Proteases as Insecticidal Agents
Abstract
:1. Toxic Proteins as Insecticidal Agents
| Target tissue | Class of protease | ||
|---|---|---|---|
| Cysteine protease | Metalloprotease | Serine protease | |
| Midgut (including peritrophic matrix) | Zea mays Mir1-CP | Baculovirus enhancins | Trypsin |
| Carica papaya and Ficus virgata latex proteases | Bacterial (B. thuringiensis) enhancins | ||
| Baculovirus V-CATH | B. thuringiensis InhA2 | ||
| Cuticle | Metarhizium anisopliae PR1A (subtilisin-like) | ||
| Beauveria bassiana CDEP1 | |||
| Hemocoel (including basement membrane and the prophenoloxidase cascade) | Sarcophaga peregrina ScathL (cathepsin L-like) | Eulophus pennicornis reprolysin | |
| Delia coarctata DcCathL | B. thuringiensis InhA | ||
| Helicoverpa armigera cathepsin B-like | |||
| Tuberaphis styraci cathepsin B | |||
2. Proteases with Toxicity towards Insects
2.1. Proteases that target the peritrophic matrix
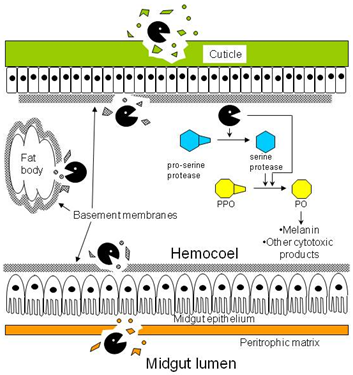
 ). Besides degrading the cuticle, basement membranes, and peritrophic matrix, proteases in the hemocoel may convert prophenoloxidase (PPO) to phenoloxidase (PO) either directly or indirectly by activating the cascade of serine proteases that lead to the conversion step.
). Besides degrading the cuticle, basement membranes, and peritrophic matrix, proteases in the hemocoel may convert prophenoloxidase (PPO) to phenoloxidase (PO) either directly or indirectly by activating the cascade of serine proteases that lead to the conversion step.
 ). Besides degrading the cuticle, basement membranes, and peritrophic matrix, proteases in the hemocoel may convert prophenoloxidase (PPO) to phenoloxidase (PO) either directly or indirectly by activating the cascade of serine proteases that lead to the conversion step.
). Besides degrading the cuticle, basement membranes, and peritrophic matrix, proteases in the hemocoel may convert prophenoloxidase (PPO) to phenoloxidase (PO) either directly or indirectly by activating the cascade of serine proteases that lead to the conversion step.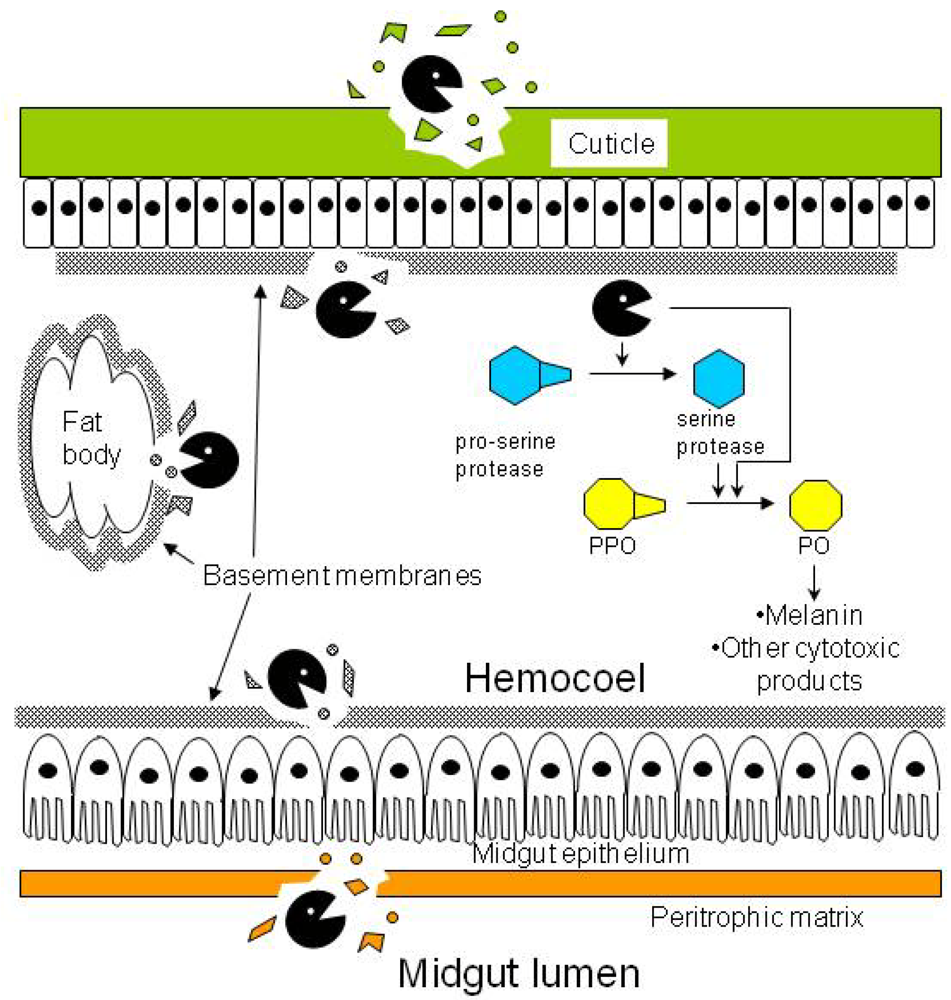
2.2. Proteases that target the cuticle
2.3. Proteases that target the basement membrane


3. Application of Proteases with Biocontrol Potential
4. Conclusions
Acknowledgements
References
- Devine, G.J.; Furlong, M.J. Insecticide use: Contexts and ecological consequences. Agric. Human Values 2007, 24, 281–306. [Google Scholar] [CrossRef]
- Schnepf, E.; Crickmore, N.; Van Rie, J.; Lereclus, D.; Baum, J.; Feitelson, J.; Zeigler, D.R.; Dean, D.H. Bacillus thuringiensis and its pesticidal crystal proteins. Microbiol. Mol. Biol. Rev. 1998, 62, 775–806. [Google Scholar]
- Bravo, A.; Gill, S.S.; Soberon, M. Mode of action of Bacillus thuringiensis Cry and Cyt toxins and their potential for insect control. Toxicon 2007, 49, 423–435. [Google Scholar] [CrossRef]
- Brookes, G.; Barfoot, P. Global impact of biotech crops: Socio-economic and environmental effects, 1996-2006. AgBioForum 2008, 11, 21–38. [Google Scholar]
- Lee, M.K.; Walters, F.S.; Hart, H.; Palekar, N.; Chen, J.S. The mode of action of the Bacillus thuringiensis vegetative insecticidal protein Vip3A differs from that of Cry1Ab delta-endotoxin. Appl. Environ. Microbiol. 2003, 69, 4648–4657. [Google Scholar]
- Hernandez-Rodriguez, C.S.; Boets, A.; Van Rie, J.; Ferre, J. Screening and identification of vip genes in Bacillus thuringiensis strains. J. Appl. Microbiol. 2009, 107, 219–225. [Google Scholar]
- Shen, Z.; Corbin, D.R.; Greenplate, J.T.; Grebenok, R.J.; Galbraith, D.W.; Purcell, J.P. Studies on the mode of action of cholesterol oxidase on insect midgut membranes. Arch. Insect Biochem. Physiol. 1997, 34, 429–442. [Google Scholar] [CrossRef]
