Iron and Zinc Nutrition in the Economically-Developed World: A Review
Abstract
:1. Introduction
2. Methods
| Search terms for iron and zinc bioavailability | Search terms for interactions between dietary iron and zinc | |
|---|---|---|
| 1 | iron | iron |
| 2 | zinc | zinc |
| 3 | absorption | interaction |
| 4 | bioavailability | supplementation |
| 5 | absorption and bioavailability | iron and zinc |
| 6 | iron and absorption and bioavailability | interaction and iron and zinc |
| 7 | zinc and absorption and bioavailability | supplementation and iron and zinc |
3. Dietary Sources and Intakes of Iron and Zinc
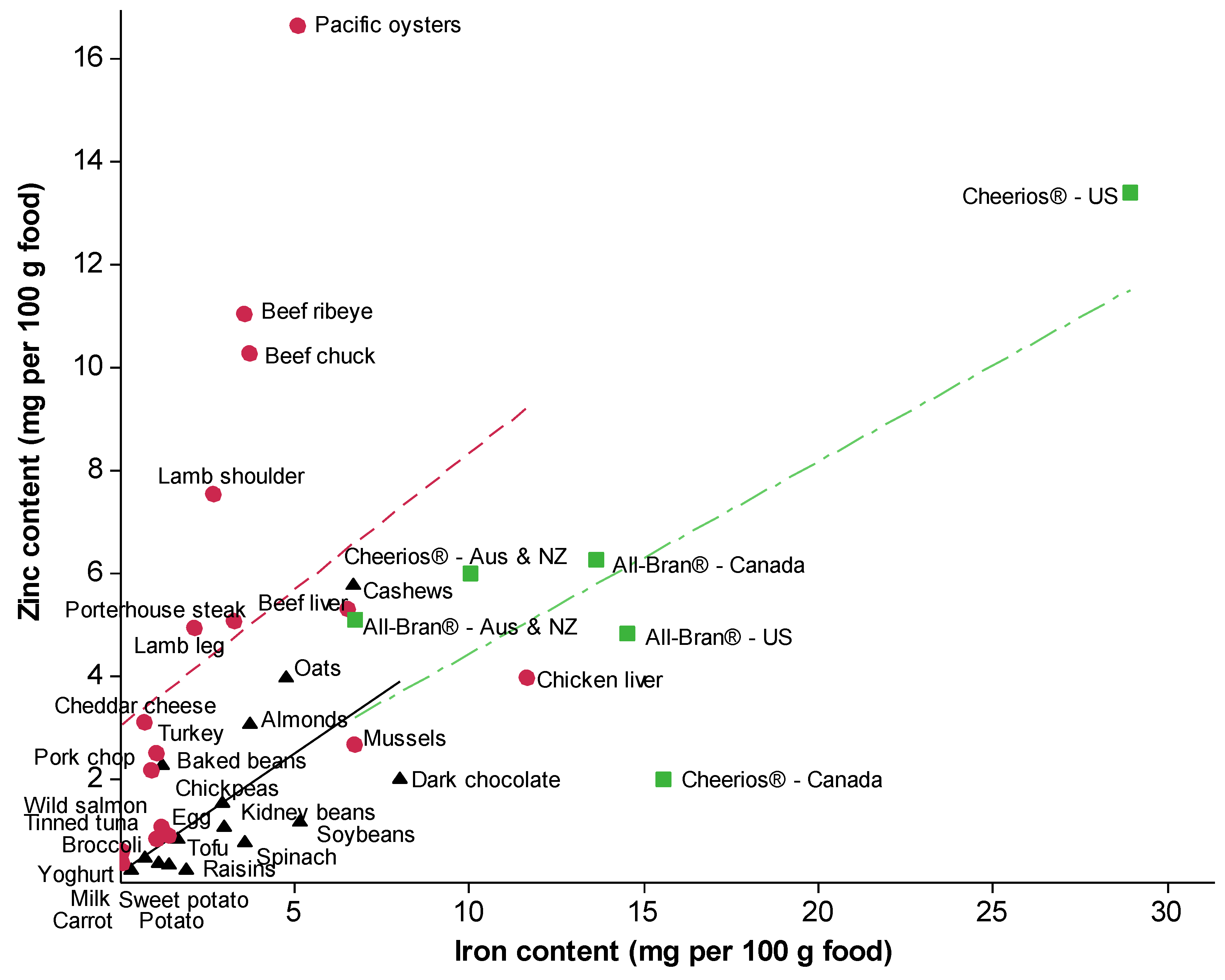
4. Recommended Intakes of Iron and Zinc
| Country | Survey | Sample | Dietary iron intake (mg/day) | Inadequate intakes (i.e., intakes < EAR) % | Top 3 Food sources | Contribution of food to iron intake % |
|---|---|---|---|---|---|---|
| Australia | 1995 National Nutrition Survey [11] | Males 19+ years (n = 5081) | Median: 15.2 a | Cereals and cereal products (includes breads, breakfast cereals, biscuits, cakes) | 41 | |
| Meat and meat products (includes poultry) | 22 | |||||
| Vegetables | 12 | |||||
| Females 19+ years (n = 5770) | Median: 11.1 a | Cereals and cereal products (includes breads, breakfast cereals, biscuits, cakes) | 38 | |||
| Meat and meat products (includes poultry) | 17 | |||||
| Vegetables | 13 | |||||
| Canada | 2004 Community Health Survey [45] | Males 19+ years (n = 8470) | Median (25%–75% IQR): 15.7 (12.9–19.1) | 0.4 (SE 0.1) | ||
| Females 19+ years (n = 10350) | Median (25%–75% IQR): 11.9 (9.9–14.3) | 19–30 years: 17 (SE 1.5) | ||||
| 31–50 years: 18 (SE 1.1) | ||||||
| 51–70 years: <3 a | ||||||
| >70 years: 2 (SE 0.4) | ||||||
| New Zealand | 2008/2009 Adult Nutrition Survey [12] | Males 15+ years (n = 2066) | Median (25%–75% IQR): 13.2 (12.8–13.6) | 1 a | Cereals and cereal products (includes breads, breakfast cereals, biscuits, cakes) | 40 |
| Meat and meat products (includes poultry) | 20 | |||||
| Vegetables | 13 | |||||
| Females 15+ years (n = 2655) | Median (25%–75% IQR): 9.9 (9.6–10.2) | 10 a | Cereals and cereal products (includes breads, breakfast cereals, biscuits, cakes) | 36 | ||
| Vegetables | 15 | |||||
| Meat and meat products (includes poultry) | 14 | |||||
| UK | Iron intake and inadequate intakes: 2008/09–2010/11 UK National Diet and Nutrition Survey [46] Food sources: 2000–2001 National Diet and Nutrition Survey [13] | Males 19–64 years (n = 346) | Median (lower 2.5 percentile-upper 2.5 percentile): 11.7 (5.7–22.3) | 1 a,b | Cereals and cereal products (includes breads, breakfast cereals, biscuits, cakes) | 44 |
| Meat and meat products (includes poultry) | 19 | |||||
| Vegetables | 9 | |||||
| Females 19–64 years (n = 461) | Median (lower 2.5 percentile-upper 2.5 percentile): 9.7 (4.0–16.3) | 22 a,b | Cereals and cereal products (includes breads, breakfast cereals, biscuits, cakes) | 45 | ||
| Meat and meat products (includes poultry) | 15 | |||||
| Vegetables | 11 | |||||
| US | Iron intake: NHANES 2009–2010 [47] | Iron intake:
Males 20+ years (n = 2789) | Mean (SE): 17.5 (0.43) | Males 19+ years <3 a | ||
| Inadequate intakes: NHANES 2001–2002 [48] | Iron intake:
Females 20+ years (n = 2973) | Mean (SE): 12.9 (0.33) | Females 19+ years 10 a | |||
| Food sources: NHANES 2003–2006 [14] | Food sources: Males and females 19+ years (n = 9490) | Cereals and cereal products (includes breads, breakfast cereals, biscuits, cakes) | 47 | |||
| Meat and meat products (includes poultry) | 10 | |||||
| Vegetables | 6 | |||||
| Country | Survey | Sample | Dietary zinc intake (mg/day) | Inadequate intakes (i.e., intakes < EAR) % | Top 3 Food sources | Contribution of food to zinc intake % |
|---|---|---|---|---|---|---|
| Australia | 1995 National Nutrition Survey [11] | Males 19+ years (n = 5081) | Median: 14.4 a | Meat and meat products (includes poultry) | 39 | |
| Cereals and cereal products (includes breads, breakfast cereals, biscuits, cakes) | 24 | |||||
| Dairy products | 12 | |||||
| Females 19+ years (n = 5770) | Median: 9.7 a | Meat and meat products (includes poultry) | 32 | |||
| Cereals and cereal products (includes breads, breakfast cereals, biscuits, cakes) | 24 | |||||
| Dairy products | 15 | |||||
| Canada | 2004 Community Health Survey [45] | Males 19+ years (n = 8470) | Median (25%–75% IQR): 11.9 (10.3–13.8) | 17 (SE 1.4) | ||
