Association between Dietary Patterns of Meat and Fish Consumption with Bone Mineral Density or Fracture Risk: A Systematic Literature
Abstract
:1. Introduction
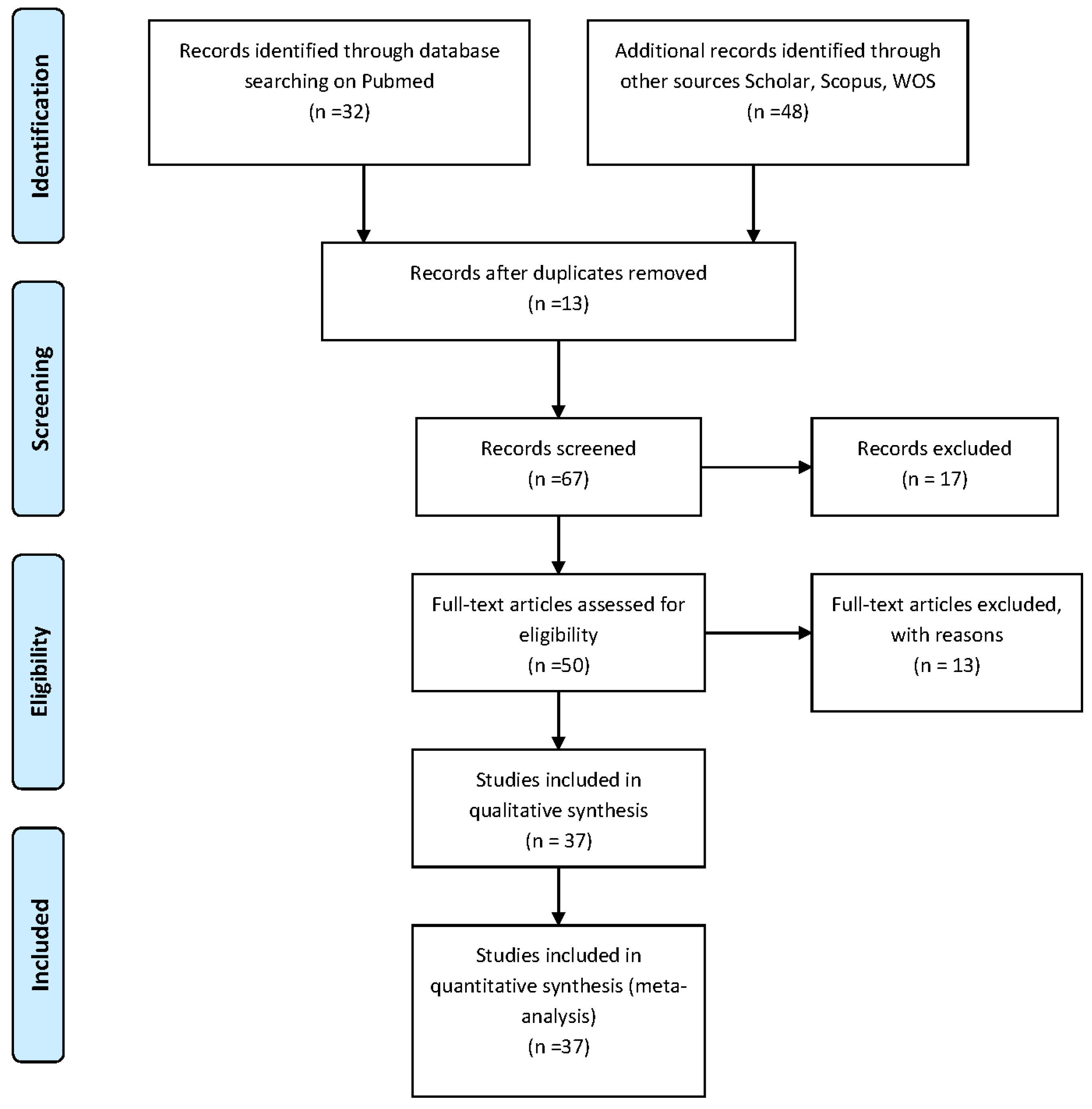
2. Methods
- (i)
- formulation of the revision question on the basis of considerations made in the abstract; and
- (ii)
- identification of relevant studies.
2.1. Inclusion and Exclusion Criteria
2.2. Data Collection
- (i)
- Author and year of publication;
- (ii)
- Number of participants for each study;
- (iii)
- Mean age of the subjects;
- (iv)
- Country;
- (v)
- Dietary patterns (for fish and meat consumption);
- (vi)
- Duration of intervention (in weeks or years);
- (vii)
- Association with BMD and Fractures Risk outcome;
- (viii)
- Results;
- (ix)
- Conclusions; and
- (x)
- Effect on BMD; and Fracture Risk.
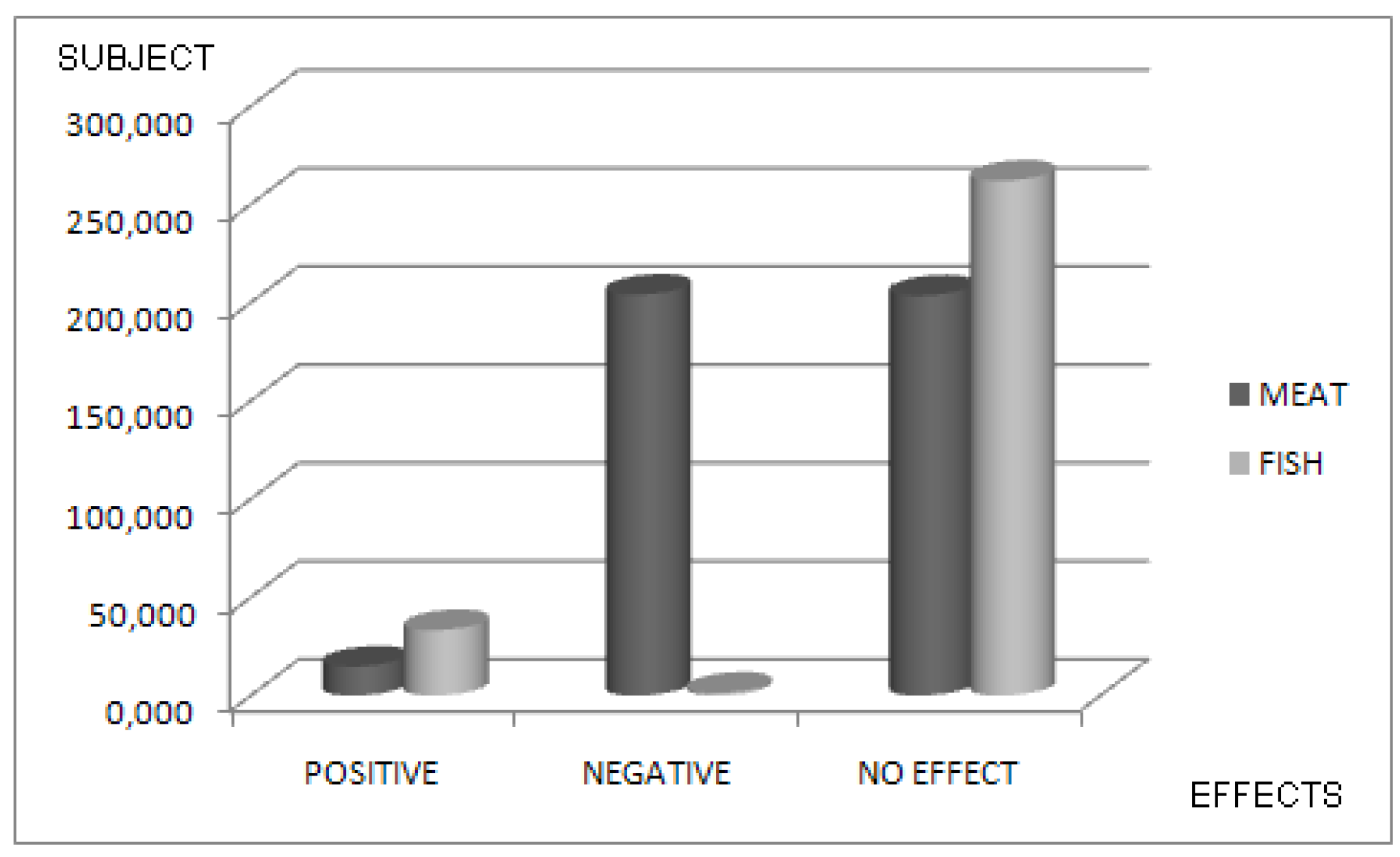
3. Results
4. Discussion
4.1. The Relevant Data
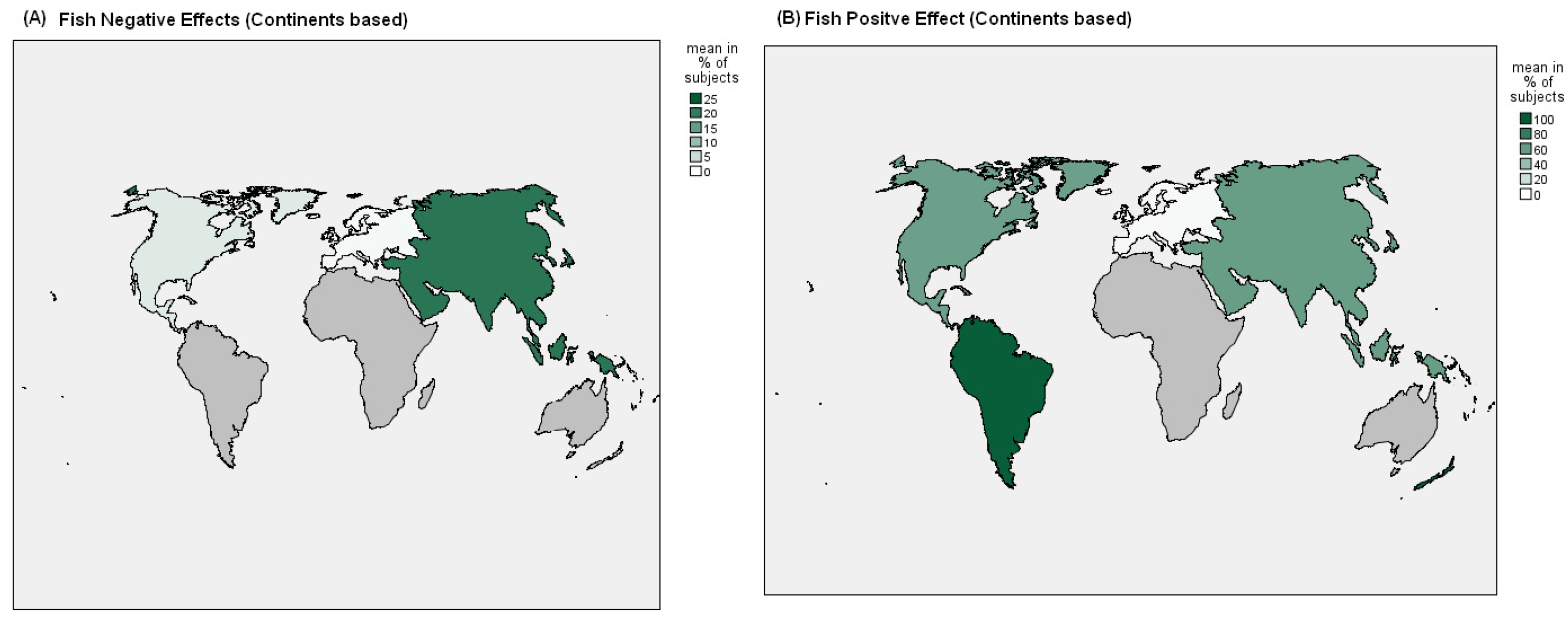
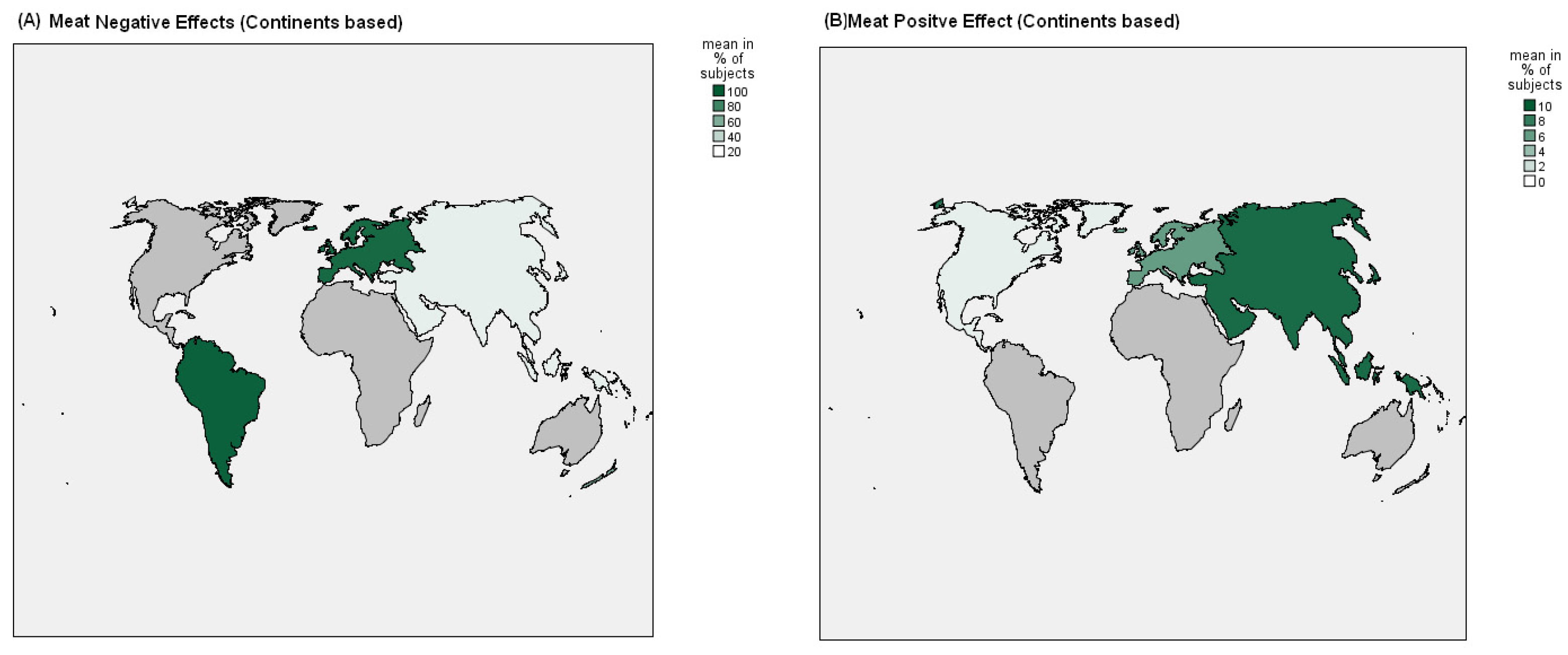
4.2. Negative Effects and Non-Compliance to Mediterranean or Asian Diet
4.3. The Positive Effects of FishDiet: Why and What?
4.4. Limitation of This Study
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Rizzoli, R.; Abraham, C.; Brandi, M.L. Nutrition and bone health: Turning knowledge and beliefs into healthy behaviour. Curr. Med. Res. Opin. 2014, 30, 131–141. [Google Scholar] [PubMed]
- Heaney, R.P. Dairy and bone health. J. Am. Coll. Nutr. 2009, 28 (Suppl. 1), 82S–90S. [Google Scholar] [PubMed]
- Nakashima, T.; Takayanagi, H. Osteoimmunology: Crosstalk between the immune and bone systems. J. Clin. Immunol. 2009, 29, 555–567. [Google Scholar] [PubMed]
- Kajarabille, N.; Díaz-Castro, J.; Hijano, S.; López-Frías, M.; LópezAliaga, I.; Ochoa, J.J. A new insight to bone turnover: Role of ω-3 polyunsaturated fatty acids. Sci. World J. 2013. [Google Scholar] [CrossRef]
- Shen, C.L.; von Bergen, V.; Chyu, M.C.; Jenkins, M.R.; Mo, H.; Chen, C.H.; Kwun, I.S. Fruits and dietary phytochemicals in bone protection. Nutr. Res. 2012, 32, 897–910. [Google Scholar] [PubMed]
