The Effects of Multivitamin Supplementation on Diurnal Cortisol Secretion and Perceived Stress
Abstract
:1. Introduction
2. Methods
2.1. Participants
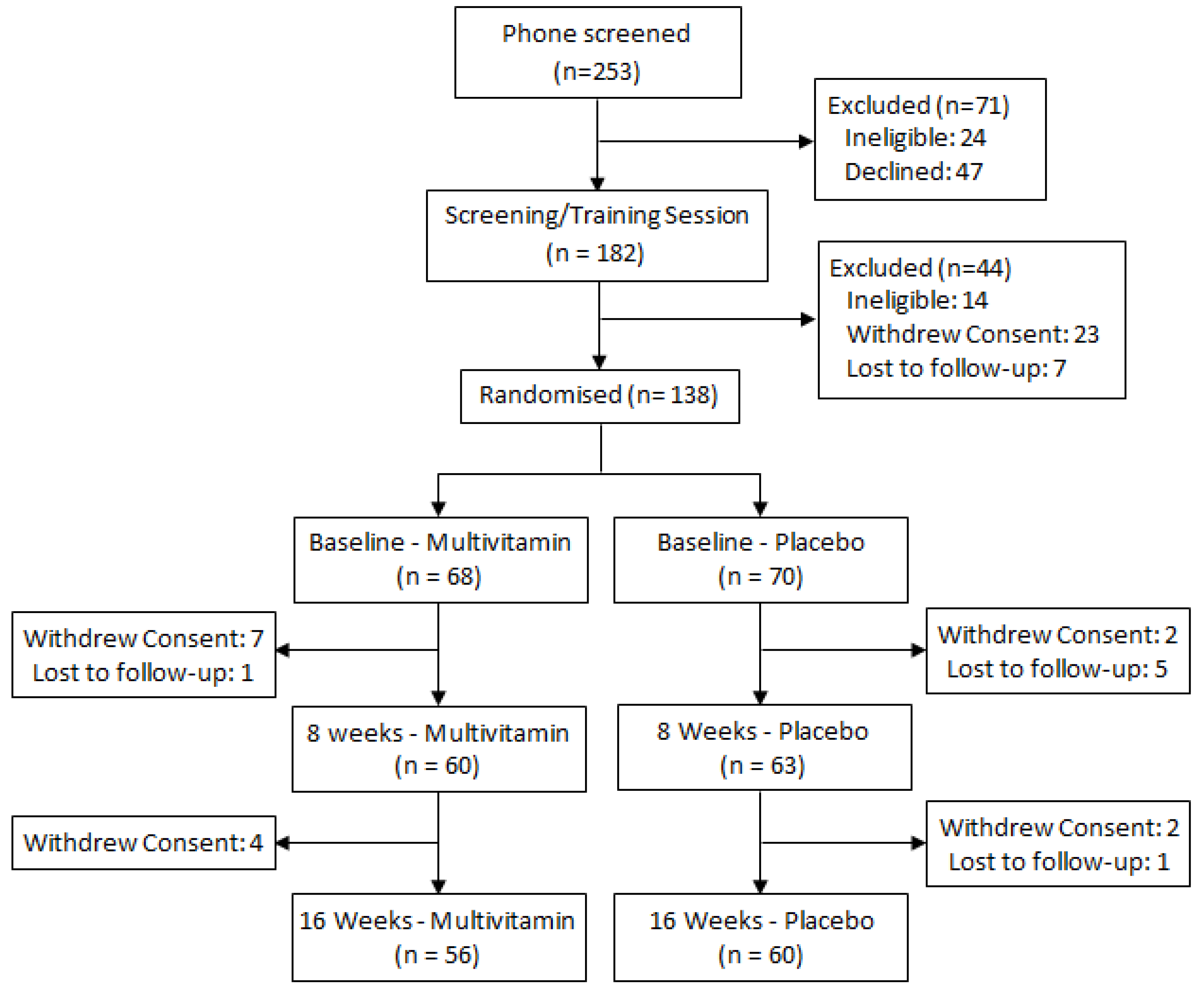
2.2. Treatments and Randomization
2.3. Salivary Cortisol Collection
2.4. Blood Collection
2.5. Perceived Stress
2.6. Statistical Analysis
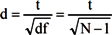
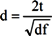
3. Results
3.1. Primary Analysis: Changes in Salivary Cortisol Parameters in Response to Multivitamin Supplementation
| Study Visit/Assay Time | Total | Males | Females | |||||
|---|---|---|---|---|---|---|---|---|
| N | M ± SE | N | M ± SE | N | M ± SE | |||
| Placebo | ||||||||
| baseline | Wake | 67 | 6.908 ± 0.55 | 29 | 6.396 ± 0.836 | 38 | 7.299 ± 0.74 | |
| 15 min | 66 | 7.653 ± 0.56 | 28 | 7.353 ± 0.846 | 38 | 7.873 ± 0.76 | ||
| 30 min | 65 | 9.388 ±0.816 | 28 | 10.20 ± 1.604 | 37 | 8.772 ± 0.77 | ||
| Bedtime | 58 | 1.337 ± 0.15 | 23 | 1.020 ± 0.149 | 35 | 1.546 ± 0.22 | ||
| 8-weeks | Wake | 60 | 7.383 ± 0.74 | 26 | 8.460 ± 1.399 | 34 | 6.559 ± 0.74 | |
| 15 min | 60 | 8.763 ± 0.75 | 25 | 9.207 ± 1.082 | 35 | 8.446 ± 1.04 | ||
| 30 min | 57 | 9.534 ± 0.94 | 23 | 8.556 ± 1.342 | 34 | 10.20 ± 1.30 | ||
| Bedtime | 48 | 2.248 ± 0.35 | 17 | 2.257 ± 0.615 | 31 | 2.244 ± 0.44 | ||
| 16-weeks | Wake | 53 | 7.983 ± 0.52 | 23 | 8.052 ± 0.897 | 30 | 7.930 ± 0.63 | |
| 15 min | 53 | 8.749 ± 0.66 | 23 | 9.444 ± 1.196 | 30 | 8.216 ± 0.73 | ||
| 30 min | 53 | 9.540 ± 0.75 | 23 | 8.616 ± 1.046 | 30 | 10.25 ± 1.06 | ||
| Bedtime | 49 | 1.453 ± 0.31 | 21 | 1.025 ± 0.105 | 28 | 1.774 ± 0.53 | ||
| Multivitamin | ||||||||
| baseline | Wake | 61 | 7.696 ± 0.66 | 27 | 6.788 ± 0.789 | 34 | 8.418 ± 0.99 | |
| 15 min | 62 | 8.093 ± 0.60 | 28 | 7.117 ±0.735 | 34 | 8.897 ± 0.89 | ||
| 30 min | 60 | 10.18 ± 0.80 | 26 | 8.694 ± 1.024 | 34 | 11.32 ± 1.16 | ||
| Bedtime | 56 | 1.597 ± 0.23 | 24 | 1.840 ± 0.473 | 32 | 1.415 ± 0.20 | ||
| 8-weeks | Wake | 53 | 6.962 ± 0.54 | 23 | 6.024 ± 0.617 | 30 | 7.682 ± 0.81 | |
| 15 min | 55 | 8.049 ± 0.64 | 24 | 6.865 ± 0.743 | 31 | 8.966 ± 0.95 | ||
