Inhibitory Effects and Mechanisms of Luteolin on Proliferation and Migration of Vascular Smooth Muscle Cells
Abstract
:1. Introduction
2. Luteolin and Atherosclerosis
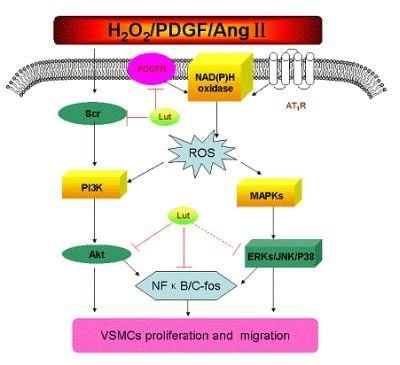
3. Luteolin Inhibited VSMCs Proliferation and Migration
3.1. Luteolin Inhibited VSMCs Proliferation
3.2. Luteolin Inhibited VSMCs Migration
3.3. Luteolin and Apoptosis of VSMCs
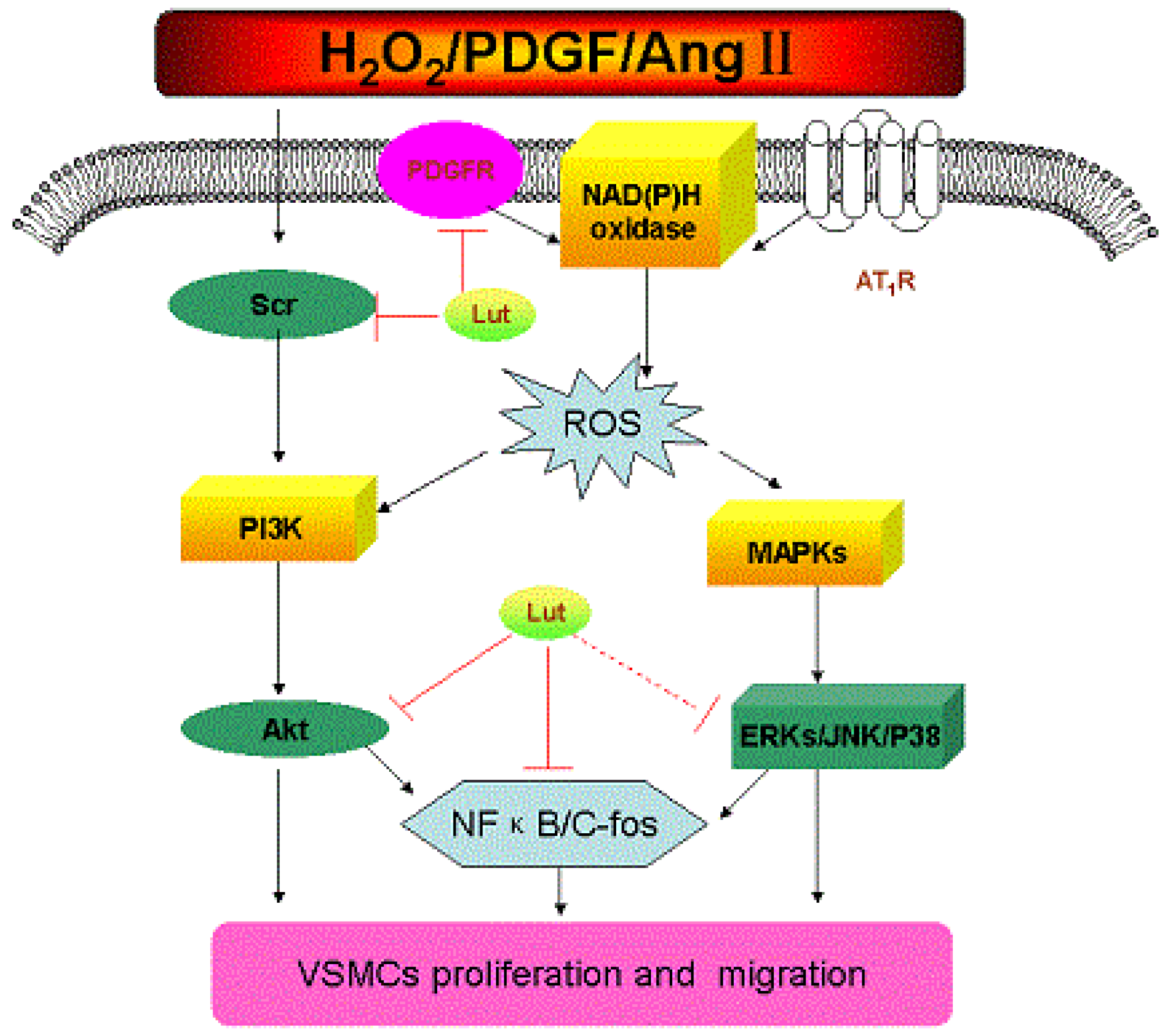
4. Conclusions and Perspective
Conflict of Interest
References
- Doran, A.C.; Meller, N.; McNamara, C.A. Role of smooth muscle cells in the initiation and early progression of atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 812–819. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, Y.; Yang, Q.; Yang, Y.; Gu, Y.; Wang, M.; Wu, K. A novel cultured tissue model of rat aorta: VSMC proliferation mechanism in relationship to atherosclerosis. Exp. Mol. Pathol. 2007, 83, 453–458. [Google Scholar] [CrossRef]
- Ross, J.S.; Stagliano, N.E.; Donovan, M.J.; Breitbart, R.E.; Ginsburg, G.S. Atherosclerosis and cancer: Common molecular pathways of disease development and progression. Ann. N. Y. Acad. Sci. 2001, 947, 271–292; discussion 292–293. [Google Scholar]
- Zernecke, A.; Weber, C. Chemokines in the vascular inflammatory response of atherosclerosis. Cardiovasc. Res. 2010, 86, 192–201. [Google Scholar] [CrossRef]
- Boyle, EM., Jr.; Lille, S.T.; Allaire, E.; Clowes, A.W.; Verrier, E.D. Endothelial cell injury in cardiovascular surgery: Atherosclerosis. Ann. Thorac. Surg. 1997, 63, 885–894. [Google Scholar] [CrossRef]
- Stoneman, V.E.; Bennett, M.R. Role of apoptosis in atherosclerosis and its therapeutic implications. Clin. Sci. (Lond.) 2004, 107, 343–354. [Google Scholar]
- Wasserman, E.J.; Shipley, N.M. Atherothrombosis in acute coronary syndromes: Mechanisms, markers, and mediators of vulnerability. Mt. Sinai J. Med. 2006, 73, 431–439. [Google Scholar]
- Bellosta, S.; Bernini, F.; Ferri, N.; Quarato, P.; Canavesi, M.; Arnaboldi, L.; Fumagalli, R.; Paoletti, R.; Corsini, A. Direct vascular effects of HMG-CoA reductase inhibitors. Atherosclerosis 1998, 137, S101–S109. [Google Scholar] [CrossRef]
- Zeng, Y.; Song, J.X.; Shen, X.C. Herbal remedies supply a novel prospect for the treatment of atherosclerosis: A review of current mechanism studies. Phytother. Res. 2012, 26, 159–167. [Google Scholar] [CrossRef]
- Kelloff, G.J. Perspectives on cancer chemoprevention research and drug development. Adv. Cancer Res. 2000, 78, 199–334. [Google Scholar] [CrossRef]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Remesy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, 230S–242S. [Google Scholar]
