Cholera-Like Enterotoxins and Regulatory T cells
Abstract
:1. Introduction
2. Structure of Cholera-Like Enterotoxins and Toxicity
3. Regulatory T cells (Figure 1)
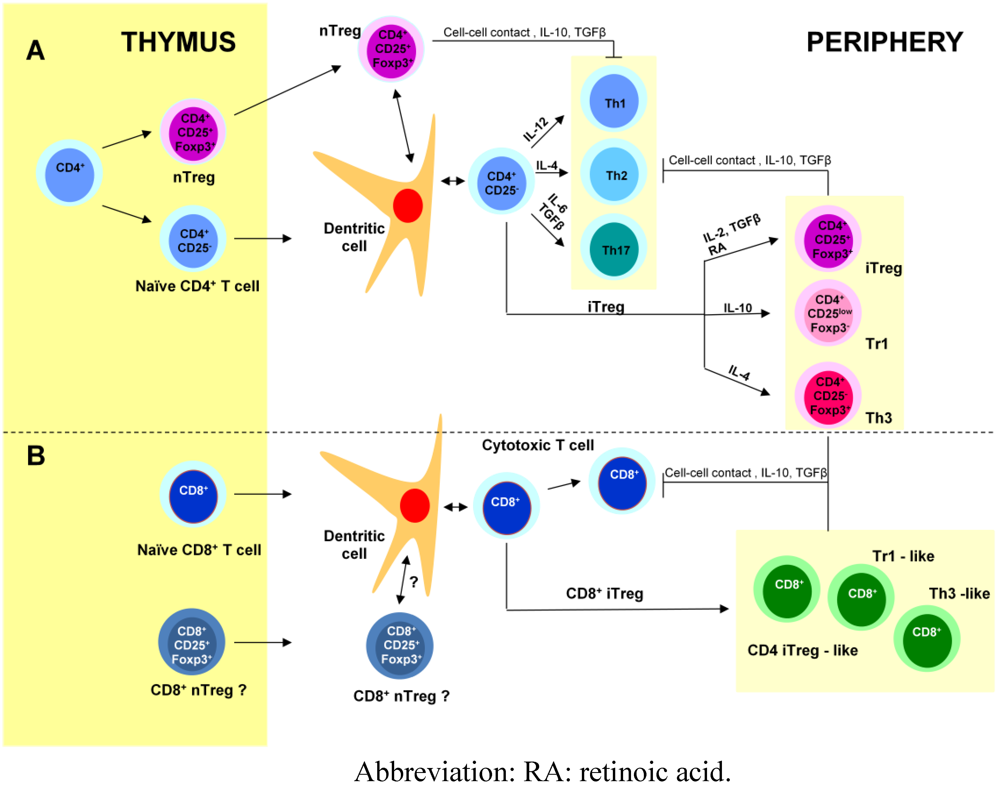
4. Adjuvanticity of Cholera-Like Enterotoxins
5. Tolerogenicity of Cholera-Like Enterotoxins
6. Cholera-Like Enterotoxins and Tregs
6.1. B Subunits
6.1.1. a-CTB
| Model | Antigen | Protocol | Effects | Proposed Mechanism for Tolerance | Reference |
|---|---|---|---|---|---|
| NOD mice Experimental diabetes | Hu insulin-CTB | Oral 1 dose 2–20 mg + transfer | Protection from clinical diabetes; Suppression of beta cell destruction | Protective T cells Non characterized | [42] |
| Transfer of splenocytes induces protection | |||||
| NOD mice Experimental diabetes | Hu insulin-CTB | Oral 1 dose 10 mg + transfer | Increase in IL-4 (Th2); decrease in IFNg (Th1) in pancreatic LN + increase in TGFb in MLN | Ag specific CD4+ Tregs in the pancreas and draining LN Non characterized | [43] |
| Transfer of CD4+ (but not CD8+) splenocytes induces protection | |||||
| NOD mice Experimental diabetes | Hu insulin- CTB | Nasal 1 dose 1 mg + transfer | Delays the incidence of diabetes ; dose-dependent effect; IL10 and TGFb increase in pancreas | CD4+ Tregs | [39] |
| Transfer of CD4+ splenocytes induces protection | Non characterized | ||||
| Dose-dependent effect | |||||
| H2d-RIP-LCMV-NP transgenic mice (LCMV induced diabetes) | Hu insulin-CTB or Porcine-insulin CTB | Oral 0.1-10 mg Biweekly (7 wks) + transfer | Transfer of splenocytes induces protection from diabetes and to bystander OVA antigen | CD4+ Tregs | [40] |
| CD4 depletion abrogates protection | Non characterized | ||||
| Dose- dependent effect (only intermetiate dosages are protective) | Dose- dependent effect | ||||
| NOD mice Experimental diabetes | CTB alone | IP 10 mg 3 times a week (4 weeks) + transfer | Decreases the development of clinical diabetes | Regulatory cells | [44] |
| Transfer of splenocytes inhibits the adoptive transfer of diabetes by spleen cells from diabetic mice into irradiated NOD mice | Non characterized | ||||
| Rat EAE | MBP-CTB | Oral 50 mg 3 doses 4, 6 and 8 days after EAE induction transfer of MLN cells | Protection from clinical EAE; Decrease in CD4, CD8, IL-2R and MHC class II in spinal cord + Decrease in IFNg, IL-12, TNFa, MCP-1 and RANTES | Protective TGFb producing regulatory T cells | [45] |
| Increase in TGFb | Non characterized | ||||
| Transfer of MLN cells induces protection | |||||
| Rat experimental uveitis | Pept-HSP60- CTB | Oral 5 doses 15mg on alternate days Transfer of MLN cells | Increase in regulatory CD4+CD45RClowRT6+ subset of Th2 memory in MLN and spleen | Regulatory subset of memory cells | [38] |
| Increase in IL-10, TGFb, decrease in IFNg and IL-12 in the MLN and the uveal tract | Shift from Th1 to Th2 and Th3 in the MLN and the uveal tract Non characterized | ||||
| Prevention of uveitis | |||||
| Transfer of MLN cells induces protection | |||||
| Apoe(-/-) mice atherosclerosis inflammatory disease model | Peptide apolipopt B-100-CTB | Nasal 15m g twice weekly12 weeks | Reduces aortic lesion size | Tr1 Tregs | [41] |
| Induction of Tregs CD4+ IL-10+ (Tr1)TGF independent |
| Model | Antigen | Protocol | Effects | Proposed Mechanism for Tolerance | Reference |
|---|---|---|---|---|---|