| 30 min | 55 | 9.399 ± 0.69 | 24 | 7.970 ± 0.795 | 31 | 10.51 ± 1.02 | ||
| Bedtime | 53 | 1.739 ± 0.27 | 24 | 2.024 ± 0.546 | 29 | 1.504 ± 0.21 | ||
| 16-weeks | Wake | 53 | 7.160 ± 0.62 | 23 | 7.140 ± 0.767 | 30 | 7.176 ± 0.93 | |
| 15 min | 51 | 9.335 ± 0.89 | 22 | 10.22 ± 1.295 | 29 | 8.661 ± 1.23 | ||
| 30 min | 54 | 11.38 ± 1.14 | 24 | 11.89 ± 1.913 | 30 | 10.98 ± 1.39 | ||
| Bedtime | 47 | 2.384 ± 0.53 | 20 | 2.732 ± 1.067 | 27 | 2.126 ± 0.51 | ||
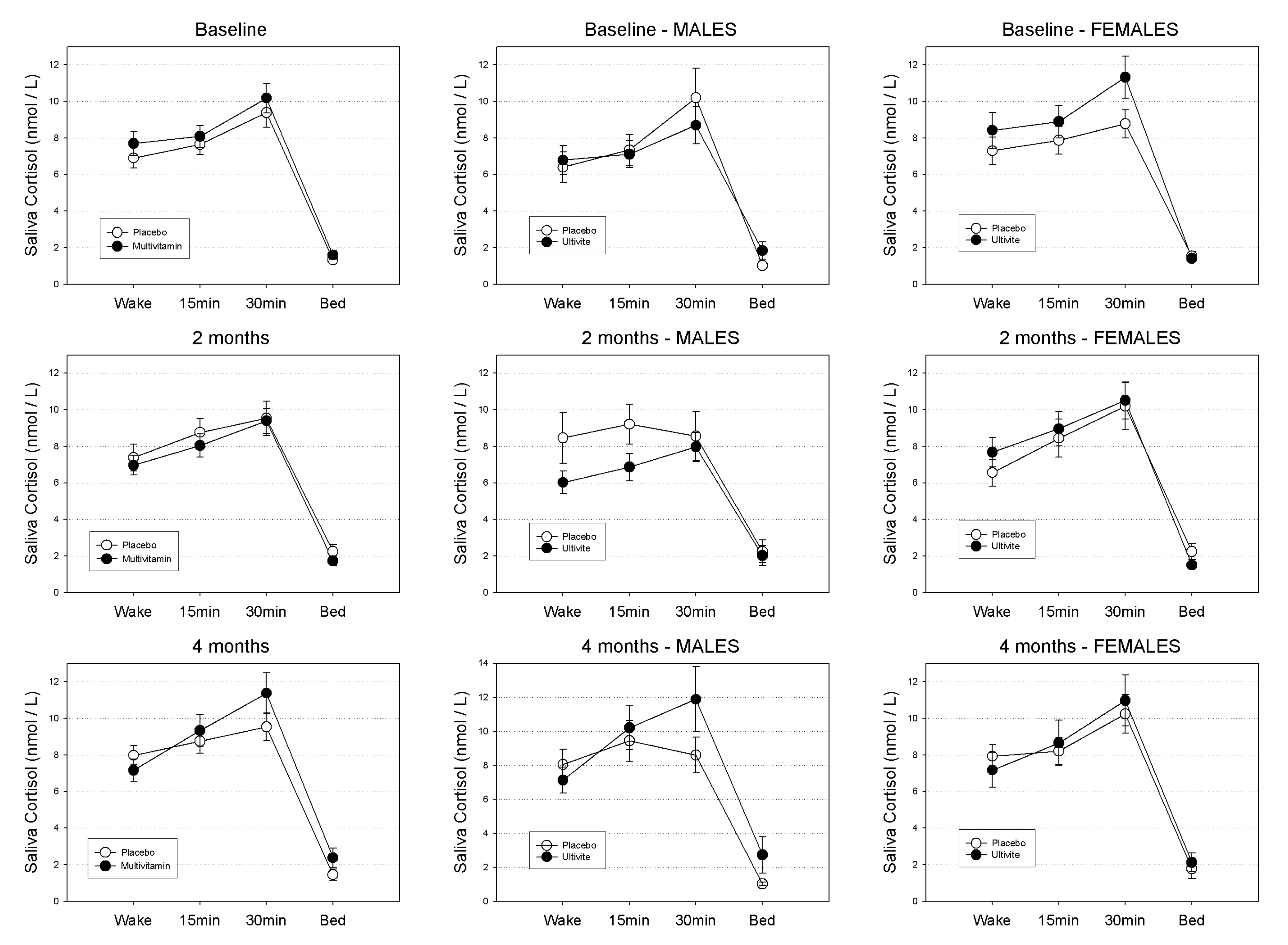
3.1.1. Waking Salivary Cortisol
3.1.2. Evening Salivary Cortisol
3.1.3. Cortisol Awakening Response (CAR)
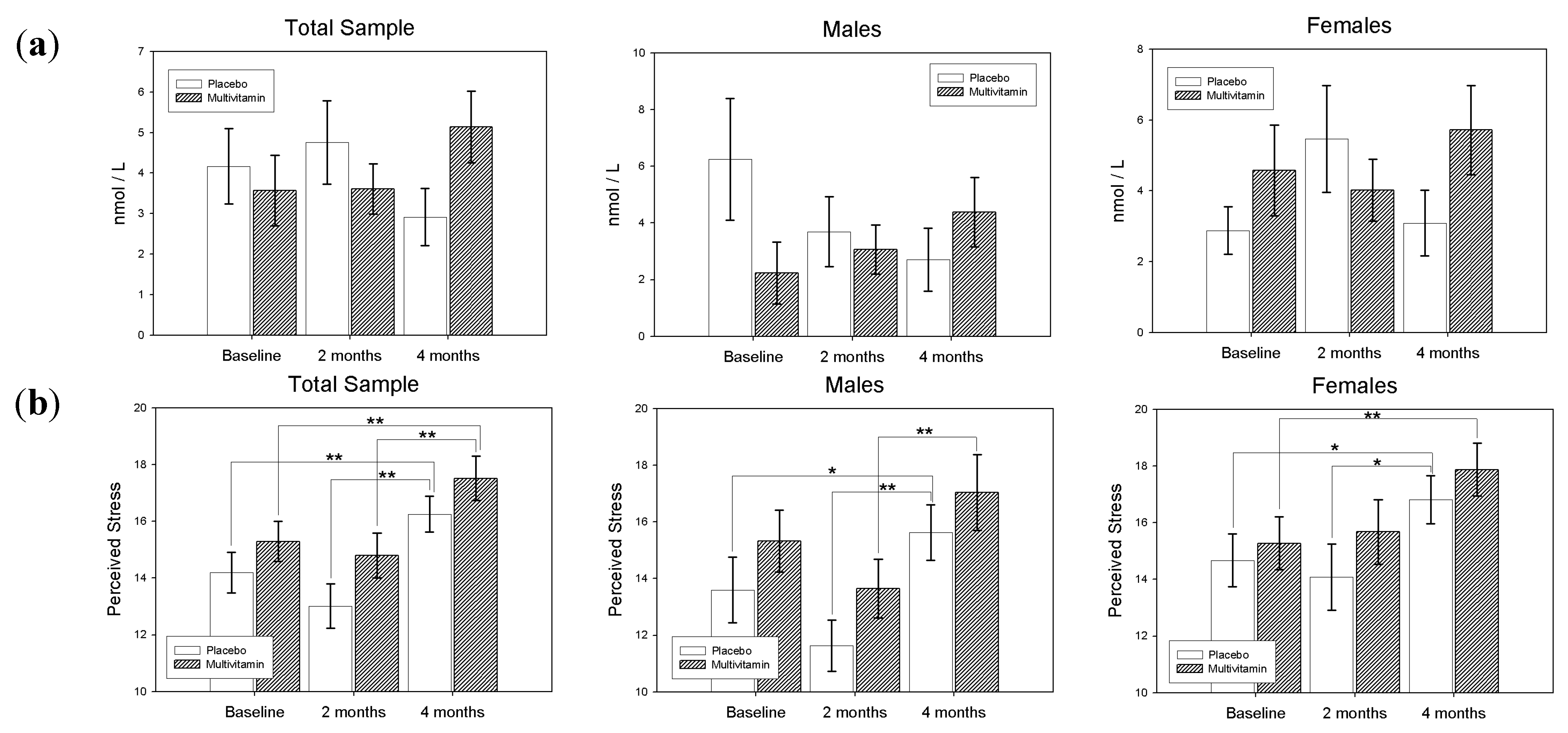
3.2. Secondary Analysis: Relationships between Salivary Cortisol, Perceived Stress and Blood Levels of B Vitamins and Homocysteine
3.2.1. Perceived Stress
| Treatment | Baseline | 8-Weeks | 16-Weeks | |||||
|---|---|---|---|---|---|---|---|---|
| N | M (SE) | N | M (SE) | N | M (SE) | |||
| Perceived Stress Scale | ||||||||
| Total | Placebo | 69 | 14.20 (0.72) | 62 | 13.02 (0.78) | 58 | 16.26 (0.64) | |
| MV | 64 | 15.30 (0.70) | 60 | 14.80 (0.79) | 56 | 17.52 (0.78) | ||
| Males | Placebo | 30 | 13.60 (1.16) | 27 | 11.63 (0.91) | 27 | 15.63 (0.98) | |
| MV | 28 | 15.32 (1.09) | 26 | 13.65 (1.03) | 24 | 17.04 (1.34) | ||
| Females | Placebo | 39 | 14.67 (0.93) | 35 | 14.09 (1.17) | 31 | 16.81 (0.85) | |
| MV | 36 | 15.28 (0.94) | 34 | 15.68 (1.14) | 32 | 17.88 (0.94) | ||
| Blood Measures | ||||||||
| B6 | Placebo | 50 | 114.08 (10.27) | 121.32 (19.32) | 130.84 (24.14) | |||
| MV | 43 | 111.70 (9.36) | 631.40 (38.17) | 567.67 (31.80) | ||||
| B12 | Placebo | 50 | 314.60 (14.48) | 323.9000 (14.84) | 318.9400 (15.46) | |||
| MV | 44 | 289.25 (12.11) | 371.0909 (18.44) | 391.9773 (15.32) | ||||
| RCF | Placebo | 49 | 944.96 (21.94) | 952.55 (26.09) | 1039.65 (32.38) | |||
| MV | 41 | 933.27 (34.99) | 1051.44 (46.11) | 1237.05 (48.43) | ||||
| HCy | Placebo | 51 | 10.6137 (0.22) | 10.8725 (0.29) | 10.4098 (0.33) | |||
| MV | 45 | 10.8444 (0.35) | 9.8578 (0.27) | 9.2244 (0.27) | ||||
3.2.2. B-Vitamins and Homocysteine
| Study Visit | Waking | Evening | CAR | PSS | |||||
|---|---|---|---|---|---|---|---|---|---|