| Females 19+ years (n = 10350) | Median (25%–75% IQR): 9.5 (8.2–11.1) | 14 (SE 1.3) | ||||
| New Zealand | 2008/2009 Adult Nutrition Survey [12] | Males 15+ years (n = 2066) | Median (25%–75% IQR): 12.9 (12.4–13.4) | 39 a | Cereals and cereal products (includes breads, breakfast cereals, biscuits, cakes) | 33 |
| Meat and meat products (includes poultry) | 28 | |||||
| Dairy products | 11 | |||||
| Females 15+ years (n = 2655) | Median (25%–75% IQR): 9.0 (8.7–9.3) | 11 a | Cereals and cereal products (includes breads, breakfast cereals, biscuits, cakes) | 30 | ||
| Meat and meat products (includes poultry) | 24 | |||||
| Dairy products | 14 | |||||
| UK | Zinc intake and inadequate intakes:2008/09–2010/11 UK National Diet and Nutrition Survey [46] Food sources:2000–2001 National Diet and Nutrition Survey [13] | Males 19–64 years (n = 346) | Median (lower 2.5 percentile-upper 2.5 percentile): 9.6 (4.7–20.1) | 8 a,b | Meat and meat products (includes poultry) | 36 |
| Cereals and cereal products (includes breads, breakfast cereals, biscuits, cakes) | 25 | |||||
| Dairy products | 16 | |||||
| Females 19–64 years (n = 461) | Median (lower 2.5 percentile-upper 2.5 percentile): 7.6 (3.7–13.1) | 3 a,b | Meat and meat products (includes poultry) | 30 | ||
| Cereals and cereal products (includes breads, breakfast cereals, biscuits, cakes) | 26 | |||||
| Dairy products | 19 | |||||
| US | Zinc intake: NHANES 2009–2010 [47] Inadequate intakes: NHANES 2001–2002 [48] Food sources: NHANES 2003–2006 [14] | Zinc intake:Males 20+ years (n = 2789) | Mean (SE): 14.2 (0.22) | Males 19+ years 11 (SE 1.0) | ||
| Zinc intake:Females 20+ years (n = 2973) | Mean (SE): 9.8 (0.17) | Females 19+ years 17 (SE 1.6) | ||||
| Food sources: Males and females 19+ years (n = 9490) | Meat and meat products (includes poultry) | 35 | ||||
| Cereals and cereal products (includes breads, breakfast cereals, biscuits, cakes) | 15 | |||||
| Dairy products | 13 | |||||
| Australia and New Zealand [49] | UK [50] | US and Canada [43] | |||||
|---|---|---|---|---|---|---|---|
| EAR (mg/day) | RDI (mg/day) | EAR (mg/day) | RNI (mg/day) | EAR (mg/day) | RDA (mg/day) | ||
| Iron | Males 19–50 years | 6 | 8 | 6.7 | 8.7 | 6 | 8 |
| Males 51+ years | 6 | 8 | 6.7 | 8.7 | 6 | 8 | |
| Females 19–50 years a | 8 | 18 | 11.4 | 14.8 | 8.1 | 18 | |
| Females 51+ years | 5 | 8 | 6.7 | 8.7 | 5 | 8 | |
| Zinc | Males 19–50 years | 12 | 14 | 7.3 | 9.5 | 9.4 | 11 |
| Males 51+ years | 12 | 14 | 7.3 | 9.5 | 9.4 | 11 | |
| Females 19–50 years a | 6.5 | 8 | 5.5 | 7.0 | 6.8 | 8 | |
| Females 51+ years | 6.5 | 8 | 5.5 | 7.0 | 6.8 | 8 | |
5. Bioavailability
6. Inhibitors and Enhancers of Absorption
6.1. Phytate
6.2. Polyphenols in Beverages
6.3. Animal Tissue
6.4. Soy Protein
6.5. Calcium
6.6. Ascorbic Acid
7. Iron and Zinc Losses
| Main routes of iron loss | Main routes of zinc loss |
|---|---|
| Urine (<1 mg/day) | Endogenous intestinal losses (0.5 to 3 mg/day) |
| Feces (from occult blood loss and shed enterocytes; 0.6 mg/day) | Urine (0.5 to 0.7 mg/day) |
| Menstrual blood (0.4 to 0.5 mg/day) | Skin cells (0.5 to 0.7 mg/day) |
| Skin cells and sweat (0.2 to 0.3 mg/day) | Semen (1 mg/ejaculate) |
8. Iron and Zinc Status
8.1. Iron Status
| Country | Survey | Sample | Ferritin (mean (95% CI) µg/L) | Low ferritin % |
|---|---|---|---|---|
| Canada | 2009–2011 Canadian Health Measures Survey [115] | Males 20–49 years a | 164 (152–177) b | 1 c |
| Males 50–64 years a | 166 (148–187) b | 1 c | ||
| Males 65–79 years a | 155 (138–175) b | 0 c | ||
| Females 20–49 years d | 41 (38–44) b | 9 c | ||
| Females 50–64 years d | 85 (73–98) b | 6 c | ||
| Females 65–79 years d | 89 (74–107) b | 6 c | ||
| New Zealand | 2008/09 New Zealand Adult Nutrition Survey [12] | Males 15+ years (n = 1470) | 177 (165–189) e | 2 f |
| Females 15+ years (n = 1878) | 79 (75–84) e | 8 f | ||
| UK | 2008/09–2010/11 UK National Diet and Nutrition Survey [116] | Males 19–64 years (n = 160) | 155 e,g | |
| Females 19–64 years (n = 231) | 53 e,g | |||
| US | NHANES 2003–2006 [117] | Females 20–39 years h (n = 1780) | 36.5 (33.5–39.9) b | Females 20–49 years (n = 2539):13 c |
| Females 40–49 years h (n = 759) | 40.0 (34.5–46.3) b |
8.2. Zinc Status
| Country | Survey | Sample | Mean zinc (μmol/L) | 2.5th Percentile (μmol/L) |
|---|---|---|---|---|
| US | NHANES II1976–1980 [121,122] | Males 10–64 years AM fasting (n = 1767) | 15.0 a | 11.3 |
