicis (82%) | muscle stimulation | 3 injections in 16 week cycles | general anesthetic |
| Tedroff et al. | 6 U/kg body weight | Ona-BoNT-A (Botox®, Allergan) | M. gastrocnemius | palpation | 2 injections with 6 month interval | without sedation |
| Graham et al. | 6 U/kg body weight to a max. dose of 16 U/kg body weight | Ona-BoNT-A (Botox®, Allergan) | M. adductor longus M. gastrocnemius | palpation | repeated injections on the basis of clinical symptoms of spasticity | mask anesthesia |
2.1.4. Summary of Results
| Study | Outcome | Timing | N | Treatment | N | Control | p |
|---|---|---|---|---|---|---|---|
| Result, Mean (±SD) | Result, Mean (±SD) | ||||||
| Olesch et al. | MTS Elbow flexors | 12 months | 11 | 34.5 (48.0) | 11 | 77.3 (56.2) | 0.070 |
| Forearm pronators | 22.7 (33.2) | 72.7 (28.7) | 0.001 | ||||
| Wrist flexors | 3.2 (7.2) | 24.1 (28.5) | 0.029 | ||||
| QUEST Dissociated movements | 12 months | 11 | 79.9 (10.9) | 11 | 74.9 (11.8) | n.r. | |
| Grasp | 73.4 (11.0) | 69.7 (14.1) | n.r. | ||||
| Weight bearing | 88.9 (11.0) | 86.1 (11.9) | n.r. | ||||
| Protective extension | 75.8 (16.5) | 60.9 (16.3) | n.r. | ||||
| Total score | 79.6 (8.0) | 72.9 (11.5) | 0.129 | ||||
| PDMS-FM | 12 months | 11 | 542.6 (36.2) | 11 | 537.2 (37.2) | 0.753 | |
| COPM Performance | 12 months | 11 | 2.5 (1.0) | 11 | 1.7 (0.6) | 0.047 | |
| Satisfaction | 2.5 (1.1) | 1.7 (0.9) | 0.090 | ||||
| GAS | 12 months | 11 | 55.8 (6.6) | 11 | 48.8 (8.6) | 0.047 | |
| Change, Mean (±SD) | Change, Mean (±SD) | ||||||
| Tedroff et al. | ROM Ankle joint | 3.5 years | 6 | 1.7 | 9 | 9 | >0.05 |
| Knee joint | 4 | 11 | 0.016 | ||||
| Ashworth score Plantar flexor muscle tone | 3.5 years | 6 | 1 | 9 | 0.3 | >0.05 | |
| Knee flexor muscle tone | 0.5 | 0.2 | 0.05 | ||||
| GMFM-66 | 3.5 years | 6 | 23.6 | 9 | 20.9 | >0.05 | |
| PEDI | 3.5 years | 6 | n.r. | 9 | n.r | >0.05 | |
| Graham et al. | Migration percentage | 3 years | 43 | 2.6 | 42 | 5.5 | 0.05 |
| Progression to surgery | 3 years | 43 | 25.6 | 42 | 52.4 | n.r |
2.1.5. Spasticity
2.1.6. Range of Movement
2.1.7. Motor Development
2.1.8. Other Aspects of Impairment
2.1.9. Adverse Effects and Complications
| Study | Symptoms | Percentage (%) | Course |
|---|---|---|---|
| Olesch et al. | maculopapular rash weakness of the index finger weakness in the finger flexors | 27.3 | completely resolved |
| Tedroff et al. | Weakness dysaesthesia of the skin pain at injection site | 50.0 | completely resolved |
| Graham et al. | major adverse effects (2 deaths) | 6.0 | |
| minor adverse effects | 16.0 | completely resolved |
2.2. Discussion
3. Experimental Section
3.1. Search Strategy
3.2. Information Sources
3.3. Eligibility Criteria and Study Selection
3.4. Data Extraction
| Study | Design | Diagnosis | Age | Treatment | n | Control | n |
|---|---|---|---|---|---|---|---|
| Olesch et al. | SB RCT | CP | 1 year 10 months to 4 year 10 months | BoNT-A and OT | 11 | OT | 11 |
| Tedroff et al. | SB RCT | CP | 11 months to 1 year 11 month | BoNT-A and ST | 6 | Control | 9 |
| Graham et al. | RCT | CP | 1 year to 5 year | BoNT-A and Bracing | 47 | Control | 44 |
3.5. Analysis
4. Conclusion and Direction of Further Research
Conflict of Interest
Acknowledgements
References
- Koman, L.A.; Mooney, J.F., III; Smith, B.; Goodman, A.; Mulvaney, T. Management of cerebral palsy with botulinum-A toxin: Preliminary investigation. J. Pediatr. Orthop. 1993, 13, 489–495. [Google Scholar] [CrossRef]
- Intiso, D. Therapeutic use of botulinum toxin in neurorehabilitation. J. Toxicol. 2012, 2012, 802893. [Google Scholar]
- Naidu, K.; Smith, K.; Sheedy, M.; Adair, B.; Yu, X.; Graham, H.K. Systemic adverse events following botulinum toxin a therapy in children with cerebral palsy. Dev. Med. Child Neurol. 2010, 52, 139–144. [Google Scholar] [CrossRef]
- Tedroff, K.; Lowing, K.; Haglund-Akerlind, Y.; Gutierrez-Farewik, E.; Forssberg, H. Botulinum toxin a treatment in toddlers with cerebral palsy. Acta Paediatr. 2010, 99, 1156–1162. [Google Scholar] [CrossRef]
- Elkamil, A.I.; Andersen, G.L.; Skranes, J.; Lamvik, T.; Vik, T. Botulinum neurotoxin treatment in children with cerebral palsy: A population-based study in norway. Eur. J. Paediatr. Neurol. 2012, 16, 522–527. [Google Scholar] [CrossRef]
- Allergan. Global in Reach. Specialed in Focus. Available online: http://www.allergan.com (accessed on 17 August 2012).