| r | p | r | p | r | p | r | p | ||
| B6 | baseline | 0.015 | ns | 0.121 | ns | 0.032 | ns | 0.054 | ns |
| 8-weeks | −0.002 | ns | −0.100 | ns | −0.092 | ns | −0.051 | ns | |
| 16-weeks | −0.138 | ns | −0.142 | ns | 0.205 + | 0.074 | 0.050 | ns | |
| RCF | baseline | 0.042 | ns | 0.059 | ns | 0.047 | ns | 0.148 + | 0.098 |
| 8-weeks | −0.177 | ns | −0.095 | ns | −0.097 | ns | −0.057 | ns | |
| 16-weeks | −0.019 | ns | −0.060 | ns | 0.268 * | 0.019 | 0.151 | ns | |
| B12 | baseline | −0.031 | ns | 0.077 | ns | −0.087 | ns | 0.130 | ns |
| 8-weeks | 0.013 | ns | −0.085 | ns | −0.102 | ns | −0.125 | ns | |
| 16-weeks | −0.124 | ns | −0.218 + | 0.060 | 0.091 | ns | −0.058 | ns | |
| HCy | baseline | 0.022 | ns | −0.132 | ns | 0.112 | ns | −0.135 | ns |
| 8-weeks | 0.313 ** | 0.003 | −0.036 | ns | −0.081 | ns | 0.052 | ns | |
| 16-weeks | −0.046 | ns | −0.187 | ns | 0.035 | ns | −0.045 | ns | |
| PSS | baseline | 0.046 | ns | −0.032 | ns | −0.136 | ns | - | - |
| 8-weeks | −0.061 | ns | −0.081 | ns | 0.090 | ns | - | - | |
| 16-weeks | 0.064 | ns | 0.144 | ns | 0.046 | ns | - | - | |
4. Discussion
5. Conclusions
Conflicts of Interest
References
- Kudielka, B.M.; Kirschbaum, C. Sex differences in HPA axis responses to stress: A review. Biol. Psychol. 2005, 69, 113–132. [Google Scholar] [CrossRef]
- Pruessner, J.C.; Wolf, O.T.; Hellhammer, D.H.; Buske-Kirschbaum, A.; von Auer, K.; Jobst, S.; Kaspers, F.; Kirschbaum, C. Free cortisol levels after awakening: A reliable biological marker for the assessment of adrenocortical activity. Life Sci. 1997, 61, 2539–2549. [Google Scholar] [CrossRef]
- Saxbe, D.E. A field (researcher’s) guide to cortisol: Tracking HPA axis functioning in everyday life. Health Psychol. Rev. 2008, 2, 163–190. [Google Scholar] [CrossRef]
- Buckley, T.M.; Schatzberg, A.F. Review: On the interactions of the hypothalamic-pituitary-adrenal (HPA) axis and sleep: Normal HPA axis activity and circadian rhythm, exemplary sleep disorders. J. Clin. Endocrinol. Metab. 2005, 90, 3106–3114. [Google Scholar] [CrossRef]