- Markwick, N.P.; Docherty, L.C.; Phung, M.M.; Lester, M.T.; Murray, C.; Yao, J.L.; Mitra, D.S.; Cohen, D.; Beuning, L.L.; Kutty-Amma, S.; Christeller, J.T. Transgenic Res. 2003, 12, 671–681. [CrossRef] [PubMed]
- ffrench-Constant, R.H.; Dowling, A.; Waterfield, N.R. Insecticidal toxins from Photorhabdus bacteria and their potential use in agriculture. Toxicon 2007, 49, 436–451. [Google Scholar] [CrossRef]
- Shahidi-Noghabi, S.; Van Damme, E.J.; Smagghe, G. Carbohydrate-binding activity of the type-2 ribosome-inactivating protein SNA-I from elderberry (Sambucus nigra) is a determining factor for its insecticidal activity. Phytochemistry 2008, 69, 2972–2978. [Google Scholar]
- Haq, S.K.; Atif, S.M.; Khan, R.H. Protein proteinase inhibitor genes in combat against insects, pests, and pathogens: Natural and engineered phytoprotection. Arch. Biochem. Biophys. 2004, 431, 145–159. [Google Scholar] [CrossRef] [PubMed]
- Arakane, Y.; Muthukrishnan, S. Insect chitinase and chitinase-like proteins. Cell. Mol. Life Sci. 2010, 67, 201–216. [Google Scholar]
- Gordon, D.; Karbat, I.; Ilan, N.; Cohen, L.; Kahn, R.; Gilles, N.; Dong, K.; Stuhmer, W.; Tytgat, J.; Gurevitz, M. The differential preference of scorpion alpha-toxins for insect or mammalian sodium channels: Implications for improved insect control. Toxicon 2007, 49, 452–472. [Google Scholar]
- Gurevitz, M.; Karbat, I.; Cohen, L.; Ilan, N.; Kahn, R.; Turkov, M.; Stankiewicz, M.; Stuhmer, W.; Dong, K.; Gordon, D. The insecticidal potential of scorpion beta-toxins. Toxicon 2007, 49, 473–489. [Google Scholar]
- Nicholson, G.M. Insect-selective spider toxins targeting voltage-gated sodium channels. Toxicon 2007, 49, 490–512. [Google Scholar] [CrossRef]
- Inceoglu, A.B.; Kamita, S.G.; Hammock, B.D. Genetically modified baculoviruses: A historical overview and future outlook. Adv. Virus Res. 2006, 68, 323–360. [Google Scholar] [CrossRef]
- St Leger, R.J.; Wang, C. Genetic engineering of fungal biocontrol agents to achieve greater efficacy against insect pests. Appl. Microbiol. Biotechnol. 2010, 85, 901–907. [Google Scholar] [CrossRef]
- Liu, S.; Li, H.; Sivakumar, S.; Bonning, B.C. Virus-derived genes for insect resistant transgenic plants. In Insect Viruses: Biotechnological Applications; Bonning, B.C., Ed.; Academic Press: San Diego, CA, USA, 2006. [Google Scholar]
- Schmidt, N.R.; Bonning, B.C. Intrahemocoelic toxins for lepidopteran pest management. In Molecular Biology and Genetics of Lepidoptera; Goldsmith, M., Marc, F., Eds.; Taylor and Francis: Baton Rouge, FL, USA, 2010; pp. 307–319. [Google Scholar]
- Elazar, M.; Levi, R.; Zlotkin, E. Targeting of an expressed neurotoxin by its recombinant baculovirus. J. Exp. Biol. 2001, 204, 2637–2645. [Google Scholar]
- Haq, S.K.; Atif, S.M.; Khan, R.H. Protein proteinase inhibitor genes in combat against insects, pests, and pathogens: Natural and engineered phytoprotection. Arch. Biochem. Biophys. 2004, 431, 145–159. [Google Scholar] [PubMed]
- Lemeire, E.; Borovsky, D.; Van Camp, J.; Smagghe, G. Effect of ace inhibitors and TMOF on growth, development, and trypsin activity of larval Spodoptera littoralis. Arch. Insect Biochem. Physiol. 2008, 69, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Borovsky, D.; Rabindran, S.; Dawson, W.O.; Powell, C.A.; Iannotti, D.A.; Morris, T.J.; Shabanowitz, J.; Hunt, D.F.; DeBondt, H.L.; DeLoof, A. Expression of Aedes trypsin-modulating oostatic factor on the virion of TMV: A potential larvicide. Proc. Natl. Acad. Sci. USA 2006, 103, 18963–18968. [Google Scholar]
- Hegedus, D.; Erlandson, M.; Gillott, C.; Toprak, U. New insights into peritrophic matrix synthesis, architecture, and function. Annu. Rev. Entomol. 2009, 54, 285–302. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Granados, R.R. Calcofluor disrupts the midgut defense system in insects. Insect Biochem. Mol. Biol. 2000, 30, 135–143. [Google Scholar] [CrossRef]
