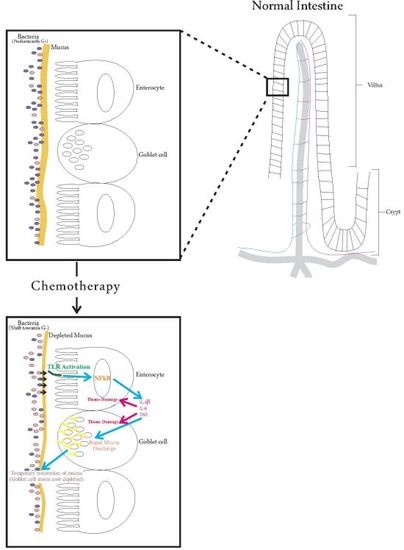
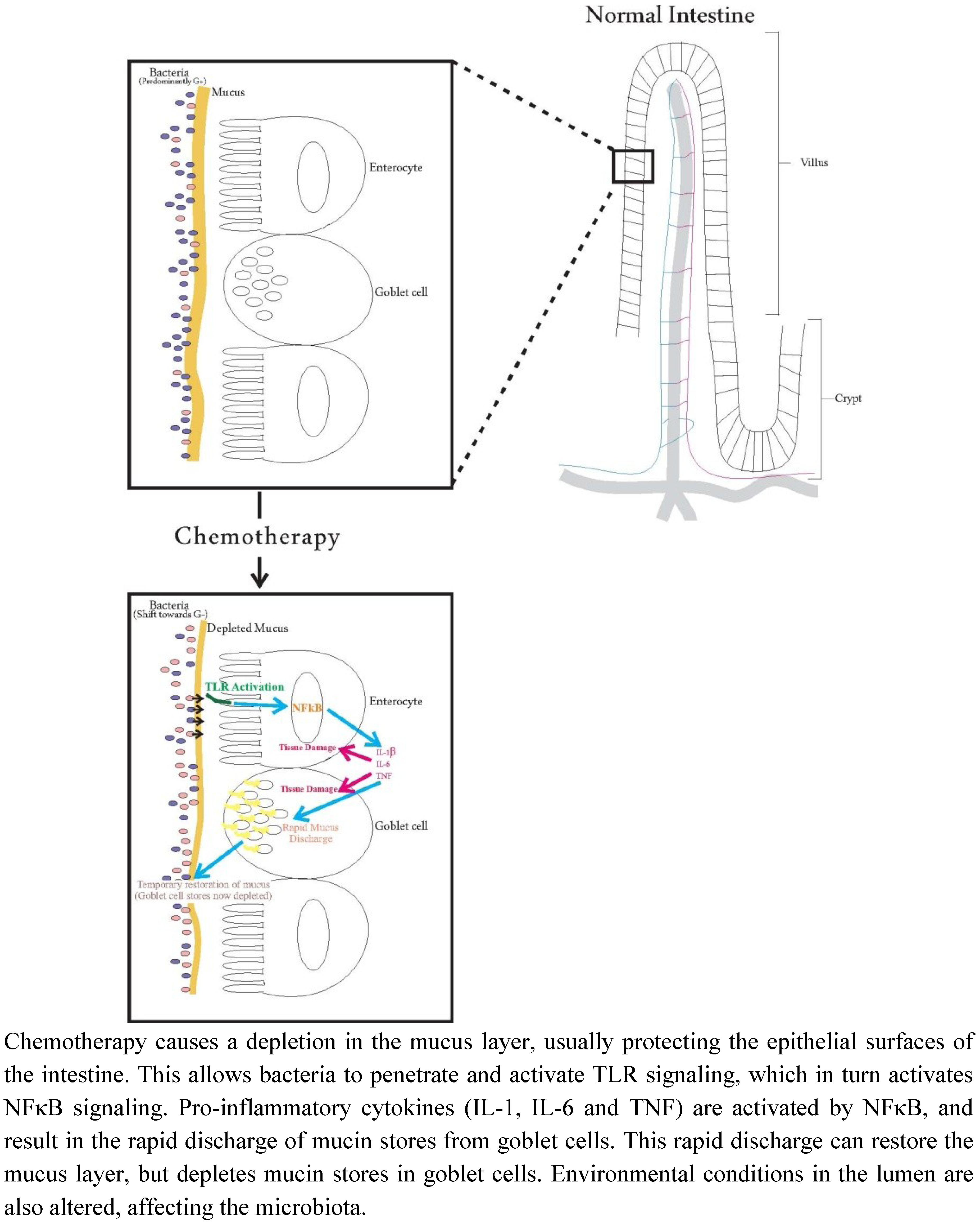
Interaction between Host Cells and Microbes in Chemotherapy-Induced Mucositis
Abstract
:1. Introduction
2. Pathobiology of Mucositis
3. Possible Roles for Microbiota in the Pathobiology of Chemotherapy-Induced Mucositis
3.1. Initiation and Generation of Reactive Oxygen Species
3.2. Nuclear Factor Kappa B (NFκB) Up-Regulation, Message Generation, and Amplification
4. Mucous Mechanisms of Modulating Microbiota
5. Effects of Chemotherapy
5.1. Effects of Chemotherapy on the Intestinal Microbiota
5.2. Effects of Chemotherapy on Mucins
5.3. Effects of Chemotherapy on Pro-Inflammatory Cytokines
6. Relationships between Intestinal Microbiota, Mucins and Pro-Inflammatory Cytokines
7. Future Directions
8. Conclusions
Conflict of Interest
References
- Keefe, D.M. Gastrointestinal mucositis: A new biological model. Support. Care Cancer 2004, 12, 6–9. [Google Scholar] [CrossRef]
- De Vita, V., Jr. Principals of Cancer Management: Chemotherapy. In Cancer: Principles and Practice of Oncology, 5th; De Vita, V., Jr., Ed.; Lippincott-Raven: Philadelphia, PA, USA, 1997; pp. 333–347. [Google Scholar]
- Cunningham, D.; Morgan, R.J.; Mills, P.R.; Nelson, L.M.; Toner, P.G.; Soukop, M.; McArdle, C.S.; Russell, R.I. Functional and structural changes of the human proximal small intestine after cytotoxic therapy. J. Clin. Pathol. 1985, 38, 265–270. [Google Scholar]
- Keefe, D.M.; Gibson, R.J.; Hauer-Jensen, M. Gastrointestinal mucositis. Semin. Oncol. Nurs. 2004, 20, 38–47. [Google Scholar] [CrossRef]
- Duncan, M.; Grant, G. Oral and intestinal mucositis—causes and possible treatments. Aliment. Pharmacol. Ther. 2003, 18, 853–874. [Google Scholar] [CrossRef]
- Takasuna, K.; Hagiwara, T.; Hirohashi, M.; Kato, M.; Nomura, M.; Nagai, E.; Yokoi, T.; Kamataki, T. Involvement of beta-glucuronidase in intestinal microflora in the intestinal toxicity of the antitumor camptothecin derivative irinotecan hydrochloride (CPT-11) in rats. Cancer Res. 1996, 56, 3752–3757. [Google Scholar]
- Ikuno, N.; Soda, H.; Watanabe, M.; Oka, M. Irinotecan (CPT-11) and characteristic mucosal changes in the mouse ileum and cecum. J. Natl. Cancer Inst. 1995, 87, 1876–1883. [Google Scholar] [CrossRef]
- Gibson, R.J.; Keefe, D.M.; Thompson, F.M.; Clarke, J.M.; Goland, G.J.; Cummins, A.G. Effect of interleukin-11 on ameliorating intestinal damage after methotrexate treatment of breast cancer in rats. Dig. Dis. Sci. 2002, 47, 2751–2757. [Google Scholar] [CrossRef]