- Berweck, S.; Heinen, F. Cerebralparese; Verlag Hans Huber: Bern, CH, Swizerland, 2008. [Google Scholar]
- Placzek, R. Botulinum toxin A in children with infantile cerebral palsy: Indications and treatment concepts. Orthopade 2010, 39, 23–30. [Google Scholar] [CrossRef]
- Placzek, R.; Siebold, D.; Funk, J.F. Development of treatment concepts for the use of botulinum toxin a in children with cerebral palsy. Toxins 2010, 2, 2258–2271. [Google Scholar] [CrossRef]
- Rosenbaum, P.L.; Walter, S.D.; Hanna, S.E.; Palisano, R.J.; Russell, D.J.; Raina, P.; Wood, E.; Bartlett, D.J.; Galuppi, B.E. Prognosis for gross motor function in cerebral palsy: Creation of motor development curves. J. Am. Med. Assoc. 2002, 288, 1357–1363. [Google Scholar]
- Russell, A.; Cotton, E. The Petö System and Its Evolution in Britain; Acorn Foundation: London, UK, 1994. [Google Scholar]
- Wijnhoven, T.M.; de Onis, M.; Onyango, A.W.; Wang, T.; Bjoerneboe, G.E.; Bhandari, N.; Lartey, A.; al Rashidi, B. Assessment of gross motor development in the who multicentre growth reference study. Food Nutr. Bull. 2004, 25, S37–S45. [Google Scholar]
- Pascual-Pascual, S.I.; Pascual-Castroviejo, I. Safety of botulinum toxin type a in children younger than 2 years. Eur. J. Paediatr. Neurol. 2009, 13, 511–515. [Google Scholar] [CrossRef]
- Olesch, C.A.; Greaves, S.; Imms, C.; Reid, S.M.; Graham, H.K. Repeat botulinum toxin-A injections in the upper limb of children with hemiplegia: A randomized controlled trial. Dev. Med. Child Neurol. 2010, 52, 79–86. [Google Scholar]
- Graham, H.K.; Boyd, R.; Carlin, J.B.; Dobson, F.; Lowe, K.; Nattrass, G.; Thomason, P.; Wolfe, R.; Reddihough, D. Does botulinum toxin a combined with bracing prevent hip displacement in children with cerebral palsy and “hips at risk”? A randomized, controlled trial. J. Bone Joint Surg. 2008, 90, 23–33. [Google Scholar]
- PEDro Database. Available online: http://www.pedro.org.au (accessed on 17 August 2012).