- Puel, C.; Coxam, V.; Davicco, M.J. Mediterranean diet and osteoporosis prevention. Med. Sci. 2007, 23, 756–760. [Google Scholar]
- Benetou, V.; Orfanos, P.; Pettersson-Kymmer, U.; Bergström, U.; Svensson, O.; Johansson, I.; Berrino, F.; Tumino, R.; Borch, K.B.; Lund, E.; et al. Mediterranean diet and incidence of hip fractures in a European cohort. Osteoporos. Int. 2013, 24, 1587–1598. [Google Scholar] [PubMed]
- Alacid, F.; Vaquero-Cristóbal, R.; Sánchez-Pato, A.; Muyor, J.M.; López-Miñarro, P.Á. Habit based consumptions in the Mediterranean diet and the relationship with anthropometric parameters in young female kayakers. Nutr. Hosp. 2014, 29, 121–127. [Google Scholar] [PubMed]
- Ahmadieh, H.; Arabi, A. Vitamins and bone health: Beyond calcium and vitamin D. Nutr. Rev. 2011, 69, 584–598. [Google Scholar] [PubMed]
- Lambert, H.; Frassetto, L.; Moore, J.B.; Torgerson, D.; Gannon, R.; Burckhardt, P.; Lanham-New, S. The effect of supplementation with alkaline potassium salts on bone metabolism: A meta-analysis. Osteoporosis. Int. 2015, 26, 1311–1318. [Google Scholar]
- Cashman, K.D. Diet, Nutrition, and Bone Health. J. Nutr. 2007, 137, 2507S–2512S. [Google Scholar] [PubMed]
- Qiu, R.; Cao, W.T.; Tian, H.Y.; He, J.; Chen, G.D.; Chen, Y.M. Greater intake of fruit and vegetables is associated with greater bone mineral density and lower osteoporosis risk in middle-aged and elderly adults. PLoS ONE 2017, 12, e0168906. [Google Scholar]
- Marini, H.; Bitto, A.; Altavilla, D.; Burnett, B.P.; Polito, F.; Di Stefano, V.; Frisina, N. Breast safety and efficacy of genistein aglycone for postmenopausal bone loss: A follow-up study. J. Clin. Endocrinol. Metab. 2008, 93, 4787–4796. [Google Scholar] [PubMed]
- Egger, M.; Davey-Smith, G.; Altman, D. Systematic Reviews in Health Care: Meta-Analysis in Context; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- Thomson Reuters. Available online: www.thomsonreuters.com (accessed on 13 September 2017).
