Coffee Consumption and Risk of Biliary Tract Cancers and Liver Cancer: A Dose–Response Meta-Analysis of Prospective Cohort Studies
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Data Extraction
2.3. Statistical Analysis
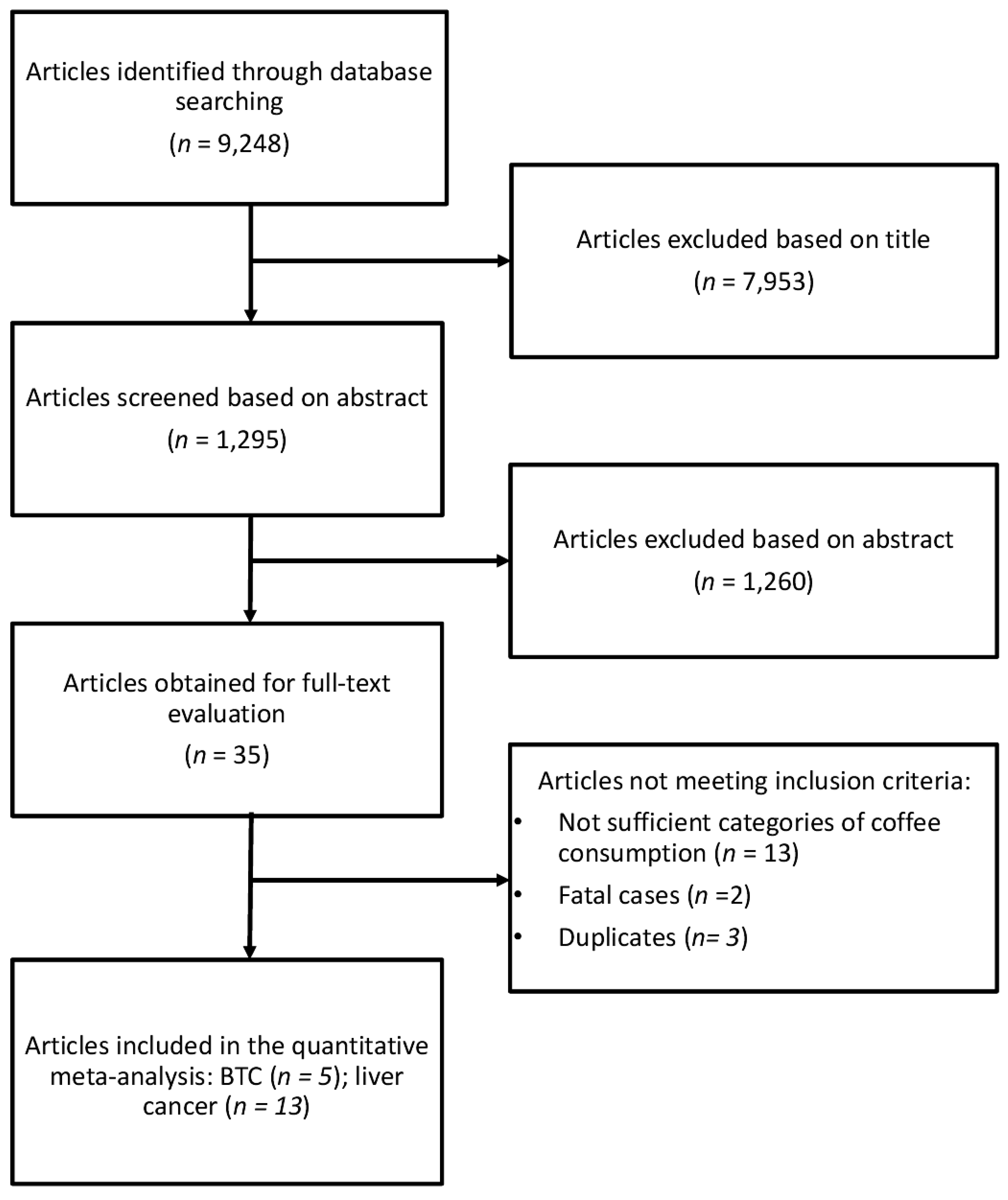
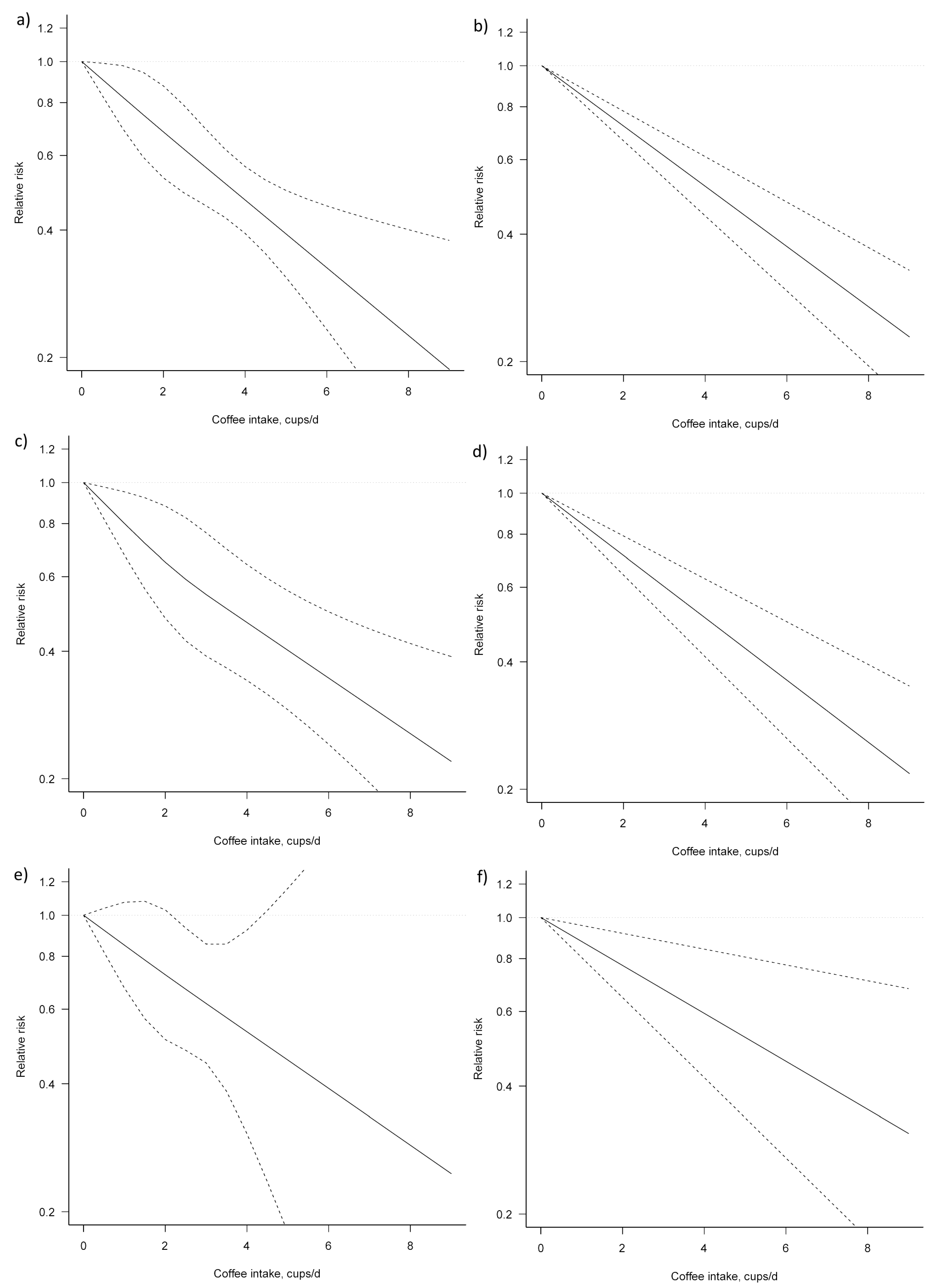
3. Results
3.1. Study Characteristics
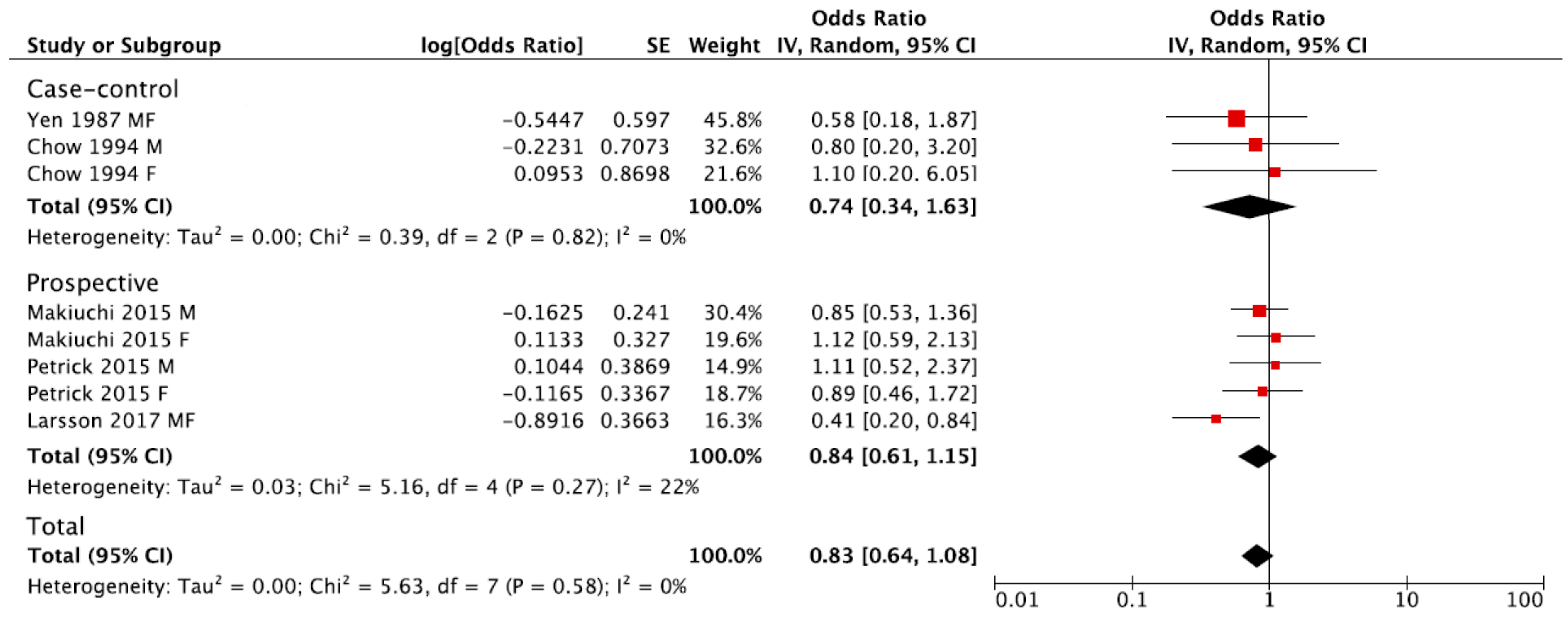
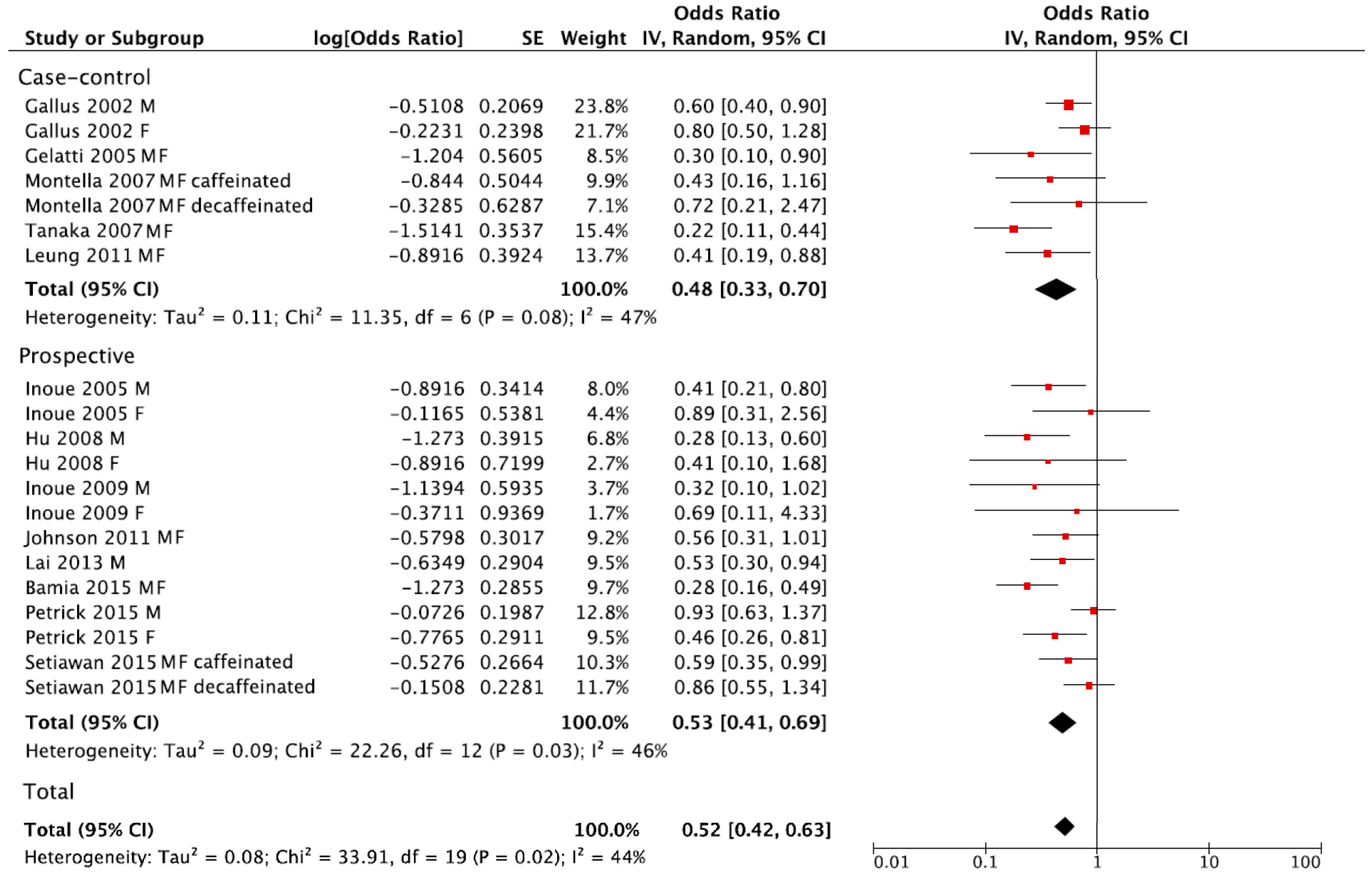
3.2. Summary Relative Risk for the Highest versus Lowest Category of Coffee Consumption
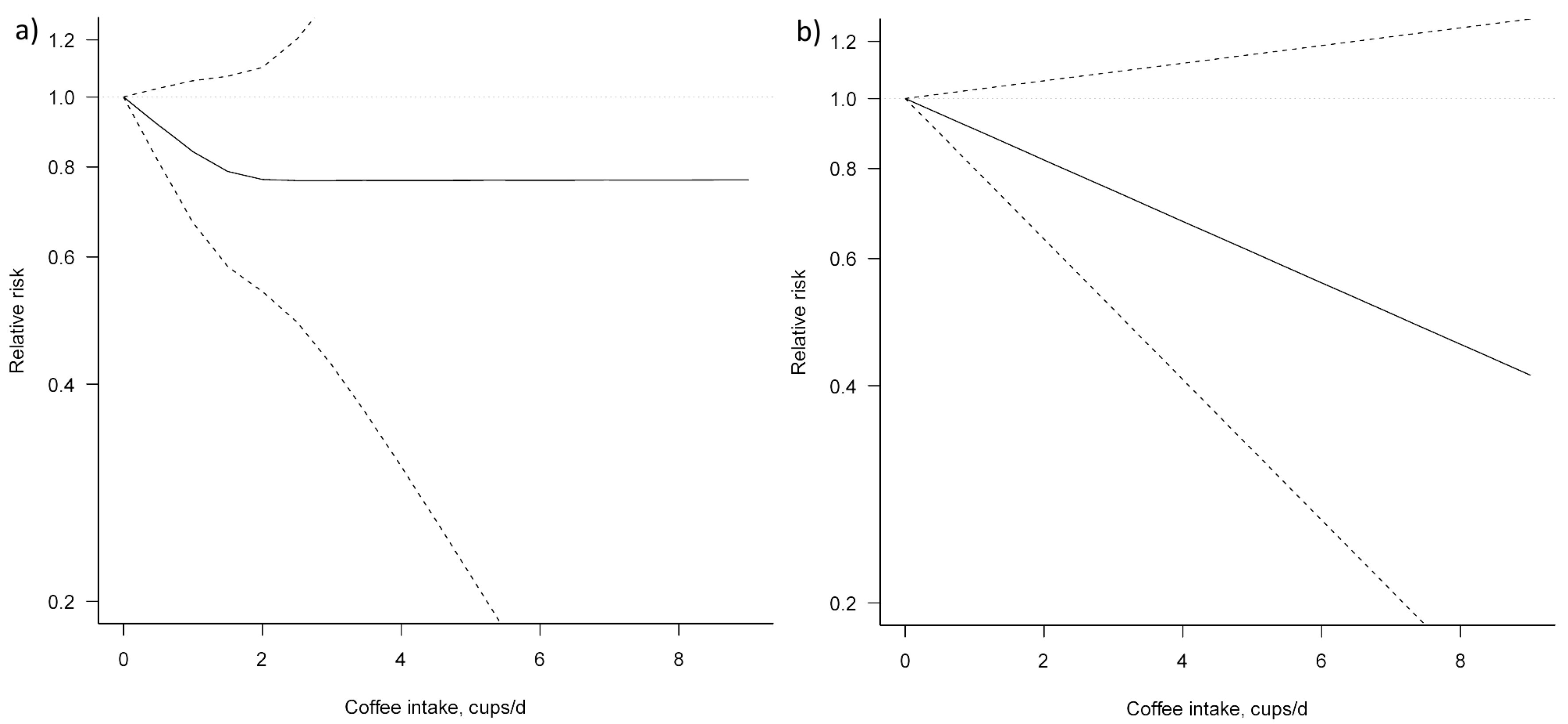
3.3. Dose–Response Meta-Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Grosso, G.; Godos, J.; Galvano, F.; Giovannucci, E.L. Coffee, caffeine, and health outcomes: An umbrella review. Annu. Rev. Nutr. 2017, 37, 131–156. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Micek, A.; Godos, J.; Sciacca, S.; Pajak, A.; Martinez-Gonzalez, M.A.; Giovannucci, E.L.; Galvano, F. Coffee consumption and risk of all-cause, cardiovascular, and cancer mortality in smokers and non-smokers: A dose-response meta-analysis. Eur. J. Epidemiol. 2016, 31, 1191–1205. [Google Scholar] [CrossRef] [PubMed]
- Buscemi, S.; Marventano, S.; Antoci, M.; Cagnetti, A.; Castorina, G.; Galvano, F.; Marranzano, M.; Mistretta, A. Coffee and metabolic impairment: An updated review of epidemiological studies. NFS J. 2016, 3, 1–7. [Google Scholar] [CrossRef]
- Caprioli, G.; Cortese, M.; Sagratini, G.; Vittori, S. The influence of different types of preparation (espresso and brew) on coffee aroma and main bioactive constituents. Int. J. Food Sci. Nutr. 2015, 66, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Koksal, E.; Yardimci, H.; Kocaadam, B.; Deniz Gunes, B.; Yilmaz, B.; Karabudak, E. Relationship between dietary caffeine intake and blood pressure in adults. Int. J. Food Sci. Nutr. 2016, 68, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Godos, J.; Pluchinotta, F.R.; Marventano, S.; Buscemi, S.; Li Volti, G.; Galvano, F.; Grosso, G. Coffee components and cardiovascular risk: Beneficial and detrimental effects. Int. J. Food Sci. Nutr. 2014, 65, 925–936. [Google Scholar] [CrossRef] [PubMed]
- Espinoza, J.A.; Bizama, C.; Garcia, P.; Ferreccio, C.; Javle, M.; Miquel, J.F.; Koshiol, J.; Roa, J.C. The inflammatory inception of gallbladder cancer. Biochim. Biophys. Acta 2016, 1865, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Hsing, A.W.; Gao, Y.T.; Han, T.Q.; Rashid, A.; Sakoda, L.C.; Wang, B.S.; Shen, M.C.; Zhang, B.H.; Niwa, S.; Chen, J.; et al. Gallstones and the risk of biliary tract cancer: A population-based study in China. Br. J. Cancer 2007, 97, 1577–1582. [Google Scholar] [CrossRef] [PubMed]
- Douglas, B.R.; Jansen, J.B.; Tham, R.T.; Lamers, C.B. Coffee stimulation of cholecystokinin release and gallbladder contraction in humans. Am. J. Clin. Nutr. 1990, 52, 553–556. [Google Scholar] [PubMed]
- Lillemoe, K.D.; Magnuson, T.H.; High, R.C.; Peoples, G.E.; Pitt, H.A. Caffeine prevents cholesterol gallstone formation. Surgery 1989, 106, 400–407. [Google Scholar] [PubMed]
- Marventano, S.; Salomone, F.; Godos, J.; Pluchinotta, F.; Del Rio, D.; Mistretta, A.; Grosso, G. Coffee and tea consumption in relation with non-alcoholic fatty liver and metabolic syndrome: A systematic review and meta-analysis of observational studies. Clin. Nutr. 2016, 35, 1269–1281. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Wang, X.; Wu, G.; Chen, L.; Hu, P.; Ren, H.; Hu, H. Coffee consumption decreases risks for hepatic fibrosis and cirrhosis: A meta-analysis. PLoS ONE 2015, 10, e0142457. [Google Scholar] [CrossRef]