- Lukban, M.B.; Rosales, R.L.; Dressler, D. Effectiveness of botulinum toxin a for upper and lower limb spasticity in children with cerebral palsy: A summary of evidence. J. Neural Transm. 2009, 116, 319–331. [Google Scholar] [CrossRef]
- Koman, L.A.; Mooney, J.F., III; Smith, B.P.; Walker, F.; Leon, J.M. Botulinum toxin type a neuromuscular blockade in the treatment of lower extremity spasticity in cerebral palsy: A randomized, double-blind, placebo-controlled trial. Botox study group. J. Pediatr. Orthop. 2000, 20, 108–115. [Google Scholar]
- Fosang, A.L.; Galea, M.P.; McCoy, A.T.; Reddihough, D.S.; Story, I. Measures of muscle and joint performance in the lower limb of children with cerebral palsy. Dev. Med. Child Neurol. 2003, 45, 664–670. [Google Scholar]
- DeMatteo, C.; Law, M.; Russell, D.; Pollock, N.; Rosenbaum, P.; Walter, S. The reliability and validity of quality of upper extremity skills test. Phys. Occup. Ther. Pediatr. 1993, 13, 1–18. [Google Scholar]
- Thorley, M.; Lannin, N.; Cusick, A.; Novak, I.; Boyd, R. Construct validity of the quality of upper extremity skills test for children with cerebral palsy. Dev. Med. Child Neurol. 2012, 54, 1037–1043. [Google Scholar] [CrossRef]
- Chien, C.W.; Bond, T.G. Measurement properties of fine motor scale of peabody developmental motor scales-second edition: A rasch analysis. Am. J. Phys. Med. Rehabil. 2009, 88, 376–386. [Google Scholar] [CrossRef]
- Maring, J.R.; Elbaum, L. Concurrent validity of the early intervention developmental profile and the peabody developmental motor scale-2. Pediatr. Phys. Ther. 2007, 19, 116–120. [Google Scholar] [CrossRef]
- Palisano, R.J.; Kolobe, T.H.; Haley, S.M.; Lowes, L.P.; Jones, S.L. Validity of the peabody developmental gross motor scale as an evaluative measure of infants receiving physical therapy. Phys. Ther. 1995, 75, 939–948. [Google Scholar]
- Wei, S.; Su-Juan, W.; Yuan-Gui, L.; Hong, Y.; Xiu-Juan, X.; Xiao-Mei, S. Reliability and validity of the gmfm-66 in 0- to 3-year-old children with cerebral palsy. Am. J. Phys. Med. Rehabil. 2006, 85, 141–147. [Google Scholar] [CrossRef]
- Steenbeek, D.; Meester-Delver, A.; Becher, J.G.; Lankhorst, G.J. The effect of botulinum toxin type a treatment of the lower extremity on the level of functional abilities in children with cerebral palsy: Evaluation with goal attainment scaling. Clin. Rehabil. 2005, 19, 274–282. [Google Scholar] [CrossRef]
- Ostensjo, S.; Oien, I.; Fallang, B. Goal-oriented rehabilitation of preschoolers with cerebral palsy—A multi-case study of combined use of the canadian occupational performance measure (copm) and the goal attainment scaling (gas). Dev. Neurorehabil. 2008, 11, 252–259. [Google Scholar] [CrossRef]
- Graham, H.K. Painful hip dislocation in cerebral palsy. Lancet 2002, 359, 907–908. [Google Scholar] [CrossRef]
- Boyd, R.N.; Dobson, F.; Parrott, J.; Love, S.; Oates, J.; Larson, A.; Burchall, G.; Chondros, P.; Carlin, J.; Nattrass, G.; et al. The effect of botulinum toxin type a and a variable hip abduction orthosis on gross motor function: A randomized controlled trial. Eur. J. Neurol. 2001, 8, 109–119. [Google Scholar]
- Ma, J.; Smith, B.P.; Smith, T.L.; Walker, F.O.; Rosencrance, E.V.; Koman, L.A. Juvenile and adult rat neuromuscular junctions: Density, distribution, and morphology. Muscle Nerve 2002, 26, 804–809. [Google Scholar] [CrossRef]
- Santafé, M.M.; Salon, I.; Garcia, N.; Lanuza, M.A.; Uchitel, O.D.; Tomàs, J. Modulation of ACh release by presynaptic muscarinic autoreceptors in the neuromuscular junction of the newborn and adult rat. Eur. J. Neurosci. 2003, 17, 119–127. [Google Scholar] [CrossRef]
- Barrett, R.S.; Lichtwark, G.A. Gross muscle morphology and structure in spastic cerebral palsy: A systematic review. Dev. Med. Child Neurol. 2010, 52, 794–804. [Google Scholar] [CrossRef]
- Ryll, U.; Bastiaenen, C.; de Bie, R.; Staal, B. Effects of leg muscle botulinum toxin a injections on walking in children with spasticity-related cerebral palsy: A systematic review. Dev. Med. Child Neurol. 2011, 53, 210–216. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Druschel, C.; Althuizes, H.C.; Funk, J.F.; Placzek, R. Off Label Use of Botulinum Toxin in Children under Two Years of Age: A Systematic Review. Toxins 2013, 5, 60-72. https://doi.org/10.3390/toxins5010060
Druschel C, Althuizes HC, Funk JF, Placzek R. Off Label Use of Botulinum Toxin in Children under Two Years of Age: A Systematic Review. Toxins. 2013; 5(1):60-72. https://doi.org/10.3390/toxins5010060
Chicago/Turabian StyleDruschel, Claudia, Henriette C. Althuizes, Julia F. Funk, and Richard Placzek. 2013. "Off Label Use of Botulinum Toxin in Children under Two Years of Age: A Systematic Review" Toxins 5, no. 1: 60-72. https://doi.org/10.3390/toxins5010060




