Plant Insecticidal Toxins in Ecological Networks
Abstract
:1. Introduction: The Diversity of Plant Insecticidal Toxins
1.1. Secondary Metabolites and Plant Defences
| Plant Insecticidal Compounds | Activity | Plant localization | Insect | References |
|---|---|---|---|---|
| C Based compounds | ||||
| Terpenoids | ||||
| Monoterpene alcohol | repellent | flowers | Lasius niger (Hymenoptera) | [2] |
| Diterpenoids | repellentantifeeding | stems | Ostrinia nubilalis (Lepidoptera) | [3] |
| Cardenolides | toxicity | aerial and subterranean parts | Danaus plexippus (Lepidoptera) | [4] |
| Iridoid glycosides | toxicity | leavesnectar | Junonia coenia (Lepidoptera) | [5,6] |
| Phenolics of low molecular weight | ||||
| Phenolic glucosides | deterrenttoxicity | aerial parts | Generalist and specialist invertebrates | [7] |
| Aromatic esters | repellent | nectar | Solenopsis xyloni (Hymenoptera) | [8] |
| Flavonoids | repellent | leaves | Spodoptera exigua (Lepidoptera) | [9] |
| Isoflavones | feeding deterrent | roots | Costelytra zealandicator (Coleoptera) | [10] |
| Furanocoumarins and coumarins | toxicity | leaves | Trichoplusia ni (Lepidoptera) | [11] |
| Tannins | toxicity (oxidation) | leaves | Orgyia leucostigma (Lepidoptera) | [12] |
| N Based compounds | ||||
| Cyanogenic glucosides | toxicity | leaves | Spodoptera frugiperda (Lepidoptera) | [13] |
| Glucosinolates | toxicity | leaves | Pieris brassicae (Lepidoptera) | [14] |
| Alkaloïds | repellent | nectar | Bee pollinators | [8,15,16] |
| Pyrrolizidine alkaloids | toxicity | leaves | Non adapted Arctiidae (Lepidoptera) | [17,18] |
| Azoglucosides | toxicity (mutagen) | leaves, seeds, cones | Rhopalotria sp. (Coleoptera) | [19] |
| Non protein amino-acid | toxicity | leaves | Invertebrates | [20] |
| Protease inhibitors | toxicity | leaves | Spodoptera littoralis (Lepidoptera) | [21] |
| Peptides (cyclotides) | toxicity | leaves, flowers, stems, roots | Invertebrates | [22] |
1.2. Diversity of Effects on Insects
1.2.1. Repellent Effect
1.2.2. Growth Inhibitor, Toxic Effects
1.2.3. Pleitropic Role of Tannins
2. Insecticidal Toxins in an Antagonistic Context
2.1. Strategies Selected in Insects to Overcome Plant Chemical Defences
2.2. The Co-evolutionary Arms Race and the Evolution of Specialisation
3. Plant Toxins in Mutualistic Interactions
3.1. Plants Use Toxins to Choose Adequate Partners
3.2. Plants Use Toxins to Control Mutualistic Partners
3.3. Toxins in Mutualisms Usually Evolve in Relation to Anti-Herbivore Defence
4. Insecticidal Toxins and Multi-Trophic Interactions
4.1. Effect of Plant Toxins on Predators and the Evolution of Aposematism
4.2. Effect of Plant Toxins on Parasitoids and Entomopathogens
4.3. Plant Toxins, Pharmacophagy, and Self-Medication in Insects
4.4. Plant Toxins and Insect Symbionts
5. Conclusions
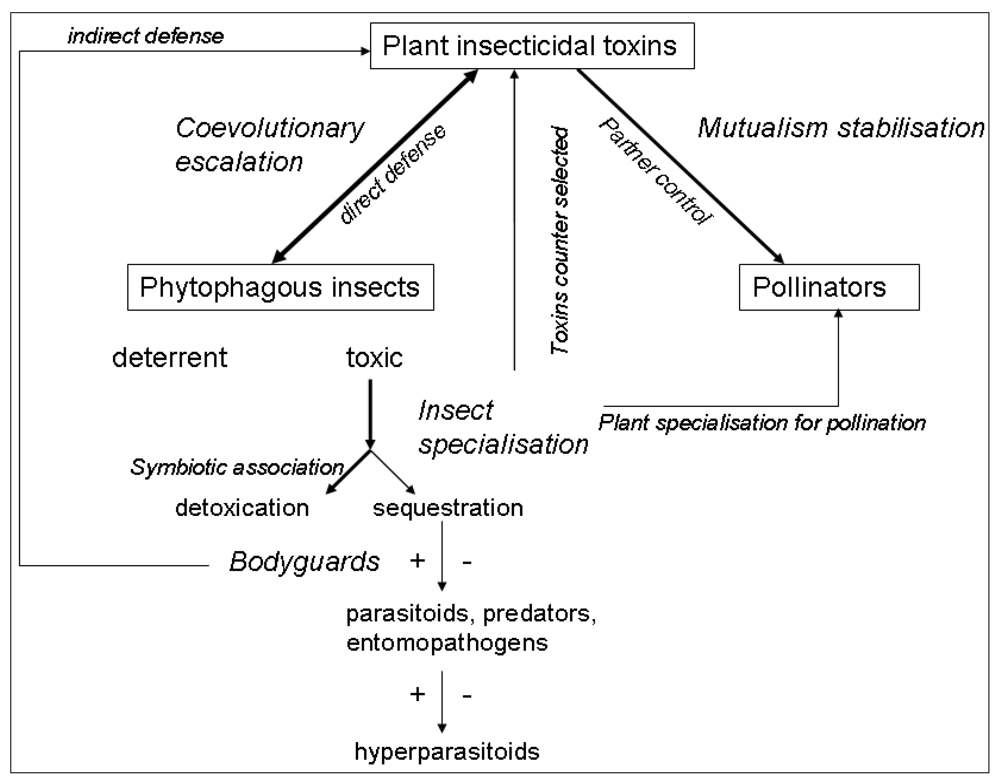
References
- Macias, F.A.; Galindo, J.L.G.; Galindo, J.C.G. Evolution and current status of ecological phytochemistry. Phytochemistry 2007, 68, 2917–2936. [Google Scholar]