- Sobotnik, J.; Kudlikova-Krizkova, I.; Vancova, M.; Munzbergova, Z.; Hubert, J. Chitin in the peritrophic membrane of Acarus siro (Acari: Acaridae) as a target for novel acaricides. J. Econ. Entomol. 2008, 101, 1028–1033. [Google Scholar]
- Wang, P.; Granados, R.R. Molecular structure of the peritrophic membrane (PM): identification of potential PM target sites for insect control. Arch. Insect Biochem. Physiol. 2001, 47, 110–118. [Google Scholar]
- Lepore, L.S.; Roelvink, P.R.; Granados, R.R. Enhancin, the granulosis virus protein that facilitates nucleopolyhedrovirus (NPV) infections, is a metalloproteas. J. Invertebr. Pathol. 1996, 68, 131–140. [Google Scholar]
- Tanada, Y. A synopsis of studies on the synergistic property of an insect baculovirus: A tribute to Edward A. Steinhaus. J. Invertebr. Pathol. 1985, 45, 125–138. [Google Scholar]
- Derksen, A.C.; Granados, R.R. Alteration of a lepidopteran peritrophic membrane by baculoviruses and enhancement of viral infectivity. Virology 1988, 167, 242–250. [Google Scholar]
- Wang, P.; Hammer, D.A.; Granados, R.R. Interaction of Trichoplusia ni granulosis virus-encoded enhancin with the midgut epithelium and peritrophic membrane of four lepidopteran insects. J. Gen. Virol. 1994, 75, 1961–1967. [Google Scholar]
- Peng, J.; Zhong, J.; Granados, R.R. A baculovirus enhancin alters the permeability of a mucosal midgut peritrophic matrix from lepidopteran larvae. J. Insect Physiol. 1999, 45, 159–166. [Google Scholar]
- Wang, P.; Granados, R.R. An intestinal mucin is the target substrate for a baculovirus enhancin. Proc. Natl. Acad. Sci. USA 1997, 94, 6977–6982. [Google Scholar]
- Cao, J.; Ibrahim, H.; Garcia, J.J.; Mason, H.; Granados, R.R.; Earle, E.D. Transgenic tobacco plants carrying a baculovirus enhancin gene slow the development and increase the mortality of Trichoplusia ni larvae. Plant Cell Rep. 2002, 21, 244–250. [Google Scholar]
- Mori, M.; Kitamura, H.; Kondo, A.; Dohi, K.; Mori, M.; Kaido, M.; Mise, K.; Shimojyo, E.; Hashimoto, Y. Expression of an enhancin gene from the Trichoplusia ni granulosis virus confers resistance to lepidopterous insect pests to rice. Plant Biotechnol. 2006, 23, 55–61. [Google Scholar] [CrossRef]
- Granados, R.R.; Fu, Y.; Corsaro, B.; Hughes, P.R. Enhancement of Bacillus thuringiensis toxicity to lepidopterous species with the enhancin from Trichoplusia ni granulovirus. Biol. Control 2001, 20, 153–159. [Google Scholar] [CrossRef]
- Galloway, C.S.; Wang, P.; Winstanley, D.; Jones, I.M. Comparison of the bacterial Enhancin-like proteins from Yersinia and Bacillus spp. with a baculovirus Enhancin. J. Invertebr. Pathol. 2005, 90, 134–137. [Google Scholar] [CrossRef] [PubMed]
- Hajaij-Ellouze, M.; Fedhila, S.; Lereclus, D.; Nielsen-LeRoux, C. The enhancin-like metalloprotease from the Bacillus cereus group is regulated by the pleiotropic transcriptional activator PlcR but is not essential for larvicidal activity. FEMS Microbiol. Lett. 2006, 260, 9–16. [Google Scholar]
- Fang, S.; Wang, L.; Guo, W.; Zhang, X.; Peng, D.; Luo, C.; Yu, Z.; Sun, M. Bacillus thuringiensis bel protein enhances the toxicity of Cry1Ac protein to Helicoverpa armigera larvae by degrading insect intestinal mucin. Appl. Environ. Microbiol. 2009, 75, 5237–5243. [Google Scholar]
- Fedhila, S.; Nel, P.; Lereclus, D. The InhA2 metalloprotease of Bacillus thuringiensis strain 407 is required for pathogenicity in insects infected via the oral route. J. Bacteriol. 2002, 184, 3296–3304. [Google Scholar]
- Fedhila, S.; Gohar, M.; Slamti, L.; Nel, P.; Lereclus, D. The Bacillus thuringiensis PlcR-regulated gene inhA2 is necessary, but not sufficient, for virulence. J. Bacteriol. 2003, 185, 2820–2825. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Siregar, U.; Willeford, K.O.; Luthe, D.S.; Williams, W.P. Association of a 33-kilodalton cysteine proteinase found in corn callus with the inhibition of fall armyworm larval growth. Plant Physiol. 1995, 108, 1631–1640. [Google Scholar]