| BALB/c mice | OVA-CTB | Intragastric 3 doses (200 μg) at 2 days of interval | Increase in the frequency and suppressive activity of Ag-specific CD25+CD4+Foxp3+ Tregs (MLN, PP and spleen) abolished by the coadministration of CT (Table 4) | Foxp3+CD25+CD4+Tregs Foxp3+and Foxp3-CD25-CD4+Tregs | [46] |
| Transfer of splenocytes and MLN cells | Generation of both Foxp3+ and Foxp3-CD25-CD4+ | ||||
| Increase in TGFβ (serum) CD25+ and CD25- T cells suppress-effector Tcell proliferation in vitro - OVA-specific T cells and DTH after adoptive transfer | |||||
| BALB/c mice | OVA-CTB | Sublingual | Increase in Tregs in CLN MLN, spleen Increase in TGFβ (serum) Suppression of proliferative responses to OVA in vitro Suppression of OVA-specific DTH responses in vivo and T-cell proliferative responses in mice immunized SC with OVA + CFA | Foxp3+CD25+CD4+ Tregs | [47] |
| One or 3 doses (40 μg) | |||||
| BALB/c mice | OVA-CTB | Sublingual | Development of OVA-specific Foxp3+CD25+CD4+ Tregs Suppression of peripheral T cell responses to OVA | CD4+CD25+Foxp3+ Tregs inhibit Teffector cell proliferation and induce Teffector cell apoptosis and depletion | [48] |
| Adoptive transfer of OVA-specific TCR transgenic CD4+ T cells 3 doses (40 μg or 60 μg) at 2-day intervals. | Apoptosis of OVA-specific T effector cells in peripheral LN, dependent on CD25+ Treg cells | ||||
| BALB/c mice | Allergen Betv1a-CTB | Nasal 3 doses (20 μg) D0,D7,D14 | Decrease in IgE, IL-5 Increase in IgG2a, IFNγ + local IgA , Th1 shift | Tolerance induction by the conjugate not associated with an increased expression of Foxp3, CTLA4 or the suppressive cytokines IL-10 and TGFβ in lymphocyte population of spleens or lungs | [49] |
| allergic sensitization with the allergen | Upregulation of Foxp3, IL-10 and TGFβ mRNA in splenocytes after pretreatment with unconjugated allergen but not with the fusion molecule |
6.1.2.b-LTB
| Model | Antigen | Protocol | Effects | Proposed Mechanism for Immuno-Modulation | Reference |
|---|---|---|---|---|---|
| DBA/1 mice CIA | LTB alone | Nasal (100 μg) or intragastric (1 μg) transfer | Protection against CIA at the induction or 25 days later (not CTB) | CD4+CD25+ Tregs | [52] |
| Decrease in IFNγ but not in IL-4 and IL-10 | |||||
| Transfer of CD4+ T cells induces protection abrogated upon depletion of the CD25+ population | |||||
| DO11.10 chimeric mice | LTB + OVA | Oral D1, 3, 5, and 7 (1 mg OVA+ 20 μg LTB) | Depressed IFNγ and enhanced TGFβ CTLA-4 up-regulation | Activated regulatory T cell populations as part of tolerance induction | [53] |
| Balb/c mice Latently infected by HSV-1 Therapeutic vaccination | LTB + HSV GP | Nasal 3 doses at 10-day intervals 10 μg HSV-1 glycoproteins + 20 μg LTB | Modulation of the Th1-dominated proinflammatory response induced upon infection | Tr1 Tregs | [54] |
| Increase in IL-10 production by proliferating T cells from LN | |||||
| Protection from HSV reactivation: decrease in incidence and severity of keratitis + reduction of virus spread + protection from encephalitis+ reduction in the incidence of recurrent herpetic corneal | |||||
| B10.RIII EAU | LTB alone | Nasal (50 μg) for 4 days, starting either 3 days before or 3 days after EAU induction | Preimmunization treatment protects from EAU decrease in Th1 | IL-10 producing Tregs (Tr1)? modulate Th17 cells Critical importance of the dynamics of infiltration | [55] |
| Treatment after induction does not protect despite decrease in IFNγ (Th1 decrease). Increases Th17 infiltration but not symptoms |
6.2. A Subunit: CTA1-DD and Mutant (Table 4)
| Model | Protocol | Effects | Proposed Mechanism for Tolerance | Reference |
