Evaluation of Bread Crumbs as a Potential Carbon Source for the Growth of Thraustochytrid Species for Oil and Omega-3 Production
Abstract
:1. Introduction
2. Experimental Section
2.1. Preparation of Seed Culture
2.2. Submerged Liquid Fermentation with Glucose Medium
2.3. Preparation of Bakery Waste Bread Crumbs (BC) for Static Fermentation
2.4. Scanning Electron Microscopy (SEM)
2.5. Fatty Acid Extraction and Gas Chromatography (GC) Analysis
2.6. Attenuated Total Reflection Fourier Transform Infrared (ATR-FTIR) Spectroscopic Analysis
2.7. Statistical Analysis
3. Results and Discussion
| Fermentation type | Biomass (mg/L) | Lipids (mg/L) | Saturated fatty acids 1 (mg/g) | Mono-unsaturated fatty acids 2 (mg/g) |
|---|---|---|---|---|
| Submerged liquid fermentation (3% glucose) | 4300 | 941.32 | 44.16 | 63.19 |
| Static fermentation (0.5% bread crumbs) | 2530 | 260.0 | 42.4 | 29.00 |
| Static fermentation (1% bread crumbs) | 4760 | 390.0 | 33.6 | 22.6 |
3.1. Fermentation Growth Using Bread Crumbs as the Carbon Source
| C16:0 | C18:0 | C18:1n9 | C18:2n6 | C18:3n3 | C20:5n3 | C22:5n3 | C22:6n3 | others |
|---|---|---|---|---|---|---|---|---|
| Unfermented bread crumbs fatty acid profile | ||||||||
| 2.20 | 1.50 | 3.10 | 1.00 | 0.00 | 0.00 | 0.00 | 0.00 | 1.50 |
| Submerged liquid fermentation fatty acid profile 2 | ||||||||
| 32.33 a | 11.82 a | 63.19 a | 2.76 a | 0.00 a | 3.03 | 4.32 | 23.74 | 33.35 |
| Static fermentation fatty acid profile 3 | ||||||||
| 25.9 b,4 | 16.5 b | 29.0 b | 12.9 b | 1.2 b | 0.00 | 0.57 | 2.40 | 14.40 |
| 20.4 b,5 | 13.2 b | 22.6 b | 11.2 b | 1.2 b | 0.00 | 0.00 | 1.30 | 11.50 |

3.2. Scanning Electron Microscopy (SEM) Observation of Cell Growth

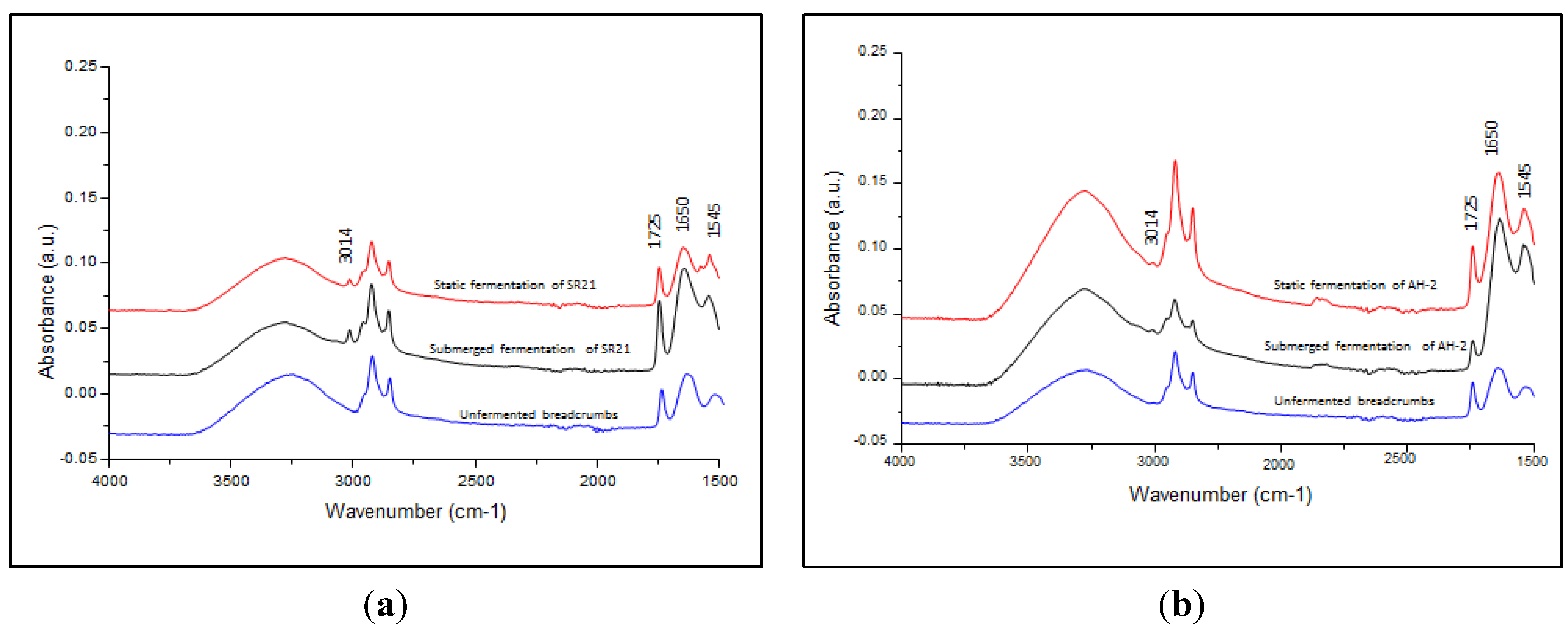
3.3. ATR-FTIR Spectroscopy Analysis of Cell Lipid Content
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Raghukumar, S. Ecology of the marine protists, the Labyrinthulomycetes (Thraustochytrids and Labyrinthulids). Eur. J. Protisol. 2002, 38, 127–145. [Google Scholar] [CrossRef]
- Gupta, A.; Barrow, C.J.; Puri, M. Omega-3 biotechnology: Thraustochytrids as a novel source of omega-3 oils. Biotechnol. Adv. 2012, 30, 1733–1745. [Google Scholar]
- Lee Chang, K.; Dunstan, G.; Abell, G.; Clementson, L.; Blackburn, S.; Nichols, P.; Koutoulis, A. Biodiscovery of new Australian thraustochytrids for production of biodiesel and long-chain omega-3 oils. Appl. Microbiol. Biotechnol. 2012, 93, 2215–2231. [Google Scholar] [CrossRef]
- Brennan, L.; Owende, P. Biofuels from microalgae A review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 14, 557–577. [Google Scholar] [CrossRef]
- Johnson, M.B.; Wen, Z. Production of biodiesel fuel from the microalga Schizochytrium limacinum by direct transesterification of algal biomass. Energy Fuels 2009, 23, 5179–5183. [Google Scholar] [CrossRef]
- Pleissner, D.; Lam, W.C.; Sun, Z.; Lin, C.S.K. Food waste as nutrient source in heterotrophic microalgae cultivation. Bioresour. Technol. 2013, 137, 139–146. [Google Scholar] [CrossRef]
- Abad, S.; Turon, X. Valorization of biodiesel derived glycerol as a carbon source to obtain added-value metabolites: Focus on polyunsaturated fatty acids. Biotechnol. Adv. 2012, 30, 733–741. [Google Scholar]
