Contrasting Nephropathic Responses to Oral Administration of Extract of Cultured Penicillium polonicum in Rat and Primate
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of fungal extract for toxicity test
2.2. Rat experimental
| Rat | Period of exposure | P. polonicum treatment format | Daily consumption of nephrotoxin, expressed as weight of shredded wheat substrate moulded, and consumed, whole or as extract in feed |
|---|---|---|---|
| 1 | 4 days | Moulded shredded wheat | 5 g |
| 2 | 4 days | Water-soluble extract fraction | 7 g |
| 3 | 4 days | Water-soluble extract fraction | 35 g |
| 4 | 5 weeks | Moulded shredded wheat | 5 g |
| 5 | 10 weeks, and more | Moulded shredded wheat | 5 g |
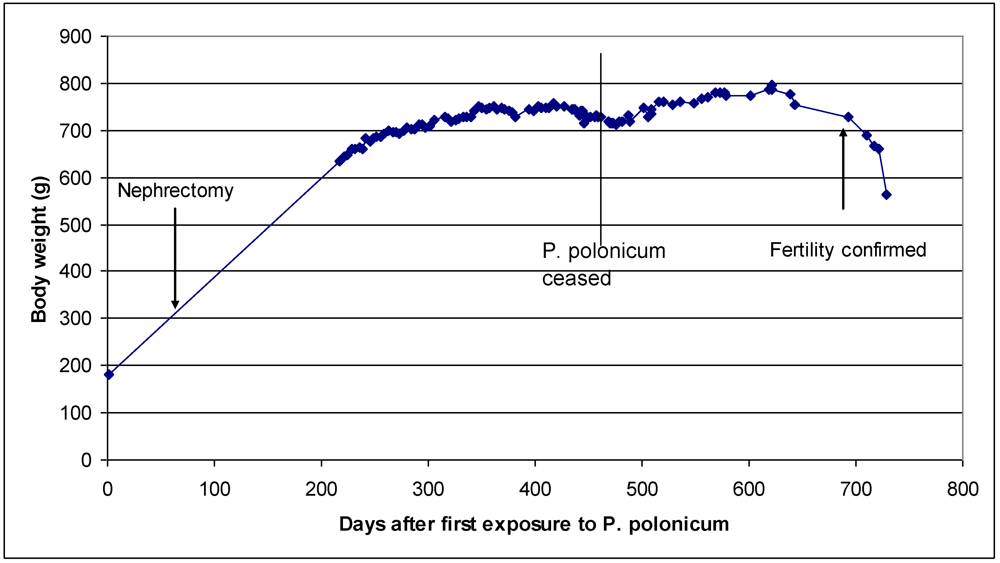
2.3. Primate experimental
3. Results
3.1. Rat: Demonstration of renal histopathogenic equivalence of whole fungal culture and the extracted fraction after short-term exposure
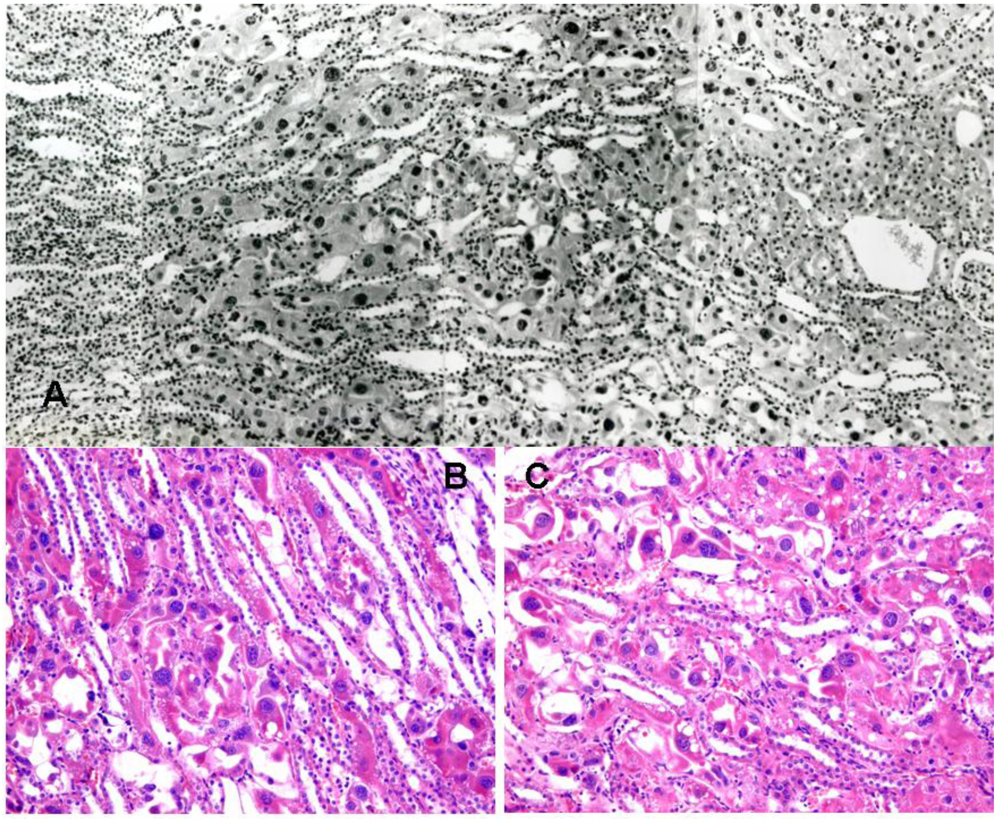
3.2. Rat: Renal histopathology after five, ten or sixty-six weeks on a diet containing a 20% component of shredded wheat moulded by P. polonicum
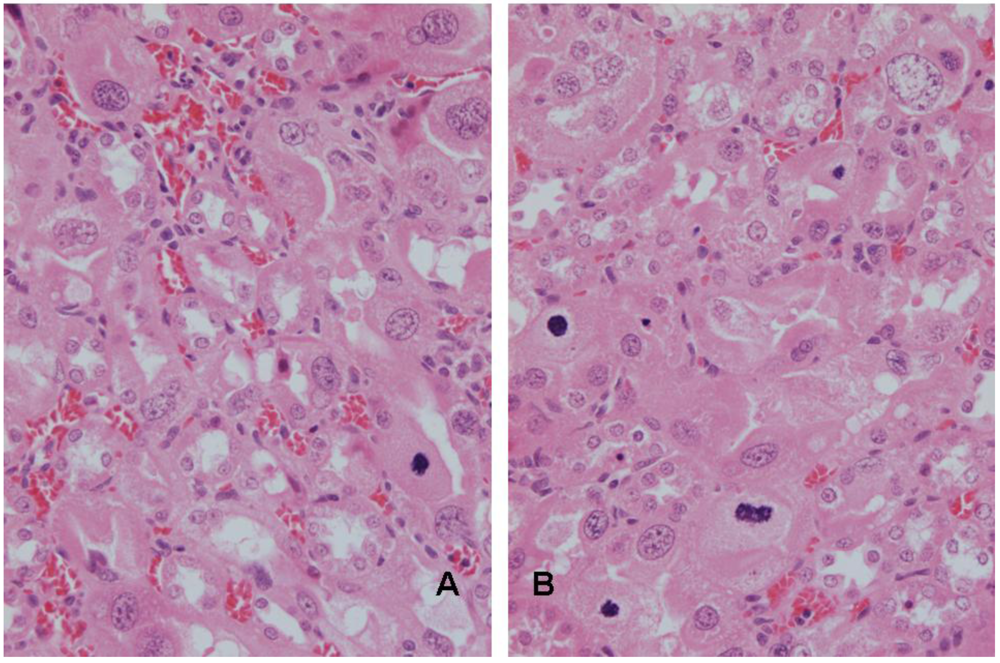
3.3. Vervet monkey
| Clinical | Before treatment | After treatment |
|---|---|---|
| Body wt (kg) | ||
| Treated | 2.40 | 2.40 |
| Control | 2.33 | 2.24 |
| Rectal temp (°C) | ||
| Treated | 39.0 | 39.0 |
| Control | 38.4 | 38.8 |
| Respiration (/min) | ||
| Treated | 42 | 40 |
| Control | 36 | 28 |
| Pulse (/min) | ||
| Treated | 180 | 180 |
| Control | 180 | 160 |
| Three weeks before treatment | Penultimate treatment day | Last treatment day | Reference data (n = 74 *) | |
|---|---|---|---|---|
| Treated | 2.72 | 2.67 | 2.99 | 2.43 ± 0.56 |
| Control | 3.09 | 2.09 | 2.69 | 2.43 ± 0.56 |
| RBC | Hb | Hct | MCV | RDW | Plt | WBC | N | L | |