- Lopez, L.; Camas, A.; Shivaji, R.; Ankala, A.; Williams, P.; Luthe, D. Mir1-CP, a novel defense cysteine protease accumulates in maize vascular tissues in response to herbivory. Planta 2007, 226, 517–527. [Google Scholar] [CrossRef]
- Pechan, T.; Jiang, B.; Steckler, D.; Ye, L.; Lin, L.; Luthe, D.S.; Williams, W.P. Characterization of three distinct cDNA clones encoding cysteine proteinases from maize (Zea mays L.) callus. Plant Mol. Biol. 1999, 40, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Pechan, T.; Ye, L.; Chang, Y.; Mitra, A.; Lin, L.; Davis, F.M.; Williams, W.P.; Luthe, D.S. A unique 33-kD cysteine proteinase accumulates in response to larval feeding in maize genotypes resistant to fall armyworm and other Lepidoptera. Plant Cell 2000, 12, 1031–1040. [Google Scholar]
- Pechan, T.; Cohen, A.; Williams, W.P.; Luthe, D.S. Insect feeding mobilizes a unique plant defense protease that disrupts the peritrophic matrix of caterpillars. Proc. Natl. Acad. Sci. USA 2002, 99, 13319–13323. [Google Scholar]
- Mohan, S.; Ma, P.W.; Pechan, T.; Bassford, E.R.; Williams, W.P.; Luthe, D.S. Degradation of the S. frugiperda peritrophic matrix by an inducible maize cysteine protease. J. Insect Physiol. 2006, 52, 21–28. [Google Scholar] [PubMed]
- Mohan, S.; Ma, P.W.; Williams, W.P.; Luthe, D.S. A naturally occurring plant cysteine protease possesses remarkable toxicity against insect pests and synergizes Bacillus thuringiensis toxin. PLoS One 2008, 3, e1786. [Google Scholar]
- Konno, K.; Hirayama, C.; Nakamura, M.; Tateishi, K.; Tamura, Y.; Hattori, M.; Kohno, K. Papain protects papaya trees from herbivorous insects: Role of cysteine proteases in latex. Plant J. 2004, 37, 370–378. [Google Scholar]
- Zhu-Salzman, K.; Luthe, D.S.; Felton, G.W. Arthropod-inducible proteins: Broad spectrum defenses against multiple herbivores. Plant Physiol. 2008, 146, 852–858. [Google Scholar]
- Slack, J.M.; Kuzio, J.; Faulkner, P. Characterization of v-cath, a cathepsin L-like proteinase expressed by the baculovirus Autographa californica multiple nuclear polyhedrosis virus. J. Genl. Virol. 1995, 76, 1091–1098. [Google Scholar] [CrossRef]
- Hawtin, R.E.; Zarkowska, T.; Arnold, K.; Thomas, C.J.; Gooday, G.W.; King, L.A.; Kuzio, J.A.; Possee, R.D. Liquefaction of Autographa californica nucleopolyhedrovirus-infected insects is dependent on the integrity of virus-encoded chitinase and cathepsin genes. Virology 1997, 238, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Li, G.; Granados, R.R. Identification of two new peritrophic membrane proteins from larval Trichoplusia ni: Structural characteristics and their functions in the protease rich insect gut. Insect Biochem. Mol. Biol. 2004, 34, 215–227. [Google Scholar] [CrossRef]
- Li, C.; Song, X.; Li, G.; Wang, P. Midgut cysteine protease-inhibiting activity in Trichoplusia ni protects the peritrophic membrane from degradation by plant cysteine proteases. Insect Biochem. Mol. Biol. 2009, 39, 726–734. [Google Scholar] [CrossRef]
- Rahbe, Y.; Febvay, G. Protein toxicity to aphids: an in vitro test on Acyrthosiphon pisum. Entomol. Exp. Appl. 1993, 67, 149–160. [Google Scholar] [CrossRef]
- Chapman, R.F. Integument. In The Insects: Structure and Function; Cambridge University Press: Cambridge, UK, 1998; pp. 415–440. [Google Scholar]
- St Leger, R.J.; Wang, C. Genetic engineering of fungal biocontrol agents to achieve greater efficacy against insect pests. Appl. Microbiol. Biotechnol. 2010, 85, 901–907. [Google Scholar] [CrossRef]