|---|---|---|---|---|
| DBA-1 mice | Nasal | Protection against CIA | Peptide specific induced Tregs Tr1? | [25] |
| 3 doses 5 μg on D5, 6, 7 | Lower serum anti-collagen antibodies | |||
| CTA1R7K-COL-DD | Decrease in IL-6, IL-17, IFNγ | |||
| after the collagen boost/induction of CIA. | Increase in IL-10 in serum and at the T cell level. | |||
| BALB/c mice | Nasal | Induction of long-lived specific tolerance to OVA, | Induction of CD4+CD25-Foxp3- Tr1 cells producing IL-10 | [24] |
| CTA1R7K-OVA-DD | Induction of IL-10 Tregs Dependent on enzymatic activity |
6.3. Whole Toxins and Mutants (Table 5)
6.4. Mechanisms
| Model | Protocol | Route | Effects | Conclusion | Reference |
|---|---|---|---|---|---|
| C57B1/6 or CB6F1 mice | CT (10 μg) or CTB (100 μg) + transfer of splenocytes from donors fed with CT+ KLH | Intragastric | IEL CD8+ depletion in the group receiving CT | Abrogation of suppressor T cell function in vivo by mucosal CT | [56] |
| T cell suppression of both secretory IgA and plasma IgG anti-KLH after KLH feeding and its abrogation by CT | Tregs non characterized | ||||
| Rat | CT (100 μg) or CTB (62 μg) | Intragastric | CT induces a transient depletion of jejunal CD8 (IEL) | Disturbance of the gut homeostasis | [57] |
| Tregs non characterized | |||||
| Balb/c mice | OVA-CTB 3 doses (200 μg) at 2 days of interval +/− CT (4 μg) | Intragastric | CT abrogates the increase in the frequency and suppressive activity of Ag-specific CD25+CD4+Foxp3+Tregs (MLN, PP and spleen) induced by CTB | CT prevents the induction of CD25+CD4+ suppressor T cells by OVA-CTB | [46] Cf Table 2 |
| CT also inhibits the normal suppressive function and Foxp3 gene expression of the mucosal CD25+ Treg cells in the OVA/CTB-treated mice | |||||
| BALB/c, C3H/HeN, and C3H/HeJ mice | D0: KLH (10 μg) or KLH (10 μg) and CT (1.0 μg) D7: KLH (20 μg) Generation of Ag specific Tcell lines and clones Cocultures of DC + LPS + CT | SC footpad | Induction of Th2 and Tr1 cells (specific for KLH); Inhibition of IFNγ production | CT promotes the induction of Tr1 cells specific for bystander Ag | [59] |
| DC + LPS + CT: CT induces maturation of DC, induces IL-10, and inhibits IL-12 production | Effect on DC | ||||
| Balb /c mice | D0: LT-R192G (10 μg) + rotavirus VLP (10 μg) D14: in vitro restimulation with Ag, LT-R192G or both | Intrarectal | LT-R192G decreases in vitro CD4+CD25+Foxp3+ from non immunized mice (nTregs?) | Effect of LT-R192G on CD4+CD25+Foxp3+ Tregs, different between a first contact and a recall | [60] |
| LT-R192G induces specific CD4+CD25+Foxp3+ and promotes the induction of CD4+CD25+Foxp3+ specific for the Ag |
6.5. Direct effect on Treg-target cell interaction
6.5.1. cAMP
6.5.2. TGFβ
6.5.3. CTLA-4
7. Conclusions
References
- Freytag, L.C.; Clements, J.D. Mucosal adjuvants. Vaccine 2005, 23, 1804–1813. [Google Scholar]
- Cox, E.; Verdonck, F.; Vanrompay, D.; Goddeeris, B. Adjuvants modulating mucosal immune responses or directing systemic responses towards the mucosa. Vet. Res. 2006, 37, 511–539. [Google Scholar]
- Sanchez, J.; Holmgren, J. Cholera toxin structure, gene regulation and pathophysiological and immunological aspects. Cell Mol. Life Sci. 2008, 65, 1347–1360. [Google Scholar]
- Williams, N.A.; Hirst, T.R.; Nashar, T.O. Immune modulation by the cholera-like enterotoxins: From adjuvant to therapeutic. Immunol. Today 1999, 20, 95–101. [Google Scholar]