- Junker, R.R.; Gershenzon, J.; Unsicker, S.B. Floral odor bouquet loses its ant repellent properties after inhibition of terpene biosynthesis. J. Chem. Ecol. 2011, 37, 1323–1331. [Google Scholar]
- Schmelz, E.A.; Kaplan, F.; Huffaker, A.; Dafoe, N.J.; Vaughan, M.M.; Ni, X.Z.; Rocca, J.R.; Alborn, H.T.; Teal, P.E. Identity, regulation, and activity of inducible diterpenoid phytoalexins in maize. Proc. Natl. Acad. Sci. USA 2011, 108, 5455–5460. [Google Scholar]
- Rasmann, S.; Agrawal, A.A. Latitudinal patterns in plant defense: Evolution of cardenolides, their toxicity and induction following herbivory. Ecol. Lett. 2010, 14, 476–483. [Google Scholar]
- Stephenson, A.G. Toxic nectar deters nectar thieves of Catalpa speciosa. Am. Midl. Nat. 1981, 105, 381–383. [Google Scholar] [CrossRef]
- Smilanich, A.M.; Dyer, L.A.; Chambers, J.Q.; Bowers, M.D. Immunological cost of chemical defence and the evolution of herbivore diet breadth. Ecol. Lett. 2009, 12, 612–621. [Google Scholar]
- Boeckler, G.A.; Gershenzon, J.; Unsicker, S.B. Phenolic glycosides of the Salicaceae and their role as anti-herbivore defenses. Phytochemistry 2011, 72, 1497–1509. [Google Scholar]
- Kessler, D.; Baldwin, I.T. Making sense of nectar scents: The effects of nectar secondary metabolites on floral visitors of Nicotiana attenuata. Plant J. 2007, 49, 840–854. [Google Scholar] [CrossRef]
- Cipollini, D.; Stevenson, R.; Enright, S.; Eyles, A.; Bonello, P. Phenolic metabolites in leaves of the invasive shrub, Lonicera maackii, and their potential phytotoxic and anti-herbivore effects. J. Chem. Ecol. 2008, 34, 144–152. [Google Scholar] [CrossRef]
- Lane, G.A.; Sutherland, O.R.W.; Skipp, R.A. Isoflavonoids as insect feeding deterrents and antifungal components from root of Lupinus angustifolius. J. Chem. Ecol. 1987, 13, 771–783. [Google Scholar] [CrossRef]
- Lampert, E.C.; Zangerl, A.R.; Berenbaum, M.R.; Ode, P.J. Generalist and specialist host-parasitoid associations respond differently to wild parsnip (Pastinaca sativa) defensive chemistry. Ecol. Entomol. 2011, 36, 52–61. [Google Scholar] [CrossRef]
- Barbehenn, R.V.; Maben, R.E.; Knoester, J.J. Linking phenolic oxidation in the midgut lumen with oxidative stress in the midgut tissues of a tree-feeding caterpillar Malacosoma disstria (Lepidoptera: Lasiocampidae). Environ. Entomol. 2008, 37, 1113–1118. [Google Scholar] [CrossRef]
- Hay-Roe, M.M.; Meagher, R.L.; Nagoshi, R.N. Effects of cyanogenic plants on fitness in two host strains of the fall armyworm (Spodoptera frugiperda). J. Chem. Ecol. 2011, 37, 1314–1322. [Google Scholar] [CrossRef]
- Harvey, J.A.; van Dam, N.M.; Gols, R. Interactions over four trophic levels: Foodplant quality affects development of a hyperparasitoid as mediated through a herbivore and its primary parasitoid. J. Anim. Ecol. 2003, 72, 520–531. [Google Scholar]