|---|---|---|---|---|---|---|---|---|---|
| T 4 weeks before treatment | 5.25 | 13.0 | 38.6 | 73.5 | 24.0 | 467 | 7.05 | 2.23 | 4.50 |
| T penultimate treatment day | 5.09 | 12.6 | 37.6 | 73.8 | 20.0 | 425 | 5.26 | 1.96 | 3.00 |
| T last treatment day | 5.02 | 11.7 | 37.4 | 74.5 | 23.3 | 429 | 6.90 | 2.66 | 3.89 |
| C 4 weeks before treatment | 5.21 | 13.2 | 40.0 | 76.7 | 23.0 | 338 | 3.26 | 1.07 | 1.92 |
| C penultimate treatment day | 5.00 | 12.8 | 38.3 | 76.5 | 18.8 | 271 | 7.52 | 6.01 | 0.97 |
| C last treatment day | 5.15 | 12.3 | 39.5 | 76.7 | 19.8 | 297 | 3.65 | 1.80 | 1.51 |
| T1 | T2 | T3 | C1 | C2 | C3 | Reference * | |
|---|---|---|---|---|---|---|---|
| Sodium mmol/L | 149 | 154 | 146 | 148 | 156 | 149 | 149.9 (2.8) |
| Potassium mmol/L | 3.8 | 3.7 | 3.5 | 3.9 | 3.6 | 3.8 | 3.5 (0.6) |
| Chloride mmol/L | 112 | 115 | 108 | 100 | 110 | 105 | 108.5 (2.2) |
| CO2 mmol/L | 27 | 27 | 26 | 26 | 23 | 31 | 25.7 (4.0) |
| Anion gap mmol/L | 10 | 12 | 12 | 13 | 23 | 13 | 15.4 (4.8) |
| Urea mmol/L | 4.6 | 8.9 | 12.5 | 3.9 | 17.1 | 10 | 5.4 (1.7) |
| Creatinine µmol/L | 71 | 71 | 71 | 71 | 124 | 80 | 67.7 (17.9) |
| Total protein g/L | 62 | 63 | 62 | 60 | 61 | 58 | 61.3 (4.6) |
| Albumin g/L | 42 | 43 | 43 | 42 | 42 | 41 | 39.6 (3.6) |
| Globulin g/L | 20 | 20 | 19 | 18 | 19 | 17 | 21.7 (5.1) |
| Calcium mmol/L | 2.33 | 2.33 | 2.30 | 2.38 | 2.33 | 2.30 | 2.23 (0.2) |
| Phosphorus mmol/L | 1.84 | 1.74 | 1.68 | 1.97 | 2.00 | 1.55 | 1.97 (0.4) |
| Magnesium mmol/L | 0.60 | 0.69 | 0.70 | 0.61 | 0.88 | 0.66 | - |
| Cholesterol mmol/L | 3.77 | 3.82 | 4.06 | 5.15 | 5.72 | 5.75 | 4.4 (1.9) |
| Bilirubin µmol/L | 3 | 3 | 3 | 3 | 7 | 5 | 3.2 (1.8) |
| Unconjugated Bilirubin µmol/L | 3 | 3 | 3 | 3 | 7 | 5 | 2.7 (1.0) |
| Glucose mmol/L | 6.5 | 4.5 | 4.7 | 5.4 | 4.4 | 4.5 | 4.1 (1.1) |