- Rappuoli, R.; Pizza, M.; Douce, G.; Dougan, G. Structure and mucosal adjuvanticity of cholera and Escherichia coli heat-labile enterotoxins. Immunol. Today 1999, 20, 493–500. [Google Scholar]
- Pickett, C.L.; Twiddy, E.M.; Belisle, B.W.; Holmes, R.K. Cloning of genes that encode a new heat-labile enterotoxin of Escherichia coli. J. Bacteriol. 1986, 165, 348–352. [Google Scholar]
- Holmes, R.K.; Twiddy, E.M.; Pickett, C.L. Purification and characterization of type II heat-labile enterotoxin of Escherichia coli. Infect. Immun. 1986, 53, 464–473. [Google Scholar]
- Kotloff, K.L.; Sztein, M.B.; Wasserman, S.S.; Losonsky, G.A.; DiLorenzo, S.C.; Walker, R.I. Safety and immunogenicity of oral inactivated whole-cell Helicobacter pylori vaccine with adjuvant among volunteers with or without subclinical infection. Infect Immun. 2001, 69, 3581–3590. [Google Scholar]
- Lapa, J.A.; Sincock, S.A.; Ananthakrishnan, M.; Porter, C.K.; Cassels, F.J.; Brinkley, C.; Hall, E.R.; van Hamont, J.; Gramling, J.D.; Carpenter, C.M.; Baqar, S.; Tribble, D.R. Randomized clinical trial assessing the safety and immunogenicity of oral microencapsulated enterotoxigenic Escherichia coli surface antigen 6 with or without heat-labile enterotoxin with mutation R192G. Clin. Vaccine Immunol. 2008, 15, 1222–1228. [Google Scholar]
- Lemere, C.A. Developing novel immunogens for a safe and effective Alzheimer's disease vaccine. Prog. Brain Res. 2009, 175, 83–93. [Google Scholar]
- Lewis, D.J.; Huo, Z.; Barnett, S.; Kromann, I.; Giemza, R.; Galiza, E.; Woodrow, M.; Thierry-Carstensen, B.; Andersen, P.; Novicki, D.; Del Giudice, G.; Rappuoli, R. Transient facial nerve paralysis (Bell's palsy) following intranasal delivery of a genetically detoxified mutant of Escherichia coli heat labile toxin. PLoS One 2009, 4, e6999. [Google Scholar]
- Peppoloni, S.; Ruggiero, P.; Contorni, M.; Morandi, M.; Pizza, M.; Rappuoli, R.; Podda, A.; Del Giudice, G. Mutants of the Escherichia coli heat-labile enterotoxin as safe and strong adjuvants for intranasal delivery of vaccines. Expert Rev. Vaccines 2003, 2, 285–293. [Google Scholar]
- Sun, J.B.; Czerkinsky, C.; Holmgren, J. Mucosally induced immunological tolerance, regulatory T cells and the adjuvant effect by cholera toxin B subunit. Scand J. Immunol. 2010, 71, 1–11. [Google Scholar]
- Stanford, M.; Whittall, T.; Bergmeier, L.A.; Lindblad, M.; Lundin, S.; Shinnick, T.; Mizushima, Y.; Holmgren, J.; Lehner, T. Oral tolerization with peptide 336-351 linked to cholera toxin B subunit in preventing relapses of uveitis in Behcet's disease. Clin. Exp. Immunol. 2004, 137, 201–208. [Google Scholar]
- Mestecky, J.; Russell, M.W.; Elson, C.O. Perspectives on mucosal vaccines: Is mucosal tolerance a barrier? J. Immunol. 2007, 179, 5633–5638. [Google Scholar] [PubMed]
- Holmgren, J. Comparison of the tissue receptors for Vibrio cholerae and Escherichia coli enterotoxins by means of gangliosides and natural cholera toxoid. Infect. Immun. 1973, 8, 851–859. [Google Scholar]
- Fukuta, S.; Magnani, J.L.; Twiddy, E.M.; Holmes, R.K.; Ginsburg, V. Comparison of the carbohydrate-binding specificities of cholera toxin and Escherichia coli heat-labile enterotoxins LTh-I, LT-IIa, and LT-IIb. Infect Immun. 1988, 56, 1748–1753. [Google Scholar]
- Liang, S.; Hosur, K.B.; Lu, S.; Nawar, H.F.; Weber, B.R.; Tapping, R.I.; Connell, T.D.; Hajishengallis, G. Mapping of a microbial protein domain involved in binding and activation of the TLR2/TLR1 heterodimer. J. Immunol. 2009, 182, 2978–2985. [Google Scholar]