- Kennedy, O.J.; Roderick, P.; Buchanan, R.; Fallowfield, J.A.; Hayes, P.C.; Parkes, J. Systematic review with meta-analysis: Coffee consumption and the risk of cirrhosis. Aliment. Pharmacol. Ther. 2016, 43, 562–574. [Google Scholar] [CrossRef]
- Bravi, F.; Tavani, A.; Bosetti, C.; Boffetta, P.; La Vecchia, C. Coffee and the risk of hepatocellular carcinoma and chronic liver disease: A systematic review and meta-analysis of prospective studies. Eur. J. Cancer Prev. 2016, 26, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Bai, K.; Cai, Q.; Jiang, Y.; Lv, L. Coffee consumption and risk of hepatocellular carcinoma: A meta-analysis of eleven epidemiological studies. Onco Targets Ther. 2016, 9, 4369–4375. [Google Scholar] [PubMed]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting, Meta-analysis of observational studies in epidemiology (moose) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses; Ottawa Health Research Institute: Ottawa, ON, Canada, 1999. [Google Scholar]
- Greenland, S. Quantitative methods in the review of epidemiologic literature. Epidemiol. Rev. 1987, 9, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Greenland, S.; Longnecker, M.P. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am. J. Epidemiol. 1992, 135, 1301–1309. [Google Scholar] [CrossRef] [PubMed]
- Orsini, N.; Bellocco, R.; Greenland, S. Generalized least squares for trend estimation of summarized dose-response data. Stata J. 2006, 6, 40–57. [Google Scholar]
- Orsini, N.; Li, R.; Wolk, A.; Khudyakov, P.; Spiegelman, D. Meta-analysis for linear and nonlinear dose-response relations: Examples, an evaluation of approximations, and software. Am. J. Epidemiol. 2012, 175, 66–73. [Google Scholar] [CrossRef]
- Chow, W.H.; McLaughlin, J.K.; Menck, H.R.; Mack, T.M. Risk factors for extrahepatic bile duct cancers: Los Angeles county, California (USA). Cancer Causes Control 1994, 5, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Giovannucci, E.L.; Wolk, A. Coffee consumption and risk of gallbladder cancer in a prospective study. J. Natl. Cancer Inst. 2017, 109, 1–3. [Google Scholar] [CrossRef]
- Makiuchi, T.; Sobue, T.; Kitamura, T.; Ishihara, J.; Sawada, N.; Iwasaki, M.; Sasazuki, S.; Yamaji, T.; Shimazu, T.; Tsugane, S. Association between green tea/coffee consumption and biliary tract cancer: A population-based cohort study in Japan. Cancer Sci. 2016, 107, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Petrick, J.L.; Freedman, N.D.; Graubard, B.I.; Sahasrabuddhe, V.V.; Lai, G.Y.; Alavanja, M.C.; Beane-Freeman, L.E.; Boggs, D.A.; Buring, J.E.; Chan, A.T.; et al. Coffee consumption and risk of hepatocellular carcinoma and intrahepatic cholangiocarcinoma by sex: The liver cancer pooling project. Cancer Epidemiol. Biomark. Prev. 2015, 24, 1398–1406. [Google Scholar] [CrossRef] [PubMed]
- Yen, S.; Hsieh, C.C.; MacMahon, B. Extrahepatic bile duct cancer and smoking, beverage consumption, past medical history, and oral-contraceptive use. Cancer 1987, 59, 2112–2116. [Google Scholar] [CrossRef]
- Gallus, S.; Bertuzzi, M.; Tavani, A.; Bosetti, C.; Negri, E.; La Vecchia, C.; Lagiou, P.; Trichopoulos, D. Does coffee protect against hepatocellular carcinoma? Br. J. Cancer 2002, 87, 956–959. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.; Tuomilehto, J.; Pukkala, E.; Hakulinen, T.; Antikainen, R.; Vartiainen, E.; Jousilahti, P. Joint effects of coffee consumption and serum gamma-glutamyltransferase on the risk of liver cancer. Hepatology 2008, 48, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Kurahashi, N.; Iwasaki, M.; Shimazu, T.; Tanaka, Y.; Mizokami, M.; Tsugane, S. Japan Public Health Center-Based Prospective Study, G. Effect of coffee and green tea consumption on the risk of liver cancer: Cohort analysis by hepatitis virus infection status. Cancer Epidemiol. Biomark. Prev. 2009, 18, 1746–1753. [Google Scholar] [CrossRef]