| AST units/L | 47 | 59 | 96 | 36 | 24 | 72 | 92.8 (69.8) |
| ALT units/L | 66 | 97 | 101 | 89 | 90 | 57 | 65.6 (53.0) |
| LD units/L | 350 | 371 | 547 | 348 | 294 | 708 | - |
| GGT units/L | 52 | 48 | 45 | 63 | 68 | 63 | 61.6 (47.3) |
| ALP units/L | 880 | 894 | 1007 | 1050 | 1314 | 1303 | 824 (515.1) |
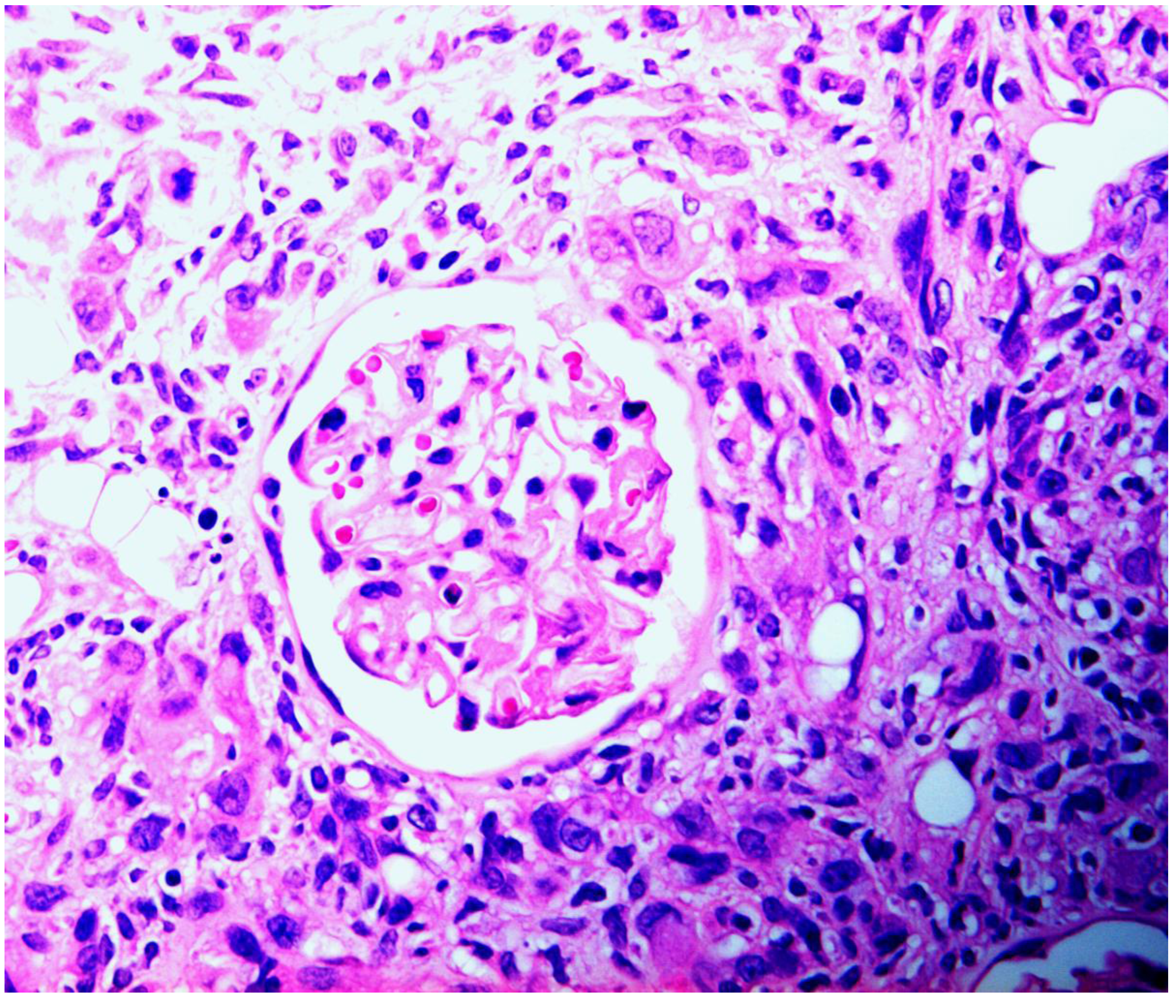
4. Discussion
Acknowledgements
References
- Mantle, P.G. Secondary metabolites of Penicillium and Acremonium. In Penicillium andAcremonium (Biotechnology Handbooks); Peberdy, J.F., Ed.; Plenum: New York, NY, USA, 1987; Volume 1. [Google Scholar]
- Frisvad, J.C.; Lund, F. Toxin and secondary metabolite production by Penicillium species growing in stored cereals. In Occurrence and Significance of Mycotoxins; Scudamore, K.A., Ed.; Central Science Laboratory: Slough, UK, 1993; pp. 146–171. [Google Scholar]
- Lund, F.; Frisvad, J.C. Chemotaxonomy of Penicillium aurantiogriseum and related species. Mycol. Res. 1994, 98, 1317–1328. [Google Scholar]
- Samson, R.A.; Frisvad, J.C. Penicillium subgenus Penicillium: new taxonomic schemes, mycotoxins and other extrolites. Stud. Mycol. 2004, 49, 1–73. [Google Scholar]
- Yeulet, S.E. A Penicillium and associated metabolites in the aetiology of Balkan nephropathy. Ph.D. Thesis, University of London, London, UK, 1986. [Google Scholar]
- Yeulet, S.E.; Mantle, P.G.; Bilton, J.N.; Rzepa, H.S.; Sheppard, R.N. Auranthine, a new benzodiazepinone metabolite of Penicillium aurantiogriseum. J. Chem. Soc.Perkin Trans. I 1986, 1891–1894. [Google Scholar]
- Boyes-Korkis, J.M.; Gurney, K.A.; Penn, J.; Mantle, P.G.; Bilton, J.N.; Sheppard, R.N. Anacine, a new benzodiazepine metabolite of Penicillium aurantiogriseum produced with other alkaloids in submerged fermentation. J. Nat. Prod. 1993, 56, 1707–1717. [Google Scholar]