- Gibson, R.J.; Keefe, D.M.; Clarke, J.M.; Regester, G.O.; Thompson, F.M.; Goland, G.J.; Edwards, B.G.; Cummins, A.G. The effect of keratinocyte growth factor on tumour growth and small intestinal mucositis after chemotherapy in the rat with breast cancer. Cancer Chemother. Pharmacol. 2002, 50, 53–58. [Google Scholar] [CrossRef]
- Keefe, D.M.; Brealey, J.; Goland, G.J.; Cummins, A.G. Chemotherapy for cancer causes apoptosis that precedes hypoplasia in crypts of the small intestine in humans. Gut 2000, 47, 632–637. [Google Scholar] [CrossRef]
- Sonis, S.T.; Elting, L.S.; Keefe, D.; Peterson, D.E.; Schubert, M.; Hauer-Jensen, M.; Bekele, B.N.; Raber-Durlacher, J.; Donnelly, J.P.; Rubenstein, E.B. Perspectives on cancer therapy-induced mucosal injury: Pathogenesis, measurement, epidemiology, and consequences for patients. Cancer 2004, 100, 1995–2025. [Google Scholar]
- Sonis, S.T. Pathobiology of mucositis. Semin. Oncol. Nurs. 2004, 20, 11–15. [Google Scholar] [CrossRef]
- Sonis, S.T. The pathobiology of mucositis. Nat. Rev. Cancer 2004, 4, 277–284. [Google Scholar] [CrossRef]
- Stringer, A.M.; Gibson, R.J.; Logan, R.M.; Bowen, J.M.; Yeoh, A.S.; Burns, J.; Keefe, D.M. Chemotherapy-induced diarrhea is associated with changes in the luminal environment in the da rat. Exp. Biol. Med. (Maywood) 2007, 232, 96–106. [Google Scholar]
- Logan, R.M.; Stringer, A.M.; Bowen, J.M.; Yeoh, A.S.; Gibson, R.J.; Sonis, S.T.; Keefe, D.M. The role of pro-inflammatory cytokines in cancer treatment-induced alimentary tract mucositis: Pathobiology, animal models and cytotoxic drugs. Cancer Treat. Rev. 2007, 33, 448–460. [Google Scholar] [CrossRef]
- Gibson, R.J.; Bowen, J.M.; Inglis, M.R.; Cummins, A.G.; Keefe, D.M. Irinotecan causes severe small intestinal damage, as well as colonic damage, in the rat with implanted breast cancer. J. Gastroenterol. Hepatol. 2003, 18, 1095–1100. [Google Scholar] [CrossRef]
- Gibson, R.J.; Keefe, D.M. Cancer chemotherapy-induced diarrhoea and constipation: Mechanisms of damage and prevention strategies. Support. Care Cancer 2006, 14, 890–900. [Google Scholar] [CrossRef]
- Yeoh, A.S.; Bowen, J.M.; Gibson, R.J.; Keefe, D.M. Nuclear factor kappaB (NFkappaB) and cyclooxygenase-2 (cox-2) expression in the irradiated colorectum is associated with subsequent histopathological changes. Int. J. Radiat Oncol. Biol. Phys. 2005, 63, 1295–1303. [Google Scholar] [CrossRef]
- Logan, R.M.; Stringer, A.M.; Bowen, J.M.; Gibson, R.J.; Sonis, S.T.; Keefe, D.M. Serum levels of NFkappaB and pro-inflammatory cytokines following administration of mucotoxic drugs. Cancer Biol. Ther. 2008, 7, 1139–1145. [Google Scholar] [CrossRef]
