Dietary Proteins and Angiogenesis
Abstract
:1. Introduction
2. Milk Angiogenin-2

3. Milk Lactoferrin

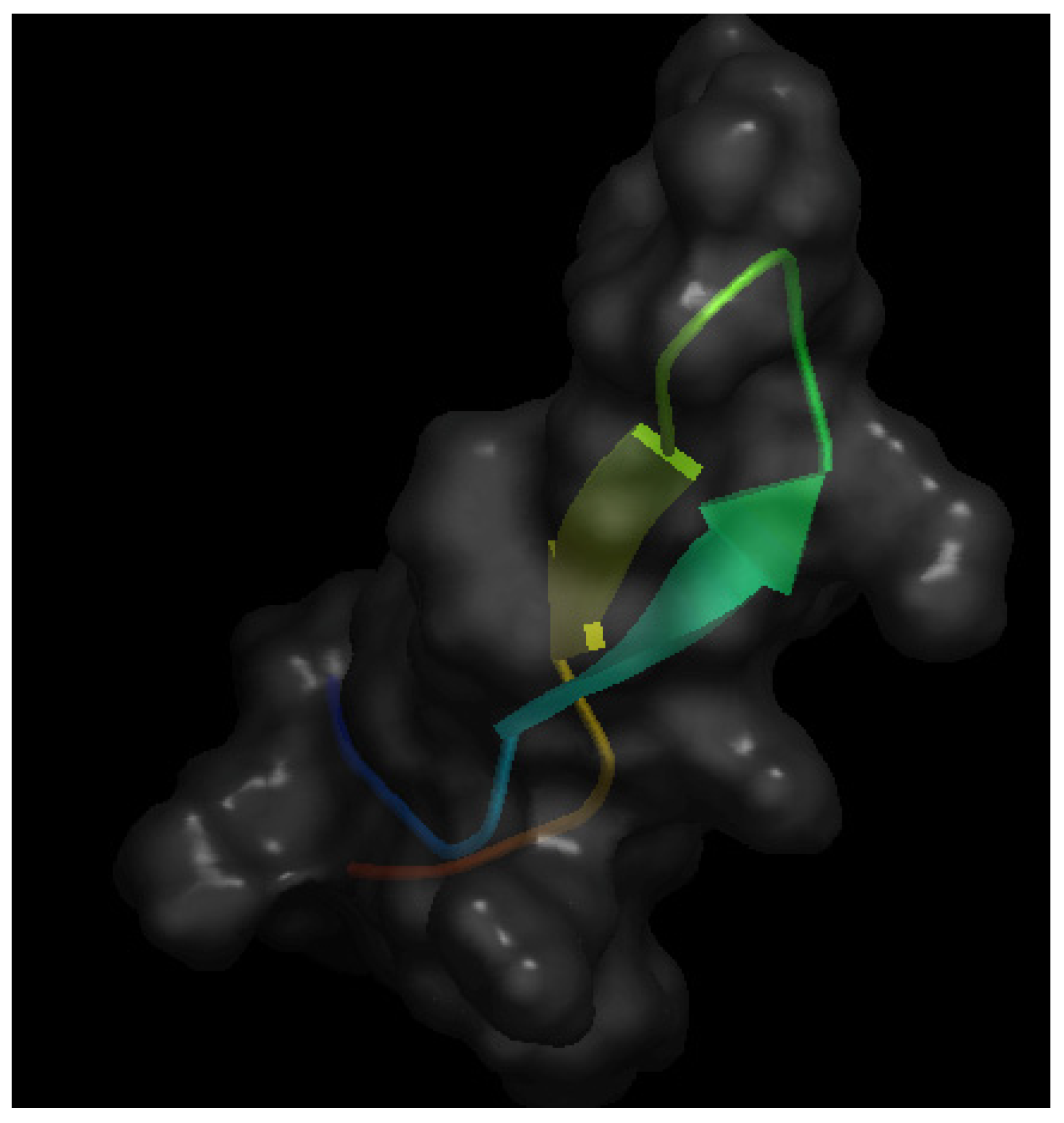
4. Milk Lactadherin
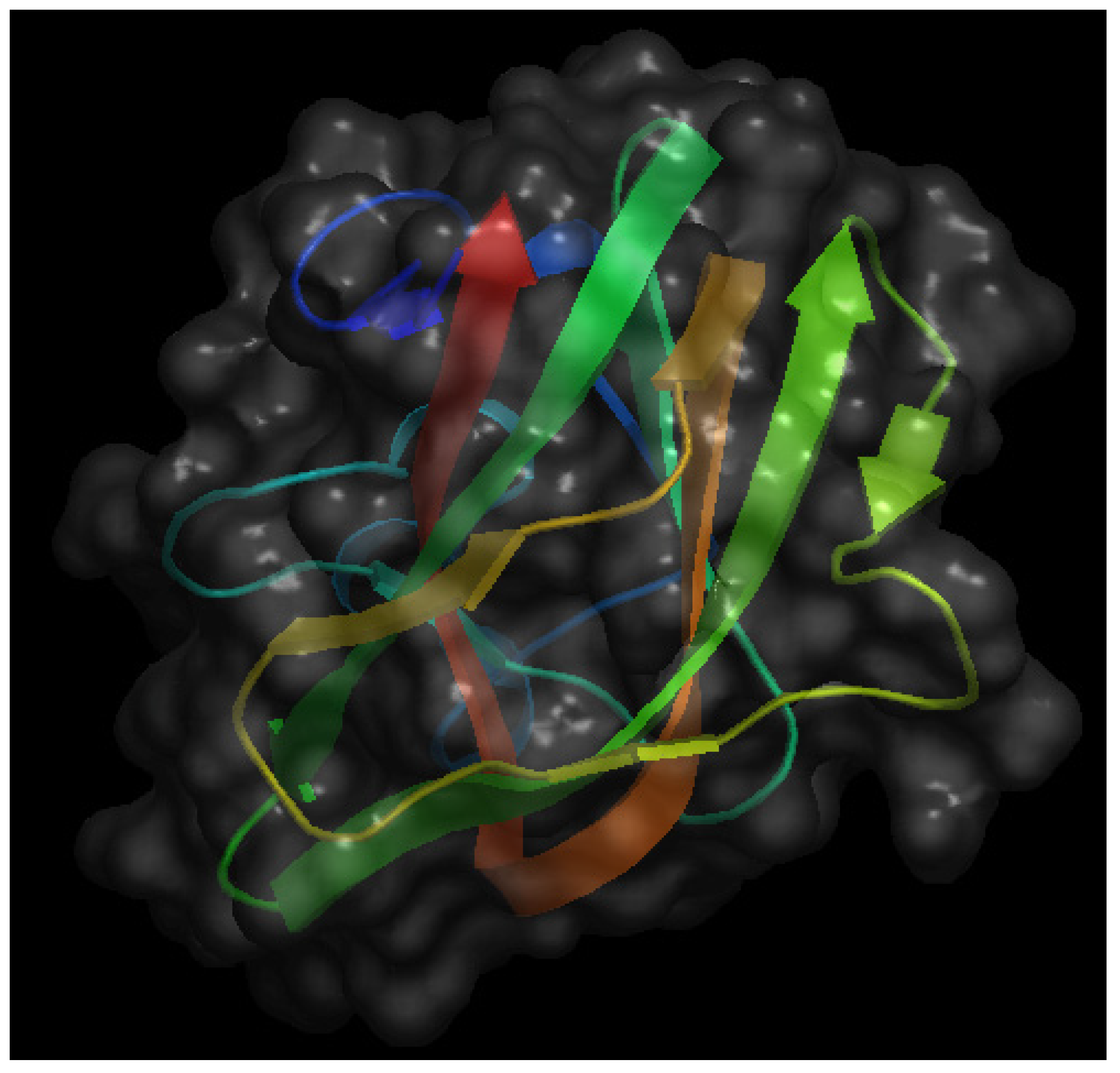
5. Lectins
6. Soy Proteins
7. High Protein Diet
8. Conclusions
Acknowledgments
Conflicts of Interest
References
- Carmeliet, P. Angiogenesis in life, disease and medicine. Nature 2005, 438, 932–936. [Google Scholar] [CrossRef]
- Medina, M.A.; Muñoz-Chápuli, R.; Quesada, A.R. Challenges of antiangiogenic cancer therapy: Trials and errors, and renewed hope. J. Cell. Mol. Med. 2007, 11, 374–382. [Google Scholar] [CrossRef]
- Quesada, A.R.; Muñoz-Chápuli, R.; Medina, M.A. Anti-angiogenic drugs: From bench to clinical trials. Med. Res. Rev. 2006, 26, 483–530. [Google Scholar] [CrossRef]
- Quesada, A.R.; Meidna, M.A.; Muñoz-Chápuli, R.; Ponce, A.L.G. Do not say ever never more: The ins and outs of antiangiogenic therapies. Curr. Pharm. Des. 2010, 16, 3932–3957. [Google Scholar] [CrossRef]
- Potente, M.; Gerhardt, H.; Carmeliet, P. Basic and therapeutic aspects of angiogenesis. Cell 2011, 146, 873–887. [Google Scholar] [CrossRef]