- Agren, L.C.; Ekman, L.; Lowenadler, B.; Nedrud, J.G.; Lycke, N.Y. Adjuvanticity of the cholera toxin A1-based gene fusion protein, CTA1-DD, is critically dependent on the ADP-ribosyltransferase and Ig-binding activity. J. Immunol. 1999, 162, 2432–2440. [Google Scholar]
- Cunningham, K.A.; Carey, A.J.; Lycke, N.; Timms, P.; Beagley, K.W. CTA1-DD is an effective adjuvant for targeting anti-chlamydial immunity to the murine genital mucosa. J. Reprod. Immunol. 2009, 81, 34–38. [Google Scholar] [Green Version]
- McNeal, M.M.; Basu, M.; Bean, J.A.; Clements, J.D.; Lycke, N.Y.; Ramne, A.; Lowenadler, B.; Choi, A.H.; Ward, R.L. Intrarectal immunization of mice with VP6 and either LT(R192G) or CTA1-DD as adjuvant protects against fecal rotavirus shedding after EDIM challenge. Vaccine 2007, 25, 6224–6231. [Google Scholar]
- Sundling, C.; Schon, K.; Morner, A.; Forsell, M.N.; Wyatt, R.T.; Thorstensson, R.; Karlsson Hedestam, G.B.; Lycke, N.Y. CTA1-DD adjuvant promotes strong immunity against human immunodeficiency virus type 1 envelope glycoproteins following mucosal immunization. J. Gen. Virol. 2008, 89, 2954–2964. [Google Scholar]
- Eliasson, D.G.; El Bakkouri, K.; Schon, K.; Ramne, A.; Festjens, E.; Lowenadler, B.; Fiers, W.; Saelens, X.; Lycke, N. CTA1-M2e-DD: A novel mucosal adjuvant targeted influenza vaccine. Vaccine 2008, 26, 1243–1252. [Google Scholar]
- Hasselberg, A.; Ekman, L.; Yrlid, L.F.; Schon, K.; Lycke, N.Y. ADP-ribosylation controls the outcome of tolerance or enhanced priming following mucosal immunization. J. Immunol. 2010, 184, 2776–2784. [Google Scholar]
- Hasselberg, A.; Schon, K.; Tarkowski, A.; Lycke, N. Role of CTA1R7K-COL-DD as a novel therapeutic mucosal tolerance-inducing vector for treatment of collagen-induced arthritis. Arthritis Rheum. 2009, 60, 1672–1682. [Google Scholar]
- Josefowicz, S.Z.; Rudensky, A. Control of regulatory T cell lineage commitment and maintenance. Immunity 2009, 30, 616–625. [Google Scholar]
- Sakaguchi, S.; Yamaguchi, T.; Nomura, T.; Ono, M. Regulatory T cells and immune tolerance. Cell 2008, 133, 775–787. [Google Scholar]
- Saurer, L.; Mueller, C. T cell-mediated immunoregulation in the gastrointestinal tract. Allergy 2009, 64, 505–519. [Google Scholar]
- Curotto de Lafaille, M.A.; Lafaille, J.J. Natural and adaptive foxp3+ regulatory T cells: More of the same or a division of labor? Immunity 2009, 30, 626–635. [Google Scholar] [CrossRef] [PubMed]
- Groux, H.; O'Garra, A.; Bigler, M.; Rouleau, M.; Antonenko, S.; de Vries, J.E.; Roncarolo, M.G. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature 1997, 389, 737–742. [Google Scholar]
- Roncarolo, M.G.; Gregori, S.; Battaglia, M.; Bacchetta, R.; Fleischhauer, K.; Levings, M.K. Interleukin-10-secreting type 1 regulatory T cells in rodents and humans. Immunol. Rev. 2006, 212, 28–50. [Google Scholar]
- Faria, A.M.; Weiner, H.L. Oral tolerance. Immunol. Rev. 2005, 206, 232–259. [Google Scholar]
- Shevach, E.M. From vanilla to 28 flavors: Multiple varieties of T regulatory cells. Immunity 2006, 25, 195–201. [Google Scholar]
- Tamayo, E.; Postigo, J.; Del Giudice, G.; Rappuoli, R.; Benito, A.; Yagita, H.; Merino, R.; Merino, J. Involvement of the intrinsic and extrinsic cell-death pathways in the induction of apoptosis of mature lymphocytes by the Escherichia coli heat-labile enterotoxin. Eur. J. Immunol. 2009, 39, 439–446. [Google Scholar]
- Lee, J.B.; Jang, J.E.; Song, M.K.; Chang, J. Intranasal delivery of cholera toxin induces th17-dominated T-cell response to bystander antigens. PLoS One 2009, 4, e5190. [Google Scholar]