- Loke, W.M.; Proudfoot, J.M.; Stewart, S.; McKinley, A.J.; Needs, P.W.; Kroon, P.A.; Hodgson, J.M.; Croft, K.D. Metabolic transformation has a profound effect on anti-inflammatory activity of flavonoids such as quercetin: Lack of association between antioxidant and lipoxygenase inhibitory activity. Biochem. Pharmacol. 2008, 75, 1045–1053. [Google Scholar] [CrossRef]
- Chen, Z.; Tu, M.; Sun, S.; Kong, S.; Wang, Y.; Ye, J.; Li, L.; Zeng, S.; Jiang, H. The exposure of luteolin is much lower than that of apigenin in oral administration of Flos Chrysanthemi extract to rats. Drug Metab. Pharmacokinet. 2012, 27, 162–168. [Google Scholar] [CrossRef]
- Cao, G.; Sofic, E.; Prior, R.L. Antioxidant and prooxidant behavior of flavonoids: Structure-activity relationships. Free Radic. Biol. Med. 1997, 22, 749–760. [Google Scholar] [CrossRef]
- Seelinger, G.; Merfort, I.; Schempp, C.M. Anti-oxidant, anti-inflammatory and anti-allergic activities of luteolin. Planta Med. 2008, 74, 1667–1677. [Google Scholar] [CrossRef]
- Yochum, L.; Kushi, L.H.; Meyer, K.; Folsom, A.R. Dietary flavonoid intake and risk of cardiovascular disease in postmenopausal women. Am. J. Epidemiol. 1999, 149, 943–949. [Google Scholar] [CrossRef]
- Middleton, E., Jr.; Kandaswami, C.; Theoharides, T.C. The effects of plant flavonoids on mammalian cells: Implications for inflammation, heart disease, and cancer. Pharmacol. Rev. 2000, 52, 673–751. [Google Scholar]
- Lopez-Lazaro, M. Distribution and biological activities of the flavonoid luteolin. Mini Rev. Med. Chem. 2009, 9, 31–59. [Google Scholar] [CrossRef]
- Lin, Y.; Shi, R.; Wang, X.; Shen, H.M. Luteolin, a flavonoid with potential for cancer prevention and therapy. Curr. Cancer Drug Targets 2008, 8, 634–646. [Google Scholar] [CrossRef]
- Mulvihill, E.E.; Huff, M.W. Antiatherogenic properties of flavonoids: Implications for cardiovascular health. Can. J. Cardiol. 2010, 26, 17A–21A. [Google Scholar] [CrossRef]
- Clarke, M.; Bennett, M. Defining the role of vascular smooth muscle cell apoptosis in atherosclerosis. Cell Cycle 2006, 5, 2329–2331. [Google Scholar] [CrossRef]
- Ross, J.A.; Kasum, C.M. Dietary flavonoids: Bioavailability, metabolic effects, and safety. Annu. Rev. Nutr. 2002, 22, 19–34. [Google Scholar]
- Agullo, G.; Gamet-Payrastre, L.; Manenti, S.; Viala, C.; Remesy, C.; Chap, H.; Payrastre, B. Relationship between flavonoid structure and inhibition of phosphatidylinositol 3-kinase: A comparison with tyrosine kinase and protein kinase C inhibition. Biochem. Pharmacol. 1997, 53, 1649–1657. [Google Scholar] [CrossRef]
- Lv, L.; Zhang, Y.; Kong, Q. Luteolin prevents LPS-induced TNF-α expression in cardiac myocytes through inhibiting NF-κB signaling pathway. Inflammation 2011, 34, 620–629. [Google Scholar]
- Wang, G.; Li, W.; Lu, X.; Bao, P.; Zhao, X. Luteolin ameliorates cardiac failure in type I diabetic cardiomyopathy. J. Diabetes Complicat. 2012, 26, 259–265. [Google Scholar] [CrossRef]