- Logan, R.M.; Gibson, R.J.; Bowen, J.M.; Stringer, A.M.; Sonis, S.T.; Keefe, D.M. Characterisation of mucosal changes in the alimentary tract following administration of irinotecan: Implications for the pathobiology of mucositis. Cancer Chemother. Pharmacol. 2008, 62, 33–41. [Google Scholar] [CrossRef]
- Stringer, A.M.; Gibson, R.J.; Bowen, J.M.; Logan, R.M.; Ashton, K.; Yeoh, A.S.; Al-Dasooqi, N.; Keefe, D.M. Irinotecan-induced mucositis manifesting as diarrhoea corresponds with an amended intestinal flora and mucin profile. Int. J. Exp. Pathol. 2009, 90, 489–499. [Google Scholar] [CrossRef]
- Stringer, A.M.; Gibson, R.J.; Logan, R.M.; Bowen, J.M.; Yeoh, A.S.; Hamilton, J.; Keefe, D.M. Gastrointestinal microflora and mucins may play a critical role in the development of 5-fluorouracil-induced gastrointestinal mucositis. Exp. Biol. Med. (Maywood) 2009, 234, 430–441. [Google Scholar]
- Stringer, A.M.; Gibson, R.J.; Logan, R.M.; Bowen, J.M.; Yeoh, A.S.; Laurence, J.; Keefe, D.M. Irinotecan-induced mucositis is associated with changes in intestinal mucins. Cancer Chemother. Pharmacol. 2009, 64, 123–132. [Google Scholar] [CrossRef]
- Sonis, S.T. A biological approach to mucositis. J. Support. Oncol. 2004, 2, 21–32; discussion 35–36. [Google Scholar]
- Graham Espey, M. Role of oxygen gradients in shaping redox relationships between the human intestine and its microbiota. Free Radic. Biol. Med. 2012. [Google Scholar] [CrossRef]
- Hartman, A.L.; Lough, D.M.; Barupal, D.K.; Fiehn, O.; Fishbein, T.; Zasloff, M.; Eisen, J.A. Human gut microbiome adopts an alternative state following small bowel transplantation. Proc. Natl. Acad. Sci. USA 2009, 106, 17187–17192. [Google Scholar]
- Lambeth, J.D. Nox enzymes, ros, and chronic disease: An example of antagonistic pleiotropy. Free Radic. Biol. Med. 2007, 43, 332–347. [Google Scholar] [CrossRef]
- Lambeth, J.D.; Kawahara, T.; Diebold, B. Regulation of Nox and Duox enzymatic activity and expression. Free Radic. Biol. Med. 2007, 43, 319–331. [Google Scholar] [CrossRef]
- Van Vliet, M.J.; Tissing, W.J.; Dun, C.A.; Meessen, N.E.; Kamps, W.A.; de Bont, E.S.; Harmsen, H.J. Chemotherapy treatment in pediatric patients with acute myeloid leukemia receiving antimicrobial prophylaxis leads to a relative increase of colonization with potentially pathogenic bacteria in the gut. Clin. Infect. Dis. 2009, 49, 262–270. [Google Scholar] [CrossRef]
- Van Vliet, M.J.; Harmsen, H.J.; de Bont, E.S.; Tissing, W.J. The role of intestinal microbiota in the development and severity of chemotherapy-induced mucositis. PLoS Pathog. 2010, 6, e1000879. [Google Scholar] [CrossRef]