- Kudielka, B.M.; Kirschbaum, C. Awakening cortisol responses are influenced by health status and awakening time but not by menstrual cycle phase. Psychoneuroendocrinology 2003, 28, 35–47. [Google Scholar] [CrossRef]
- Lovell, B.; Moss, M.; Wetherell, M.A. Perceived stress, common health complaints and diurnal patterns of cortisol secretion in young, otherwise healthy individuals. Horm. Behav. 2011, 60, 301–305. [Google Scholar] [CrossRef]
- García-Prieto, M.D.; Tébar, F.J.; Nicolás, F.; Larqué, E.; Zamora, S.; Garaulet, M. Cortisol secretary pattern and glucocorticoid feedback sensitivity in women from a Mediterranean area: Relationship with anthropometric characteristics, dietary intake and plasma fatty acid profile. Clin. Endocrinol. 2007, 66, 185–191. [Google Scholar] [CrossRef]
- Heaney, J.L.J.; Phillips, A.C.; Carroll, D. Aging, health behaviors, and the diurnal rhythm and awakening response of salivary cortisol. Exp. Aging Res. 2012, 38, 295–314. [Google Scholar] [CrossRef]
- Michels, N.; Sioen, I.; Braet, C.; Huybrechts, I.; Vanaelst, B.; Wolters, M.; de Henauw, S. Relation between salivary cortisol as stress biomarker and dietary pattern in children. Psychoneuroendocrinology 2013, 38, 1512–1520. [Google Scholar] [CrossRef]
- Berg, A.L.; Rafnsson, A.T.; Johannsson, M.; Hultberg, B.; Arnadottir, M. The effects of adrenocorticotrophic hormone and cortisol on homocysteine and vitamin B concentrations. Clin. Chem. Lab. Med. 2006, 44, 628–631. [Google Scholar]
- Kennedy, D.O.; Veasey, R.; Watson, A.; Dodd, F.; Jones, E.; Maggini, S.; Haskell, C.F. Effects of high-dose B vitamin complex with vitamin C and minerals on subjective mood and performance in healthy males. Psychopharmacology 2010, 211, 55–68. [Google Scholar] [CrossRef]
- Haskell, C.F.; Scholey, A.B.; Jackson, P.A.; Elliott, J.M.; Defeyter, M.A.; Greer, J.; Robertson, B.C.; Buchanan, T.; Tiplady, B.; Kennedy, D.O. Cognitive and mood effects in healthy children during 12 weeks’ supplementation with multi-vitamin/minerals. Br. J. Nutr. 2008, 100, 1086–1096. [Google Scholar] [CrossRef]