- Kim, J.H.; Jin, Y.R.; Park, B.S.; Kim, T.J.; Kim, S.Y.; Lim, Y.; Hong, J.T.; Yoo, H.S.; Yun, Y.P. Luteolin prevents PDGF-BB-induced proliferation of vascular smooth muscle cells by inhibition of PDGF β-receptor phosphorylation. Biochem. Pharmacol. 2005, 69, 1715–1721. [Google Scholar] [CrossRef]
- Lang, Y.; Chen, D.; Li, D.; Zhu, M.; Xu, T.; Zhang, T.; Qian, W.; Luo, Y. Luteolin inhibited hydrogen peroxide-induced vascular smooth muscle cells proliferation and migration by suppressing the Src and Akt signalling pathways. J. Pharm. Pharmacol. 2012, 64, 597–603. [Google Scholar] [CrossRef]
- Zhu, M.; Chen, D.; Li, D.; Ding, H.; Zhang, T.; Xu, T.; Zhang, Y. Luteolin inhibits angiotensin II-induced human umbilical vein endothelial cell proliferation and migration through downregulation of src and akt phosphorylation. Circ. J. 2012, 77, 772–779. [Google Scholar]
- Ando, C.; Takahashi, N.; Hirai, S.; Nishimura, K.; Lin, S.; Uemura, T.; Goto, T.; Yu, R.; Nakagami, J.; Murakami, S.; et al. Luteolin, a food-derived flavonoid, suppresses adipocyte-dependent activation of macrophages by inhibiting JNK activation. FEBS Lett. 2009, 583, 3649–3654. [Google Scholar] [CrossRef]
- Qi, L.; Pan, H.; Li, D.; Fang, F.; Chen, D.; Sun, H. Luteolin improves contractile function and attenuates apoptosis following ischemia-reperfusion in adult rat cardiomyocytes. Eur. J. Pharmacol. 2011, 668, 201–207. [Google Scholar]
- Sun, D.; Huang, J.; Zhang, Z.; Gao, H.; Li, J.; Shen, M.; Cao, F.; Wang, H. Luteolin limits infarct size and improves cardiac function after myocardium ischemia/reperfusion injury in diabetic rats. PLoS One 2012, 7, e33491. [Google Scholar] [CrossRef]
- Fang, F.; Li, D.; Pan, H.; Chen, D.; Qi, L.; Zhang, R.; Sun, H. Luteolin inhibits apoptosis and improves cardiomyocyte contractile function through the PI3K/Akt pathway in simulated ischemia/reperfusion. Pharmacology 2011, 88, 149–158. [Google Scholar] [CrossRef]
- Song, J.; Liu, K.; Yi, J.; Zhu, D.; Liu, G.; Liu, B. Luteolin inhibits lysophosphatidylcholine-induced apoptosis in endothelial cells by a calcium/mitocondrion/caspases-dependent pathway. Planta Med. 2010, 76, 433–438. [Google Scholar] [CrossRef]
- Muslin, A.J. MAPK signalling in cardiovascular health and disease: Molecular mechanisms and therapeutic targets. Clin. Sci. (Lond.) 2008, 115, 203–218. [Google Scholar] [CrossRef]
- Muto, A.; Fitzgerald, T.N.; Pimiento, J.M.; Maloney, S.P.; Teso, D.; Paszkowiak, J.J.; Westvik, T.S.; Kudo, F.A.; Nishibe, T.; Dardik, A. Smooth muscle cell signal transduction: Implications of vascular biology for vascular surgeons. J. Vasc. Surg. 2007, 45 (Suppl. A), A15–A24. [Google Scholar]