- Scopus. Available online: www.scopus.com (accessed on 13 September 2017).
- ISI Web of Science. Available online: www.webofknowledge.com (accessed on 13 September 2017).
- Google Scholar. Available online: www.scholar.google.com (accessed on 13 September 2017).
- Genant, H.K.; Cooper, C.; Poor, G.; Reid, I.; Ehrlich, G.; Kanis, J.; Dawson-Hughes, B. Interim report and recommendations of the World Health Organization task-force for osteoporosis. Osteoporos. Int. 1999, 10, 259–264. [Google Scholar] [PubMed]
- Casciaro, S.; Conversano, F.; Pisani, P.; Muratore, M. New perspectives in echographic diagnosis of osteoporosis on hip and spine. Clin. Cases Miner. Bone Metab. 2015, 12, 142. [Google Scholar] [PubMed]
- Langsetmo, L.; Hanley, D.A.; Prior, J.C.; Barr, S.I.; Anastassiades, T.; Towheed, T.; Goltzman, D.; Morin, S.; Poliquin, S.; Kreiger, N.; et al. Dietary patterns and incident low-trauma fractures in postmenopausal women and men aged ≥50 y: A population-based cohort study. Am. J. Clin. Nutr. 2011, 93, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Karamati, M.; Yousefian-Sanni, M.; Shariati-Bafghi, S.E.; Rashidkhani, B. Major nutrient patterns and bone mineral density among postmenopausal Iranian women. Calcif. Tissue Int. 2014, 94, 648–658. [Google Scholar] [PubMed]
- Langsetmo, L.; Poliquin, S.; Hanley, D.A.; Prior, J.C.; Barr, S.; Anastassiades, T.; Towheed, T.; Goltzman, D.; Kreiger, N. Dietary patterns in Canadian men and women ages 25 and older: Relationship to demographics, body mass index, and bone mineral density. BMC Musculoskelet. Disord. 2010, 11, 1471–2474. [Google Scholar]
- Wosje, K.S.; Khoury, P.R.; Claytor, R.P.; Copeland, K.A.; Hornung, R.W.; Daniels, S.R.; Kalkwarf, H.J. Dietary patterns associated with fat and bone mass in young children. Am. J. Clin. Nutr. 2010, 92, 294–303. [Google Scholar] [PubMed]
- Fairweather-Tait, S.J.; Skinner, J.; Guile, G.R.; Cassidy, A.; Spector, T.D.; MacGregor, A.J. Diet and bone mineral density study in postmenopausal women from the TwinsUK registry shows a negative association with a traditional English dietary pattern and a positive association with wine. Am. J. Clin. Nutr. 2011, 94, 1371–1375. [Google Scholar] [PubMed]
- Shin, S.; Joung, H. A dairy and fruit dietary pattern is associated with a reduced likelihood of osteoporosis in Korean postmenopausal women. Br. J. Nutr. 2013, 110, 1926–1933. [Google Scholar] [PubMed]
- Shin, S.; Sung, J.; Joung, H. A fruit, milk and whole grain dietary pattern is positively associated with bone mineral density in Korean healthy adults. Eur. J. Clin. Nutr. 2015, 69, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Park, S.J.; Joo, S.E.; Min, H.; Park, J.K.; Kim, Y.; Kim, S.S.; Ahn, Y. Dietary patterns and osteoporosis risk in postmenopausal Korean women. Osong Public Health Res. Perspect. 2012, 3, 199–205. [Google Scholar] [PubMed]
- Go, G.; Tserendejid, Z.; Lim, Y.; Jung, S.; Min, Y.; Park, H. The association of dietary quality and food group intake patterns with bone health status among Korean postmenopausal women: A study using the 2010 Korean National Health and Nutrition Examination Survey Data. Nutr. Res. Pract. 2014, 8, 662–669. [Google Scholar] [PubMed]
- Chan, R.; Leung, J.; Woo, J. Dietary patterns and risk of frailty in Chinese community-dwelling older people in Hong Kong: a prospective cohort study. Nutrients 2015, 7, 7070–7084. [Google Scholar] [PubMed]
- Choi, E.; Park, Y. The association between the consumption of fish/shellfish and the risk of osteoporosis in men and postmenopausal women aged 50 years or older. Nutrients 2016, 8, 113. [Google Scholar] [PubMed]
- De França, N.A.G.; Camargo, M.B.R.; Lazaretti-Castro, M.; Peters, B.S.E.; Martini, L.A. Dietary patterns and bone mineral density in Brazilian postmenopausal women with osteoporosis: A cross-sectional study. Eur. J. Clin. Nutr. 2016, 70, 85–90. [Google Scholar] [PubMed]
- Nieves, J.W.; Melsop, K.; Curtis, M.; Kelsey, J.L.; Bachrach, L.K.; Greendale, G.; Sowers, M.F.; Sainani, K.L. Nutritional factors that influence change in bone density and stress fracture risk among young female cross-country runners. Am. Acad. Phys. Med. Rehabil. 2010, 2, 740–750. [Google Scholar]
- McNaughton, S.A.; Wattanapenpaiboon, N.; Wark, J.D.; Nowson, C.A. An energy-dense, nutrient-poor dietary pattern is inversely associated with bone health in women. J. Nutr. Nutr. Epidemiol. 2011. [Google Scholar] [CrossRef]
- Monjardino, T.; Lucas, R.; Ramos, E.; Barros, H. Associations between a priori-defined dietary patterns and longitudinal changes in bone mineral density in adolescents. Public Health Nutr. 2014, 17, 195–205. [Google Scholar] [PubMed]
- Monjardino, T.; Lucas, R.; Ramos, E.; Lopes, C.; Gaio, R.; Barros, H. Associations between a posteriori defined dietary patterns and bone mineral density in adolescents. Eur. J. Nutr. 2015, 54, 273–282. [Google Scholar] [PubMed]
- Zeng, F.F.; Wu, B.H.; Fan, F.; Xie, H.L.; Xue, W.Q.; Zhu, H.L.; Chen, Y.M. Dietary patterns and the risk of hip fractures in elderly Chinese: A matched case-control study. J. Clin. Endocrinol. Metab. 2013, 98, 2347–2355. [Google Scholar] [CrossRef]
- Petersen, S.B.; Rasmussen, M.A.; Olsen, S.F.; Vestergaard, P.; Mølgaard, C.; Halldorsson, T.I.; Strøm, M. Maternal dietary patterns during pregnancy in relation to offspring forearm fractures: prospective study from the Danish national birth cohort. Nutrients 2015, 7, 2382–2400. [Google Scholar] [PubMed]
- Van den Hooven, E.H.; Ambrosini, G.L.; Huang, R.C.; Mountain, J.; Straker, L.; Walsh, J.P.; Zhu, K.; Oddy, W.H. Identification of a dietary pattern prospectively associated with bone mass in Australian young adults. Am. J. Clin. Nutr. 2015, 102, 1035–1043. [Google Scholar] [PubMed]
- Silva, T.R.; Franz, R.; Maturana, M.A.; Spritzer, P.M. Associations between body composition and lifestyle factors with bone mineral density according to time since menopause in women from Southern Brazil: A cross-sectional study. BMC Endocr. Disord. 2015, 15, 71. [Google Scholar]
- Haring, B.; Crandall, C.J.; Wu, C.; LeBlanc, E.S.; Shikany, J.M.; Carbone, L.; Orchard, T.; Thomas, F.; Wactawaski-Wende, J.; Li, W.; et al. Dietary patterns and fractures in postmenopausal women results from the women’s health initiative. JAMA Intern. Med. 2016, 176, 645–652. [Google Scholar] [CrossRef] [PubMed]
- Denova-Gutiérrez, E.; Clark, P.; Tucker, K.L.; Muñoz-Aguirre, P.; Salmerón, J. Dietary patterns are associated with bone mineral density in an urban Mexican adult population. Osteoporos. Int. 2016, 27, 3033–3040. [Google Scholar] [PubMed]
- De Jonge, E.A.L.; Rivadeneira, F.; Erler, N.S.; Hofman, A.; Uitterlinden, A.G.; Franco, O.H.; Kiefte-de Jong, J.C. Dietary patterns in an elderly population and their relation with bone mineral density: The Rotterdam Study. Eur. J. Nutr. 2016. [Google Scholar] [CrossRef]
- Hardcastle, A.C.; Aucott, L.; Fraser, W.D.; Reid, D.M.; Macdonald, H.M. Dietary patterns, bone resorption and bone mineral density in early post-menopausal Scottish women. Eur. J. Clin. Nutr. 2011, 65, 378–385. [Google Scholar] [PubMed]