| Males 10–64 years AM non-fasting (n = 1750) | 14.4 a | 10.7 | ||
| Males 10–64 years PM (n = 2765) | 12.5 a | 9.3 | ||
| Females 10–70+ years AM fasting (n = 1576) | 14.1 a | 10.7 | ||
| Females 10–70+ years AM non-fasting (n = 1658) | 12.5 a | 10.1 | ||
| Females 10–70+ years PM (n = 2343) | 11.6 a | 9.0 | ||
| UK | 2008/09–2010/11 UK National Diet and Nutrition Survey [116] | Males 19–64 years AM fasting (n = 160) | 15.8 b | 10.3 |
| Females 19–64 years AM fasting (n = 231) | 15.5 b | 10.0 |
8.3. Correlation between Iron and Zinc Status
9. Effect of Iron Intake on Zinc Status and Zinc Absorption
10. Effect of Zinc Intake on Iron Status and Iron Absorption
11. Conclusions
Acknowledgments
Conflict of Interest
References
- King, J.; Cousins, R. Zinc. In Modern Nutrition in Health and Disease, 10th ed.; Shils, M., Ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2006; pp. 271–285. [Google Scholar]
- Wood, R.; Ronnenberg, A. Iron. In Modern Nutrition in Health and Disease, 10th ed.; Shils, M., Ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2006; pp. 248–270. [Google Scholar]
- Kordas, K.; Stoltzfus, R.J. New evidence of iron and zinc interplay at the enterocyte and neural tissues. J. Nutr. 2004, 134, 1295–1298. [Google Scholar]
- Sandstead, H.H. Causes of iron and zinc deficiencies and their effects on brain. J. Nutr. 2000, 130, 347S–349S. [Google Scholar]
- Hunt, J.R. Moving toward a plant-based diet: Are iron and zinc at risk? Nutr. Rev. 2002, 60, 127–134. [Google Scholar]
- Donovan, U.M.; Gibson, R.S. Iron and zinc status of young women aged 14 to 19 years consuming vegetarian and omnivorous diets. J. Am. Coll. Nutr. 1995, 14, 463–472. [Google Scholar]
- Gibson, R.; Heath, A.L.; Prosser, N.; Parnell, W.; Donovan, U.; Green, T.; McLaughlin, K.; O’Connor, D.; Bettger, W.; Skeaff, C. Are Young Women with Low Iron Stores at Risk of Zinc as Well as Iron Deficiency? In Trace Elements in Man and Animals 10; Roussel, A.M., Favier, A.E., Anderson, R.A., Eds.; Kluwer Academic Publishers: New York, NY, USA, 1999; pp. 323–328. [Google Scholar]
- Hunt, J.R. Bioavailability of iron, zinc, and other trace minerals from vegetarian diets. Am. J. Clin. Nutr. 2003, 78, 633S–639S. [Google Scholar]
- Nair, K.M.; Brahmam, G.N.; Radhika, M.S.; Dripta, R.C.; Ravinder, P.; Balakrishna, N.; Chen, Z.; Hawthorne, K.M.; Abrams, S.A. Inclusion of guava enhances non-heme iron bioavailability but not fractional zinc absorption from a rice-based meal in adolescents. J. Nutr. 2013, 143, 852–858. [Google Scholar]
- Minitab, Minitab 15; State College: Pennsylvania, PA, USA, 2007.
- McLennan, W.; Podger, A. National Nutrition Survey: Nutrient Intakes and Physical Measurements, Australia, 1995; Australian Bureau of Statistics: Canberra, Australia, 1998. [Google Scholar]
- University of Otago and Ministry of Health, A Focus on Nutrition: Key Findings of the 2008/09 New Zealand Adult Nutrition Survey; Ministry of Health: Wellington, New Zealand, 2011.
- Henderson, L.; Irving, K.; Gregory, J.; Bates, C.; Prentice, A.; Perks, J.; Swan, G.; Farron, M. The National Diet and Nutrition Survey: Adults Aged 19 to 64 Years—Vitamin and Mineral Intake and Urinary Analytes; The Stationery Office: London, UK, 2003. [Google Scholar]
- O’Neil, C.E.; Keast, D.R.; Fulgoni, V.L.; Nicklas, T.A. Food sources of energy and nutrients among adults in the US: NHANES 2003–2006. Nutrients 2012, 4, 2097–2120. [Google Scholar]
- Kellogg Canada All-Bran® Original Cereal, 2012. Available online: http://www.all-bran.ca/en/cereal/All-Bran_Original_cereal.aspx (accessed on 24 May 2013).
- Kellogg Company Kellogg (Aust) All-Bran® Original Cereal, 2012. Available online: http://www.all-bran.com.au/products/all-bran-original.aspx (accessed on 24 May 2013).
- Kellogg NA Co Kellogg’s® All-Bran® Original Cereal, 2013. Available online: http://www.all-bran.com/products/original-cereal.aspx (accessed on 24 May 2013).
- General Mills Cheerios®, 2013. Available online: http://www.cheerios.com/Products/Cheerios#.UZ63DrT4V8E (accessed on 24 May 2013).
- General Mills (Canada) Cheerios®, 2012. Available online: http://www.generalmillsfoodservice.ca/en/pdf/pos/CHEERIOS_ENG.pdf (accessed on 24 May 2013).
- US Department of Agriculture, Agricultural Research Service; USDA National Nutrient Database for Standard Reference; Release 25; 2012. Available online: http://www.ars.usda.gov/ba/bhnrc/ndl (accessed 20 April 2013).