- Barnes, J.M.; Carter, R.L.; Peristianis, G.C.; Austwick, P.K.C.; Flynn, F.V.; Aldridge, W.N. Balkan (endemic) nephropathy and a toxin-producing strain of Penicillium verrucosum var. cyclopium, an experimental model in rats. Lancet 1977, 289, 671–675. [Google Scholar]
- Yeulet, S.E.; Mantle, P.G.; Rudge, M.S.; Greig, J.B. Nephrotoxicity of Penicillium aurantiogriseum, a possible factor in the aetiology of Balkan endemic nephropathy. Mycopathologia 1988, 102, 21–30. [Google Scholar]
- Macgeorge, K.M.; Mantle, P.G. Nephrotoxicity of Penicillium aurantiogriseum and P. commune from an endemic nephropathy area of Yugoslavia. Mycopathologia 1990, 112, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Macgeorge, K.M.; Mantle, P.G. Nephrotoxic fungi in a Yugoslavian community in which Balkan Nephropathy is hyperendemic. Mycol. Res. 1991, 95, 660–664. [Google Scholar]
- Mantle, P.G.; McHugh, K.M.; Adatia, R.; Gray, T.; Turner, D.R. Persistent karyomegaly caused by Penicillium nephrotoxins in the rat. Proc. R. Soc. Lond. B 1991, 246, 251–259. [Google Scholar]
- Mantle, P.G. Renal histopathological responses to nephrotoxic Penicillium aurantiogriseum in the rat during pregnancy, lactation and after weaning. Nephron 1994, 66, 93–98. [Google Scholar]
- Adatia, R.; Heaton, J.M.; Macgeorge, K.M.; Mantle, P.G. Acute histopathological changes produced by Penicillium aurantiogriseum nephrotoxin in the rat. Int. J. Exp. Pathol. 1991, 72, 47–53. [Google Scholar]
- Mantle, P.G.; McHugh, K.M.; Adatia, R.; Heaton, J.M.; Gray, T.; Turner, D.R. Penicillium aurantiogriseum-induced, persistent renal histopathological changes in rats; an experimental model for Balkan endemic nephropathy in competition with ochratoxin A. In Mycotoxins, Endemic Nephropathy and Urinary Tract Tumours; Castegnaro, M., Plestina, R., Dirheimer, G., Chernozemsky, I.N., Bartsch, H., Eds.; International Agency for Research on Cancer Scientific Publications: Lyon, France, 1991; pp. 119–127. [Google Scholar]