- Mu, M.; Wang, S.F.; Sheng, J.; Zhao, Y.; Wang, G.X.; Liu, K.Y.; Hu, C.L.; Tao, F.B.; Wang, H.L. Dietary patterns are associated with body mass index and bone mineral density in Chinese freshmen. J. Am. Coll. Nutr. 2014, 33, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Melaku, Y.A.; Gill, T.K.; Adams, R.; Shi, Z. Association between dietary patterns and low bone mineral density among adults aged 50 years and above: Findings from the North West Adelaide Health Study (NWAHS). Br. J. Nutr. 2016, 116, 1437–1446. [Google Scholar] [PubMed]
- Kontogianni, M.D.; Melistas, L.; Yannakoulia, M.; Malagaris, I.; Panagiotakos, D.B.; Yiannakouris, N. Association between dietary patterns and indices of bone mass in a sample of Mediterranean women. Nutrition 2009, 25, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Tucker, K.L.; Chen, H.; Hannan, M.T.; Cupples, L.A.; Wilson, P.W.F.; Felson, D.; Kiel, D.P. Bone mineral density and dietary patterns in older adults: The Framingham Osteoporosis Study. Am. J. Clin. Nutr. 2002, 76, 245–252. [Google Scholar] [PubMed]
- Whittle, C.R.; Woodside, J.V.; Cardwell, C.R.; McCourt, H.J.; Young, I.S.; Murray, L.J.; Boreham, C.A.; Gallagher, A.M.; Neville, C.E.; McKinley, M.C. Dietary patterns and bone mineral status in young adults: The Northern Ireland Young Hearts Project. Br. J. Nutr. 2012, 108, 1494–1504. [Google Scholar] [CrossRef] [PubMed]
- Mangano, K.M.; Sahni, S.; Kiel, D.P.; Tucker, K.L.; Dufour, A.B.; Hannan, M.T. Dietary protein is associated with musculoskeletal health independently of dietary pattern: The Framingham Third Generation Study. Am. J. Clin. Nutr. 2017. [Google Scholar] [CrossRef]
- Monma, Y.; Niu, K.; Iwasaki, K.; Tomita, N.; Nakaya, N.; Hozawa, A.; Kuriyama, S.; Takayama, S.; Seki, T.; Takeda, T.; et al. Dietary patterns associated with fall-related fracture in elderly Japanese: A population based prospective study. BMC Geriatr. 2010, 10, 31. [Google Scholar]
- Okubo, H.; Sasaki, S.; Horiguchi, H.; Oguma, E.; Miyamoto, K.; Hosoi, Y.; Kim, M.; Kayama, F. Dietary patterns associated with bone mineral density in premenopausal Japanese farmwomen. Am. J. Clin. Nutr. 2006, 83, 1185–1192. [Google Scholar] [PubMed]
- Yang, Y.; Hu, X.M.; Chen, T.J.; Bai, M.J. Rural-urban differences of dietary patterns, overweight, and bone mineral status in Chinese students. Nutrients 2016, 8, 537. [Google Scholar] [CrossRef]
- Muraki, S.; Yamamoto, S.; Ishibashi, H.; Oka, H.; Yoshimura, N.; Kawaguchi, H.; Nakamura, K. Diet and lifestyle associated with increased bone mineral density: Cross-sectional study of Japanese elderly women at an osteoporosis outpatient. J. Orthopae. Sci. 2007. [Google Scholar] [CrossRef]
- De Jonge, E.A.; Kiefte-de Jong, J.C.; Hofman, A.; Uitterlinden, A.G.; Kieboom, B.C.; Voortman, T.; Franco, O.H.; Rivadeneira, F. Dietary patterns explaining differences in bone mineral density and hip structure in the elderly: The Rotterdam Study. Am. J. Clin. Nutr. 2017, 105, 203–211. [Google Scholar] [PubMed]
- Fung, T.T.; Feskanich, D. Dietary patterns and risk of hip fractures in postmenopausal women and men over 50 years. Osteoporos. Int. 2015, 26, 1825–1830. [Google Scholar] [PubMed]
- Mangano, K.M.; Sahni, S.; Kerstetter, J.E. Dietary protein is beneficial to bone health under conditions of adequate calcium intake: An update on clinical research. Curr. Opin. Clin. Nutr. Metab. Care 2014, 17, 69. [Google Scholar] [PubMed]
- Corwin, R.L.; Hartman, T.J.; Maczuga, S.A.; Graubard, B.I. Dietary saturated fat intake is inversely associated with bone density in humans: Analysis of NHANES III. J. Nutr. 2006, 136, 159–165. [Google Scholar] [PubMed]
- Atteh, J.O.; Leeson, S. Effects of dietary saturated or unsaturated fatty acids and calcium levels on performance and mineral metabolism of broiler chicks. Poult. Sci. 1984, 63, 2252–2260. [Google Scholar] [PubMed]
- Parhami, F.; Tintut, Y.; Beamer, W.G.; Gharavi, N.; Goodman, W.; Demer, L.L. Atherogenic high-fat diet reduces bone mineralization in mice. J. Bone Miner. Res. 2001, 16, 182–188. [Google Scholar] [PubMed]
- Tintut, Y.; Parhami, F.; Tsingotjidou, A.; Tetradis, S.; Territo, M.; Demer, L.L. 8-Isoprostaglandin E2 enhances receptor-activated NFκB ligand (RANKL)-dependent osteoclastic potential of marrow hematopoietic precursors via the cAMP pathway. J. Biol. Chem. 2002, 277, 14221–14226. [Google Scholar] [PubMed]
- Maurer, M.; Riesen, W.; Muser, J.; Hulter, H.N.; Krapf, R. Neutralization of Western diet inhibits bone resorption independently of K intake and reduces cortisol secretion in humans. Am. J. Physiol. Renal Physiol. 2003, 284, F32–F40. [Google Scholar] [PubMed]
- Sebastian, A.; Harris, S.T.; Ottaway, J.H.; Todd, K.M.; Morris, R.C., Jr. Improved mineral balance and skeletal metabolism in postmenopausal women treated with potassium bicarbonate. N. Engl. J. Med. 1994, 330, 1776–1781. [Google Scholar] [PubMed]
- Sahni, S.; Mangano, K.M.; McLean, R.R.; Hannan, M.T.; Kiel, D.P. Dietary approaches for bone health: Lessons from the Framingham osteoporosis study. Curr. Osteoporos. Rep. 2015, 13, 245–255. [Google Scholar] [PubMed]
- Teucher, B.; Dainty, J.R.; Spinks, C.A. Sodium and bone health: Impact of moderately high and low salt intakes on calcium metabolism in postmenopausal women. J. Bone Miner. Res. 2008, 23, 1477–1485. [Google Scholar] [PubMed]
- Harrington, M.; Cashman, K.D. High salt intake appears to increase bone resorption in postmenopausal women but high potassium intake ameliorates this adverse effect. Nutr. Rev. 2003, 61, 179–183. [Google Scholar] [PubMed]
- Papoutsi, Z.; Kassi, E.; Chinou, I.; Halabalaki, M.; Skaltsounis, L.A. Walnut extract (Juglans regia L.) and its component ellagic acid exhibit anti-inflammatory activity in human aorta endothelial cells and osteoblastic activity in the cell line KS483. Br. J. Nutr. 2008, 99, 715–722. [Google Scholar] [PubMed]
- Díaz-Castro, J.; Kajarabille, N.; Pulido-Morán, M.; Moreno-Fernández, J.; López-Frías, M.; Ochoa, J.J. Influence of omega-3 fatty acids on bone turnover. Omega-3 Fatty Acids 2016. [Google Scholar] [CrossRef]
- Orchard, T.S.; Steven, W.; Lu, B.; Belury, M.A.; Johnson, K.; Wactawski-Wende, J.; Jackson, R.D. The association of red blood cell n-3 and n-6 fatty acids with bone mineral density and hip fracture risk in the women’s health initiative. J. Bone Miner. Res. 2013, 28, 505–515. [Google Scholar] [PubMed]
- Chin, K.Y.; Ima-Nirwana, S. Can soy prevent male osteoporosis? A review of the current evidence. Curr. Drug Targets 2013, 14, 1632–1641. [Google Scholar] [PubMed]
| References | Number of Participants | Age (Years) | Country | Duration of the Study | Dietary Patterns Association with Osteoporosis Outcome Results | Conclusions | Summary of Effect on BMD | Study Design (Level of Evidence) | |
|---|---|---|---|---|---|---|---|---|---|
| Meat | Fish | ||||||||
| Langsetmo, L. et al. (2011) [21] | 5188 subjects (1649 men; 3539 women) | ≥50 | Canada | 2 years | Energy-dense factor (EDF) (representing energy-dense foods such as processed meat) are associated with HRs for risk of fractures of: 1.01 (p = NS) in women and of 1.08 (p = NS) in men. | NR | The intake of processed meat was not associated with fractures. | Meat: no effect on BMD; Fish: NR. | Retrospective cohort study |
| Karamati, M. et al. (2014) [22] | 151 women | 60.3 (59.1–61.6) | Iran | 3 years | NR | Pattern 2: includes fish intake. Pairwise difference between Lumbar spine: −0.01 g/cm2 (p = NS); Femoral neck: 0.01 g/cm2 (p = NS). | Pattern 2: (fish intake) was not associated with lumbar spine or femoral neck BMD. | Meat: NR; Fish: no effect on BMD. | Cross-sectional study |