- Foster, M.; Chu, A.; Petocz, P.; Samman, S. Effect of vegetarian diets on zinc status: A systematic review and meta-analysis of studies in humans. J. Sci. Food Agric. 2013, 93, 2362–2371. [Google Scholar]
- Ball, M.J.; Bartlett, M.A. Dietary intake and iron status of Australian vegetarian women. Am. J. Clin. Nutr. 1999, 70, 353–358. [Google Scholar]
- Reddy, S.; Sanders, T.A. Haematological studies on pre-menopausal Indian and Caucasian vegetarians compared with Caucasian omnivores. Br. J. Nutr. 1990, 64, 331–338. [Google Scholar]
- Worthington-Roberts, B.S.; Breskin, M.W.; Monsen, E.R. Iron status of premenopausal women in a university community and its relationship to habitual dietary sources of protein. Am. J. Clin. Nutr. 1988, 47, 275–279. [Google Scholar]
- Deriemaeker, P.; Aerenhouts, D.; de Ridder, D.; Hebbelinck, M.; Clarys, P. Health aspects, nutrition and physical characteristics in matched samples of institutionalized vegetarian and non-vegetarian elderly (>65years). Nutr. Metab. 2011, 8, 37. [Google Scholar]
- Li, D.; Sinclair, A.J.; Mann, N.J.; Turner, A.; Ball, M.J. Selected micronutrient intake and status in men with differing meat intakes, vegetarians and vegans. Asia Pac. J. Clin. Nutr. 2000, 9, 18–23. [Google Scholar]
- American Dietetic Association. Dietitians of Canada. Position of the American Dietetic Association and Dietitians of Canada: Vegetarian diets. J. Am. Diet Assoc. 2003, 103, 748–765.
- Allen, L.; de Benoist, B.; Dary, O.; Hurrell, R. Guidelines on Food Fortification with Micronutrients; World Health Organisation/Food and Agricultural Organisation: Geneva, Switzerland, 2006. [Google Scholar]
- Institute of Medicine, Food and Nutrition Board, Dietary Reference Intakes: Guiding Principles for Nutrition Labeling and Fortification; National Academies Press: Washington, DC, USA, 2003.
- Scientific Advisory Committee on Nutrition, Iron and Health; The Stationery Office: London, UK, 2010.
- Flour Fortification Initiative. Flour Fortification Initiative: Country Profiles, 2012. Available online: http://www.ffinetwork.org/country_profiles/index.php (accessed on 1 March 2013).
- Brown, K.H.; Hambidge, K.M.; Ranum, P. Zinc fortification of cereal flours: Current recommendations and research needs. Food Nutr. Bull. 2010, 31, S62–S74. [Google Scholar]
- Food Standards Australia New Zealand, Australia New Zealand Food Standards Code—Standard 1.3.2: Vitamins and Minerals, 2013. Available online: http://www.comlaw.gov.au/Details/F2013C00099 (accessed on 26 April 2013).
- Health Canada. Food and Drug Regulations, 2013. Available online: http://laws-lois.justice.gc.ca/PDF/C.R.C.,_c._870.pdf (accessed on 26 April 2013).
- Commission of the European Communities, Regulation (EC). No. 1925/2006 of the European Parliament and of the Council of 20 December 2006 on the addition of vitamins and minerals and of certain other substances to foods. Off. J. Eur. Union 2006, 404, 26–38.
- Schwitters, B.; Achanta, G.; van der Vlies, D.; Bast, A.; Hanekamp, J.C. The European regulation of food supplements and food fortification. Environ. Law. Manag. 2007, 19, 19–29. [Google Scholar]
- Food Standards Agency, Safe Upper Levels for Vitamins and Minerals. Report of Expert Group on Vitamins and Minerals, 2003. Available online: http://www.food.gov.uk/multimedia/pdfs/vitmin2003.pdf (accessed on 26 April 2013).
- UK Parliament, Food Safety Act, 1990. Available online: http://www.legislation.gov.uk/ukpga/1990/16/contents (accessed on 26 April 2013).
- Kohgo, Y.; Ikuta, K.; Ohtake, T.; Torimoto, Y.; Kato, J. Body iron metabolism and pathophysiology of iron overload. Int. J. Hematol. 2008, 88, 7–15. [Google Scholar]
- Fosmire, G.J. Zinc toxicity. Am. J. Clin. Nutr. 1990, 51, 225–227. [Google Scholar]
- Lawrence, M. Food Fortification: The Evidence, Ethics, and Politics of Adding Nutrients to Food; Oxford University Press: Oxford, UK, 2013. [Google Scholar]
- Lawrence, M.; Robertson, A. Reference Standards and Guidelines. In Public Health Nutrition—From Principle to Practice; Lawrence, M., Worsley, T., Eds.; Allen & Unwin: Crows Nest, Australia, 2007; pp. 39–70. [Google Scholar]
- Institute of Medicine, Food and Nutrition Board, Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; National Academies Press: Washington, DC, USA, 2001.
- Dhonukshe-Rutten, R.A.; Timotijevic, L.; Cavelaars, A.E.; Raats, M.M.; de Wit, L.S.; Doets, E.L.; Tabacchi, G.; Roman, B.; Ngo-de la Cruz, J.; Gurinovic, M.; et al. European micronutrient recommendations aligned: A general framework developed by EURRECA. Eur. J. Clin. Nutr. 2010, 64, S2–S10. [Google Scholar]
- Statistics Canada, Canadian Community Health Survey Cycle 2.2, Nutrition (2004); Minister of Health Canada: Ottawa, Canada, 2009.
- UK Department of Health, National Diet and Nutrition Survey: Headline Results from Years 1 and 2 (Combined) of the Rolling Programme, 2008/09–2009/10; Headline Results from Years 1 and 2 (Combined) Tables, 2011. Available online: https://www.gov.uk/government/ uploads/system/uploads/attachment_data/file/152237/dh_128556.pdf (accessed on 27 April 2013).
- US Department of Agriculture, Agricultural Research Service; 2009–2010 What We Eat In America, NHANES Tables 1–40, 2010. Available online: http://www.ars.usda.gov/SP2UserFiles/Place/12355000/pdf/0910/tables_1–40_2009–2010.pdf (accessed on 29 April 2013).
- Moshfegh, A.; Goldman, J.; Cleveland, L. What We Eat in America, NHANES 2001–2002: Usual Nutrient Intakes From Food Compared to Dietary Reference Intakes; US Department of Agriculture, Agricultural Research Service: Washington, DC, USA, 2005. [Google Scholar]
- National Health and Medical Research Council, Nutrient Reference Values for Australia and New Zealand Including Recommended Dietary Intakes; National Health and Medical Research Council: Canberra, Australia, 2006.
- Department of Health, Dietary Reference Values for Food Energy and Nutrients for the United Kingdom; Her Majesty’s Stationary Office: London, UK, 1991.