- Fotsis, T.; Pepper, M.; Adlercreutz, H.; Fleischmann, G.; Hase, T.; Montesano, R.; Schweigerer, L. Genistein, a dietary-derived inhibitor of in vitro angiogenesis. Proc. Natl. Acad. Sci. USA 1993, 90, 2690–2694. [Google Scholar] [CrossRef]
- Cao, Y.; Cao, R. Angiogenesis inhibited by drinking tea. Nature 1999, 398, 381. [Google Scholar] [CrossRef]
- Brakenhielm, E.; Caom, R.; Cao, Y. Suppression of angiogenesis, tumor growth, and wound healing by resveratrol, a natural compound in red wine and grapes. FASEB J. 2001, 15, 1798–1800. [Google Scholar]
- Cárdenas, C.; Quesada, A.R.; Medina, M.A. Anti-angiogenic and anti-inflammatory properties of kahweol, a coffee diterpene. PLoS One 2011, 6, e23407. [Google Scholar] [CrossRef]
- Einagar, A.Y.; Sylvester, P.W.; El Sayed, K.A. (−)-Oleocanthal as a c-Met inhibitor for the control of metastatic breast and prostate cancer. Planta Med. 2011, 77, 1013–1019. [Google Scholar] [CrossRef]
- Fortes, C.; García-Vilas, J.A.; Quesada, A.R.; Medina, M.A. Evaluation of the anti-angiogenic potential of hydroxytyrosol and tyrosol, two bio-active phenolic compounds of extra virgin olive oil, in endothelial cell cultures. Food Chem. 2012, 134, 134–140. [Google Scholar] [CrossRef]
- López-Jiménez, A.; García-caballero, M.; Medina, M.A.; Quesada, A.R. Anti-angiogenic properties of carnosol and carnosic acid, two major dietary compounds from rosemary. Eur. J. Nutr. 2013, 52, 85–95. [Google Scholar] [CrossRef]
- Fett, J.W.; Strydom, D.J.; Lobb, R.R.; Alderman, E.M.; Bethune, J.L.; Riordan, J.F.; Vallee, B.L. Isolation and characterization of angiogenin, an angiogenic protein from human carcinoma cells. Biochemistry 1985, 24, 5480–5486. [Google Scholar] [CrossRef]
- Shapiro, R.; Vallee, B.L. Identification of functional arginines in human angiogenin by site-directed mutagenesis. Biochemistry 1992, 31, 12477–12485. [Google Scholar] [CrossRef]
- Strydom, D.J.; Bond, M.D.; Vallee, B.L. An angiogenic protein from bovine serum and milk. Purification and primary structure of angiogenin-2. Eur. J. Biochem. 1997, 247, 535–544. [Google Scholar]
- Protein Data Bank in Europe. Available online: www.ebi.ac.uk/pdbe/ (accessed on 30 October 2013).