| Langsetmo, L. et al. (2010) [23] | 6539 subjects (1928 men; 4611 women) | Men: 58.8 (±13.5) women: 61.2 (±12.2) | Canada | 5 years (secondary outcome); 2 years (primary outcome) | Energy dense food (included meat). R2 for dietary patterns and energy intake as predictors of femoral neck BMD (g/cm2). The parameter estimates are for each 1 SD increase of the nutrient dense factor score, the energy dense factor score, the difference between energy dense and nutrient dense factor score, and the log-transformed energy intake (1 SD is roughly 36% change in energy intake). p-Values for null hypothesis (from top to bottom). Adult Men: p = NS. Older Men: p = 0.007 (decrease 1 SD with meat). Premenopausal Women: p = NS. Postmenopausal Women: p = 0.032 (decrease 1 SD with meat). | Nutrient dense score food (included fish). R2 for dietary patterns and energy intake as predictors of femoral neck BMD (g/cm2). The parameter estimates are for each 1 SD increase of the nutrient dense factor score, the energy dense factor score, the difference between energy dense and nutrient dense factor score, and the log-transformed energy intake (1 SD is roughly 36% change in energy intake). p-values for null hypothesis (from top to bottom). Younger Men: p = 0.028 (increase 1 SD with fish); Older men: p = NS. Premenopausal Women: p = NS. Postmenopausal Women: p = NS. | Fish increased BMD in younger men. In older men Meat decreased BMD in older men and in postmenopausal women. | Meat: decreased BMD; Fish: increased BMD. | Longitudinal cohort study |
| Wosje K. et al. (2010) [24] | 325 children | 3.8–7.8 | USA | 4 years | Pattern 1: (meat, poultry, processed meat) high intake of meat. Bone mass increase during the quartile in 1,3 and 4 year (p < 0.01). | Pattern 2: high intake of fish. Bone mass increase during the quartile in year 1 and 3 (p < 0.01) and decrease during the quartile 4. | Pattern 1 (meat) was significantly associated with higher bone mass. Pattern 2 (fish) data related to bone mass were contradictory | Meat: increased BMD; Fish: no effect on BMD. | Longitudinal study |
| Fairweather-Tait S.J. et al. (2011) [25] | 2464 women | 56.3 (±11.9) | UK | 11 years | Traditional English pattern score (high intake of fish): Spine: BMD ∆: −0.035 g/cm2 (p < 0.05); Total hip: BMD ∆: −0.039 g/cm2 (p < 0.01); Hip Neck: BMD ∆: −0.055 g/cm2 (p < 0.01). | High intakes of fried fish, fried potatoes, legumes (e.g., baked beans), red and processed meat, vegetables was associated with a lower BMD. | Meat: decreased BMD; Fish: decreased BMD. | Co-twin control study | |
| Shin S. et al. (2013) [26] | 3735 women | 64.1 (±9.5) | South Korea | 3 years | Factor 1: meat consumption. CC among Factor 1 and BMD: Total femur: 0.005 CC; Trochanter: 0.008 CC; Intertrochanter: 0.004 CC; Femoral neck: 0.003 CC; Ward: 0.005 CC; Lumbar spine: 0.031 CC; (p = NS) Risk for osteoporosis of the femoral neck and lumbar spine across the quintile (Q) categories in factor 1: Femoral neck odds ratio (OR): 1.01 (p = NS); Lumbar spine: OR: 0.72 (p = NS). | Factor 3: Seaweed consumption. Correlation coefficients (CC) among Factor 3 and bone mineral density (BMD): Total femur 0.006 CC; Trochanter 0.011 CC; Intertrochanter −0.009 CC; Femoral neck −0.014 CC; Ward −0.040 CC; Lumbar spine −0.040 CC; Risk for osteoporosis of the femoral neck and lumbar spine across the quintile (Q) categories in factor 3: Femoral neck odds ratio (OR): 0.70 (p = NS); Lumbar spine: OR: 0.94 (p = NS). | Seaweed pattern (Factor 3) had a 40% higher risk of osteoporosis in the lumbar spine. | Meat: no effect on BMD; Fish: decreased BMD. | The Korea National Health and Nutrition Examination Survey (KNHANES: nationwide cross-sectional survey) |
| Shin S. et al. (2015) [27] | 1818 subjects (716 men; 1102 women) | 46.4 (±12.3) | South Korea | 2 years and 6 month | Factor 2: meat consumption (Meat/poultry/processed meats). Correlation coefficients (CC) among Factor 2 and BMD: Whole arm (g/cm2) −0.048 * CC; Whole leg (g/cm2): −0.041 CC; Whole pelvis (g/cm2): −0.020 CC; Whole spine (g/cm2): −0.023 CC; Whole body (g/cm2): 0.035 * CC. | Factor 1: fish Consumption. CC among Factor 1 and BMD: Whole arm (g/cm2) 0.088 *** CC; Whole leg (g/cm2) 0.050 * CC; Whole pelvis (g/cm2): 0.038 CC; Whole spine (g/cm2): 0.045 CC; Whole body (g/cm2): 0.017 CC. | The dietary pattern characterized by the consumption of fish and shellfish was significantly associated with whole-arm BMD only and not with other BMD measurements. | Meat: decreased BMD; Fish: increased BMD. | Healthy Twins Cohort, cross-sectional survey |
| Park S.J. e al. (2012) [28] | 1464 women | 58.8 (±6.7) | South Korea | 4 years | Factor 3 (Western diet): meat consumption: Radius RR (Risk Ratio): 1.46 (p < 0.05) Tibia RR: 1.46 (p = NS) | Factor 1 (Traditional diet): fish and seaweed consumption: Radius RR (Risk Ratio): 1.46 (p < 0.05); Tibia RR: 1.82 (p < 0.05). | Traditional diet with high intake of fish and Western with high intake of meat dietary patterns were associated with greater risk for osteoporosis in postmenopausal Korean women. | Meat: decreases BMD; Fish: decreases BMD. | The Korean Genome and Epidemiology Study (KoGES) is a longitudinal cohort study |
| Go G. et al. (2014) [29] | 847 women | NR | South Korea | 1 year | Food group with intake of meat (excluding dairy products, and including grain, vegetables and fruits): GMdVF (Grain, Meat, Dairy, Vegetable, Fruit Capital letter indicates eating a certain amount from the food group; lower case letter means not eating a certain amount from the food group): Normal (n = 136): GMdVF: 73 (53.7% n. of subject); Osteopenia (n = 413): GMdVF: 216 (53.0%); Osteoporosis (n = 298): GMdVF: 166 (59.5%). | NR | Meat consumption does not increase BMD. High number of subjects in group of osteoporosis versus normal (59% vs. 53.7%) | Meat: decreases BMD; Fish: NR. | The Korea National Health and Nutrition Examination Survey (KNHANES: nationwide cross-sectional survey) |
| Chan R. et al. (2015) [30] | 2724 women | 71.8 (±4.8) | Hong Kong | 2 years | Factor 3: (Meat-Fish) OR: 0.86 (IC 95%: 0.59–1.24) (p = NS) | Factor 3: (Fish-meat) OR: 0.86 (IC 95%: 0.59–1.24) (p = NS) | There was no association of “meat-fish” pattern with incident frailty | Meat: no effect on BMD; Fish: no effect on BMD. | Prospective cohort study |
| Choi E. et al. (2016) [31] | 9812 women | 60 | South Korea United States | 3 years | NR | In NHANES: p = NS; In KNHANES: p < 0.05; Correlation coefficients (CC) between bone mineral density (g/cm2) of: Total femur (g/cm2) men: 0.0748 CC; women: 0.1611 CC; Femoral neck (g/cm2) men: 0.0768 CC; women: 0.1806 CC; Lumbar spine (g/cm2) men: 0.0465 CC; women: 0.1630 CC. | A positive association between the consumption of fish and shellfish and bone health among men and postmenopausal women over 50 years old in Koreans but not in Americans | Meat: NR; Fish: increases BMD. | KNHANES and the NHANES |