- Kennedy, D.O.; Haskell, C.F.; Robertson, B.; Reay, J.; Brewster-Maund, C.; Luedemann, J.; Maggini, S.; Ruf, M.; Zangara, A.; Scholey, A.B. Improved cognitive performance and mental fatigue following a multi-vitamin and mineral supplement with added guaraná (Paullinia cupana). Appetite 2008, 50, 506–513. [Google Scholar] [CrossRef]
- Benton, D.; Haller, J.; Fordy, J. Vitamin supplementation for 1 year improves mood. Neuropsychobiology 1995, 32, 98–105. [Google Scholar] [CrossRef]
- Schlebusch, L.; Bosch, B.A.; Polglase, G.; Kleinschmidt, I.; Pillay, B.J.; Cassimjee, M.H. A double-blind, placebo-controlled, double-centre study of the effects of an oral multivitamin-mineral combination on stress. S. Afr. Med. J. 2000, 90, 1216–1223. [Google Scholar]
- Carroll, D.; Ring, C.; Suter, M.; Willemsen, G. The effects of an oral multivitamin combination with calcium, magnesium, and zinc on psychological well-being in healthy young male volunteers: A double-blind placebo-controlled trial. Psychopharmacology 2000, 150, 220–225. [Google Scholar] [CrossRef]
- Haskell, C.F.; Robertson, B.; Jones, E.; Forster, J.; Jones, R.; Wilde, A.; Maggini, S.; Kennedy, D.O. Effects of a multi-vitamin/mineral supplement on cognitive function and fatigue during extended multi-tasking. Hum. Psychopharmacol. 2010, 25, 448–461. [Google Scholar] [CrossRef]
- Wetherell, M.A.; Sidgreaves, M.C. Short communication: Secretory immunoglobulin-A reactivity following increases in workload intensity using the Defined Intensity Stressor Simulation (DISS). Stress Health 2005, 21, 99–106. [Google Scholar] [CrossRef]
- Harris, E.; Kirk, J.; Rowsell, R.; Vitetta, L.; Sali, A.; Scholey, A.B.; Pipingas, A. The effect of multivitamin supplementation on mood and stress in healthy older men. Hum. Psychopharmacol. 2011, 26, 560–567. [Google Scholar] [CrossRef]
- Stough, C.; Scholey, A.; Lloyd, J.; Spong, J.; Myers, S.; Downey, L.A. The effect of 90 day administration of a high dose vitamin B-complex on work stress. Hum. Psychopharmacol. 2011, 26, 470–476. [Google Scholar] [CrossRef]
- Rucklidge, J.J.; Andridge, R.; Gorman, B.; Blampied, N.; Gordon, H.; Boggis, A. Shaken but unstirred? Effects of micronutrients on stress and trauma after an earthquake: RCT evidence comparing formulas and doses. Hum. Psychopharmacol. Clin. Exp. 2012, 27, 440–454. [Google Scholar] [CrossRef]