- Gegear, R.J.; Manson, J.S.; Thomson, J.D. Ecological context influences pollinator deterrence by alkaloids in floral nectar. Ecol. Lett. 2007, 10, 375–382. [Google Scholar]
- Irwin, R.E.; Adler, L.S. Nectar secondary compounds affect self-pollen transfer: Implications for female and male reproduction. Ecology 2008, 89, 2207–2217. [Google Scholar]
- Singer, M.S.; Mace, K.C.; Bernays, E.A. Self-Medication as adaptive plasticity: Increased ingestion of plant toxins by parasitized caterpillars. PLoS One 2009, 4, e4796. [Google Scholar]
- Smilanich, A.M.; Mason, P.A.; Sprung, L.; Chase, T.R.; Singer, M.S. Complex effects of parasitoids on pharmacophagy and diet choice of a polyphagous caterpillar. Oecologia 2011, 165, 995–1005. [Google Scholar]
- Schneider, D.; Wink, M.; Sporer, F.; Lounibos, P. Cycads: Their evolution, toxins, herbivores and insect pollinators. Naturwissenschaften 2002, 89, 281–294. [Google Scholar] [CrossRef]
- Bown, A.W.; MacGregor, K.B.; Shelp, B.J. Gamma-aminobutyrate: Defense against invertebrate pests? Trends Plant Sci. 2006, 11, 424–427. [Google Scholar] [CrossRef]
- Hartl, M.; Giri, A.P.; Kaur, H.; Baldwin, I.T. Serine protease inhibitors specifically defend Solanum nigrum against generalist herbivores but do not influence plant growth and development. Plant Cell 2010, 22, 4158–4175. [Google Scholar] [CrossRef]
- Gruber, W.C.; Cemazar, M.; Anderson, M.A.; Craik, D.J. Insecticidal plant cyclotides and related cystine knot toxins. Toxicon 2007, 49, 561–575. [Google Scholar]
- Sampedro, L.; Moreira, X.; Zas, R. Costs of constitutive and herbivore-induced chemical defences in pine trees emerge only under low nutrient availability. J. Ecol. 2011, 99, 818–827. [Google Scholar] [Green Version]
- Roberts, M.R.; Paul, N.D. Seduced by the dark side: Integrating molecular and ecological perspectives on the inflence of light on plant defence against pests and pathogens. New Phytol. 2006, 170, 677–699. [Google Scholar]
- Gutbrodt, B.; Mody, K.; Dorn, S. Drought changes plant chemistry and causes contrasting responses in lepidopteran herbivores. Oikos 2011, 120, 1732–1740. [Google Scholar]
- Baldwin, I.T. Inducible nicotine production in native Nicotiana as an example of adaptive phenotypic plasticity. J. Chem. Ecol. 1999, 25, 3–30. [Google Scholar] [CrossRef]
- Baldwin, I.T. Jasmonate-induced responses are costly but benefit plants under attack in native populations. Proc. Natl. Acad. Sci. USA 1998, 95, 8113–8118. [Google Scholar]
- Heil, M.; Baldwin, I.T. Fitness costs of induced resistance: Emerging experimental support for a slippery concept. Trends Plant Sci. 2002, 7, 61–67. [Google Scholar]
- Strauss, S.Y.; Rudgers, J.A.; Lau, J.A.; Irwin, R.E. Direct and ecological costs of resistance to herbivory. Trends Ecol. Evol. 2002, 17, 278–285. [Google Scholar]
- Koricheva, J. Meta-analysis of sources of variation in fitness costs of plant antiherbivore defenses. Ecology 2002, 83, 176–190. [Google Scholar]