| De Franca N.A.G. et al. (2016) [32] | 156 women | 68.4 (±9) | Brazil | 3 years | Meat included in “Red meat and refined cereals” dietary pattern Factor-loading matrix in red meat pattern for meat; 0.666 score Results of adjusted linear regression analysis (β-coefficient), and 95% confidence interval (95% CI) of the dietary patterns (score values) and body mineral density (g/cm2)): Lumbar spine (β:−0.094) (95% CI: −0.031 to 0.010) Femoral neck (β: −0.005) (95% CI IC: −0.016 to 0.015) Total femur (β:0.038) (95% CI: −0.014 to 0.022) Total body ( β:−0.019) (95% CI: −0.023 to 0.018) (p = NS) | Fish included in “Red meat and refined cereals” dietary pattern. Factor-loading matrix in red meat pattern for fish: −0.472 score. Results of adjusted linear regression analysis (β-coefficient), and 95% confidence interval of the dietary patterns (score values) and body mineral density (g/cm2): Lumbar spine (β:−0.094) (95% CI: −0.031 to 0.010); Femoral neck (β:−0.005) (95% CI: −0.016 to 0.015); Total femur (β: 0.038) (95% CI: −0.014 to 0.022); Total body (β:−0.019) (95% CI: −0.023 to 0.018); (p = NS) | No effects on BMD were observed with meat and fish consumption. | Meat: no effect on BMD; Fish: no effect on BMD. | Cross-sectional study |
| Nieves J.W. et al. (2010) [33] | 125 women | 22.1 (±2.6) | USA | 2 years | Dietary Pattern 3 (high animal proteins, high fat, low fruit and vegetables, low fiber): Dietary Pattern 3 HR:1.06 (95% CI: 0.54–2.09) (p = NS) Dietary Pattern 4 (high protein) are associated with HR of risk of fractures of: Dietary Pattern 4 HR:1.54 (95% CI: 0.31–7.48) (p = NS) Animal protein (SD) g/day/kg body Weight and Whole-body BMD (g/cm2/year ± SE) 0.00602 ± 0.00219 (p < 0.01) | Protein intake, specifically animal protein, was related to small but significantly greater increases in total body bone mass. | Meat: increases BMD Fish: increases BMD | Prospective cohort study | |
| McNaughton S.A. et al. (2011) [34] | 527 women | 18–65 | Australia | 10 years | Pattern 1 (Sausages and processed meat), Factor loading: 0.33 score BMC (g/cm2) β: −15.07 (p < 0.05) Hip BMD (g/cm2) β: 0.0013 (p = NS) Lumbar spine BMD (g/cm2) β:−0.0017 (p = NS) Pattern 2 (red meat), Factor loading: 0.27 score BMC (g/cm2) β: 3.14 (p = NS) Hip BMD (g/cm2) β: −0.0009 (p = NS) Lumbar spine BMD (g/cm2) β: −0.0017 (p = NS) | Pattern 3 (Fish) Factor loading: 0.23 score; BMC (g/cm2) β: 4.60 (p = NS); Hip BMD β: −0.0006 (p = NS); Lumbar spine BMD (g/cm2) β: −0.0001 (p = NS); Pattern 4 (Seafood) Factor loading: 0.48 score; BMC (g/cm2) β: 15.20 (p = NS); Hip BMD (g/cm2) β: 0.0022 (p < 0.05); Lumbar spine BMD (g/cm2) β: 0.0037 (p < 0.05). | Pattern 1(Sausages and processed meat) was inversely associated with total body BMC. Pattern 4 (Seafood) was directly associated with regional BMD and total BMC | Meat: decreases BMD Fish: increases BMD | Cross-sectional study |
| Monjardino T. et al. (2014) [35] | 1023 subjects (474 boys; 549 girls) | 13–17 | Portugal | 2 years | MD (Mediterranean diet) pattern: Meat Girls annual BMD variation (mg/cm2 per year): 0.028 (p = NS) Boys annual BMD variation (mg/cm2 per year): −0.012 (p = NS) DASH diet (Dietary Approaches to Stop Hypertension) Girls annual BMD variation (mg/cm2 per year): −0.002 (p = NS) Boys annual BMD variation (mg/cm2 per year): −0.026 (p = NS) | MD pattern: Fish; Girls annual BMD variation (mg/cm2 per year): 0.028 (p = NS); Boys annual BMD variation (mg/cm2 per year): −0.012 (p = NS); DASH diet (Dietary Approaches to Stop Hypertension): Girls annual BMD variation (mg/cm2 per year): −0.002 (p = NS); Boys annual BMD variation (mg/cm2 per year): −0.026 (p = NS). | The selected dietary patterns may not capture the elements of diet that are truly important in determining adolescent bone quality | Meat: no effect on BMD; Fish: no effect on BMD | Epidemiological Health Investigation of Teenagers in Porto (EPITeen population based cohort) |
| Monjardino T. et al. (2015) [36] | 1007 subjects (543 girls; 464 boys) | 13–17 | Portugal | 2 years | Lower intake (red meat): Girls annual BMD variation (mg/cm2 per year): −0.381 (p = NS); Boys annual BMD variation (mg/cm2 per year): 0.333 (p = NS) | Lower intake (fish): Girls annual BMD variation (mg/cm2 per year): −0.381 (p = NS); Boys annual BMD variation (mg/cm2 per year): 0.333 (p = NS). | There were no consistent associations between dietary patterns and forearm BMD in adolescents. | Meat: no effect on BMD; Fish: no effect on BMD. | Epidemiological Health Investigation of Teenagers in Porto (EPITeen population based cohort) |
| Zeng F.F. et al. (2013) [37] | 581 subjects (148 men; 433 women) | 71 (±7) | China | 3 years | Healthy Dietary Pattern (Poultry): OR of Hip Fractures (g/cm2) for Tertiles: (T3 vs. T1) OR: 0.42 (95% CI: 0.24-0.73), (p < 0.01) Prudent Dietary Pattern (Red meat): OR of Hip Fractures (g/cm2) for Tertiles: (T3 vs. T1) OR: 0.51 (95% CI: 0.28–0.90),(p < 0.05) Traditional Dietary Pattern (Processed meat, animal organ meat): OR of Hip Fractures (g/cm2) for Tertiles: (T3 vs. T1) OR: 0.83 (95% CI: 0.49–1.43), (p = NS); High Fat Dietary Pattern (Red meat, Poultry, Animal organ meat): OR of Hip Fractures (g/cm2) for Tertiles: (T3 vs. T1) OR: 2.25 (95% CI: 1.38–3.69), (p < 0.01) | Healthy Dietary Pattern (Freshwater fish): OR (Odds Ratio) of Hip Fractures (g/cm2) for Tertiles: (T3 vs. T1) OR: 0.42 (95% CI: 0.24–0.73) (p < 0.01); Prudent Dietary Pattern (shellfish, sea fish, processed fish): OR of Hip Fractures (g/cm2) for Tertiles: (T3 vs. T1) OR: 0.51 (95% CI: 0.28–0.90) (p < 0.05); Traditional Dietary Pattern (shellfish, processed fish): OR of Hip Fractures (g/cm2) for Tertiles: (T3 vs. T1) OR: 0.83 (95% CI: 0.49–1.43) (p = NS); High Fat Dietary Pattern (shellfish) OR of Hip Fractures (g/cm2) for Tertiles: (T3 vs. T1) OR: 2.25 (95% CI: 1.38–3.69) (p < 0.01) | The findings suggest that dietary patterns that feature a high intake of fish and low-fat poultry and a low intake of saturated fat may protect against hip fracture. | Meat: increases BMD Fish: increases BMD | 1:1 matched case-control study |
| Petersen S.B. et al. (2015) [38] | 53,922 children | <16 | Denmark | 6 years | Western (meat): HRs:1.03 (p = NS) Traditional (meat–poultry): HRs: 1.00 (p = NS). | Seafood (fish–shellfish): HRs: 0.94 (p = NS). | There were indications that maternal Western diet was associated with offspring forearm fractures. However, it was not possible to identify any single food item in the Western pattern that appeared to be of importance for offspring forearm fracture risk. | Meat: no effect on BMD Fish: no effect on BMD | Prospective study (Danish National Birth Cohort (DNBC)) |
| van den Hooven E.H. et al. (2015) [39] | 1024 young adults | 14–20 | Australia | 2 years | Pattern 2 (high-protein, low-calcium, low-potassium): Factor loading: Meat 0.24; Poultry 0.36; Red meat 0.42; Processed meat 0.29 BMD (mg/cm2): −0.2 (p = NS); BCM (g): −0.5 (p = NS). | Pattern 1 (high-protein, high-calcium, high-potassium) Factor loading: Fish 0.18; BMD (mg/cm2): 8.6 (p < 0.05); BCM (g): 21.9 (p < 0.05). | A dietary pattern characterized by high intake of protein and low intakes of calcium and potassium was not associated with later bone outcomes. A dietary pattern characterized by high-protein, high-calcium, high-potassium was associated with higher BMD and BMC | Meat: no effect on BMD Fish: increases BMD | Longitudinal study based on Western Australian Pregnancy Cohort (Raine) Study |
| Silva T.R. et al. (2015) [40] | 99 women | 55.2 (±4.9) | Brazil | 2 years | OR for low bone mass: Meat and eggs (<96 g/day): OR 2.30 (p = NS). | NR | Meat intake did not interfere with BMD, but participants were mostly sedentary | Meat: no effect on BMD Fish: NR | Cross-sectional study |