- Freimoser, F.M.; Screen, S.; Bagga, S.; Hu, G.; St Leger, R.J. Expressed sequence tag (EST) analysis of two subspecies of Metarhizium anisopliae reveals a plethora of secreted proteins with potential activity in insect hosts. Microbiology 2003, 149, 239–247. [Google Scholar]
- Freimoser, F.M.; Hu, G.; St Leger, R.J. Variation in gene expression patterns as the insect pathogen Metarhizium anisopliae adapts to different host cuticles or nutrient deprivation in vitro. Microbiology 2005, 151, 361–371. [Google Scholar]
- Cho, E.M.; Boucias, D.; Keyhani, N.O. EST analysis of cDNA libraries from the entomopathogenic fungus Beauveria (Cordyceps) bassiana. II. Fungal cells sporulating on chitin and producing oosporein. Microbiology 2006, 152, 2855–2864. [Google Scholar] [CrossRef] [PubMed]
- Bagga, S.; Hu, G.; Screen, S.E.; St Leger, R.J. Reconstructing the diversification of subtilisins in the pathogenic fungus Metarhizium anisopliae. Gene 2004, 324, 159–169. [Google Scholar] [CrossRef]
- St Leger, R.J.; Charnley, A.K.; Cooper, R.M. Characterization of cuticle-degrading proteases produced by the entomopathogen Metarhizium anisopliae. Arch. Biochem. Biophys. 1987, 253, 221–232. [Google Scholar]
- St Leger, R.J.; Durrands, P.K.; Charnley, A.K.; Cooper, R.M. Role of extracellular chymoelastase in the virulence of Metarhizium anisopliae for Manduca sexta. J. Invertebr. Pathol. 1988, 52, 285–293. [Google Scholar] [CrossRef]
- St Leger, R.J.; Frank, D.C.; Roberts, D.W.; Staples, R.C. Molecular cloning and regulatory analysis of the cuticle-degrading-protease structural gene from the entomopathogenic fungus Metarhizium anisopliae. Eur. J. Biochem. 1992, 204, 991–1001. [Google Scholar] [CrossRef]
- St Leger, R.J.; Durrands, P.K.; Cooper, R.M.; Charnley, A.K. Regulation of production of proteolytic enzymes by the entomopathogenic fungus Metarhizium anisopliae. Arch. Microbiol. 1988, 150, 413–416. [Google Scholar] [CrossRef]
- Paterson, I.C.; Charnley, A.K.; Cooper, R.M.; Clarkson, J.M. Partial characterization of specific inducers of a cuticle-degrading protease from the insect pathogenic fungus Metarhizium anisopliae. Microbiology 1994, 140, 3153–3159. [Google Scholar]
- St Leger, R.; Joshi, L.; Bidochka, M.J.; Roberts, D.W. Construction of an improved mycoinsecticide overexpressing a toxic protease. Proc. Natl. Acad. Sci. USA 1996, 93, 6349–6354. [Google Scholar]
- Wang, C.; Hu, G.; St Leger, R.J. Differential gene expression by Metarhizium anisopliae growing in root exudate and host (Manduca sexta) cuticle or hemolymph reveals mechanisms of physiological adaptation. Fungal Genet. Biol. 2005, 42, 704–718. [Google Scholar] [CrossRef]
- Marmaras, V.J.; Charalambidis, N.D.; Zervas, C.G. Immune response in insects: The role of phenoloxidase in defense reactions in relation to melanization and sclerotization. Arch. Insect Biochem. Physiol. 1996, 31, 119–133. [Google Scholar] [CrossRef]
- Cerenius, L.; Lee, B.L.; Soderhall, K. The proPO-system: Pros and cons for its role in invertebrate immunity. Trends Immunol. 2008, 29, 263–271. [Google Scholar]
- Carton, Y.; Nappi, A.J. Drosophila cellular immunity against parasitoids. Parasitol. Today 1997, 13, 218–227. [Google Scholar] [CrossRef]
- Ashida, M.; Brey, P.T. Recent advances in research on the insect phenoloxidase cascade. In Molecular Mechanisms of Immune Responses in Insects; Brey, P.T., Hultmark, D., Eds.; Chapman & Hall: London, UK, 1997; pp. 135–172. [Google Scholar]
- Lu, D.; Pava-Ripoll, M.; Li, Z.; Wang, C. Insecticidal evaluation of Beauveria bassiana engineered to express a scorpion neurotoxin and a cuticle degrading protease. Appl. Microbiol. Biotechnol. 2008, 81, 515–522. [Google Scholar] [CrossRef]
- Fang, W.; Feng, J.; Fan, Y.; Zhang, Y.; Bidochka, M.J.; Leger, R.J.; Pei, Y. Expressing a fusion protein with protease and chitinase activities increases the virulence of the insect pathogen Beauveria bassiana. J. Invertebr. Pathol. 2009, 102, 155–159. [Google Scholar] [CrossRef]