- Rakoff-Nahoum, S.; Paglino, J.; Eslami-Varzaneh, F.; Edberg, S.; Medzhitov, R. Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis. Cell 2004, 118, 229–241. [Google Scholar] [CrossRef]
- Cario, E. Innate immune signalling at intestinal mucosal surfaces: A fine line between host protection and destruction. Curr. Opin. Gastroenterol. 2008, 24, 725–732. [Google Scholar] [CrossRef]
- Cario, E. Barrier-protective function of intestinal epithelial toll-like receptor 2. Mucosal Immunol. 2008, 1, S62–S66. [Google Scholar] [CrossRef]
- Hedl, M.; Li, J.; Cho, J.H.; Abraham, C. Chronic stimulation of Nod2 mediates tolerance to bacterial products. Proc. Natl. Acad. Sci. USA 2007, 104, 19440–19445. [Google Scholar] [CrossRef]
- Cario, E. Bacterial interactions with cells of the intestinal mucosa: Toll-like receptors and Nnod2. Gut 2005, 54, 1182–1193. [Google Scholar] [CrossRef]
- Kelly, D.; Campbell, J.I.; King, T.P.; Grant, G.; Jansson, E.A.; Coutts, A.G.; Pettersson, S.; Conway, S. Commensal anaerobic gut bacteria attenuate inflammation by regulating nuclear-cytoplasmic shuttling of PPAR-gamma and RelA. Nat. Immunol. 2004, 5, 104–112. [Google Scholar] [CrossRef]
- Beg, A.A. Comppartmentalizing NF-kappaB in the gut. Nat. Immunol. 2004, 5, 14–16. [Google Scholar] [CrossRef]
- Ewaschuk, J.B.; Diaz, H.; Meddings, L.; Diederichs, B.; Dmytrash, A.; Backer, J.; Looijer-van Langen, M.; Madsen, K.L. Secreted bioactive factors from bifidobacterium infantis enhance epithelial cell barrier function. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 295, G1025–G1034. [Google Scholar] [CrossRef]
- Sokol, H.; Pigneur, B.; Watterlot, L.; Lakhdari, O.; Bermudez-Humaran, L.G.; Gratadoux, J.J.; Blugeon, S.; Bridonneau, C.; Furet, J.P.; Corthier, G.; et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of crohn disease patients. Proc. Natl. Acad. Sci. USA 2008, 105, 16731–16736. [Google Scholar] [CrossRef]
- Stringer, A.M.; Gibson, R.J.; Bowen, J.M.; Logan, R.M.; Yeoh, A.S.; Keefe, D.M. Chemotherapy-induced mucositis: The role of gastrointestinal microflora and mucins in the luminal environment. J. Support. Oncol. 2007, 5, 259–267. [Google Scholar]
- Specian, R.D.; Oliver, M.G. Functional biology of intestinal goblet cells. Am. J. Physiol. 1991, 260, C183–C193. [Google Scholar]
- Robbe, C.; Capon, C.; Coddeville, B.; Michalski, J.C. Structural diversity and specific distribution of O-glycans in normal human mucins along the intestinal tract. Biochem. J. 2004, 384, 307–316. [Google Scholar] [CrossRef]
- Deplancke, B.; Gaskins, H.R. Microbial modulation of innate defense: Goblet cells and the intestinal mucus layer. Am. J. Clin. Nutr. 2001, 73, 1131S–1141S. [Google Scholar]