| Haring B. et al. (2016) [41] | 796 women | 63.6 (±7.4) | USA | 5 years | Mediterranean Diet (aMED) (red and processed meats): HRs: 0.80 (p = NS); Dietary Approaches to Stop Hypertension (DASH) (Red and processed meat): HRs: 0.89 (p = NS). | aMED (Fish) Hazard ratios (HRs): 0.80 (p = NS) Healthy Eating Index 2010 (HEI-2010) (seafood) HRs: 0.87 (p = NS) Alternate Healthy Eating Index 2010 (AHEI-2010) (long-chain ω-3 polyunsaturated fatty acids, polyunsaturated fatty acids) Hazard ratios (HRs): 0.94 (p = NS) | There were no consistent associations between dietary patterns and BMD. | Meat: no effect on BMD; Fish: no effect on BMD. | Women’s Health Initiative observational study (WHI-OS) |
| Denova-Gutiérrez E. (2016) [42] | 6915 subjects (1948 men; 4967 women) | 20–80 | Mexico | NR | Westernized dietary pattern (red meat): Odds ratios (OR): Total BMD (g/cm2): Q2 1.54; Q5 1.74 (p < 0.05); Hip BMD (g/cm2): Q2 1.40; Q5 1.91 (p < 0.01); Spine BMD (g/cm2): Q2 1.47; Q5 1.61(p < 0.05). | “Dairy and fish” dietary pattern (Fish and sea food) Odds ratios (OR) Total BMD (g/cm2): Q2 0.69; Q5 0.51 (p < 0.001) Hip BMD (g/cm2): Q2 0.99; Q5 0.86 (p = NS) Spine BMD (g/cm2): Q2 0.87; Q5 0.69 (p < 0.001) | A “dairy and fish” dietary pattern may contribute to better BMD. In contrast, a Westernized dietary pattern was significantly associated with higher likelihood of low BMD. | Meat: decreases BMD Fish: increases BMD | Cross-sectional analysis (Health Workers Cohort Study (HWCS)) |
| De Jonge E.A.L. et al. (2016) [43] | 5144 men and women | ≥55 | Netherlands | 11 years | Traditional dietary pattern (meat): BMD of the femoral neck: 0.01 g/cm2 (p = NS); Processed dietary pattern (processed meat): BMD of the femoral neck: −0.03 g/cm2 (p = NS); Health dietary pattern (poultry): BMD of the femoral neck: 0.04 g/cm2 (p = 0.01). | Health dietary pattern (fish): BMD of the femoral neck: 0.04 g/cm2 (p = 0.01). | Health dietary pattern has benefits for BMD; in contrast, adherence to a Processed dietary pattern may pose a risk for low BMD. | Meat: increases BMD Fish: increases BMD | The Rotterdam Study (population-based cohort study) |
| Hardcastle A.C. et al. (2011) [44] | 3236 women | 55.1 (±2.2) | Scotland | 9 years | Healthy pattern (meat). Multiple linear regression associated with the two bone resorption markers, fPYD/Cr (free deoxypyridinoline expressed relative to creatinine) and fDPD/Cr (free pyridinoline expressed relative to creatinine), HRT use and menopausal status. fPYD/Cr: Unstandardised β: 3.42 (95% CI: 3.13, 3.72) (p < 0001); fDPD/Cr: Unstandardised β: 2.07 (95% CI: 1.76, 2.39) (p < 0001). | Healthy pattern (fish). Multiple linear regression associated with the two bone resorption markers, fPYD/Cr (free deoxypyridinoline expressed relative to creatinine) and fDPD/Cr (free pyridinoline expressed relative to creatinine), HRT (hormone replacement therapy) use and menopausal status. fPYD/Cr: Unstandardised β: 3.42 (95% CI: 3.13, 3.72) (p < 0001); fDPD/Cr: Unstandardised β: 2.07 (95% CI: 1.76, 2.39) (p < 0001). | White meat, white and oily fish and dairy products contain nutrients that are associated with good bone health. | Meat: increases BMD Fish: increases BMD | Cross-sectional study |
| Mu M. et al. (2014) [45] | 1319 men | 18.1 (±1.2) | China | 1 month | Animal Protein Pattern: Meat (Lard, fat and lean meat) (Chicken, duck, goose): Hazard ratios (HR): 1.04 (p = NS) | Animal Protein Pattern: Fish (Carp, grass carp, silver carp, herring, shrimp) (Kelp laver, sea fish, seaweed) Hazard ratios (HR): 1.04 (p = NS) | The animal protein pattern was not associated with a decreased or increase risk of osteopenia or osteoporosis | Meat: no effect on BMD Fish: no effect on BMD | Cross-sectional study |
| Melaku Y.A. et al. (2016) [46] | 1182 men and women | median 62 years | South Australia | 11 years | Pattern 2 (“Western pattern”) includes high levels of processed and red meat, poultry: PR for the association between tertiles of food patterns and low bone mineral density. PR: 1.68 (95% CI: 1.02–2.77) (p < 0.05) | Pattern 1 (“prudent pattern”) includes fish PR for the association between tertiles of food patterns and low bone mineral density. PR: 0.52 (95% CI: 0.33–0.83) (p < 0.01). | Western pattern characterized by high intakes of processed and red meat was inversely associated with BMD Prudent pattern characterized by high intakes of fish was associated with higher BMD | Meat: decreases BMD Fish: increases BMD | The North West Adelaide Health Study (NWAHS) |
| Kontogianni M.D. et al. (2009) [47] | 196 women | 48 (±12) | Greek Greece | NR | A pattern characterized by high consumption of poultry (coefficient score 0.855) (component 4) BMD (lumbar bone mineral density) (g/cm2) β: 0.054 (p = NS) | A pattern characterized by high consumption of fish (coefficient score 0.867) (component 3); lumbar BMD (g/cm2) β: 0.185 (p < 0.05) | A dietary pattern characterized by high consumption of fish and low red meat intake was associated with higher BMD | Meat: no effect on BMD Fish: increases BMD | Cross-sectional study |
| Tucker K.L. et al. (2002) [48] | 907 women and men | Men 75.1 (±4.9) Women 75.3 (±4.8) | USA | 2 years | “Meat, dairy, and bread” group (n = 313) including p < 0.05: red meat, chicken BMD (±SE): Femoral neck 0.86 g/cm2 (p = 0.001); in men and in women 0.74 g/cm2 (p = NS); “Meat and sweet baked products” group (n = 260) including% p < 0.05: red meat, processed meat Adjusted mean (±SE) bone mineral density (BMD) “Sweet baked products” group (n = 69) including% p < 0.05: chicken Adjusted mean (±SE) bone mineral density (BMD) | “Meat, dairy, and bread” group (n = 313) including% p < 0.05: fish Adjusted mean (±SE) BMD “Sweet baked products” group (n = 69) including% p < 0.05: fish Adjusted mean (±SE) bone mineral density (BMD) “Candy” group (n = 75) including% p < 0.05: fish Adjusted mean (±SE) bone mineral density (BMD) | Men with a diet high in fruit, vegetables, and cereal (red meat and processed meat) had significantly greater BMD than did men with other dietary patterns. In contrast, those consuming the most candy (fish) had significantly lower BMD than did most other groups. | Meat: increases BMD Fish: decreases BMD | Longitudinal cohort study (The Framingham Heart Study) |
| Whittle C.R. et al. (2012) [49] | 489 women and men | Men 22.4 (±1.6) Women 22.8 (±1.7) | Northern Ireland | 2 years | Factor loading for men Factor 1: “Healthy” included meat dishes: −0.365 CC. BMD for the quintiles (Q) group of Healthy pattern determined by a posteriori principal component analysis. LS BMD (g/cm2) Q1–Q5 (p = NS). FN BMD (g/cm2) Q1–Q5 (p = NS). LS BMC (g) Q1–Q5 (p = NS). FN BMC (g) Q1–Q5 (p = NS); Factor 2: “Traditional” included red meat: 0,398 CC. Included poultry : –0.272 CC. LS BMD (g/cm2) Q1–Q5 (p = NS). FN BMD (g/cm2) Q1-Q5 (p = NS). LS BMC (g) Q1–Q5 (p = NS). FN BMC (g) Q1–Q5 (p = NS); Factor 3: “Refined” included meat dishes: 0,257 CC. LS BMD (g/cm2) Q1–Q5 (p = NS). FN (Femoral Neck) BMD (g/cm2) Q1–Q5 (p = NS). LS BMC (g) Q1–Q5 (p = NS). FN BMC (g) Q1–Q5 (p = NS) Adjusted (p < 0.05). Factor loading for women Factor 1: “Healthy” included meat dishes: −0.319 CC. LS BMD (g/cm2) Q1–Q5 (p = NS). FN BMD (g/cm2) Q1–Q5 (p = NS). LS BMC (g) Q1–Q5 (p = NS). FN BMC (g) Q1–Q5 (p = NS). Factor 2: “Traditional” included red meat: 0.299 included poultry: 0.337 LS BMD (g/cm2) Q1–Q5 (p = NS). FN BMD (g/cm2) Q1–Q5 (p = NS). LS BMC (g) Q1–Q5 (p = NS). FN BMC (g) Q1–Q5 (p = NS); Factor 3: “Nuts and Meat” included meat dishes: 0.372 LS BMD (g/cm2) Q1–Q5 (p = NS); FN (Femoral Neck) BMD (g/cm2) Q1–Q5 (p = NS) Adjusted (p < 0.05). LS BMC (g) Q1–Q5 (p = NS). FN BMC (g) Q1–Q5 (p = NS) Adjusted (p < 0.05). | Factor loading for men:Factor 4 “Social” included white fish: 0.436 CC. LS BMD (g/cm2) Q1–Q5 (p = NS); FN BMD (g/cm2) Q1–Q5 (p = NS); LS BMC (g) Q1–Q5 (p = NS). FN BMC (g) Q1–Q5 (p < 0.05) Adjusted (p = NS); Factor loading for women: Factor 1: “Healthy” included white fish: 0,325 CC. LS BMD (g/cm2) Q1–Q5 (p = NS). FN BMD (g/cm2) Q1–Q5 (p = NS); LS BMC (g) Q1–Q5 (p = NS). FN BMC (g) Q1–Q5 (p = NS). | “Refined” group scores (Factor 3 for men) (meat dishes) and “Nuts and Meat” group scores (Factor 3 for women) (meat dishes) were associated with higher FN BMC and in women also FN BMD. “Social” group scores were associated with higher FN BMC but when further adjusted were not significant. | Meat: increases BMD Fish: no effect on BMD | Longitudinal study (The Northern Ireland Young Hearts Project) |