- Gibson, R.S. Principles of Nutritional Assessment, 2nd ed.; Oxford University Press: New York, NY, USA, 2005. [Google Scholar]
- Knutson, M.D. Iron-sensing proteins that regulate hepcidin and enteric iron absorption. Annu. Rev. Nutr. 2010, 30, 149–171. [Google Scholar]
- Conrad, M.E.; Umbreit, J.N. Iron absorption and transport—An update. Am. J. Hematol. 2000, 64, 287–298. [Google Scholar]
- Gibson, R.S. The role of diet- and host-related factors in nutrient bioavailability and thus in nutrient-based dietary requirement estimates. Food Nutr. Bull. 2007, 28, S77–S100. [Google Scholar]
- Solomons, N.W.; Jacob, R.A.; Pineda, O.; Viteri, F. Studies on the bioavailability of zinc in man. II. Absorption of zinc from organic and inorganic sources. J. Lab. Clin. Med. 1979, 94, 335–343. [Google Scholar]
- Sandström, B. Bioavailability of zinc. Eur. J. Clin. Nutr. 1997, 51, S17–S19. [Google Scholar]
- Lönnerdal, B. Dietary factors influencing zinc absorption. J. Nutr. 2000, 130, 1378S–1383S. [Google Scholar]
- Hurrell, R.; Egli, I. Iron bioavailability and dietary reference values. Am. J. Clin. Nutr. 2010, 91, 1461S–1467S. [Google Scholar] [CrossRef]
- Layrisse, M.; Martínez-Torres, C. Model for measuring dietary absorption of heme iron: Test with a complete meal. Am. J. Clin. Nutr. 1972, 25, 401–411. [Google Scholar]
- Cook, J.D.; Layrisse, M.; Martinez-Torres, C.; Walker, R.; Monsen, E.; Finch, C.A. Food iron absorption measured by an extrinsic tag. J. Clin. Investig. 1972, 51, 805–815. [Google Scholar]
- Fairweather-Tait, S.J.; Dainty, J. Use of stable isotopes to assess the bioavailability of trace elements: A review. Food Addit. Contam. 2002, 19, 939–947. [Google Scholar]
- Cosgrove, D. The chemistry and biochemistry of inositol phosphates. Rev. Pure Appl. Chem. 1966, 16, 209–224. [Google Scholar]
- Reddy, N.; Sathe, S. Introduction. In Food Phytates; Reddy, N.R., Sathe, S., Eds.; CRC Press: Boca Raton, FL, USA, 2001; pp. 1–6. [Google Scholar]
- Shears, S.; Turner, B. Nomenclature and Terminology of Inositol Phosphates: Clarification and a Glossary of Terms. In Inositol Phosphates. Linking Agriculture and the Environment; Turner, B., Richardson, A., Mullaney, E., Eds.; CAB International: Wallingford, England, UK, 2007; pp. 1–6. [Google Scholar]
- Hallberg, L.; Brune, M.; Rossander, L. Iron absorption in man: Ascorbic acid and dose-dependent inhibition by phytate. Am. J. Clin. Nutr. 1989, 49, 140–144. [Google Scholar]
- Hallberg, L.; Rossander, L.; Skanberg, A. Phytates and the inhibitory effect of bran on iron absorption in man. Am. J. Clin. Nutr. 1987, 45, 988–996. [Google Scholar]
- Hurrell, R.F.; Reddy, M.B.; Juillerat, M.A.; Cook, J.D. Degradation of phytic acid in cereal porridges improves iron absorption by human subjects. Am. J. Clin. Nutr. 2003, 77, 1213–1219. [Google Scholar]
- Larsson, M.; Rossander-Hulthen, L.; Sandstrom, B.; Sandberg, A.S. Improved zinc and iron absorption from breakfast meals containing malted oats with reduced phytate content. Br. J. Nutr. 1996, 76, 677–688. [Google Scholar]
- Sandberg, A.S.; Brune, M.; Carlsson, N.G.; Hallberg, L.; Skoglund, E.; Rossander-Hulthen, L. Inositol phosphates with different numbers of phosphate groups influence iron absorption in humans. Am. J. Clin. Nutr. 1999, 70, 240–246. [Google Scholar]
- Fredlund, K.; Isaksson, M.; Rossander-Hulthen, L.; Almgren, A.; Sandberg, A.S. Absorption of zinc and retention of calcium: Dose-dependent inhibition by phytate. J. Trace Elem. Med. Biol. 2006, 20, 49–57. [Google Scholar]
- Reddy, N.R. Occurrence, Distribution, Content, and Dietary Intake of Phytate. In Food Phytates; Reddy, N.R., Sathe, S.K., Eds.; CRC Press: Boca Raton, FL, USA, 2001; pp. 25–52. [Google Scholar]
- Weaver, C.; Kannan, S. Phytate and Mineral Bioavailability. In Food Phytates; Reddy, N.R., Sathe, S.K., Eds.; CRC Press: Boca Raton, USA, 2001; pp. 211–219. [Google Scholar]
- Bach Kristensen, M.; Tetens, I.; Alstrup Jørgensen, A.B.; Dal Thomsen, A.; Milman, N.; Hels, O.; Sandström, B.; Hansen, M. A decrease in iron status in young healthy women after long-term daily consumption of the recommended intake of fibre-rich wheat bread. Eur. J. Nutr. 2005, 44, 334–340. [Google Scholar]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar]
- Disler, P.B.; Lynch, S.R.; Charlton, R.W.; Torrance, J.D.; Bothwell, T.H.; Walker, R.B.; Mayet, F. The effect of tea on iron absorption. Gut 1975, 16, 193–200. [Google Scholar]
- Hallberg, L.; Rossander, L. Effect of different drinks on the absorption of non-heme iron from composite meals. Hum. Nutr. Appl. Nutr. 1982, 36, 116–123. [Google Scholar]
- Hurrell, R.F.; Reddy, M.; Cook, J.D. Inhibition of non-haem iron absorption in man by polyphenolic-containing beverages. Br. J. Nutr. 1999, 81, 289–295. [Google Scholar]
- Morck, T.A.; Lynch, S.R.; Cook, J.D. Inhibition of food iron absorption by coffee. Am. J. Clin. Nutr. 1983, 37, 416–420. [Google Scholar]
- Thankachan, P.; Walczyk, T.; Muthayya, S.; Kurpad, A.V.; Hurrell, R.F. Iron absorption in young Indian women: The interaction of iron status with the influence of tea and ascorbic acid. Am. J. Clin. Nutr. 2008, 87, 881–886. [Google Scholar]
- Kim, E.Y.; Ham, S.K.; Shigenaga, M.K.; Han, O. Bioactive dietary polyphenolic compounds reduce nonheme iron transport across human intestinal cell monolayers. J. Nutr. 2008, 138, 1647–1651. [Google Scholar]
- Peterson, J.; Dwyer, J.; Bhagwat, S.; Haytowitz, D.; Holden, J.; Eldridge, A.; Beecher, G.; Aladesanmi, J. Major flavonoids in dry tea. J. Food Compost. Anal. 2005, 18, 487–501. [Google Scholar]