- Mack, D.R.; Michail, S.; Wei, S.; McDougall, L.; Hollingsworth, M.A. Probiotics inhibit enteropathogenic e. Coli adherence in vitro by inducing intestinal mucin gene expression. Am. J. Physiol. 1999, 276, G941–G950. [Google Scholar]
- Enss, M.L.; Cornberg, M.; Wagner, S.; Gebert, A.; Henrichs, M.; Eisenblatter, R.; Beil, W.; Kownatzki, R.; Hedrich, H.J. Proinflammatory cytokines trigger MUC gene expression and mucin release in the intestinal cancer cell line LS180. Inflamm. Res. 2000, 49, 162–169. [Google Scholar] [CrossRef]
- Jarry, A.; Vallette, G.; Branka, J.E.; Laboisse, C. Direct secretory effect of interleukin-1 via type I receptors in human colonic mucous epithelial cells (HT29-C1.16E). Gut 1996, 38, 240–242. [Google Scholar] [CrossRef]
- Stringer, A.M.; Gibson, R.J.; Logan, R.M.; Bowen, J.M.; Yeoh, A.S.; Keefe, D.M. Faecal microflora and beta-glucuronidase expression are altered in an irinotecan-induced diarrhea model in rats. Cancer Biol. Ther. 2008, 7, 1919–1925. [Google Scholar] [CrossRef]
- Stringer, A.M.; Gibson, R.J.; Bowen, J.M.; Keefe, D.M. Chemotherapy-induced modifications to gastrointestinal microflora: Evidence and implications of change. Curr. Drug Metab. 2009, 10, 79–83. [Google Scholar] [CrossRef]
- Lin, X.B.; Dieleman, L.A.; Ketabi, A.; Bibova, I.; Sawyer, M.B.; Xue, H.; Field, C.J.; Baracos, V.E.; Ganzle, M.G. Irinotecan (CPT-11) chemotherapy alters intestinal microbiota in tumour bearing rats. PLoS One 2012, 7, e39764. [Google Scholar]
- Bowen, J.M.; Gibson, R.J.; Tsykin, A.; Stringer, A.M.; Logan, R.M.; Keefe, D.M. Gene expression analysis of multiple gastrointestinal regions reveals activation of common cell regulatory pathways following cytotoxic chemotherapy. Int. J. Cancer 2007, 121, 1847–1856. [Google Scholar] [CrossRef]
- Bowen, J.M.; Gibson, R.J.; Cummins, A.G.; Tyskin, A.; Keefe, D.M. Irinotecan changes gene expression in the small intestine of the rat with breast cancer. Cancer Chemother. Pharmacol. 2007, 59, 337–348. [Google Scholar]
- Von Bultzingslowen, I.; Adlerberth, I.; Wold, A.E.; Dahlen, G.; Jontell, M. Oral and intestinal microflora in 5-fluorouracil treated rats, translocation to cervical and mesenteric lymph nodes and effects of probiotic bacteria. Oral Microbiol. Immunol. 2003, 18, 278–284. [Google Scholar] [CrossRef]
- Zwielehner, J.; Lassl, C.; Hippe, B.; Pointner, A.; Switzeny, O.J.; Remely, M.; Kitzweger, E.; Ruckser, R.; Haslberger, A.G. Changes in human fecal microbiota due to chemotherapy analyzed by TaqMan-PCR, 454 sequencing and PCR-DGGE fingerprinting. PLoS One 2011, 6, e28654. [Google Scholar]