- The Protein Model Portal. Available online: www.proteinmodelportal.org (accessed on 16 December 2013).
- Yoo, Y.C.; Watanabe, S.; Watanabe, R.; Hata, K.; Shimazaki, K.; Azuma, I. Bovine lactoferrin and lactoferricin, a peptide derived from bovine lactoferrin, inhibit tumor metastasis in mice. Jpn. J. Cancer Res. 1997, 88, 184–190. [Google Scholar] [CrossRef]
- Masson, P.L.; Heremans, J.F.; Schonne, E. Lactoferrin, an iron-binding protein in neutrophilic leukocytes. J. Exp. Med. 1969, 130, 643–658. [Google Scholar] [CrossRef]
- Sánchez, L.; Calvo, M.; Brock, J.H. Biological role of lactoferrin. Arch. Dis. Child. 1992, 67, 657–661. [Google Scholar] [CrossRef]
- Arnold, R.R.; Cole, M.F.; McGhee, J.R. A bactericidal effect for human lactoferrin. Science 1977, 197, 263–265. [Google Scholar]
- Vorland, L.H. Lactoferrin: A multifunctional glycoprotein. APMIS 1999, 107, 971–981. [Google Scholar] [CrossRef]
- Kanyshova, T.G.; Buneva, V.N.; Nevinsky, G.A. Lactoferrin and its biological functions. Biochemistry 2001, 66, 1–7. [Google Scholar]
- Hayashida, K.; Kaneko, T.; Takeuchi, T.; Shimizu, H.; Ando, K.; Harada, E. Oral administration of lactoferrin inhibits inflammation and nociception in rat adjuvant-induced arthritis. J. Vet. Med. Sci. 2004, 66, 149–154. [Google Scholar] [CrossRef]
- Wolf, J.S.; Li, G.; Varadhachary, A.; Petrak, K.; Schneyer, M.; Li, D.; Ongkasuwan, J.; Zhang, X.; Taylor, R.J.; Strome, S.E.; et al. Oral lactoferrin results in T cell-dependent tumor inhibition of head and neck squamous cell carcinoma in vivo. Clin. Cancer Res. 2007, 13, 1601–1610. [Google Scholar] [CrossRef]
- Legrand, D.; Mazurier, J.A. A critical review of the roles of host lactoferrin in immunity. Biometals 2010, 23, 365–376. [Google Scholar] [CrossRef]
- Shimamura, M.; Yamamoto, Y.; Ashino, H.; Oikawa, T.; Hazato, T.; Tsuda, H. Bovine lactoferrin inhibits tumor-induced angiogenesis. Int. J. Cancer 2004, 111, 111–116. [Google Scholar] [CrossRef]
- Norrby, K.; Mattsby-Baltzer, I.; Innocenti, M.; Tuneberg, S. Orally administered bovine lactoferrin systematically inhibits VEGF165-mediated angiogenesis in the rat. Int. J. Cancer 2001, 91, 236–240. [Google Scholar]
- Norrby, K. Human apo-lactoferrin enhances angiogenesis mediated by vascular endothelial growth factor A in vivo. J. Vasc. Res. 2004, 41, 293–304. [Google Scholar] [CrossRef]
- Hwang, P.M.; Zhou, N.; Shan, X.; Arrowsmith, C.H.; Vogel, H.J. Three-dimensional solution structure of lactoferricin B, an antimicrobial peptide derived from bovine lactoferrin. Biochemsitry 1997, 37, 4288–4298. [Google Scholar]
- Mader, J.S.; Smyth, D.; Marshall, J.; Hoskin, D.W. Bovine lactoferricin inhibits basic fibroblast growth factor- and vascular endothelial growth factor165-induced angiogenesis by competing for heparin-like binding sites on endothelial cells. Am. J. Pathol. 2006, 169, 1753–1766. [Google Scholar] [CrossRef]