- Timpl, R.; Brown, J.C. Supramolecular assembly of basement membranes. Bioessays 1996, 18, 123–132. [Google Scholar] [CrossRef]
- Grimstad, P.R.; Walker, E.D. Aedes triseriatus (Diptera: Culicidae) and La Crosse virus. IV. Nutritional deprivation of larvae affects the adult barriers to infection and transmission. J. Med. Entomol. 1991, 28, 378–386. [Google Scholar] [PubMed]
- Huard, J.; Feero, W.G.; Watkins, S.C.; Hoffman, E.P.; Rosenblatt, D.J.; Glorioso, J.C. The basal lamina is a physical barrier to herpes simplex virus-mediated gene delivery to mature muscle fibers. J. Virol. 1996, 70, 8117–8123. [Google Scholar]
- Peiffer, M.L.; Gildow, F.E.; Gray, S.M. Two distinct mechanisms regulate luteovirus transmission efficiency and specificity at the aphid salivary gland. J. Gen. Virol. 1997, 78, 495–503. [Google Scholar]
- Weeks, B.S.; Ramchandran, R.S.; Hopkins, J.J.; Friedman, H.M. Herpes simplex virus type-1 and -2 pathogenesis is restricted by the epidermal basement membrane. Arch. Virol. 2000, 145, 385–396. [Google Scholar]
- Romoser, W.S.; Turell, M.J.; Lerdthusnee, K.; Neira, M.; Dohm, D.; Ludwig, G.; Wasieloski, L. Pathogenesis of Rift Valley fever virus in mosquitoes-tracheal conduits & the basal lamina as an extra-cellular barrier. Arch. Virol. Suppl. 2005, 89–100. [Google Scholar]
- Reddy, J.T.; Locke, M. The size limited penetration of gold particles through insect basal laminae. J. Insect Physiol. 1990, 36, 397–407. [Google Scholar] [CrossRef]
- Jehle, J.A.; Blissard, G.W.; Bonning, B.C.; Cory, J.S.; Herniou, E.A.; Rohrmann, G.F.; Theilmann, D.A.; Thiem, S.M.; Vlak, J.M. On the classification and nomenclature of baculoviruses: A proposal for revision. Arch. Virol. 2006, 151, 1257–1266. [Google Scholar]
- Hess, R.T.; Falcon, L.A. Temporal events in the invasion of the codling moth, Cydia pomonella, by a granulosis virus. J. Invertebr. Pathol. 1987, 50, 85–105. [Google Scholar] [CrossRef]
- Engelhard, E.K.; Kam-Morgan, L.N.; Washburn, J.O.; Volkman, L.E. The insect tracheal system: A conduit for the systemic spread of Autographa californica M nuclear polyhedrosis virus. Proc. Nat. Acad. Sci. USA 1994, 91, 3224–3227. [Google Scholar]
- Smith-Johannsen, H.; Witkiewicz, H.; Iatrou, K. Infection of silkmoth follicular cells with Bombyx mori nuclear polyhedrosis virus. J. Invertebr. Pathol. 1986, 48, 74–84. [Google Scholar] [CrossRef]
- van Beek, N.A.M.; Hughes, P.R. The response time of insect larvae infected with recombinant baculoviruses. J. Invertebr. Pathol. 1998, 72, 338–347. [Google Scholar] [CrossRef]
- Keddie, B.A.; Aponte, G.W.; Volkman, L.E. The pathway of infection of Autographa californica nuclear polyhedrosis virus in an insect host. Science 1989, 243, 1728–1730. [Google Scholar]
- Harrison, R.L.; Bonning, B.C. Use of proteases to improve the insecticidal activity of baculoviruses. Biol. Control 2001, 20, 199–209. [Google Scholar] [CrossRef]
- Fujii-Taira, I.; Tanaka, Y.; Homma, K.J.; Natori, S. Hydrolysis and synthesis of substrate proteins for cathepsin L in the brain basement membranes of Sarcophaga during metamorphosis. J. Biochem. 2000, 128, 539–542. [Google Scholar] [PubMed]
- Homma, K.; Kurata, S.; Natori, S. Purification, characterization, and cDNA cloning of procathepsin L from the culture medium of NIH-Sape-4, an embryonic cell line of Sarcophaga peregrina (flesh fly), and its involvement in the differentiation of imaginal discs. J. Biol. Chem. 1994, 269, 15258–15264. [Google Scholar] [PubMed]
- Homma, K.; Natori, S. Identification of substrate proteins for cathepsin L that are selectively hydrolyzed during the differentiation of imaginal discs of Sarcophaga peregrina. Eur. J. Biochem. 1996, 240, 443–447. [Google Scholar] [CrossRef]