- Herms, D.A.; Mattson, W.J. The dilemma of plants—to grow or defend. Q. Rev. Biol. 1992, 67, 283–335. [Google Scholar]
- Lankau, R.A.; Kliebenstein, D.J. Competition, herbivory and genetics interact to determine the accumulation and fitness consequences of a defence metabolite. J. Ecol. 2009, 97, 78–88. [Google Scholar]
- van Dam, N.M. Belowground herbivory and plant defenses. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 373–391. [Google Scholar]
- Newton, E.L.; Bullock, J.M.; Hodgson, D.J. Glucosinolate polymorphism in wild cabbage (Brassica oleracea) influences the structure of herbivore communities. Oecologia 2009, 160, 63–76. [Google Scholar] [CrossRef]
- Wang, S.-D.; Liu, W.; Xue, C.-B.; Luo, W.-C. The effects of luteolin on phenoloxidase and the growth of Spodoptera exigua (Hubner) larvae (Lepidoptera: Noctuidae). J. Pestic. Sci. 2010, 35, 483–487. [Google Scholar] [CrossRef]
- Feeny, P.P.; Bostock, H. Seasonal changes in tannin content of oak leaves. Phytochemistry 1968, 7, 871–880. [Google Scholar]
- Salminen, J.-P.; Karonen, M. Chemical ecology of tannins and other phenolics: We need a change in approach. Funct. Ecol. 2011, 25, 325–338. [Google Scholar]
- Salminen, J.P.; Lempa, K. Effects of hydrolysable tannins on a herbivorous insect: Fate of individual tannins in insect digestive tract. Chemoecology 2002, 12, 203–211. [Google Scholar]
- Barbehenn, R.V.; Constabel, C.P. Tannins in plant-herbivore interactions. Phytochemistry 2011, 72, 1551–1565. [Google Scholar]
- Despres, L.; David, J.P.; Gallet, C. The evolutionary ecology of insect resistance to plant chemicals. Trends Ecol. Evol. 2007, 22, 298–307. [Google Scholar]
- Hagele, B.F.; Rowell-Rahier, M. Dietary mixing in three generalist herbivores: Nutrient complementation or toxin dilution? Oecologia 1999, 119, 521. [Google Scholar] [CrossRef]
- Dillon, R.; Charnley, K. Mutualism between the desert locust Schistocerca gregaria and its gut microbiota. Res. Microbiol. 2002, 153, 503–509. [Google Scholar] [CrossRef]
- Ode, P.J. Plant chemistry and natural enemy fitness: Effects on herbivore and natural enemy interactions. Annu. Rev. Entomol. 2006, 51, 163–185. [Google Scholar]
- Nishida, R. Sequestration of defensive substances from plants by Lepidoptera. Annu. Rev. Entomol. 2002, 47, 57–92. [Google Scholar]
- Eisner, T.; Meinwald, J. Defense-mechanisms of Arthropods. 129. The chemistry of sexual selection. Proc. Natl. Acad. Sci. USA 1995, 92, 50–55. [Google Scholar] [CrossRef]
- Ehrlich, P.R.; Raven, P.H. Butterflies and plants—A study in coevolution. Evolution 1964, 18, 586–608. [Google Scholar]
- Agrawal, A.A.; Fishbein, M. Phylogenetic escalation and decline of plant defense strategies. Proc. Natl. Acad. Sci. USA 2008, 105, 10057–10060. [Google Scholar]
- Adler, L.S. The ecological significance of toxic nectar. Oikos 2000, 91, 409–420. [Google Scholar]
- Baker, H.G. Non-sugar chemical constituents of nectar. Apidologie 1977, 8, 349–356. [Google Scholar]