- Fernandez-Hernando, C.; Jozsef, L.; Jenkins, D.; di Lorenzo, A.; Sessa, W.C. Absence of Akt1 reduces vascular smooth muscle cell migration and survival and induces features of plaque vulnerability and cardiac dysfunction during atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 2033–2040. [Google Scholar] [CrossRef]
- Stintzing, S.; Ocker, M.; Hartner, A.; Amann, K.; Barbera, L.; Neureiter, D. Differentiation patterning of vascular smooth muscle cells (VSMC) in atherosclerosis. Virchows Arch. 2009, 455, 171–185. [Google Scholar]
- Brown, X.Q.; Bartolak-Suki, E.; Williams, C.; Walker, M.L.; Weaver, V.M.; Wong, J.Y. Effect of substrate stiffness and PDGF on the behavior of vascular smooth muscle cells: Implications for atherosclerosis. J. Cell. Physiol. 2010, 225, 115–122. [Google Scholar] [CrossRef]
- Perez-Vizcaino, F.; Bishop-Bailley, D.; Lodi, F.; Duarte, J.; Cogolludo, A.; Moreno, L.; Bosca, L.; Mitchell, J.A.; Warner, T.D. The flavonoid quercetin induces apoptosis and inhibits JNK activation in intimal vascular smooth muscle cells. Biochem. Biophys. Res. Commun. 2006, 346, 919–925. [Google Scholar] [CrossRef]
- Kobayashi, T.; Nakata, T.; Kuzumaki, T. Effect of flavonoids on cell cycle progression in prostate cancer cells. Cancer Lett. 2002, 176, 17–23. [Google Scholar] [CrossRef]
- Lim, D.Y.; Jeong, Y.; Tyner, A.L.; Park, J.H. Induction of cell cycle arrest and apoptosis in HT-29 human colon cancer cells by the dietary compound luteolin. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 292, G66–G75. [Google Scholar]
- Sriram, V.; Patterson, C. Cell cycle in vasculoproliferative diseases: Potential interventions and routes of delivery. Circulation 2001, 103, 2414–2419. [Google Scholar] [CrossRef]
- Wessely, R. Atherosclerosis and cell cycle: Put the brakes on! Critical role for cyclin-dependent kinase inhibitors. J. Am. Coll. Cardiol. 2010, 55, 2269–2271. [Google Scholar] [CrossRef]
- Zettler, M.E.; Pierce, G.N. Cell cycle proteins and atherosclerosis. Herz 2000, 25, 100–107. [Google Scholar] [CrossRef]
- Ferguson, J.E., III; Patterson, C. Break the cycle: The role of cell-cycle modulation in the prevention of vasculoproliferative diseases. Cell Cycle 2003, 2, 211–219. [Google Scholar]
- Willis, A.I.; Pierre-Paul, D.; Sumpio, B.E.; Gahtan, V. Vascular smooth muscle cell migration: Current research and clinical implications. Vasc. Endovasc. Surg. 2004, 38, 11–23. [Google Scholar]
- Lamy, S.; Bédard, V.; Labbé, D.; Sartelet, H.; Barthomeuf, C.; Gingras, D.; Béliveau, R. The dietary flavones apigenin and luteolin impair smooth muscle cell migration and VEGF expression through inhibition of PDGFR-β phosphorylation. Cancer Prev. Res. (Phila.) 2008, 1, 452–459. [Google Scholar]