- Yeom, M.; Park, J.; Lee, B.; Choi, S.Y.; Kim, K.S.; Lee, H.; Hahm, D.H. Lactoferrin inhibits the inflammatory and angiogenic activation of bovine aortic endothelial cells. Inflamm. Res. 2011, 60, 475–482. [Google Scholar] [CrossRef]
- Tung, Y.T.; Chen, H.L.; Yen, C.C.; Lee, P.Y.; Tsai, H.C.; Lin, M.F.; Chen, C.M. Bovine lactoferrin inhibits lung cancer grwoth through suppression of both inflammation and expression of vascular endothelial growth factor. J. Dairy Sci. 2013, 96, 2095–2106. [Google Scholar] [CrossRef]
- Stubbs, J.D.; Lekutis, C.; Singer, K.L.; Bui, A.; Yuzuki, D.; Srinivasan, U.; Parry, G. cDNA cloning of a mouse mammary epithelial cell surface protein reveals the existence of epidermal growth factor-like domains linked to factor VIII-like sequences. Proc. Natl. Acad. Sci. USA 1990, 87, 8417–8421. [Google Scholar] [CrossRef]
- Larocca, D.; Peterson, J.A.; Urrea, R.; Kuniyoshi, J.; Bistrain, A.M.; Ceriani, R.L. A Mr 46,000 human milk fat globule protein that is highly expressed in human breast tumors contains factor VIII-like domains. Cancer Res. 1991, 51, 4994–4998. [Google Scholar]
- Raymond, A.; Ensslin, M.A.; Shur, B.D. SED1/MFG-E8: A bi-motif protein that orchestrates diverse cellular interactions. J. Cell. Biochem. 2009, 106, 957–966. [Google Scholar] [CrossRef]
- Hidai, C.; Zupancic, T.; Penta, K.; Mikhail, A.; Kawana, M.; Quertemous, E.E.; Aoka, Y.; Fukagawa, M.; Matsui, Y.; Platika, D.; et al. Cloning and characterization of developmental endothelial locus-1: An embryonic endothelial cell protein that binds the αvβ3 integrin receptor. Genes Dev. 1998, 12, 21–33. [Google Scholar] [CrossRef]
- Sylvestre, J.S.; Théry, C.; Hamard, G.; Boddaert, J.; Aguilar, B.; Delcayre, A.; Houbron, C.; Tamarat, R.; Blanc-Brude, O.; Heeneman, S.; et al. Lactadherin promotes VEGF-dependent neovascularization. Nat. Med. 2005, 11, 499–506. [Google Scholar] [CrossRef]
- Sugano, G.; Bernard-Pierrot, I.; Laé, M.; Battail, C.; Allory, Y.; Stransky, N.; Krumeich, S.; Lepage, M.L.; Maille, P.; Donnadieu, M.H.; et al. Milk fat globule-epidermal growth factor-factor VIII (MFGE8)/lactadherin promotes bladder tumor development. Oncogene 2011, 30, 642–653. [Google Scholar] [CrossRef]
- Neutzner, M.; López, T.; Feng, X.; Bergmann-Leitner, E.S.; Leitner, W.W.; Udey, M.C. MFH-E8/lactadherin promotes tumor growth in an angiogenesis-dependent transgenic mouse model of multistage carcinogenesis. Cancer Res. 2007, 67, 6777–6785. [Google Scholar]
- Fens, M.H.A.M.; Mastrobattista, E.; de Graaff, A.M.; Flesch, F.M.; Ultee, A.; Rasmussen, J.T.; Molema, G.; Storm, G.; Schiffelers, R.M. Angiogenic endothelium shows lactadherin-dependent phagocytosis of aged erythrocytes and apoptotic cells. Blood 2008, 111, 4542–4550. [Google Scholar] [CrossRef]