- Inoue, M.; Yoshimi, I.; Sobue, T.; Tsugane, S.; Group, J.S. Influence of coffee drinking on subsequent risk of hepatocellular carcinoma: A prospective study in Japan. J. Natl. Cancer Inst. 2005, 97, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Lai, G.Y.; Weinstein, S.J.; Albanes, D.; Taylor, P.R.; McGlynn, K.A.; Virtamo, J.; Sinha, R.; Freedman, N.D. The association of coffee intake with liver cancer incidence and chronic liver disease mortality in male smokers. Br. J. Cancer 2013, 109, 1344–1351. [Google Scholar] [CrossRef] [PubMed]
- Bamia, C.; Lagiou, P.; Jenab, M.; Trichopoulou, A.; Fedirko, V.; Aleksandrova, K.; Pischon, T.; Overvad, K.; Olsen, A.; Tjonneland, A.; et al. Coffee, tea and decaffeinated coffee in relation to hepatocellular carcinoma in a European population: Multicentre, prospective cohort study. Int. J. Cancer 2015, 136, 1899–1908. [Google Scholar] [CrossRef] [PubMed]
- Montella, M.; Polesel, J.; La Vecchia, C.; Dal Maso, L.; Crispo, A.; Crovatto, M.; Casarin, P.; Izzo, F.; Tommasi, L.G.; Talamini, R.; et al. Coffee and tea consumption and risk of hepatocellular carcinoma in Italy. Int. J. Cancer 2007, 120, 1555–1559. [Google Scholar] [CrossRef] [PubMed]
- Setiawan, V.W.; Wilkens, L.R.; Lu, S.C.; Hernandez, B.Y.; Le Marchand, L.; Henderson, B.E. Association of coffee intake with reduced incidence of liver cancer and death from chronic liver disease in the US multiethnic cohort. Gastroenterology 2015, 148, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Hara, M.; Sakamoto, T.; Higaki, Y.; Mizuta, T.; Eguchi, Y.; Yasutake, T.; Ozaki, I.; Yamamoto, K.; Onohara, S.; et al. Inverse association between coffee drinking and the risk of hepatocellular carcinoma: A case-control study in Japan. Cancer Sci. 2007, 98, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Gelatti, U.; Covolo, L.; Franceschini, M.; Pirali, F.; Tagger, A.; Ribero, M.L.; Trevisi, P.; Martelli, C.; Nardi, G.; Donato, F.; et al. Coffee consumption reduces the risk of hepatocellular carcinoma independently of its aetiology: A case-control study. J. Hepatol. 2005, 42, 528–534. [Google Scholar] [CrossRef] [PubMed]
- Leung, W.W.; Ho, S.C.; Chan, H.L.; Wong, V.; Yeo, W.; Mok, T.S. Moderate coffee consumption reduces the risk of hepatocellular carcinoma in hepatitis b chronic carriers: A case-control study. J. Epidemiol. Community Health 2011, 65, 556–558. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.; Koh, W.P.; Wang, R.; Govindarajan, S.; Yu, M.C.; Yuan, J.M. Coffee consumption and reduced risk of hepatocellular carcinoma: Findings from the singapore chinese health study. Cancer Causes Control 2011, 22, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Giovannucci, E.; Wolk, A. Coffee consumption and stomach cancer risk in a cohort of Swedish women. Int. J. Cancer 2006, 119, 2186–2189. [Google Scholar] [CrossRef] [PubMed]
- Kurahashi, N.; Inoue, M.; Iwasaki, M.; Sasazuki, S.; Tsugane, S. Japan Public Health Center Study, G. Coffee, green tea, and caffeine consumption and subsequent risk of bladder cancer in relation to smoking status: A prospective study in Japan. Cancer Sci. 2009, 100, 294–291. [Google Scholar] [CrossRef]