- Johnson, S.D.; Hargreaves, A.L.; Brown, M. Dark, bitter-tasting nectar functions as a filter of flower visitors in a bird-pollinated plant. Ecology 2006, 87, 2709–2716. [Google Scholar]
- Adler, L.S.; Irwin, R.E. Ecological costs and benefits of defenses in nectar. Ecology 2005, 86, 2968–2978. [Google Scholar]
- Stephenson, A.G. Iridoid glycosides in the nectar of Catalpa speciosa are unpalatable to nectar thieves. J. Chem. Ecol. 1982, 8, 1025–1034. [Google Scholar] [CrossRef]
- Elliott, S.E.; Irwin, R.E.; Adler, L.S.; Williams, N.M. The nectar alkaloid, gelsemine, does not affect offspring performance of a native solitary bee, Osmia lignaria (Megachilida). Ecol. Entomol. 2008, 33, 298–304. [Google Scholar] [CrossRef]
- Manson, J.S.; Otterstatter, M.C.; Thomson, J.D. Consumption of a nectar alkaloid reduces pathogen load in bumble bees. Oecologia 2010, 162, 81–89. [Google Scholar]
- Pellmyr, O.; Huth, C.J. Evolutionary stability of mutualism between Yuccas and Yucca Moths. Nature 1994, 372, 257–260. [Google Scholar]
- Gallet, C.; Ibanez, S.; Zinger, L.; Taravel, F.R.; Trierweiler, M.; Jeacomine, I.; Despres, L. Plant chemical defense induced by a seed-eating pollinator mutualist. J. Chem. Ecol. 2007, 33, 2078–2089. [Google Scholar]
- Ibanez, S.; Gallet, C.; Dommanget, F.; Despres, L. Plant chemical defence: A partner control mechanism stabilising plant—Seed-eating pollinator mutualisms. BMC Evol. Biol. 2009, 9, 261. [Google Scholar]
- Louca, S.; Ibanez, S.; Piau, D.; Despres, L. Specialized nursery pollination mutualisms as evolutionary traps stabilized by antagonistic traits. J. Theor. Biol. 2012, 296, 65–83. [Google Scholar]
- Armbruster, W.S. Exaptations link evolution of plant-herbivore and plant-pollinator interactions: A phylogenetic inquiry. Ecology 1997, 78, 1661–1672. [Google Scholar]
- Armbruster, W.S.; Howard, J.J.; Clausen, T.P.; Debevec, E.M.; Loquvam, J.C.; Matsuki, M.; Cerendolo, B.; Andel, F. Do biochemical exaptations link evolution of plant defense and pollination systems? Historical hypotheses and experimental tests with Dalechampia vines. Am. Nat. 1997, 149, 461–484. [Google Scholar]
- Armbruster, W.S.; Lee, J.; Baldwin, B.G. Macroevolutionary patterns of defense and pollination in Dalechampia vines: Adaptation, exaptation, and evolutionary novelty. Proc. Natl. Acad. Sci. USA 2009, 106, 18085–18090. [Google Scholar]
- Yang, S.; Ruuhola, T.; Haviola, S.; Rantala, M.J. Effects of host-plant shift on immune and other key life-history traits of an eruptive Geometrid, Epirrita autumnata (Borkhausen). Ecol. Entomol. 2008, 33, 510–516. [Google Scholar] [CrossRef]
- Janmaat, A.F.; Myers, J.H. The cost of resistance to Bacillus thuringiensis varies with the host plant of Trichoplusia ni. Proc. Royal Soc. B-Biol. Sci. 2005, 272, 1031–1038. [Google Scholar] [CrossRef]
- Ludlum, C.T.; Felton, G.W.; Duffey, S.S. Plant defences—Chlorogenic acid and polyphenol oxidase enhance toxicity of Bacillus thuringiensis subsp kurstaki to Heliothis zea. J. Chem. Ecol. 1991, 17, 217–237. [Google Scholar] [CrossRef]