- Hard, G.C.; Greig, J.B. Comparative acute nephrotoxicity of Penicillium aurantiogriseum in rats and hamsters. In Mycotoxins, Endemic Nephropathy and Urinary Tract Tumours; Castegnaro, M., Plestina, R., Dirheimer, G., Chernozemsky, I.N., Bartsch, H., Eds.; International Agency for Research on Cancer Scientific Publications: Lyon, France, 1991; pp. 113–117. [Google Scholar]
- Mantle, P.G.; Miljkovic, A.; Udupa, V.; Dobrota, M. Does apoptosis cause renal atrophy in Balkan endemic nephropathy? Lancet 1998, 352, 1118–1119. [Google Scholar] [CrossRef] [PubMed]
- Stoev, S.D.; Denev, S.; Dutton, M.F.; Njobeh, P.B.; Mosonik, J.S.; Steenkamp, P.A.; Petkov, I. Complex etiology and pathology of mycotoxic nephropathy in South African pigs. Mycotoxin Res. 2010, 10, 31–46. [Google Scholar]
- Stoev, S.D.; Dutton, M.F.; Njobeh, P.B.; Mosonik, J.S.; Steenkamp, P.A. Mycotoxic nephropathy in Bulgarian pigs and chickens: complex etiology and similarity to Balkan Endemic Nephropathy. Food Addit. Contam. 2010, 27, 72–88. [Google Scholar]
- Mantle, P.G.; McHugh, K.M. Nephrotoxic fungi in foods from nephropathy households in Bulgaria. Mycol. Res. 1993, 97, 205–212. [Google Scholar]
- Njobeh, P.B.; Dutton, M.F.; Chuurgoon, A.A.; Koch, S.H.; Steenkamp, P.A.; Stoev, S.D. Identification of novel metabolite and its cytotoxic effect on human lymphocyte cells in comparison to other mycotoxins. Int. J. Biol. Chem. Sci. 2009, 3, 524–531. [Google Scholar]
- Stoev, S.D.; Paskalev, M.; Macdonald, S.; Mantle, P.G. Experimental one year ochratoxin A toxicosis in pigs. Exp. Toxicol. Pathol. 2002, 53, 481–487. [Google Scholar]