- Clarke, M.; Bennett, M. The emerging role of vascular smooth muscle cell apoptosis in atherosclerosis and plaque stability. Am. J. Nephrol. 2006, 26, 531–535. [Google Scholar] [CrossRef]
- Clarke, M.C.; Figg, N.; Maguire, J.J.; Davenport, A.P.; Goddard, M.; Littlewood, T.D.; Bennett, M.R. Apoptosis of vascular smooth muscle cells induces features of plaque vulnerability in atherosclerosis. Nat. Med. 2006, 12, 1075–1080. [Google Scholar] [CrossRef]
- Horinaka, M.; Yoshida, T.; Shiraishi, T.; Nakata, S.; Wakada, M.; Nakanishi, R.; Nishino, H.; Matsui, H.; Sakai, T. Luteolin induces apoptosis via death receptor 5 upregulation in human malignant tumor cells. Oncogene 2005, 24, 7180–7189. [Google Scholar] [CrossRef]
- Chang, J.; Hsu, Y.; Kuo, P.; Kuo, Y.; Chiang, L.; Lin, C. Increase of Bax/Bcl-XL ratio and arrest of cell cycle by luteolin in immortalized human hepatoma cell line. Life Sci. 2005, 76, 1883–1893. [Google Scholar] [CrossRef]
- Guerrero, J.A.; Lozano, M.L.; Castillo, J.; Benavente-Garcia, O.; Vicente, V.; Rivera, J. Flavonoids inhibit platelet function through binding to the thromboxane A2 receptor. J. Thromb. Haemost. 2005, 3, 369–376. [Google Scholar] [CrossRef]
- Bagli, E.; Stefaniotou, M.; Morbidelli, L.; Ziche, M.; Psillas, K.; Murphy, C.; Fotsis, T. Luteolin inhibits vascular endothelial growth factor-induced angiogenesis; inhibition of endothelial cell survival and proliferation by targeting phosphatidylinositol 3′-kinase activity. Cancer Res. 2004, 64, 7936–7946. [Google Scholar]
- Hiremath, S.P.; Badami, S.; Hunasagatta, S.K.; Patil, S.B. Antifertility and hormonal properties of flavones of Striga orobanchioides. Eur. J. Pharmacol. 2000, 391, 193–197. [Google Scholar] [CrossRef]
- Xu, Y.C.; Leung, S.W.; Yeung, D.K.; Hu, L.H.; Chen, G.H.; Che, C.M.; Man, R.Y. Structure-activity relationships of flavonoids for vascular relaxation in porcine coronary artery. Phytochemistry 2007, 68, 1179–1188. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jiang, D.; Li, D.; Wu, W. Inhibitory Effects and Mechanisms of Luteolin on Proliferation and Migration of Vascular Smooth Muscle Cells. Nutrients 2013, 5, 1648-1659. https://doi.org/10.3390/nu5051648
Jiang D, Li D, Wu W. Inhibitory Effects and Mechanisms of Luteolin on Proliferation and Migration of Vascular Smooth Muscle Cells. Nutrients. 2013; 5(5):1648-1659. https://doi.org/10.3390/nu5051648
Chicago/Turabian StyleJiang, Dehua, Dongye Li, and Wanling Wu. 2013. "Inhibitory Effects and Mechanisms of Luteolin on Proliferation and Migration of Vascular Smooth Muscle Cells" Nutrients 5, no. 5: 1648-1659. https://doi.org/10.3390/nu5051648
APA StyleJiang, D., Li, D., & Wu, W. (2013). Inhibitory Effects and Mechanisms of Luteolin on Proliferation and Migration of Vascular Smooth Muscle Cells. Nutrients, 5(5), 1648-1659. https://doi.org/10.3390/nu5051648