- Ganji, V.; Kies, C.V. Zinc bioavailability and tea consumption: Studies in healthy humans consuming self-selected and laboratory-controlled diets. Plant Foods Hum. Nutr. 1994, 46, 267–276. [Google Scholar]
- Bjorn-Rasmussen, E.; Hallberg, L. Effect of animal proteins on the absorption of food iron in man. Nutr. Metab. 1979, 23, 192–202. [Google Scholar]
- Cook, J.D.; Monsen, E.R. Food iron absorption in human subjects. III. Comparison of the effect of animal proteins on nonheme iron absorption. Am. J. Clin. Nutr. 1976, 29, 859–867. [Google Scholar]
- Hurrell, R.F.; Lynch, S.R.; Trinidad, T.P.; Dassenko, S.A.; Cook, J.D. Iron absorption in humans as influenced by bovine milk proteins. Am. J. Clin. Nutr. 1989, 49, 546–552. [Google Scholar]
- Navas-Carretero, S.; Perez-Granados, A.M.; Sarria, B.; Carbajal, A.; Pedrosa, M.M.; Roe, M.A.; Fairweather-Tait, S.J.; Vaquero, M.P. Oily fish increases iron bioavailability of a phytate rich meal in young iron deficient women. J. Am. Coll. Nutr. 2008, 27, 96–101. [Google Scholar]
- Bæch, S.B.; Hansen, M.; Bukhave, K.; Jensen, M.; Sørensen, S.S.; Kristensen, L.; Purslow, P.P.; Skibsted, L.H.; Sandström, B. Nonheme-iron absorption from a phytate-rich meal is increased by the addition of small amounts of pork meat. Am. J. Clin. Nutr. 2003, 77, 173–179. [Google Scholar]
- Reddy, M.B.; Hurrell, R.F.; Cook, J.D. Meat consumption in a varied diet marginally influences nonheme iron absorption in normal individuals. J. Nutr. 2006, 136, 576–581. [Google Scholar]
- Sandström, B.; Almgren, A.; Kivisto, B.; Cederblad, A. Effect of protein level and protein source on zinc absorption in humans. J. Nutr. 1989, 119, 48–53. [Google Scholar]
- Hurrell, R.F.; Juillerat, M.A.; Reddy, M.B.; Lynch, S.R.; Dassenko, S.A.; Cook, J.D. Soy protein, phytate, and iron absorption in humans. Am. J. Clin. Nutr. 1992, 56, 573–578. [Google Scholar]
- Lönnerdal, B.; Cederblad, A.; Davidsson, L.; Sandstrom, B. The effect of individual components of soy formula and cows’ milk formula on zinc bioavailability. Am. J. Clin. Nutr. 1984, 40, 1064–1070. [Google Scholar]
- Cook, J.D.; Morck, T.A.; Lynch, S.R. The inhibitory effect of soy products on nonheme iron absorption in man. Am. J. Clin. Nutr. 1981, 34, 2622–2629. [Google Scholar]
- Davidsson, L.; Almgren, A.; Sandstrom, B.; Juillerat, M.; Hurrell, R.F. Zinc absorption in adult humans: The effect of protein sources added to liquid test meals. Br. J. Nutr. 1996, 75, 607–613. [Google Scholar]
- Hallberg, L.; Brune, M.; Erlandsson, M.; Sandberg, A.; Rossander-Hulten, L. Calcium: Effect of different amounts on nonheme- and heme-iron absorption in humans. Am. J. Clin. Nutr. 1991, 53, 112–119. [Google Scholar]
- Galan, P.; Cherouvrier, F.; Preziosi, P.; Hercberg, S. Effects of the increasing consumption of dairy products upon iron absorption. Eur. J. Clin. Nutr. 1991, 45, 553–559. [Google Scholar]
- Gleerup, A.; Rossander-Hulthen, L.; Gramatkovski, E.; Hallberg, L. Iron absorption from the whole diet: Comparison of the effect of two different distributions of daily calcium intake. Am. J. Clin. Nutr. 1995, 61, 97–104. [Google Scholar]
- Reddy, M.; Cook, J. Effect of calcium intake on nonheme-iron absorption from a complete diet. Am. J. Clin. Nutr. 1997, 65, 1820–1825. [Google Scholar]
- Minihane, A.M.; Fairweather-Tait, S.J. Effect of calcium supplementation on daily nonheme-iron absorption and long-term iron status. Am. J. Clin. Nutr. 1998, 68, 96–102. [Google Scholar]
- Cook, J.D.; Dassenko, S.A.; Whittaker, P. Calcium supplementation: Effect on iron absorption. Am. J. Clin. Nutr. 1991, 53, 106–111. [Google Scholar]
- Hallberg, L.; Rossander-Hulthen, L.; Brune, M.; Gleerup, A. Inhibition of haem-iron absorption in man by calcium. Br. J. Nutr. 1993, 69, 533–540. [Google Scholar]
- Sokoll, L.J.; Dawson-Hughes, B. Calcium supplementation and plasma ferritin concentrations in premenopausal women. Am. J. Clin. Nutr. 1992, 56, 1045–1048. [Google Scholar]
- Dawson-Hughes, B.; Seligson, F.H.; Hughes, V.A. Effects of calcium carbonate and hydroxyapatite on zinc and iron retention in postmenopausal women. Am. J. Clin. Nutr. 1986, 44, 83–88. [Google Scholar]
- Spencer, H.; Kramer, L.; Norris, C.; Osis, D. Effect of calcium and phosphorus on zinc metabolism in man. Am. J. Clin. Nutr. 1984, 40, 1213–1218. [Google Scholar]
- McKenna, A.A.; Ilich, J.Z.; Andon, M.B.; Wang, C.; Matkovic, V. Zinc balance in adolescent females consuming a low- or high-calcium diet. Am. J. Clin. Nutr. 1997, 65, 1460–1464. [Google Scholar]
- Hallberg, L.; Brune, M.; Rossander, L. Effect of ascorbic acid on iron absorption from different types of meals: Studies with ascorbic-acid-rich foods and synthetic ascorbic acid given in different amounts with different meals. Hum. Nutr. Appl. Nutr. 1986, 40, 97–113. [Google Scholar]
- Diaz, M.; Rosado, J.L.; Allen, L.H.; Abrams, S.; Garcia, O.P. The efficacy of a local ascorbic acid-rich food in improving iron absorption from Mexican diets: A field study using stable isotopes. Am. J. Clin. Nutr. 2003, 78, 436–440. [Google Scholar]
- Cook, J.D.; Reddy, M.B. Effect of ascorbic acid intake on nonheme-iron absorption from a complete diet. Am. J. Clin. Nutr. 2001, 73, 93–98. [Google Scholar]
- Beck, K.; Conlon, C.A.; Kruger, R.; Coad, J.; Stonehouse, W. Gold kiwifruit consumed with an iron-fortified breakfast cereal meal improves iron status in women with low iron stores: A 16-week randomised controlled trial. Br. J. Nutr. 2011, 105, 101–109. [Google Scholar]