- Tamayo, E.; Postigo, J.; Gonzalez, J.; Fernandez-Rey, M.; Iglesias, M.; Santiuste, I.; Riccardi, C.; Rappuoli, R.; Del Giudice, G.; Merino, R.; Merino, J. GITR contributes to the systemic adjuvanticity of the Escherichia coli heat-labile enterotoxin. Eur. J. Immunol. 2009, 40, 754–763. [Google Scholar]
- Wang, J.; Lu, Z.H.; Gabius, H.J.; Rohowsky-Kochan, C.; Ledeen, R.W.; Wu, G. Cross-linking of GM1 ganglioside by galectin-1 mediates regulatory T cell activity involving TRPC5 channel activation: Possible role in suppressing experimental autoimmune encephalomyelitis. J. Immunol. 2009, 182, 4036–4045. [Google Scholar]
- Phipps, P.A.; Stanford, M.R.; Sun, J.B.; Xiao, B.G.; Holmgren, J.; Shinnick, T.; Hasan, A.; Mizushima, Y.; Lehner, T. Prevention of mucosally induced uveitis with a HSP60-derived peptide linked to cholera toxin B subunit. Eur. J. Immunol. 2003, 33, 224–232. [Google Scholar]
- Aspord, C.; Thivolet, C. Nasal administration of CTB-insulin induces active tolerance against autoimmune diabetes in non-obese diabetic (NOD) mice. Clin. Exp. Immunol. 2002, 130, 204–211. [Google Scholar]
- Petersen, J.S.; Bregenholt, S.; Apostolopolous, V.; Homann, D.; Wolfe, T.; Hughes, A.; De Jongh, K.; Wang, M.; Dyrberg, T.; Von Herrath, M.G. Coupling of oral human or porcine insulin to the B subunit of cholera toxin (CTB) overcomes critical antigenic differences for prevention of type I diabetes. Clin. Exp. Immunol. 2003, 134, 38–45. [Google Scholar]
- Klingenberg, R.; Lebens, M.; Hermansson, A.; Fredrikson, G.N.; Strodthoff, D.; Rudling, M.; Ketelhuth, D.F.; Gerdes, N.; Holmgren, J.; Nilsson, J.; Hansson, G.K. Intranasal Immunization With an Apolipoprotein B-100 Fusion Protein Induces Antigen-Specific Regulatory T Cells and Reduces Atherosclerosis. Arterioscler Thromb. Vasc. Biol. 2010, 30, 94–52. [Google Scholar]
- Bergerot, I.; Ploix, C.; Petersen, J.; Moulin, V.; Rask, C.; Fabien, N.; Lindblad, M.; Mayer, A.; Czerkinsky, C.; Holmgren, J.; Thivolet, C. A cholera toxoid-insulin conjugate as an oral vaccine against spontaneous autoimmune diabetes. Proc. Natl. Acad. Sci. USA 1997, 94, 4610–4614. [Google Scholar]
- Ploix, C.; Bergerot, I.; Durand, A.; Czerkinsky, C.; Holmgren, J.; Thivolet, C. Oral administration of cholera toxin B-insulin conjugates protects NOD mice from autoimmune diabetes by inducing CD4+ regulatory T-cells. Diabetes 1999, 48, 2150–2156. [Google Scholar]
- Sobel, D.O.; Yankelevich, B.; Goyal, D.; Nelson, D.; Mazumder, A. The B-subunit of cholera toxin induces immunoregulatory cells and prevents diabetes in the NOD mouse. Diabetes 1998, 47, 186–191. [Google Scholar]
- Sun, J.B.; Xiao, B.G.; Lindblad, M.; Li, B.L.; Link, H.; Czerkinsky, C.; Holmgren, J. Oral administration of cholera toxin B subunit conjugated to myelin basic protein protects against experimental autoimmune encephalomyelitis by inducing transforming growth factor-beta-secreting cells and suppressing chemokine expression. Int. Immunol. 2000, 12, 1449–1457. [Google Scholar]
- Sun, J.B.; Raghavan, S.; Sjoling, A.; Lundin, S.; Holmgren, J. Oral tolerance induction with antigen conjugated to cholera toxin B subunit generates both Foxp3+CD25+ and Foxp3-CD25- CD4+ regulatory T cells. J. Immunol. 2006, 177, 7634–7644. [Google Scholar]