- Li, H.; Tang, H.; Harrison, R.L.; Bonning, B.C. Impact of a basement membrane-degrading protease on dissemination and secondary infection of Autographa californica multiple nucleopolyhedrovirus in Heliothis virescens (Fabricus). J. Gen. Virol. 2007, 88, 1109–1119. [Google Scholar]
- Tang, H.; Li, H.; Lei, S.M.; Harrison, R.L.; Bonning, B.C. Tissue specificity of a baculovirus-expressed, basement membrane-degrading protease in larvae of Heliothis virescens. Tissue Cell 2007, 39, 431–443. [Google Scholar]
- Philip, J.M.; Fitches, E.; Harrison, R.L.; Bonning, B.; Gatehouse, J.A. Characterisation of functional and insecticidal properties of a recombinant cathepsin L-like proteinase from flesh fly (Sarcophaga peregrina), which plays a role in differentiation of imaginal discs. Insect Biochem. Mol. Biol. 2007, 37, 589–600. [Google Scholar] [CrossRef]
- Li, H.; Tang, H.; Sivakumar, S.; Philip, J.; Harrison, R.L.; Gatehouse, J.A.; Bonning, B.C. Insecticidal activity of a basement membrane-degrading protease against Heliothis virescens (Fabricius) and Acyrthosiphon pisum (Harris). J. Insect Physiol. 2008, 54, 777–789. [Google Scholar] [CrossRef] [PubMed]
- Pyati, P.S.; Bell, H.A.; Fitches, E.; Price, D.R.; Gatehouse, A.M.; Gatehouse, J.A. Cathepsin L-like cysteine proteinase (DcCathL) from Delia coarctata (wheat bulb fly): Basis of insecticidal activity. Insect Biochem. Mol. Biol. 2009, 39, 535–546. [Google Scholar] [CrossRef]
- Hong-Lian, S.; Du-Juan, D.; Jin-Dong, H.; Jin-Xin, W.; Xiao-Fan, Z. Construction of the recombinant baculovirus AcMNPV with cathepsin B-like proteinase and its insecticidal activity against Helicoverpa armigera. Pestic. Biochem. Physiol. 2008, 91, 141–146. [Google Scholar]
- Yang, X.M.; Hou, L.J.; Dong, D.J.; Shao, H.L.; Wang, J.X.; Zhao, X.F. Cathepsin B-like proteinase is involved in the decomposition of the adult fat body of Helicoverpa armigera. Arch. Insect. Biochem. Physiol. 2006, 62, 1–10. [Google Scholar] [CrossRef]
- Yang, X.M.; Hou, L.J.; Wang, J.X.; Zhao, X.F. Expression and function of cathepsin B-like proteinase in larval hemocytes of Helicoverpa armigera during metamorphosis. Arch. Insect Biochem. Physiol. 2007, 64, 164–174. [Google Scholar]
- Zhao, X.F.; An, X.M.; Wang, J.X.; Dong, D.J.; Du, X.J.; Sueda, S.; Kondo, H. Expression of the Helicoverpa cathepsin B-like proteinase during embryonic development. Arch. Insect Biochem. Physiol. 2005, 58, 39–46. [Google Scholar] [CrossRef]
- Veiga, S.S.; Zanetti, V.C.; Braz, A.; Mangili, O.C.; Gremski, W. Extracellular matrix molecules as targets for brown spider venom toxins. Braz. J. Med. Biol. Res. 2001, 34, 843–850. [Google Scholar]
- da Silveira, R.B.; dos Santos Filho, J.F.; Mangili, O.C.; Veiga, S.S.; Gremski, W.; Nader, H.B.; von Dietrich, C.P. Identification of proteases in the extract of venom glands from brown spiders. Toxicon 2002, 40, 815–822. [Google Scholar]
- Devaraja, S.; Nagaraju, S.; Mahadeswaraswamy, Y.H.; Girish, K.S.; Kemparaju, K.A. low molecular weight serine protease: Purification and characterization from Hippasa agelenoides (funnel web) spider venom gland extract. Toxicon 2008, 52, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Malta, M.B.; Lira, M.S.; Soares, S.L.; Rocha, G.C.; Knysak, I.; Martins, R.; Guizze, S.P.; Santoro, M.L.; Barbaro, K.C. Toxic activities of Brazilian centipede venoms. Toxicon 2008, 52, 255–263. [Google Scholar]
- Trevisan-Silva, D.; Gremski, L.H.; Chaim, O.M.; da Silveira, R.B.; Meissner, G.O.; Mangili, O.C.; Barbaro, K.C.; Gremski, W.; Veiga, S.S.; Senff-Ribeiro, A. Astacin-like metalloproteases are a gene family of toxins present in the venom of different species of the brown spider (genus Loxosceles). Biochimie 2010, 92, 21–32. [Google Scholar]