- Boggs, D.A.; Palmer, J.R.; Stampfer, M.J.; Spiegelman, D.; Adams-Campbell, L.L.; Rosenberg, L. Tea and coffee intake in relation to risk of breast cancer in the black women’s health study. Cancer Causes Control 2010, 21, 1941–1948. [Google Scholar] [CrossRef] [PubMed]
- Dominianni, C.; Huang, W.Y.; Berndt, S.; Hayes, R.B.; Ahn, J. Prospective study of the relationship between coffee and tea with colorectal cancer risk: The plco cancer screening trial. Br. J. Cancer 2013, 109, 1352–1359. [Google Scholar] [CrossRef] [PubMed]
- Gierach, G.L.; Freedman, N.D.; Andaya, A.; Hollenbeck, A.R.; Park, Y.; Schatzkin, A.; Brinton, L.A. Coffee intake and breast cancer risk in the nih-aarp diet and health study cohort. Int. J. Cancer 2012, 131, 452–460. [Google Scholar] [CrossRef] [PubMed]
- Groessl, E.J.; Allison, M.A.; Larson, J.C.; Ho, S.B.; Snetslaar, L.G.; Lane, D.S.; Tharp, K.M.; Stefanick, M.L. Coffee consumption and the incidence of colorectal cancer in women. J. Cancer Epidemiol. 2016, 2016, 6918431. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.A.; Parker, E.D.; Folsom, A.R. Coffee consumption and risk of type 2 diabetes mellitus: An 11-year prospective study of 28 812 postmenopausal women. Arch. Intern. Med. 2006, 166, 1311–1316. [Google Scholar] [CrossRef] [PubMed]
- Cavin, C.; Marin-Kuan, M.; Langouet, S.; Bezencon, C.; Guignard, G.; Verguet, C.; Piguet, D.; Holzhauser, D.; Cornaz, R.; Schilter, B. Induction of NRF2-mediated cellular defenses and alteration of Phase I activities as mechanisms of chemoprotective effects of coffee in the liver. Food Chem. Toxicol. 2008, 46, 1239–1248. [Google Scholar] [CrossRef] [PubMed]
- Higgins, L.G.; Cavin, C.; Itoh, K.; Yamamoto, M.; Hayes, J.D. Induction of cancer chemopreventive enzymes by coffee is mediated by transcription factor NRF2. Evidence that the coffee-specific diterpenes cafestol and kahweol confer protection against acrolein. Toxicol. Appl. Pharmacol. 2008, 226, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Nishikawa, A.; Shima, H.; Sugie, S.; Shinoda, T.; Yoshimi, N.; Iwata, H.; Mori, H. Inhibitory effects of chlorogenic acid, reserpine, polyprenoic acid (e-5166), or coffee on hepatocarcinogenesis in rats and hamsters. Basic Life Sci. 1990, 52, 429–440. [Google Scholar] [PubMed]
- Mori, H.; Tanaka, T.; Shima, H.; Kuniyasu, T.; Takahashi, M. Inhibitory effect of chlorogenic acid on methylazoxymethanol acetate-induced carcinogenesis in large intestine and liver of hamsters. Cancer Lett. 1986, 30, 49–54. [Google Scholar] [CrossRef]
- Miura, Y.; Furuse, T.; Yagasaki, K. Inhibitory effect of serum from rats administered with coffee on the proliferation and invasion of rat ascites hepatoma cells. Cytotechnology 1997, 25, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Miura, Y.; Ono, K.; Okauchi, R.; Yagasaki, K. Inhibitory effect of coffee on hepatoma proliferation and invasion in culture and on tumor growth, metastasis and abnormal lipoprotein profiles in hepatoma-bearing rats. J. Nutr. Sci. Vitaminol. (Tokyo) 2004, 50, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Chan, E.S.; Montesinos, M.C.; Fernandez, P.; Desai, A.; Delano, D.L.; Yee, H.; Reiss, A.B.; Pillinger, M.H.; Chen, J.F.; Schwarzschild, M.A.; et al. Adenosine a(2a) receptors play a role in the pathogenesis of hepatic cirrhosis. Br. J. Pharmacol. 2006, 148, 1144–1155. [Google Scholar] [CrossRef] [PubMed]
- Gressner, O.A.; Lahme, B.; Rehbein, K.; Siluschek, M.; Weiskirchen, R.; Gressner, A.M. Pharmacological application of caffeine inhibits tgf-beta-stimulated connective tissue growth factor expression in hepatocytes via ppargamma and smad2/3-dependent pathways. J. Hepatol. 2008, 49, 758–767. [Google Scholar] [CrossRef] [PubMed]