- Garcia, O.P.; Diaz, M.; Rosado, J.L.; Allen, L.H. Ascorbic acid from lime juice does not improve the iron status of iron-deficient women in rural Mexico. Am. J. Clin. Nutr. 2003, 78, 267–273. [Google Scholar]
- Gillooly, M.; Bothwell, T.H.; Torrance, J.D.; MacPhail, A.P.; Derman, D.P.; Bezwoda, W.R.; Mills, W.; Charlton, R.W.; Mayet, F. The effects of organic acids, phytates and polyphenols on the absorption of iron from vegetables. Br. J. Nutr. 1983, 49, 331–342. [Google Scholar]
- Sandström, B.; Cederblad, A. Effect of ascorbic acid on the absorption of zinc and calcium in man. Int. J. Vitam. Nutr. Res. 1987, 57, 87–90. [Google Scholar]
- Nemeth, E.; Ganz, T. Regulation of iron metabolism by hepcidin. Annu. Rev. Nutr. 2006, 26, 323–342. [Google Scholar] [CrossRef]
- Brown, K.H.; Rivera, J.A.; Bhutta, Z.; Gibson, R.S.; King, J.C.; Lonnerdal, B.; Ruel, M.T.; Sandstrom, B.; Wasantwisut, E.; Hotz, C. International Zinc Nutrition Consultative Group (IZiNCG) technical document #1. Assessment of the risk of zinc deficiency in populations and options for its control. Food Nutr. Bull. 2004, 25, S99–S203. [Google Scholar]
- Sian, L.; Mingyan, X.; Miller, L.V.; Tong, L.; Krebs, N.F.; Hambidge, K.M. Zinc absorption and intestinal losses of endogenous zinc in young Chinese women with marginal zinc intakes. Am. J. Clin. Nutr. 1996, 63, 348–353. [Google Scholar]
- Cooper, M.; Greene-Finestone, L.; Lowell, H.; Levesque, J.; Robinson, S. Iron Sufficiency of Canadians; Minister of Industry: Ottawa, Canada, 2012. [Google Scholar]
- UK Department of Health, National Diet and Nutrition Survey: Headline Results from Years 1 and 2 (combined) of the Rolling Programme, 2008/09–2009/10; Supplementary Report: Blood Analytes, 2011. Available online: https://www.gov.uk/government/publications/national-diet-and-nutrition-survey-headline-results-from-years-1-and-2-combined-of-the-rolling-programme-2008-09–2009-10-supplementary-report-blood-analytes (accessed on 27 April 2013).
- US Centers for Disease Control and Prevention, Second National Report on Biochemical Indicators of Diet and Nutrition in the US Population 2012, 2012. Available online: http://www.cdc.gov/nutritionreport/report.html (accessed 29 April 2013).
- Lowe, N.M.; Fekete, K.; Decsi, T. Methods of assessment of zinc status in humans: A systematic review. Am. J. Clin. Nutr. 2009, 89, 2040S–2051S. [Google Scholar]
- Hess, S.Y.; Peerson, J.M.; King, J.C.; Brown, K.H. Use of serum zinc concentration as an indicator of population zinc status. Food Nutr. Bull. 2007, 28, S403–S429. [Google Scholar]
- Gibson, R.S.; Hess, S.Y.; Hotz, C.; Brown, K.H. Indicators of zinc status at the population level: A review of the evidence. Br. J. Nutr. 2008, 99, S14–S23. [Google Scholar]
- Hotz, C.; Brown, K.H. Assessment of the risk of zinc deficiency in populations and options for its control. Food Nutr. Bull. 2004, 25, S94–S203. [Google Scholar]
- Hotz, C.; Peerson, J.M.; Brown, K.H. Suggested lower cutoffs of serum zinc concentrations for assessing zinc status: Reanalysis of the second National Health and Nutrition Examination Survey data (1976–1980). Am. J. Clin. Nutr. 2003, 78, 756–764. [Google Scholar]
- Yokoi, K.; Sandstead, H.H.; Egger, N.G.; Alcock, N.W.; Sadagopa Ramanujam, V.M.; Dayal, H.H.; Penland, J.G. Association between zinc pool sizes and iron stores in premenopausal women without anaemia. Br. J. Nutr. 2007, 98, 1214–1223. [Google Scholar]
- Cole, C.R.; Grant, F.K.; Swaby-Ellis, E.D.; Smith, J.L.; Jacques, A.; Northrop-Clewes, C.A.; Caldwell, K.L.; Pfeiffer, C.M.; Ziegler, T.R. Zinc and iron deficiency and their interrelations in low-income African American and Hispanic children in Atlanta. Am. J. Clin. Nutr. 2010, 91, 1027–1034. [Google Scholar]
- Hettiarachchi, M.; Liyanage, C.; Wickremasinghe, R.; Hilmers, D.C.; Abrahams, S.A. Prevalence and severity of micronutrient deficiency: A cross-sectional study among adolescents in Sri Lanka. Asia Pac. J. Clin. Nutr. 2006, 15, 56–63. [Google Scholar]
- Yokoi, K.; Alcock, N.W.; Sandstead, H.H. Iron and zinc nutriture of premenopausal women: Associations of diet with serum ferritin and plasma zinc disappearance and of serum ferritin with plasma zinc and plasma zinc disappearance. J. Lab. Clin. Med. 1994, 124, 852–861. [Google Scholar]
- Solomons, N.W. Competitive interaction of iron and zinc in the diet: Consequences for human nutrition. J. Nutr. 1986, 116, 927–935. [Google Scholar]
- Olivares, M.; Pizarro, F.; Ruz, M.; Lopez de Romana, D. Acute inhibition of iron bioavailability by zinc: Studies in humans. Biometals 2012, 25, 657–664. [Google Scholar] [CrossRef]
- Solomons, N.W.; Jacob, R.A. Studies on the bioavailability of zinc in humans: Effects of heme and nonheme iron on the absorption of zinc. Am. J. Clin. Nutr. 1981, 34, 475–482. [Google Scholar]
- Solomons, N.W.; Pineda, O.; Viteri, F.; Sandstead, H.H. Studies on the bioavailability of zinc in humans: Mechanism of the intestinal interaction of nonheme iron and zinc. J. Nutr. 1983, 113, 337–349. [Google Scholar]
- Meadows, N.J.; Grainger, S.L.; Ruse, W.; Keeling, P.W.; Thompson, R.P. Oral iron and the bioavailability of zinc. Br. Med. J. (Clin. Res. Ed.). 1983, 287, 1013–1014. [Google Scholar]
- Solomons, N.W. Dietary sources of zinc and factors affecting its bioavailability. Food Nutr. Bull. 2001, 22, 138–154. [Google Scholar]
- Dijkhuizen, M.A.; Wieringa, F.T.; West, C.E.; Martuti, S. Muhilal effects of iron and zinc supplementation in Indonesian infants on micronutrient status and growth. J. Nutr. 2001, 131, 2860–2865. [Google Scholar]