- Ferreira, M.L.; Santos, M.P.; Moro, T.A.; Basto, G.; Andrade, R.S.; Gonçalves, É.B.A. Formulation and characterization of functional foods based on fruit and vegetable residue flour. J. Food Sci. 2013, 22, 498–508. [Google Scholar]
- Anikwe, M.A.N.; Nwobodo, K.C.A. Long term effect of municipal waste disposal on soil properties and productivity of sites used for urban agriculture in Abakaliki, Nigeria. Bioresour. Technol. 2002, 83, 241–250. [Google Scholar] [CrossRef]
- Rawat, I.; Ranjith Kumar, R.; Mutanda, T.; Bux, F. Biodiesel from microalgae: A critical evaluation from laboratory to large scale production. Appl. Energy 2013, 103, 444–467. [Google Scholar] [CrossRef]
- Fan, K.W.; Chen, F.; Jones, E.B.G.; Vrijmoed, L.L.P. Utilization of food processing waste by Thraustochytrids. Fungal Divers. 2000, 5, 185–194. [Google Scholar]
- Jang, H.-D.; Yang, S.-S. Polyunsaturated fatty acids production with a solid-state column reactor. Bioresour. Technol. 2008, 99, 6181–6189. [Google Scholar] [CrossRef]
- Fan, K.W.; Chen, F.; Jones, E.B.G.; Vrijmoed, L.L.P. Eicosapentaenoic and docosahexaenoic acids production by and okara-utilizing potential of thraustochytrids. J. Ind. Microbiol. Biotechnol. 2001, 27, 199–202. [Google Scholar] [CrossRef]
- Quilodran, B.; Hinzpeter, I.; Quiroz, A.; Shene, C. Evaluation of liquid residues from beer and potato processing for the production of docosahexaenoic acid (C22:6n-3, DHA) by native thraustochytrid strains. World J. Microbiol. Biotechnol. 2009, 25, 2121–2128. [Google Scholar] [CrossRef]
- Liang, Y.; Sarkany, N.; Cui, Y.; Yesuf, J.; Trushenski, J.; Blackburn, J.W. Use of sweet sorghum juice for lipid production by Schizochytrium limacinum SR21. Bioresour. Technol. 2010, 101, 3623–3627. [Google Scholar] [CrossRef]
- Unagul, P.; Assantachai, C.; Phadungruengluij, S.; Suphantharika, M.; Tanticharoen, M.; Verduyn, C. Coconut water as a medium additive for the production of docosahexaenoic acid (C22:6 n3) by Schizochytrium mangrovei Sk-02. Bioresour. Technol. 2007, 98, 281–287. [Google Scholar] [CrossRef]
- Jung, I.S.; Lovitt, R.W. Integrated production of long chain polyunsaturated fatty acids (PUFA)-rich Schizochytrium biomass using a nutrient supplemented marine aquaculture wastewater. Aquac. Eng. 2010, 43, 51–61. [Google Scholar] [CrossRef]
- Pyle, D.J.; Garcia, R.A.; Wen, Z. Producing docosahexaenoic acid (DHA)-rich algae from biodiesel-derived crude glycerol: Effects of impurities on DHA production and algal biomass composition. J. Agric. Food Chem. 2008, 56, 3933–3939. [Google Scholar] [CrossRef]
- Li, Q.; Chen, G.-Q.; Fan, K.-W.; Lu, F.-P.; Aki, T.; Jiang, Y. Screening and Characterization of Squalene-Producing Thraustochytrids from Hong Kong Mangroves. J. Agric. Food Chem. 2009, 57, 4267–4272. [Google Scholar] [CrossRef]
- Lewis, T.; Nichols, P.D.; McMeekin, T.A. Evaluation of extraction methods for recovery of fatty acids from lipid-producing microheterotrophs. J. Microbiol. Methods 2000, 43, 107–116. [Google Scholar] [CrossRef]