- Gordillo-Bastidas, D.; Oceguera-Contreras, E.; Salazar-Montes, A.; Gonzalez-Cuevas, J.; Hernandez-Ortega, L.D.; Armendariz-Borunda, J. NRF2 and snail-1 in the prevention of experimental liver fibrosis by caffeine. World J. Gastroenterol. 2013, 19, 9020–9033. [Google Scholar] [CrossRef] [PubMed]
- Gressner, O.A.; Lahme, B.; Siluschek, M.; Gressner, A.M. Identification of paraxanthine as the most potent caffeine-derived inhibitor of connective tissue growth factor expression in liver parenchymal cells. Liver Int. 2009, 29, 886–897. [Google Scholar] [CrossRef] [PubMed]
- Arauz, J.; Moreno, M.G.; Cortes-Reynosa, P.; Salazar, E.P.; Muriel, P. Coffee attenuates fibrosis by decreasing the expression of TGF-beta and CTGF in a murine model of liver damage. J. Appl. Toxicol. 2013, 33, 970–979. [Google Scholar] [CrossRef] [PubMed]
- Furtado, K.S.; Prado, M.G.; Aguiar, E.S.M.A.; Dias, M.C.; Rivelli, D.P.; Rodrigues, M.A.; Barbisan, L.F. Coffee and caffeine protect against liver injury induced by thioacetamide in male wistar rats. Basic Clin. Pharmacol. Toxicol. 2012, 111, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Vitaglione, P.; Morisco, F.; Mazzone, G.; Amoruso, D.C.; Ribecco, M.T.; Romano, A.; Fogliano, V.; Caporaso, N.; D’Argenio, G. Coffee reduces liver damage in a rat model of steatohepatitis: The underlying mechanisms and the role of polyphenols and melanoidins. Hepatology 2010, 52, 1652–1661. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Dong, L.; Bai, Y.; Zhao, J.; Zhang, Y.; Zhang, L. Chlorogenic acid against carbon tetrachloride-induced liver fibrosis in rats. Eur. J. Pharmacol. 2009, 623, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Dong, L.; Jiang, J.; Zhao, J.; Zhao, G.; Dang, X.; Lu, X.; Jia, M. Chlorogenic acid reduces liver inflammation and fibrosis through inhibition of toll-like receptor 4 signaling pathway. Toxicology 2013, 303, 107–114. [Google Scholar] [CrossRef]
- Monente, C.; Ludwig, I.A.; Stalmach, A.; de Pena, M.P.; Cid, C.; Crozier, A. In vitro studies on the stability in the proximal gastrointestinal tract and bioaccessibility in caco-2 cells of chlorogenic acids from spent coffee grounds. Int. J. Food Sci. Nutr. 2015, 66, 657–664. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Wang, X.F.; Shi, J.J.; Li, Y.P.; Yang, N.; Zhai, S.; Dang, S.S. Caffeic acid phenethyl ester inhibits liver fibrosis in rats. World J. Gastroenterol. 2015, 21, 3893–3903. [Google Scholar] [CrossRef] [PubMed]
- Pang, C.; Zheng, Z.; Shi, L.; Sheng, Y.; Wei, H.; Wang, Z.; Ji, L. Caffeic acid prevents acetaminophen-induced liver injury by activating the keap1-NRF2 antioxidative defense system. Free Radic. Biol. Med. 2016, 91, 236–246. [Google Scholar] [CrossRef] [PubMed]
- Yazgan, U.C.; Elbey, B.; Kus, S.; Baykal, B.; Keskin, I.; Yilmaz, A.; Sahin, A. Effect of caffeic acid phenethyl ester on oxidant and anti-oxidant status of liver and serum in a rat model with acute methanol intoxication. Ir. J. Med. Sci. 2016, 186, 519–523. [Google Scholar] [CrossRef]