- Janmaat, A.F.; Myers, J.H. Rapid evolution and the cost of resistance to Bacillus thuringiensis in greenhouse populations of cabbage loopers, Trichoplusia ni. Proc Royal Soc. Lond. Ser. B-Biol. Sci. 2003, 270, 2263–2270. [Google Scholar] [CrossRef]
- Ojala, K.; Julkunen-Tiito, R.; Lindstrom, L.; Mappes, J. Diet affects the immune defence and life-history traits of an Arctiid moth Parasemia plantaginis. Evol. Ecol. Res. 2005, 7, 1153–1170. [Google Scholar]
- Manson, J.S.; Thomson, J.D. Post-ingestive effects of nectar alkaloids depend on dominance status of bumblebees. Ecol. Entomol. 2009, 34, 421–426. [Google Scholar]
- Cory, J.S.; Hoover, K. Plant-mediated effects in insect-pathogen interactions. Trends Ecol. Evol. 2006, 21, 278–286. [Google Scholar]
- Cook, S.P.; Webb, R.E.; Podgwaite, J.D.; Reardon, R.C. Increased mortality of gypsy moth Lymantria dispar (L.) (Lepidoptera: Lymantriidae) exposed to gypsy moth nuclear polyhedrosis virus in combination with the phenolic gycoside salicin. J. Econ. Entomol. 2003, 96, 1662–1667. [Google Scholar] [CrossRef]
- Karban, R.; Karban, C.; Huntzinger, M.; Pearse, I.; Crutsinger, G. Diet mixing enhances the performance of a generalist caterpillar, Platyprepia virginalis. Ecol. Entomol. 2010, 35, 92–99. [Google Scholar] [CrossRef]
- Boppre, M. Redefining pharmacophagy. J. Chem. Ecol. 1984, 10, 1151–1154. [Google Scholar]
- Chapuisat, M.; Oppliger, A.; Magliano, P.; Christe, P. Wood ants use resin to protect themselves against pathogens. Proc. Royal Soc. B-Biol. Sci. 2007, 274, 2013–2017. [Google Scholar] [CrossRef]
- Bankova, V.S.; de Castro, S.L.; Marcucci, M.C. Propolis: Recent advances in chemistry and plant origin. Apidologie 2000, 31, 3–15. [Google Scholar]
- Karban, R.; English-Loeb, G. Tachinid parasitoids affect host plant choice by caterpillars to increase caterpillar survival. Ecology 1997, 78, 603–611. [Google Scholar]
- Feldhaar, H. Bacterial symbionts as mediators of ecologically important traits of insect hosts. Ecol. Entomol. 2011, 36, 533–543. [Google Scholar]
- Dowd, P.F. Insect fungal symbionts—A promising source of detoxifying enzymes. J. Ind. Microbiol. 1992, 9, 149–161. [Google Scholar]
- Bezemer, T.M.; van Dam, N.M. Linking aboveground and belowground interactions via induced plant defenses. Trends Ecol. Evol. 2005, 20, 617–624. [Google Scholar] [Green Version]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ibanez, S.; Gallet, C.; Després, L. Plant Insecticidal Toxins in Ecological Networks. Toxins 2012, 4, 228-243. https://doi.org/10.3390/toxins4040228
Ibanez S, Gallet C, Després L. Plant Insecticidal Toxins in Ecological Networks. Toxins. 2012; 4(4):228-243. https://doi.org/10.3390/toxins4040228
Chicago/Turabian StyleIbanez, Sébastien, Christiane Gallet, and Laurence Després. 2012. "Plant Insecticidal Toxins in Ecological Networks" Toxins 4, no. 4: 228-243. https://doi.org/10.3390/toxins4040228