- Stoev, S.D.; Hald, B.; Mantle, P.G. Porcine nephropathy in Bulgaria: a progressive syndrome of complex or uncertain (mycotoxin) etiology. Vet. Rec. 1998, 142, 190–194. [Google Scholar]
- van der Merwe, K.J.; Steyn, P.S.; Fourie, L.; Scott, D.B.; Theron, J.J. Ochratoxin A, a toxic metabolite produced by Aspergillus ochraceus Wilh. Nature 1965, 205, 1112–1113. [Google Scholar]
- Brown, A.L.; Odell, E.W.; Mantle, P.G. DNA ploidy distribution in renal tumours induced in male rats by dietary ochratoxin A. Exp. Toxicol. Pathol. 2007, 59, 85–95. [Google Scholar]
- Turner, D.R.; Mantle, P.G.; McHugh, K.M.; Adatia, R.; Gray, T. The effect of Penicillium moulds on the kidney and their relationship with Balkan nephropathy. Proceedings of the 14th European Pathology Congress, Innsbruck, Austria; September 1993. [Google Scholar]
- Mantle, P.G.; Faucet-Marquis, V.; Manderville, R.A.; Squillaci, B.; Pfohl-Leszkowicz, A. Structures of covalent adducts between DNA and ochratoxin A: a new factor in debate about genotoxicity and human risk assessment. Chem. Res. Toxicol. 2010, 23, 89–98. [Google Scholar]
- Marin-Kuan, M.; Cavin, C.; Delatour, T.; Schilter, B. Ochratoxin A carcinogenicity involves a complex network of epigenetic mechanisms. Toxicon 2008, 52, 195–202. [Google Scholar]
- Marasas, W.F.O.; Kriek, N.P.K.; Fincham, J.E.; Van Rensburg, S.J. Primary liver cancer and oesophageal basal cell hyperplasia in rats caused by Fusarium moniliforme. Int. J. Cancer 1984, 34, 383–387. [Google Scholar]
- U.S. Department of Health and Human Services. Toxicology and carcinogenesis studies of fumonisin B1 in F344/N rats and B6C3F1 mice (feed studies). NTP Technical Report 496. National Toxicology Program: Research Triangle Park, NC, USA, 2001. Available online: http://ntp.niehs.nih.gov/ntp/htdocs/LT_rpts/tr496.pdf (Accessed on 23 June 2010).