- Dos Santos, P.R.; Ferrari, G.S.; Ferrari, C.K. Diet, sleep and metabolic syndrome among a legal amazon population, Brazil. Clin. Nutr. Res. 2015, 4, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Driessen, M.T.; Koppes, L.L.; Veldhuis, L.; Samoocha, D.; Twisk, J.W. Coffee consumption is not related to the metabolic syndrome at the age of 36 years: The amsterdam growth and health longitudinal study. Eur. J. Clin. Nutr. 2009, 63, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.S.; Chang, Y.F.; Liu, P.Y.; Chen, C.Y.; Tsai, Y.S.; Wu, C.H. Smoking, habitual tea drinking and metabolic syndrome in elderly men living in rural community: The tianliao old people (top) study 02. PLoS ONE 2012, 7, e38874. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Marventano, S.; Galvano, F.; Pajak, A.; Mistretta, A. Factors associated with metabolic syndrome in a mediterranean population: Role of caffeinated beverages. J. Epidemiol. 2014, 24, 327–333. [Google Scholar] [CrossRef]
- Grosso, G.; Stepaniak, U.; Micek, A.; Topor-Madry, R.; Pikhart, H.; Szafraniec, K.; Pajak, A. Association of daily coffee and tea consumption and metabolic syndrome: Results from the polish arm of the hapiee study. Eur. J. Nutr. 2015, 54, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Lutsey, P.L.; Steffen, L.M.; Stevens, J. Dietary intake and the development of the metabolic syndrome: The atherosclerosis risk in communities study. Circulation 2008, 117, 754–761. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, H.; Mure, K.; Nishio, N.; Kitano, N.; Nagai, N.; Takeshita, T. Relationship between coffee consumption and prevalence of metabolic syndrome among Japanese civil servants. J. Epidemiol. 2012, 22, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Nordestgaard, A.T.; Thomsen, M.; Nordestgaard, B.G. Coffee intake and risk of obesity, metabolic syndrome and type 2 diabetes: A mendelian randomization study. Int. J. Epidemiol. 2015, 44, 551–565. [Google Scholar] [CrossRef] [PubMed]
- Takami, H.; Nakamoto, M.; Uemura, H.; Katsuura, S.; Yamaguchi, M.; Hiyoshi, M.; Sawachika, F.; Juta, T.; Arisawa, K. Inverse correlation between coffee consumption and prevalence of metabolic syndrome: Baseline survey of the Japan Multi-Institutional Collaborative Cohort (J-MICC) study in Tokushima, Japan. J. Epidemiol. 2013, 23, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Suliga, E.; Koziel, D.; Ciesla, E.; Rebak, D.; Gluszek, S. Coffee consumption and the occurrence and intensity of metabolic syndrome: A cross-sectional study. Int. J. Food Sci. Nutr. 2016, 68, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Salomone, F.; Galvano, F.; Li Volti, G. Molecular bases underlying the hepatoprotective effects of coffee. Nutrients 2017, 9, 85. [Google Scholar] [CrossRef] [PubMed]
| Author, Year | Cohort Name, Country | Years of Study, Follow-up | Cases; Controls/Total Population | Age Range, Gender | Adjustments |
|---|---|---|---|---|---|
| Biliary Tract Cancer | |||||
| Prospective | |||||
| Petrick, 2015 | LCPP, USA | Multicentre | 260; 1,212,893 | MF | Age, sex, race, cohort, BMI, smoking status, cigarette smoking intensity, alcohol. |
| Makiuchi, 2016 | JPHC, Japan | 1990–2010 Cohort I, 1993–2010 Cohort II, 20 years (maximum) Cohort I, 17 years (maximum) Cohort II | 267; 89,555 | 40–69 years, MF | Age, sex, study area, BMI, history of cholelithiasis, history of diabetes mellitus, history of chronic hepatitis or cirrhosis, history of smoking, drinking frequency, physical activity by METs/day score, total energy consumption, energy-adjusted consumption of fish, red meat, and vegetable and fruit, and green tea. |
| Larsson, 2017 | SMC, COSM, Sweden | 1998–2012, 13.3 years | 74; 72,680 | 45–83 years, MF | Age, sex, education, smoking, BMI, and diabetes. |
| Case-Control | |||||
| Yen, 1987 | USA | 1975–1979, NA | 67/272 | 50–79 years, MF | Sex and age in decades. |
| Chow, 1994 | USA | 1985–1989, NA | 98/226 | 30–84 years, MF | Age, ethnic origin, and smoking status. |
| Liver Cancer | |||||