- Sun, J.B.; Cuburu, N.; Blomquist, M.; Li, B.L.; Czerkinsky, C.; Holmgren, J. Sublingual tolerance induction with antigen conjugated to cholera toxin B subunit induces Foxp3+CD25+CD4+ regulatory T cells and suppresses delayed-type hypersensitivity reactions. Scand. J. Immunol. 2006, 64, 251–259. [Google Scholar]
- Sun, J.B.; Czerkinsky, C.; Holmgren, J. Sublingual 'oral tolerance' induction with antigen conjugated to cholera toxin B subunit generates regulatory T cells that induce apoptosis and depletion of effector T cells. Scand. J. Immunol. 2007, 66, 278–286. [Google Scholar]
- Bublin, M.; Hoflehner, E.; Wagner, B.; Radauer, C.; Wagner, S.; Hufnagl, K.; Allwardt, D.; Kundi, M.; Scheiner, O.; Wiedermann, U.; Breiteneder, H. Use of a genetic cholera toxin B subunit/allergen fusion molecule as mucosal delivery system with immunosuppressive activity against Th2 immune responses. Vaccine 2007, 25, 8395–8404. [Google Scholar]
- Wiedermann, U.; Jahn-Schmid, B.; Lindblad, M.; Rask, C.; Holmgren, J.; Kraft, D.; Ebner, C. Suppressive versus stimulatory effects of allergen/cholera toxoid (CTB) conjugates depending on the nature of the allergen in a murine model of type I allergy. Int. Immunol. 1999, 11, 1717–1724. [Google Scholar]
- George Chandy, A.; Hultkrantz, S.; Raghavan, S.; Czerkinsky, C.; Lebens, M.; Telemo, E.; Holmgren, J. Oral tolerance induction by mucosal administration of cholera toxin B-coupled antigen involves T-cell proliferation in vivo and is not affected by depletion of CD25+ T cells. Immunology 2006, 118, 311–320. [Google Scholar]
- Luross, J.A.; Heaton, T.; Hirst, T.R.; Day, M.J.; Williams, N.A. Escherichia coli heat-labile enterotoxin B subunit prevents autoimmune arthritis through induction of regulatory CD4+ T cells. Arthritis Rheum. 2002, 46, 1671–1682. [Google Scholar]
- Plant, A.; Williams, R.; Jackson, M.E.; Williams, N.A. The B subunit of Escherichia coli heat labile enterotoxin abrogates oral tolerance, promoting predominantly Th2-type immune responses. Eur. J. Immunol. 2003, 33, 3186–3195. [Google Scholar]
- Richards, C.M.; Case, R.; Hirst, T.R.; Hill, T.J.; Williams, N.A. Protection against recurrent ocular herpes simplex virus type 1 disease after therapeutic vaccination of latently infected mice. J. Virol. 2003, 77, 6692–6699. [Google Scholar]
- Raveney, B.J.; Richards, C.; Aknin, M.L.; Copland, D.A.; Burton, B.R.; Kerr, E.; Nicholson, L.B.; Williams, N.A.; Dick, A.D. The B subunit of Escherichia coli heat-labile enterotoxin inhibits Th1 but not Th17 cell responses in established experimental autoimmune uveoretinitis. Invest. Ophthalmol. Vis. Sci. 2008, 49, 4008–4017. [Google Scholar]
- Elson, C.O.; Holland, S.P.; Dertzbaugh, M.T.; Cuff, C.F.; Anderson, A.O. Morphologic and functional alterations of mucosal T cells by cholera toxin and its B subunit. J. Immunol. 1995, 154, 1032–1040. [Google Scholar]
- Flach, C.F.; Lange, S.; Jennische, E.; Lonnroth, I.; Holmgren, J. Cholera toxin induces a transient depletion of CD8+ intraepithelial lymphocytes in the rat small intestine as detected by microarray and immunohistochemistry. Infect. Immun. 2005, 73, 5595–5602. [Google Scholar]
- Lavelle, E.C.; Jarnicki, A.; McNeela, E.; Armstrong, M.E.; Higgins, S.C.; Leavy, O.; Mills, K.H. Effects of cholera toxin on innate and adaptive immunity and its application as an immunomodulatory agent. J. Leukoc. Biol. 2004, 75, 756–763. [Google Scholar]
- Lavelle, E.C.; McNeela, E.; Armstrong, M.E.; Leavy, O.; Higgins, S.C.; Mills, K.H. Cholera toxin promotes the induction of regulatory T cells specific for bystander antigens by modulating dendritic cell activation. J. Immunol. 2003, 171, 2384–2392. [Google Scholar]