- Sheldon, W.L.; Aspillaga, M.O.; Smith, P.A.; Lind, T. The effects of oral iron supplementation on zinc and magnesium levels during pregnancy. Br. J. Obstet. Gynaecol. 1985, 92, 892–898. [Google Scholar] [CrossRef]
- Yip, R.; Reeves, J.D.; Lonnerdal, B.; Keen, C.L.; Dallman, P.R. Does iron supplementation compromise zinc nutrition in healthy infants? Am. J. Clin. Nutr. 1985, 42, 683–687. [Google Scholar]
- Friel, J.K.; Aziz, K.; Andrews, W.L.; Harding, S.V.; Courage, M.L.; Adams, R.J. A double-masked, randomized control trial of iron supplementation in early infancy in healthy term breast-fed infants. J. Pediatr. 2003, 143, 582–586. [Google Scholar]
- Makrides, M.; Crowther, C.A.; Gibson, R.A.; Gibson, R.S.; Skeaff, C.M. Efficacy and tolerability of low-dose iron supplements during pregnancy: A randomized controlled trial. Am. J. Clin. Nutr. 2003, 78, 145–153. [Google Scholar]
- Dawson, E.B.; Albers, J.; McGanity, W.J. Serum zinc changes due to iron supplementation in teen-age pregnancy. Am. J. Clin. Nutr. 1989, 50, 848–852. [Google Scholar]
- Prosser, N.R.; Heath, A.L.; Williams, S.M.; Gibson, R.S. Influence of an iron intervention on the zinc status of young adult New Zealand women with mild iron deficiency. Br. J. Nutr. 2010, 104, 742–750. [Google Scholar] [CrossRef]
- Whittaker, P. Iron and zinc interactions in humans. Am. J. Clin. Nutr. 1998, 68, 442S–446S. [Google Scholar]
- Fairweather-Tait, S.J. Iron-zinc and calcium-Fe interactions in relation to Zn and Fe absorption. Proc. Nutr. Soc. 1995, 54, 465–473. [Google Scholar] [CrossRef]
- Valberg, L.S.; Flanagan, P.R.; Chamberlain, M.J. Effects of iron, tin, and copper on zinc absorption in humans. Am. J. Clin. Nutr. 1984, 40, 536–541. [Google Scholar]
- Sandström, B.; Davidsson, L.; Cederblad, A.; Lonnerdal, B. Oral iron, dietary ligands and zinc absorption. J. Nutr. 1985, 115, 411–414. [Google Scholar]
- Davidsson, L.; Almgren, A.; Sandstrom, B.; Hurrell, R.F. Zinc absorption in adult humans: The effect of iron fortification. Br. J. Nutr. 1995, 74, 417–425. [Google Scholar] [CrossRef]
- Donangelo, C.M.; Woodhouse, L.R.; King, S.M.; Viteri, F.E.; King, J.C. Supplemental zinc lowers measures of iron status in young women with low iron reserves. J. Nutr. 2002, 132, 1860–1864. [Google Scholar]
- Yadrick, M.K.; Kenney, M.A.; Winterfeldt, E.A. Iron, copper, and zinc status: Response to supplementation with zinc or zinc and iron in adult females. Am. J. Clin. Nutr. 1989, 49, 145–150. [Google Scholar]
- Fahmida, U.; Rumawas, J.S.; Utomo, B.; Patmonodewo, S.; Schultink, W. Zinc-iron, but not zinc-alone supplementation, increased linear growth of stunted infants with low haemoglobin. Asia Pac. J. Clin. Nutr. 2007, 16, 301–309. [Google Scholar]
- Wuehler, S.E.; Sempertegui, F.; Brown, K.H. Dose-response trial of prophylactic zinc supplements, with or without copper, in young Ecuadorian children at risk of zinc deficiency. Am. J. Clin. Nutr. 2008, 87, 723–733. [Google Scholar]
- Rosado, J.L.; Diaz, M.; Gonzalez, K.; Griffin, I.; Abrams, S.A.; Preciado, R. The addition of milk or yogurt to a plant-based diet increases zinc bioavailability but does not affect iron bioavailability in women. J. Nutr. 2005, 135, 465–468. [Google Scholar]
- Penny, M.E.; Marin, R.M.; Duran, A.; Peerson, J.M.; Lanata, C.F.; Lonnerdal, B.; Black, R.E.; Brown, K.H. Randomized controlled trial of the effect of daily supplementation with zinc or multiple micronutrients on the morbidity, growth, and micronutrient status of young Peruvian children. Am. J. Clin. Nutr. 2004, 79, 457–465. [Google Scholar]
- Brown, K.H.; Peerson, J.M.; Baker, S.K.; Hess, S.Y. Preventive zinc supplementation among infants, preschoolers, and older prepubertal children. Food Nutr. Bull. 2009, 30, S12–S40. [Google Scholar]
- Friel, J.K.; Serfass, R.E.; Fennessey, P.V.; Miller, L.V.; Andrews, W.L.; Simmons, B.S.; Downton, G.F.; Kwa, P.G. Elevated intakes of zinc in infant formulas do not interfere with iron absorption in premature infants. J. Pediatr. Gastroenterol. Nutr. 1998, 27, 312–316. [Google Scholar] [CrossRef]
- Rossander-Hulten, L.; Brune, M.; Sandstrom, B.; Lonnerdal, B.; Hallberg, L. Competitive inhibition of iron absorption by manganese and zinc in humans. Am. J. Clin. Nutr. 1991, 54, 152–156. [Google Scholar]
- Olivares, M.; Pizarro, F.; Ruz, M. Zinc inhibits nonheme iron bioavailability in humans. Biol. Trace Elem. Res. 2007, 117, 7–14. [Google Scholar] [CrossRef]
- Lopez de Romana, D.; Ruz, M.; Pizarro, F.; Landeta, L.; Olivares, M.A. Supplementation with zinc between meals has no effect on subsequent iron absorption or on iron status of Chilean women. Nutrition 2008, 24, 957–963. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lim, K.H.C.; Riddell, L.J.; Nowson, C.A.; Booth, A.O.; Szymlek-Gay, E.A. Iron and Zinc Nutrition in the Economically-Developed World: A Review. Nutrients 2013, 5, 3184-3211. https://doi.org/10.3390/nu5083184
Lim KHC, Riddell LJ, Nowson CA, Booth AO, Szymlek-Gay EA. Iron and Zinc Nutrition in the Economically-Developed World: A Review. Nutrients. 2013; 5(8):3184-3211. https://doi.org/10.3390/nu5083184
Chicago/Turabian StyleLim, Karen H. C., Lynn J. Riddell, Caryl A. Nowson, Alison O. Booth, and Ewa A. Szymlek-Gay. 2013. "Iron and Zinc Nutrition in the Economically-Developed World: A Review" Nutrients 5, no. 8: 3184-3211. https://doi.org/10.3390/nu5083184
APA StyleLim, K. H. C., Riddell, L. J., Nowson, C. A., Booth, A. O., & Szymlek-Gay, E. A. (2013). Iron and Zinc Nutrition in the Economically-Developed World: A Review. Nutrients, 5(8), 3184-3211. https://doi.org/10.3390/nu5083184