| Prospective | |||||
| Inoue, 2005 | JPHC, Japan | 1990–2001 Cohort I, 1993–2001 Cohort II, 9.7 years (average) | 334; 90,452 | 40–69 years, MF | Sex, age, study area, tobacco-smoking status, ethanol intake, green vegetable intake, green tea drinking. |
| Hu, 2008 | Finland | 1972–2002, 19.3 years (average) | 128; 60,323 | 25–74 years, MF | Age, sex, study year, alcohol consumption, education, smoking, diabetes and chronic liver disease at baseline and during follow-up, and BMI. |
| Inoue, 2009 | JPHC, Japan | 1993–2006 Cohort II, 12.7 years (average) | 110; 18,815 | 40–69 years, MF | Sex, age, area, smoking status, weekly ethanol intake, BMI, history of diabetes mellitus, green tea consumption, serum ALT level, HCV infection status, and HBV infection status. |
| Johnson, 2011 | SCHS, China | 1993–2006, 13 years (maximum) | 362; 61,321 | 45–74 years, MF | Age at recruitment, gender, dialect group, year of recruitment, BMI, level of education, consumption of alcoholic beverages, cigarette smoking, frequency of black tea and green tea intake, history of diabetes. |
| Lai, 2013 | ATBC, Finland | 1994–2009, 18.2 years (median) | 194; 27,037 | ~57 years (median), M | ATBC intervention arm, age, BMI, education, marital status, history of diabetes, years of smoking, cigarettes smoked per day, alcohol, tea intake, and serum cholesterol. |
| Bamia, 2015 | EPIC, Multicentre Europe | 1992–2010, 11 years (median) | 201; 486,799 | 25–70 years, MF | Age at recruitment, centre, sex, diabetes mellitus, education, BMI, smoking, physical activity, alcohol intake, energy intake, simultaneously including tea. |
| Petrick, 2015 | LCPP, USA | Consortium (AARP, AHS, USRT, PLCO, WHS, CPSII, IWHS, BWHS, WHI) | 860; 1,212,893 | MF | Sex, age, race, cohort, BMI, smoking status, cigarette smoking intensity, alcohol. |
| Setiawan, 2015 | MEC, USA | 1993–2010, 18 years (median) | 451; 162,022 | 45–75 years, MF | Age, sex, and race/ethnicity, education, BMI, alcohol intake, smoking status, and diabetes. |
| Case-Control | |||||
| Gallus, 2002 | Greece, Italy | I study (Italy) 1984–1997, II study (Greece) 1995–1998, NA | 333/360 Greece; 501/1552 Italy | 20–79 years, MF | Age, sex, smoking, tobacco smoking, alcohol drinking, BMI, history of diabetes and hepatitis. |
| Gelatti, 2005 | Italy | 1994–2003, NA | 250/500 | 50–79 years, MF | HBV infection, HCV infection, alcohol intake, sex and age. |
| Montella, 2007 | Italy | 1999–2002, NA | 185/412 | 43–84 years, MF | Gender, age, centre, education, smoking habits, maximal lifetime alcohol intake and serological evidence of HCV and/or HBV infection. |
| Tanaka, 2007 | Japan | 2001–2004, NA | 209/1308 | 40–79 years, MF | Sex, age, heavy alcohol use and smoking status. |
| Leung, 2011 | China | 2007–2008, NA | 109/125 | MF | Age, gender, cigarette smoking, alcohol use, tea consumption, physical activity. |
| Liver Cancer | ||||
|---|---|---|---|---|
| Subgroup | No. of Datasets | RR (95% CI) | I2 | Pheterogeneity |
| Total | 20 | 0.52 (0.42, 0.63) | 44% | 0.02 |
| Study design | ||||
| Prospective | 13 | 0.53 (0.41, 0.69) | 46% | 0.03 |
| Case-control | 7 | 0.48 (0.33, 0.70) | 47% | 0.08 |
| Gender | ||||
| Men | ||||
| Prospective | 5 | 0.49 (0.30, 0.80) | 64% | 0.02 |
| Case-control | 1 | 0.60 (0.40, 0.80) | NA | NA |
| Women | ||||
| Prospective | 4 | 0.53 (0.33, 0.83) | 0% | 0.71 |
| Case-control | 1 | 0.70 (0.50, 0.90) | NA | NA |
| Geographical location | ||||
| North America | 4 | 0.72 (0.52, 0.98) | 42% | 0.16 |
| Asia | 7 | 0.42 (0.30, 0.58) | 10% | 0.35 |