- Long, S.-J.; Benton, D. Effects of vitamin and mineral supplementation on stress, mild psychiatric symptoms, and mood in nonclinical samples: A meta-analysis. Psychosom. Med. 2013, 75, 144–153. [Google Scholar] [CrossRef]
- Miller, A.L. The methionine-homocysteine cycle and its effects on cognitive diseases. Altern. Med. Rev. 2003, 8, 7–19. [Google Scholar]
- Kruman, I.I.; Culmsee, C.; Chan, S.L.; Kruman, Y.; Guo, Z.; Penix, L.; Mattson, M.P. Homocysteine elicits a DNA damage response in neurons that promotes apoptosis and hypersensitivity to excitotoxicity. J. Neurosci. 2000, 20, 6920–6926. [Google Scholar]
- Kang, M.G.; Koh, S.B.; Cha, B.S.; Park, J.K.; Baik, S.K.; Chang, S.J. Job stress and cardiovascular risk factors in male workers. Prev. Med. 2005, 40, 583–588. [Google Scholar] [CrossRef]
- Stoney, C.M. Plasma homocysteine levels increase in women during psychological stress. Life Sci. 1999, 64, 2359–2365. [Google Scholar] [CrossRef]
- Bottiglieri, T. Folate, vitamin B12, and neuropsychiatric disorders. Nutr. Rev. 1996, 54, 382–390. [Google Scholar] [CrossRef]
- Papakostas, G.I.; Alpert, J.E.; Fava, M. S-adenosyl-methionine in depression: A comprehensive review of the literature. Curr. Psychiatry Rep. 2003, 5, 460–466. [Google Scholar] [CrossRef]
- Williams, A.L.; Girard, C.; Jui, D.; Sabina, A.; Katz, D.L. S-adenosylmethionine (SAMe) as treatment for depression: A systematic review. Clin. Investig. Med. 2005, 28, 132–139. [Google Scholar]
- Pipingas, A.; Camfield, D.A.; Stough, C.; Cox, K.H.; Fogg, E.; Tiplady, B.; Sarris, J.; White, D.J.; Sali, A.; Wetherell, M.A.; et al. The effects of multivitamin supplementation on mood and general well-being in healthy young adults. A laboratory and at-home mobile phone assessment. Appetite 2013, 69, 123–136. [Google Scholar] [CrossRef]
- Department of Health and Ageing; National Health and Medical Research Council. Nutrient Reference Values for Australia and New Zealand Including Recommended Dietary Intakes; Commonwealth of Australia: Canberra, Australia, 2006.