- William, W.C.; Han, X. Lipid Technology—Isolation, Separation, Identification and Lipidomic Analysis, 4th ed.; The Oily Press: Bridgwater, UK, 2010; Volume 22, p. 119. [Google Scholar]
- Ackman, R.G. The gas chromatograph in practical analyses of common and uncommon fatty acids for the 21st century. Anal. Chim. Acta 2002, 465, 175–192. [Google Scholar] [CrossRef]
- Kanchana, R.; Ushadevi, M.; Raghukumar, S. Alkaline lipase activity from the marine protists, thraustochytrids. World J. Microbiol. Biotechnol. 2011, 27, 2125–2131. [Google Scholar] [CrossRef]
- Bajpai, P.K.; Bajpai, P.; Ward, O.P. Optimization of production of docosahexaenoic acid (DHA) by Thraustochytrium aureum ATCC 34304. J. Am. Oil Chem. Soc. 1991, 68, 509–514. [Google Scholar] [CrossRef]
- Scanlon, M.G.; Zghal, M.C. Bread properties and crumb structure. Food Res. Int. 2001, 34, 841–864. [Google Scholar] [CrossRef]
- Shene, C.; Leyton, A.; Esparza, Y.; Flores, L.; Quilodran, B.; Hinzpeter, I.; Rubilar, M. Microbial oils and fatty acids: Effect of carbon source on docosahexaenoic acid (C22:6n3, DHA) production by thraustochytrid strains. J. Plant Nutr. Soil Sci. 2010, 10, 207–216. [Google Scholar]
- Holdsworth, J.E.; Ratledge, C. Lipid turnover in oleaginous yeasts. J. Gen. Microbiol. 1988, 134, 339–346. [Google Scholar]
- Vongsvivut, J.; Heraud, P.; Zhang, W.; Kralovec, J.A.; McNaughton, D.; Barrow, C.J. Quantitative determination of fatty acid compositions in micro-encapsulated fish-oil supplements using Fourier transform infrared (FTIR) spectroscopy. Food Chem. 2012, 135, 603–609. [Google Scholar] [CrossRef]
- Vongsvivut, J.; Heraud, P.; Gupta, A.; Puri, M.; McNaughton, D.; Barrow, C.J. FTIR microspectroscopy for rapid screening and monitoring of polyunsaturated fatty acid production in commercially valuable marine yeasts and protists. Analyst 2013, 138, 6016–6031. [Google Scholar] [CrossRef]
- Guillen, M.D.; Cabo, N. Characterization of edible oils and lard by fourier transform infrared spectroscopy. Relationships between composition and frequency of concrete bands in the fingerprint region. J. Am. Oil Chem. Soc. 1997, 74, 1281–1286. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Thyagarajan, T.; Puri, M.; Vongsvivut, J.; Barrow, C.J. Evaluation of Bread Crumbs as a Potential Carbon Source for the Growth of Thraustochytrid Species for Oil and Omega-3 Production. Nutrients 2014, 6, 2104-2114. https://doi.org/10.3390/nu6052104
Thyagarajan T, Puri M, Vongsvivut J, Barrow CJ. Evaluation of Bread Crumbs as a Potential Carbon Source for the Growth of Thraustochytrid Species for Oil and Omega-3 Production. Nutrients. 2014; 6(5):2104-2114. https://doi.org/10.3390/nu6052104
Chicago/Turabian StyleThyagarajan, Tamilselvi, Munish Puri, Jitraporn Vongsvivut, and Colin J. Barrow. 2014. "Evaluation of Bread Crumbs as a Potential Carbon Source for the Growth of Thraustochytrid Species for Oil and Omega-3 Production" Nutrients 6, no. 5: 2104-2114. https://doi.org/10.3390/nu6052104
APA StyleThyagarajan, T., Puri, M., Vongsvivut, J., & Barrow, C. J. (2014). Evaluation of Bread Crumbs as a Potential Carbon Source for the Growth of Thraustochytrid Species for Oil and Omega-3 Production. Nutrients, 6(5), 2104-2114. https://doi.org/10.3390/nu6052104