- Hard, G.C.; Howard, P.C.; Kovatch, R.M.; Bucci, T.J. Rat kidney pathology induced by chronic exposure of fumonisin B1 includes rare variants of renal tubule tumor. Toxicol. Pathol. 2001, 29, 379–386. [Google Scholar]
- Mantle, P.G.; Dobrota, M.; Gillett, C.E.; Odell, E.W.; Pinder, S.E. Oncological outcomes in rats given nephrocarcinogenic exposure to dietary ochratoxin A, followed by the tumour promoter sodium barbital for life: a pilot study. Toxins 2010, 2, 552–571. [Google Scholar]
- Gelderblom, W.C.A.; Seier, J.V.; Snijman, P.W.; Van Schalkwyk, D.J.; Shepard, G.S.; Marasas, W.F.O. Toxicity of cultured material of Fusarium verticillioides strain MRC 826 to non-human primates. Environ. Health Perspect. 2001, 109, 267–276. [Google Scholar]
- Stander, M.A.; Nieuwoudt, T.W.; Steyn, P.S.; Shephard, G.S.; Creppy, E.E.; Sewram, V. Toxicokinetics of ochratoxin A in vervet monkeys (Cercopithecus aethiops). Arch. Toxicol. 2001, 75, 262–269. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mantle, P.G.; McHugh, K.M.; Fincham, J.E. Contrasting Nephropathic Responses to Oral Administration of Extract of Cultured Penicillium polonicum in Rat and Primate. Toxins 2010, 2, 2083-2097. https://doi.org/10.3390/toxins2082083
Mantle PG, McHugh KM, Fincham JE. Contrasting Nephropathic Responses to Oral Administration of Extract of Cultured Penicillium polonicum in Rat and Primate. Toxins. 2010; 2(8):2083-2097. https://doi.org/10.3390/toxins2082083
Chicago/Turabian StyleMantle, Peter G., Katharine M. McHugh, and John E. Fincham. 2010. "Contrasting Nephropathic Responses to Oral Administration of Extract of Cultured Penicillium polonicum in Rat and Primate" Toxins 2, no. 8: 2083-2097. https://doi.org/10.3390/toxins2082083