- Cohen, S.; Kamarck, T.; Mermelstein, R. A global measure of perceived stress. J. Health Soc. Behav. 1983, 24, 385–396. [Google Scholar] [CrossRef]
- Burns, V.E.; Drayson, M.; Ring, C.; Carroll, D. Perceived stress and psychological well-being are associated with antibody status after meningitis C conjugate vaccination. Psychosom. Med. 2002, 64, 963–970. [Google Scholar] [CrossRef]
- The SAS System for Windows, release 9.2; SAS Institute: Cary, NC, USA, 2011.
- Rosenthal, R.; Rosnow, R.L. Essentials of Behavioral Research: Methods and Data Analysis, 2nd ed.; McGraw Hill: New York, NY, USA, 1991. [Google Scholar]
- Pruessner, J.C.; Hellhammer, D.H.; Kirschbaum, C. Burnout, perceived stress, and cortisol responses to awakening. Psychosom. Med. 1999, 61, 197–204. [Google Scholar]
- Schulz, P.; Kirschbaum, C.; Prüsner, J.; Hellhammer, D. Increased free cortisol secretion after awakening in chronically stressed individuals due to work overload. Stress Med. 1998, 14, 91–97. [Google Scholar] [CrossRef]
- Wüst, S.; Wolf, J.; Hellhammer, D.H.; Federenko, I.; Schommer, N.; Kirschbaum, C. The cortisol awakening response—Normal values and confounds. Noise Health 2000, 7, 77–85. [Google Scholar]
- Steptoe, A.; Cropley, M.; Griffith, J.; Kirschbaum, C. Job strain and anger expression predict early morning elevations in salivary cortisol. Psychosom. Med. 2000, 62, 286–292. [Google Scholar]
- Chida, Y.; Steptoe, A. Cortisol awakening response and psychosocial factors: A systematic review and meta-analysis. Biol. Psychol. 2009, 80, 265–278. [Google Scholar] [CrossRef]
- Clow, A.; Thorn, L.; Evans, P.; Hucklebridge, F. The awakening cortisol response: Methodological issues and significance. Stress 2004, 7, 29–37. [Google Scholar] [CrossRef]
- Powell, D.J.; Schlotz, W. Daily life stress and the cortisol awakening response: Testing the anticipation hypothesis. PLoS One 2012, 7, e52067. [Google Scholar] [CrossRef]
- Kunz-Ebrecht, S.R.; Kirschbaum, C.; Marmot, M.; Stepto, A. Differences in cortisol awakening response on work days and weekends in women and men from the Whitehall II cohort. Psychoneuroendocrinology 2004, 29, 516–528. [Google Scholar] [CrossRef]
- Rohleder, N.; Beulen, S.E.; Chen, E.; Wolf, J.M.; Cirschbaum, C. Stress on the dance floor: The cortisol stress response to social-evaluative threat in competitive ballroom dancers. Personal. Soc. Psychol. Bull. 2007, 33, 69–84. [Google Scholar] [CrossRef]
- Izawa, S.; Saito, K.; Shirotsuki, K.; Sugaya, N.; Nomura, S. Effects of prolonged stress on salivary cortisol and dehydroepiandrosterone: A study of a two-week teaching practice. Psychoneuroendocrinology 2012, 37, 852–858. [Google Scholar] [CrossRef]
- Walker, S.; O’Connor, D.B.; Schaefer, A.; Talbot, D.; Hendrickx, H. The cortisol awakening response: Associations with trait anxiety and stress reactivity. Personal. Individ. Differ. 2011, 51, 123–127. [Google Scholar] [CrossRef]
- Adam, E.K.; Hawkley, L.C.; Kudielka, B.M.; Cacioppo, J.T. Day-to-day dynamics of experience-cortisol associations in a population-based sample of older adults. Proc. Natl. Acad. Sci. USA 2006, 103, 17058–17063. [Google Scholar] [CrossRef]
- Thorn, L.; Hucklebridge, F.; Esgate, A.; Evans, P.; Clow, A. The effect of dawn simulation on the cortisol response to awakening in healthy participants. Psychoneuroendocrinology 2004, 29, 925–930. [Google Scholar] [CrossRef]
- Dahlgren, A.; Kecklund, G.; Theorell, T.; Akerstedt, T. Day-to-day variation in saliva cortisol-relation with sleep, stress and self-rated health. Biol. Psychol. 2009, 82, 149–155. [Google Scholar] [CrossRef]
- Powell, D.J.H.; Liossi, C.; Moss-Morris, R.; Schlotz, W. Unstimulated cortisol secretory activity in everyday life and its relationship with fatigue and chronic fatigue syndrome: A systematic review and subset meta-analysis. Psychoneuroendocrinology 2013, 38, 2405–2422. [Google Scholar] [CrossRef]