- Çalışkan, F.; Sivas, H.; Şahin, Y. A preliminary study for the detection of gelatinolytic proteases from the scorpion Androctonus crassicauda (Turkish Black Scorpion) venom. Turk. J. Biochem. 2009, 34, 148–153. [Google Scholar]
- Price, D.R.; Bell, H.A.; Hinchliffe, G.; Fitches, E.; Weaver, R.; Gatehouse, J.A. A venom metalloproteinase from the parasitic wasp Eulophus pennicornis is toxic towards its host, tomato moth (Lacanobia oleracae). Insect Mol. Biol. 2009, 18, 195–202. [Google Scholar] [CrossRef]
- Kutsukake, M.; Nikoh, N.; Shibao, H.; Rispe, C.; Simon, J.C.; Fukatsu, T. Evolution of soldier-specific venomous protease in social aphids. Mol. Biol. Evol. 2008, 25, 2627–2641. [Google Scholar] [CrossRef]
- Miyoshi, S.; Shinoda, S. Microbial metalloproteases and pathogenesis. Microbes Infect. 2000, 2, 91–98. [Google Scholar] [CrossRef]
- Dalhammar, G.; Steiner, H. Characterization of inhibitor A, a protease from Bacillus thuringiensis which degrades attacins and cecropins, two classes of antibacterial proteins in insect. Eur. J. Biochem. 1984, 139, 247–252. [Google Scholar]
- Assenga, S.P.; You, M.; Shy, C.H.; Yamagishi, J.; Sakaguchi, T.; Zhou, J.; Kibe, M.K.; Xuan, X.; Fujisaki, K. The use of a recombinant baculovirus expressing a chitinase from the hard tick Haemaphysalis longicornis and its potential application as a bioacaricide for tick control. Parasitol. Res. 2006, 98, 111–118. [Google Scholar] [CrossRef]
- Kim, J.S.; Roh, J.Y.; Choi, J.Y.; Wang, Y.; Shim, H.J.; Je, Y.H. Correlation of the aphicidal activity of Beauveria bassiana SFB-205 supernatant with enzymes. Fungal Biol. 2010, 114, 120–128. [Google Scholar] [CrossRef]
- Jeffers, L.A.; Michael Roe, R. The movement of proteins across the insect and tick digestive system. J. Insect Physiol. 2008, 54, 319–332. [Google Scholar] [CrossRef]
- Fitches, E.; Audsley, N.; Gatehouse, J.A.; Edwards, J.P. Fusion proteins containing neuropeptides as novel insect contol agents: Snowdrop lectin delivers fused allatostatin to insect haemolymph following oral ingestion. Insect Biochem. Mol. Biol. 2002, 32, 1653–1661. [Google Scholar]
- Fitches, E.; Edwards, M.G.; Mee, C.; Grishin, E.; Gatehouse, A.M.; Edwards, J.P.; Gatehouse, J.A. Fusion proteins containing insect-specific toxins as pest control agents: snowdrop lectin delivers fused insecticidal spider venom toxin to insect haemolymph following oral ingestion. J. Insect Physiol. 2004, 50, 61–71. [Google Scholar]
- Down, R.E.; Fitches, E.C.; Wiles, D.P.; Corti, P.; Bell, H.A.; Gatehouse, J.A.; Edwards, J.P. Insecticidal spider venom toxin fused to snowdrop lectin is toxic to the peach-potato aphid, Myzus persicae (Hemiptera: Aphididae) and the rice brown planthopper, Nilaparvata lugens (Hemiptera: Delphacidae). Pest Manag. Sci. 2006, 62, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Pham Trung, N.; Fitches, E.; Gatehouse, J.A. A fusion protein containing a lepidopteran-specific toxin from the South Indian red scorpion (Mesobuthus tamulus) and snowdrop lectin shows oral toxicity to target insects. BMC Biotechnol. 2006, 6, 18. [Google Scholar] [PubMed]
- Fitches, E.C.; Bell, H.A.; Powell, M.E.; Back, E.; Sargiotti, C.; Weaver, R.J.; Gatehouse, J.A. Insecticidal activity of scorpion toxin (ButaIT) and snowdrop lectin (GNA) containing fusion proteins towards pest species of different orders. Pest Manag. Sci. 2009, 66, 74–83. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Harrison, R.L.; Bonning, B.C. Proteases as Insecticidal Agents. Toxins 2010, 2, 935-953. https://doi.org/10.3390/toxins2050935
Harrison RL, Bonning BC. Proteases as Insecticidal Agents. Toxins. 2010; 2(5):935-953. https://doi.org/10.3390/toxins2050935
Chicago/Turabian StyleHarrison, Robert L., and Bryony C. Bonning. 2010. "Proteases as Insecticidal Agents" Toxins 2, no. 5: 935-953. https://doi.org/10.3390/toxins2050935