- De Koning, B.A.; Sluis, M.; Lindenbergh-Kortleve, D.J.; Velcich, A.; Pieters, R.; Buller, H.A.; Einerhand, A.W.; Renes, I.B. Methotrexate-induced mucositis in mucin 2-deficient mice. J. Cell. Physiol. 2007, 210, 144–152. [Google Scholar] [CrossRef]
- Saegusa, Y.; Ichikawa, T.; Iwai, T.; Goso, Y.; Okayasu, I.; Ikezawa, T.; Shikama, N.; Saigenji, K.; Ishihara, K. Changes in the mucus barrier of the rat during 5-fluorouracil-induced gastrointestinal mucositis. Scand. J. Gastroenterol. 2008, 43, 59–65. [Google Scholar] [CrossRef]
- Verburg, M.; Renes, I.B.; Meijer, H.P.; Taminiau, J.A.; Buller, H.A.; Einerhand, A.W.; Dekker, J. Selective sparing of goblet cells and paneth cells in the intestine of methotrexate-treated rats. Am. J. Physiol. Gastrointest. Liver Physiol. 2000, 279, G1037–G1047. [Google Scholar]
- Logan, R.M.; Stringer, A.M.; Bowen, J.M.; Gibson, R.J.; Sonis, S.T.; Keefe, D.M. Is the pathobiology of chemotherapy-induced alimentary tract mucositis influenced by the type of mucotoxic drug administered? Cancer Chemother. Pharmacol. 2009, 63, 239–251. [Google Scholar] [CrossRef]
- Melo, M.L.; Brito, G.A.; Soares, R.C.; Carvalho, S.B.; Silva, J.V.; Soares, P.M.; Vale, M.L.; Souza, M.H.; Cunha, F.Q.; Ribeiro, R.A. Role of cytokines (TNF-alpha, IL-1beta and KC) in the pathogenesis of CPT-11-induced intestinal mucositis in mice: Effect of pentoxifylline and thalidomide. Cancer Chemother. Pharmacol. 2008, 61, 775–784. [Google Scholar] [CrossRef]
- Curra, M.; Martins, M.A.; Lauxen, I.S.; Pellicioli, A.C.; Sant’ana Filho, M.; Pavesi, V.C.; Carrard, V.C.; Martins, M.D. Effect of topical chamomile on immunohistochemical levels of IL-1beta and TNF-alpha in 5-fluorouracil-induced oral mucositis in hamsters. Cancer Chemother. Pharmacol. 2013, 71, 293–299. [Google Scholar] [CrossRef]
- Soares, P.M.; Mota, J.M.; Souza, E.P.; Justino, P.F.; Franco, A.X.; Cunha, F.Q.; Ribeiro, R.A.; Souza, M.H. Inflammatory intestinal damage induced by 5-fluorouracil requires IL-4. Cytokine 2013, 61, 46–49. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Stringer, A.M. Interaction between Host Cells and Microbes in Chemotherapy-Induced Mucositis. Nutrients 2013, 5, 1488-1499. https://doi.org/10.3390/nu5051488
Stringer AM. Interaction between Host Cells and Microbes in Chemotherapy-Induced Mucositis. Nutrients. 2013; 5(5):1488-1499. https://doi.org/10.3390/nu5051488
Chicago/Turabian StyleStringer, Andrea M. 2013. "Interaction between Host Cells and Microbes in Chemotherapy-Induced Mucositis" Nutrients 5, no. 5: 1488-1499. https://doi.org/10.3390/nu5051488