- Manthey, L.; Leeds, C.; Giltay, E.J.; van Veen, T.; Vreeburg, S.A.; Penninx, B.W.; Zitman, F.G. Antidepressant use and salivary cortisol in depressive and anxiety disorders. Eur. Neuropsychopharmacol. 2011, 21, 691–699. [Google Scholar] [CrossRef]
- Vreeburg, S.A.; Hoogendijk, W.J.; van Pelt, J.; Derijk, R.H.; Verhagen, J.C.; van Dyck, R.; Smit, J.H.; Zitman, F.G.; Penninx, B.W. Major depressive disorder and hypothalamic-pituitary-adrenal axis activity: Results from a large cohort study. Arch. Gen. Psychiatry 2009, 66, 617–626. [Google Scholar] [CrossRef]
- Doane, L.D.; Kremen, W.S.; Eaves, L.J.; Eisen, S.A.; Hauger, R.; Hellhammer, D.; Levine, S.; Lupien, S.; Lyons, M.J.; Mendoza, S.; et al. Associations between jet lag and cortisol diurnal rhythms after domestic travel. Health Psychol. 2010, 29, 117–123. [Google Scholar] [CrossRef]
- Brand, S.; Holsboer-Trachsler, E.; Naranjo, J.R.; Schmidt, S. Influence of mindfulness practice on cortisol and sleep in long-term and short-term meditators. Neuropsychobiology 2012, 65, 109–118. [Google Scholar] [CrossRef]
- Peters, E.M.; Anderson, R.; Nieman, D.C.; Fickl, H.; Jogessar, V. Vitamin C supplementation attenuates the increases in circulating cortisol, adrenaline and anti-inflammatory polypeptides following ultramarathon running. Int. J. Sports Med. 2001, 22, 537–543. [Google Scholar] [CrossRef]
- Jezova, D.; Duncko, R.; Lassanova, M.; Kriska, M.; Moncek, F. Reduction of rise in blood pressure and cortisol release during stress by Ginkgo biloba extract (EGb 761) in healthy volunteers. J. Physiol. Pharmacol. 2002, 53, 337–348. [Google Scholar]
- Panossian, A.; Wagner, H. Stimulating effect of adaptogens: An overview with particular reference to their efficacy following single dose administration. Phytother. Res. 2005, 19, 819–838. [Google Scholar] [CrossRef]
- Schaffler, K.; Wolf, O.T.; Burkart, M. No benefit adding eleutherococcus senticosus to stress management training in stress-related fatigue/weakness, impaired work or concentration, a randomized controlled study. Pharmacopsychiatry 2013, 46, 181–190. [Google Scholar] [CrossRef]
- Rai, D.; Bhatia, G.; Sen, T.; Palit, G. Anti-stress effects of Ginkgo biloba and Panax ginseng: A comparative study. J. Pharmacol. Sci. 2003, 93, 458–464. [Google Scholar] [CrossRef]
- Stalder, T.; Hucklebridge, F.; Evans, P.; Clow, A. Use of a single case study design to examine state variation in the cortisol awakening response: Relationship with time of awakening. Psychoneuroendocrinology 2009, 34, 607–614. [Google Scholar] [CrossRef]
- Elder, G.J.; Wetherell, M.A.; Barclay, N.L.; Ellis, J.G. The cortisol awakening response—Applications and implications for sleep medicine. Sleep Med. Rev. 2013. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Camfield, D.A.; Wetherell, M.A.; Scholey, A.B.; Cox, K.H.M.; Fogg, E.; White, D.J.; Sarris, J.; Kras, M.; Stough, C.; Sali, A.; et al. The Effects of Multivitamin Supplementation on Diurnal Cortisol Secretion and Perceived Stress. Nutrients 2013, 5, 4429-4450. https://doi.org/10.3390/nu5114429
Camfield DA, Wetherell MA, Scholey AB, Cox KHM, Fogg E, White DJ, Sarris J, Kras M, Stough C, Sali A, et al. The Effects of Multivitamin Supplementation on Diurnal Cortisol Secretion and Perceived Stress. Nutrients. 2013; 5(11):4429-4450. https://doi.org/10.3390/nu5114429
Chicago/Turabian StyleCamfield, David A., Mark A. Wetherell, Andrew B. Scholey, Katherine H. M. Cox, Erin Fogg, David J. White, Jerome Sarris, Marni Kras, Con Stough, Avni Sali, and et al. 2013. "The Effects of Multivitamin Supplementation on Diurnal Cortisol Secretion and Perceived Stress" Nutrients 5, no. 11: 4429-4450. https://doi.org/10.3390/nu5114429
APA StyleCamfield, D. A., Wetherell, M. A., Scholey, A. B., Cox, K. H. M., Fogg, E., White, D. J., Sarris, J., Kras, M., Stough, C., Sali, A., & Pipingas, A. (2013). The Effects of Multivitamin Supplementation on Diurnal Cortisol Secretion and Perceived Stress. Nutrients, 5(11), 4429-4450. https://doi.org/10.3390/nu5114429