| Mangano K.M. et al. (2017) [50] | 2986 women and men | 40.6 (±8.7) | USA | 3 years | Red Meat Femoral neck (g/cm2) (n = 2903) 0.989 ± 0.006 (p = NS); Trochanter (g/cm2) (n = 2903) 0.800 ± 0.006 (p = NS); Total femur (g/cm2) (n = 2903) 1.012 ± 0.006 Lumbar spine (g/cm2) (p = NS) (n = 2831) 1.227 ± 0.009 (p = NS); Chicken Femoral neck (g/cm2) (n = 2903) 1.002 ± 0.006 (p = NS); Trochanter (g/cm2) (n = 2903) 0.806 ± 0.006 (p = NS); Total femur (g/cm2) (n = 2903) 1.022 ± 0.006 (p = NS); Lumbar spine (g/cm2) (n = 2831) 1.233 ± 0.008 (p = NS) | Fish: Femoral neck: 1.000 ± 0.006 g/cm2 (n = 2903) (p = NS); Trochanter: 0.805 ± 0.006 g/cm2 (n = 2903) (p = NS); Total femur 1.016 ± 0.007 g/cm2 (n = 2903) (p = NS); Lumbar spine: 1.239 ± 0.009 g/cm2 (n = 2831) (p = NS). | No differences at any BMD site were observed across the protein food clusters in either crude models or adjusted models. | Meat: no effect on BMD; Fish: no effect on BMD | The Framingham Third Generation Study; Longitudinal cohort study |
| Monma Y. et al. (2010) [51] | 877 women and men | 80.7 (±5.2) | Japan | 4 years | Factor 2: “Meat” pattern included Pork, beef, ham, liver, Chicken. HR (95% CI) of fall-related fracture in each dietary pattern. T2 (moderately confirmed) HR: 0.36 (95% CI: 0.14–0.96); T3 (confirmed) HR: 0.36 (95% CI: 0.12–1.06) (p = NS) | Factor 2: “Meat” pattern included Shellfish, Cuttlefish, Octopus, Shrimp. HR (95% CI) of fall-related fracture in each dietary pattern. T2 (moderately confirmed) HR: 0.36 (95% CI: 0.14–0.96) T3 (confirmed) HR: 0.36 (95% CI: 0.12–1.06) (p = NS) | The “Meat” pattern had a tendency towards reduced risk of fall-related fracture. | Meat: increases BMD Fish: increases BMD | Prospective study |
| Okubo H. et al. (2006) [52] | 291 women | 40–55 | Japan | 3 years | Factor 3: “Western” (Processed meats and meats); Q1:0.501–0.006 g/cm2 and Q5: 0.482–0.007 g/cm2; (p = NS) | Factor 1: “Healthy” (Fish and shellfish and processed fish) Q1: 0.476–0.006 g/cm2; Q5: 0.498–0.006 g/cm2; (p < 0.05) | Healthy pattern (fish) had a significantly higher BMD. No significant association was observed in the Western pattern (meat) for premenopausal women. | Meat: no effect on BMD Fish: increases BMD | Japanese Multi-centred Environmental Toxicant Study (JMETS) |
| Yang Y. et al. (2016) [53] | 1590 boys and girls | 15.1 (±1.3) | China | NR | “Meat” diet Low Bone Quality OR: T2 OR: 0.911 (95% CI 0.620–0.255); T3 OR: 0.920 (95% CI 0.626–1.354); (p = NS) | “Chinese and Western” Low Bone Quality OR: T2 OR: 0.621 (95% CI 0.512–0.832); T3 OR: 0.558 (95% CI 0.414–0.901); (p < 0.05) | The risk of low bone mineral quality could be reduced by the Chinese and Western structure. | Meat: no effect on BMD; Fish: increases BMD. | Cross-sectional study |
| Muraki S. et al. (2007) [54] | 632 women | 71.8 (±7.5) | Japan | NR | NR | Fish consumption BMD (g/cm2) 0.791 ± 0.192 T score −1.73 ± 1.59 (p = NS) | Consumption or exclusion of fish in the diet has no significant effect on bone health | Meat: NR Fish: no effect on BMD | Cross-sectional study |
| de Jonge E.A. et al. (2017) [55] | 4028 subjects (1705 men; 2323 women) | Men 66 (61–72) Women 66 (61–73) | Netherlands | NR | Pattern: ”Sweets, animal fat, and low meat”: Osteoporotic fractures HR: 1.10 (95% CI: 1.06–1.15) (p < 0.05). HRs represent the difference in instantaneous risk of fracture per 1 z score difference in dietary pattern adherence. Hip fractures HR: 1.10 (95% CI: 1.01–1.19) (p < 0.05). | NR | Each z score of adherence to the sweets, animal fat, and low meat pattern was associated with higher bone width | Meat: increases BMD Fish: NR | Cross-sectional associations (Rotterdam Study) |
| Fung T.T. et al. (2015) [56] | 112,845 subjects (38,305 men; 74,540 women) | Women: 30–55; Men: 40–75 | USA | 2 years | Relative risk (RR) (95% CI) for hip fractures according quintiles of dietary patterns: Prudent pattern (poultry and red meat): Women Q1:1–Q5: RR: 1.14 (95% CI: 0.96–1.36) (p = NS); Men Q1:1–Q5: RR: 0.86 (95% CI: 0.64-1.16) (p = NS). Western pattern (poultry and red meat): Women Q1:1–Q5: RR: 1.05 (95% CI: 0.87–1.26) (p = NS); Men Q1:1–Q5: RR: 1.03 (95% CI: 0.73–1.46) (p = NS). | NR | Neither the Prudent nor the Western dietary pattern was associated with risk of hip fractures in postmenopausal women or men over 50 years of age. | Meat: no effect on BMD Fish: NR | The Nurses’ Health Study and the Health Professionals Follow-up Study |
| Benetou V. et al. (2013) [7] | 188,795 subjects (48,814 men; 139,981 women) | 48.6 (±10.8) | Germany, Greece, Italy, Netherlands, Norway, Spain, Sweden, UK | 8 years | HR for incident hip fracture per indicated increments of intake with 95% CI in overall sample: HR per 1-unit increment: 1.18 (95% CI: 1.06–1.3); men HR per 1-unit increment 1.10 (95% CI: 0.92–1.32); women HR per 1-unit increment 1.14 (95% CI: 0.99–1.31). | HR for incident hip fracture per indicated increments of intake with 95% CI in: overall sample HR per 1-unit increment 0.96 (95% CI: 0.86–1.07) men HR per 1-unit increment 0.89 (95% CI: 0.73–1.09) women HR per 1-unit increment 0.97 (95% CI: 0.85–1.12) | High meat intake was associated with increased hip fracture incidence Higher fish consumption was weakly, although not significantly, associated with lower hip fracture incidence | Meat: decrease BMD; Fish: no effect on BMD | Prospective study |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perna, S.; Avanzato, I.; Nichetti, M.; D’Antona, G.; Negro, M.; Rondanelli, M. Association between Dietary Patterns of Meat and Fish Consumption with Bone Mineral Density or Fracture Risk: A Systematic Literature. Nutrients 2017, 9, 1029. https://doi.org/10.3390/nu9091029
Perna S, Avanzato I, Nichetti M, D’Antona G, Negro M, Rondanelli M. Association between Dietary Patterns of Meat and Fish Consumption with Bone Mineral Density or Fracture Risk: A Systematic Literature. Nutrients. 2017; 9(9):1029. https://doi.org/10.3390/nu9091029
Chicago/Turabian StylePerna, Simone, Ilaria Avanzato, Mara Nichetti, Giuseppe D’Antona, Massimo Negro, and Mariangela Rondanelli. 2017. "Association between Dietary Patterns of Meat and Fish Consumption with Bone Mineral Density or Fracture Risk: A Systematic Literature" Nutrients 9, no. 9: 1029. https://doi.org/10.3390/nu9091029