- Thiam, F.; di Martino, C.; Bon, F.; Charpilienne, A.; Cachia, C.; Poncet, D.; Clements, J.D.; Basset, C.; Kohli, E. Unexpected modulation of recall B and T cell responses after immunization with rotavirus- like particles in the presence of LT-R192G. Toxins 2010. submitted.. [Google Scholar]
- Negri, D.R.; Pinto, D.; Vendetti, S.; Patrizio, M.; Sanchez, M.; Riccomi, A.; Ruggiero, P.; Del Giudice, G.; De Magistris, M.T. Cholera toxin and Escherichia coli heat-labile enterotoxin, but not their nontoxic counterparts, improve the antigen-presenting cell function of human B lymphocyte. Infect. Immun. 2009, 77, 1924–1935. [Google Scholar]
- Anjuere, F.; Luci, C.; Lebens, M.; Rousseau, D.; Hervouet, C.; Milon, G.; Holmgren, J.; Ardavin, C.; Czerkinsky, C. In vivo adjuvant-induced mobilization and maturation of gut dendritic cells after oral administration of cholera toxin. J. Immunol. 2004, 173, 5103–5111. [Google Scholar]
- D'Ambrosio, A.; Colucci, M.; Pugliese, O.; Quintieri, F.; Boirivant, M. Cholera toxin B subunit promotes the induction of regulatory T cells by preventing human dendritic cell maturation. J. Leukoc. Biol. 2008, 84, 661–668. [Google Scholar]
- Sun, J.B.; Flach, C.F.; Czerkinsky, C.; Holmgren, J. B lymphocytes promote expansion of regulatory T cells in oral tolerance: Powerful induction by antigen coupled to cholera toxin B subunit. J. Immunol. 2008, 181, 8278–8287. [Google Scholar]
- Ogier, A.; Franco, M.A.; Charpilienne, A.; Cohen, J.; Pothier, P.; Kohli, E. Distribution and phenotype of murine rotavirus-specific B cells induced by intranasal immunization with 2/6 virus-like particles. Eur. J. Immunol. 2005, 35, 2122–2130. [Google Scholar]
- Vendetti, S.; Patrizio, M.; Riccomi, A.; De Magistris, M.T. Human CD4+ T lymphocytes with increased intracellular cAMP levels exert regulatory functions by releasing extracellular cAMP. J. Leukoc. Biol. 2006, 80, 880–888. [Google Scholar]
- Bopp, T.; Becker, C.; Klein, M.; Klein-Hessling, S.; Palmetshofer, A.; Serfling, E.; Heib, V.; Becker, M.; Kubach, J.; Schmitt, S.; Stoll, S.; Schild, H.; Staege, M.S.; Stassen, M.; Jonuleit, H.; Schmitt, E. Cyclic adenosine monophosphate is a key component of regulatory T cell-mediated suppression. J. Exp. Med. 2007, 204, 1303–1310. [Google Scholar]
- Nakamura, K.; Kitani, A.; Strober, W. Cell contact-dependent immunosuppression by CD4(+)CD25(+) regulatory T cells is mediated by cell surface-bound transforming growth factor beta. J. Exp. Med. 2001, 194, 629–644. [Google Scholar]
- Read, S.; Greenwald, R.; Izcue, A.; Robinson, N.; Mandelbrot, D.; Francisco, L.; Sharpe, A.H.; Powrie, F. Blockade of CTLA-4 on CD4+CD25+ regulatory T cells abrogates their function in vivo. J. Immunol. 2006, 177, 4376–4383. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Basset, C.; Thiam, F.; Di Martino, C.; Holton, J.; Clements, J.D.; Kohli, E. Cholera-Like Enterotoxins and Regulatory T cells. Toxins 2010, 2, 1774-1795. https://doi.org/10.3390/toxins2071774
Basset C, Thiam F, Di Martino C, Holton J, Clements JD, Kohli E. Cholera-Like Enterotoxins and Regulatory T cells. Toxins. 2010; 2(7):1774-1795. https://doi.org/10.3390/toxins2071774
Chicago/Turabian StyleBasset, Christelle, Fatou Thiam, Cyrille Di Martino, John Holton, John D. Clements, and Evelyne Kohli. 2010. "Cholera-Like Enterotoxins and Regulatory T cells" Toxins 2, no. 7: 1774-1795. https://doi.org/10.3390/toxins2071774