| Europe | 9 | 0.48 (0.36, 0.64) | 35% | 0.14 |
| Coffee type | ||||
| Caffeinated | 3 | 0.65 (0.49, 0.86) | 0% | 0.59 |
| Decaffeinated | 4 | 0.85 (0.63, 1.14) | 0% | 0.96 |
| Smoking status | ||||
| Never/former smoker | 4 | 0.61 (0.43, 0.88) | 32% | 0.22 |
| Current smoker | 5 | 0.54 (0.36, 0.81) | 61% | 0.04 |
| Chronic hepatitis | ||||
| Yes | 7 | 0.56 (0.39, 0.80) | 0% | 0.87 |
| No | 5 | 0.60 (0.48, 0.75) | 0% | 0.71 |
| No. of Datasets (No. of Studies) | Coffee Intake (Cups/Day) | I2 (%) | Pheterogeneity | Pnon-linearity | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | |||||
| Total analysis | ||||||||||||
| Non-linear | 3 (3) | Reference | 0.84 (0.67, 1.05) | 0.77 (0.54, 1.10) | 0.77 (0.43, 1.38) | 0.77 (0.31, 1.91) | 0.77 (0.22, 2.70) | 0.77 (0.15, 3.86) | 0.77 (0.11, 5.54) | 0.54 | 0.15 | 0.46 |
| Linear | 3 (3) | Reference | 0.91 (0.80, 1.03) | 0.82 (0.64, 1.06) | 0.75 (0.51, 1.09) | 0.68 (0.41, 1.12) | 0.61 (0.33, 1.15) | 0.56 (0.26, 1.18) | 0.50 (0.21, 1.22) | 0.18 | 0.13 | NA |
| No. of Datasets (No. of Studies) | Coffee Intake (Cups/Day) | I2 (%) | Pheterogeneity | Pnon-linearity | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | |||||
| Total analysis | ||||||||||||
| Non-linear | 7 (6) | Reference | 0.82 (0.70, 0.98) | 0.68 (0.53, 0.88) | 0.57 (0.46, 0.7) | 0.47 (0.39, 0.56) | 0.39 (0.31, 0.5) | 0.33 (0.23, 0.46) | 0.27 (0.17, 0.43) | 54.18 | 0.010 | 0.954 |
| Linear | 7 (6) | Reference | 0.85 (0.82, 0.88) | 0.72 (0.66, 0.78) | 0.61 (0.54, 0.69) | 0.52 (0.44, 0.61) | 0.44 (0.36, 0.54) | 0.58 (0.34, 0.98) | 0.32 (0.24, 0.42) | 17.54 | 0.296 | NA |
| Male | ||||||||||||
| Non-linear | 5 (5) | Reference | 0.73 (0.57, 0.94) | 0.56 (0.36, 0.85) | 0.47 (0.30, 0.72) | 0.42 (0.29, 0.60) | 0.38 (0.27, 0.53) | 0.33 (0.23, 0.46) | 0.30 (0.19, 0.48) | 72.9 | 0.000 | 0.286 |
| Linear | 4 (4) | Reference | 0.84 (0.80, 0.89) | 0.71 (0.64, 0.79) | 0.60 (0.51, 0.71) | 0.51 (0.41, 0.63) | 0.43 (0.33, 0.56) | 0.55 (0.47, 0.63) | 0.31 (0.21, 0.44) | 15.48 | 0.314 | NA |
| Female | ||||||||||||
| Non-linear | 4 (4) | Reference | 0.87 (0.72, 1.06) | 0.76 (0.56, 1.03) | 0.65 (0.46, 0.92) | 0.56 (0.31, 1.01) | 0.48 (0.19, 1.22) | 0.32 (0.22, 0.49) | 0.35 (0.06, 1.90) | 0 | 0.586 | 0.938 |
| Linear | 3 (3) | Reference | 0.88 (0.80, 0.96) | 0.77 (0.65, 0.92) | 0.68 (0.52, 0.88) | 0.59 (0.42, 0.84) | 0.52 (0.34, 0.81) | 0.53 (0.44, 0.65) | 0.40 (0.22, 0.74) | 0 | 0.692 | NA |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Godos, J.; Micek, A.; Marranzano, M.; Salomone, F.; Rio, D.D.; Ray, S. Coffee Consumption and Risk of Biliary Tract Cancers and Liver Cancer: A Dose–Response Meta-Analysis of Prospective Cohort Studies. Nutrients 2017, 9, 950. https://doi.org/10.3390/nu9090950
Godos J, Micek A, Marranzano M, Salomone F, Rio DD, Ray S. Coffee Consumption and Risk of Biliary Tract Cancers and Liver Cancer: A Dose–Response Meta-Analysis of Prospective Cohort Studies. Nutrients. 2017; 9(9):950. https://doi.org/10.3390/nu9090950
Chicago/Turabian StyleGodos, Justyna, Agnieszka Micek, Marina Marranzano, Federico Salomone, Daniele Del Rio, and Sumantra Ray. 2017. "Coffee Consumption and Risk of Biliary Tract Cancers and Liver Cancer: A Dose–Response Meta-Analysis of Prospective Cohort Studies" Nutrients 9, no. 9: 950. https://doi.org/10.3390/nu9090950
APA StyleGodos, J., Micek, A., Marranzano, M., Salomone, F., Rio, D. D., & Ray, S. (2017). Coffee Consumption and Risk of Biliary Tract Cancers and Liver Cancer: A Dose–Response Meta-Analysis of Prospective Cohort Studies. Nutrients, 9(9), 950. https://doi.org/10.3390/nu9090950