- Jinushi, M.; Sato, M.; Kanamoto, A.; Itoh, A.; Nagai, S.; Koyasu, S.; Dranoff, G.; Tahara, H. Milk fat globule epidermal growth factor-8 blockade triggers tumor destruction through coordinated cell-autonomous and immune-mediated mechanisms. J. Exp. Med. 2009, 206, 1317–1326. [Google Scholar] [CrossRef]
- Nyegaard, S.; Novakovic, V.A.; Rasmussen, J.T.; Gilbert, G.E. Lactadherin inhibits secretory phospholipase A2 activity on pre-apoptotic leukemia cells. PLoS One 2013, 8, e77143. [Google Scholar]
- Sharon, N. Glycioriteubs biw abd tgeb: A personal account. Acta Anat. 1998, 161, 7–17. [Google Scholar] [CrossRef]
- González de Mejía, E.; Prisecaru, V.I. Lectins as biactive plant proteins: A potential in cancer treatment. Crit. Rev. Food Sci. Nutr. 2005, 45, 425–445. [Google Scholar] [CrossRef]
- Kelsall, A.; FitzGerald, A.J.; Howard, C.V.; Evans, R.C.; Singh, R.; Rhodes, J.M.; Goodlad, R.A. Dietary lectins can stimulate pancreatic growth in the rat. Int. J. Exp. Pathol. 2002, 83, 203–208. [Google Scholar] [CrossRef]
- Park, W.B.; Lyu, S.Y.; Kim, J.H.; Choi, S.H.; Chung, H.K.; Ahn, S.H.; Hong, S.Y.; Yoon, T.J.; Choi, M.J. Inhibition of tumor growth and metastasis by Korean mistletoe lectin is associated with apoptosis and antiangiogenesis. Cancer Biother. Radiopharm. 2001, 16, 439–447. [Google Scholar] [CrossRef]
- Frigolet, M.I.; Torres, N.; Tovar, A.R. Soya protein attenuates abnormalities of the renin-angiotensin system in adipose tissue from obese rats. Br. J. Nutr. 2012, 107, 36–44. [Google Scholar] [CrossRef]
- Larsen, T.M.; Dlaskov, S.M.; van Baak, M.; Jebb, S.A.; Papadaki, A.; Pfeiffer, A.F.H.; Martínez, A.; Handjieva-Darlenska, T.; Kunesová, M.; Pihlsgard, M.; et al. Diets with high or low protein content and glycemic index for weight-loss maintenance. N. Engl. J. Med. 2010, 363, 2102–2113. [Google Scholar] [CrossRef]
- Rizkalla, S.W.; Prifti, E.; Cotillard, A.; Pelloux, V.; Rouault, C.; Allouche, R.; Laromiguière, M.; Kong, L.C.; Darakhshan, F.; Massiera, F.; et al. Differential effects of macronutrient content in 2 energy-restricted diets on cardiovascular risk factors and adipose tissue cell size in moderately obese individuals: A randomized controlled trial. Am. J. Clin. Nutr. 2012, 95, 49–63. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Medina, M.Á.; Quesada, A.R. Dietary Proteins and Angiogenesis. Nutrients 2014, 6, 371-381. https://doi.org/10.3390/nu6010371
Medina MÁ, Quesada AR. Dietary Proteins and Angiogenesis. Nutrients. 2014; 6(1):371-381. https://doi.org/10.3390/nu6010371
Chicago/Turabian StyleMedina, Miguel Ángel, and Ana R. Quesada. 2014. "Dietary Proteins and Angiogenesis" Nutrients 6, no. 1: 371-381. https://doi.org/10.3390/nu6010371




