Processing, Valorization and Application of Bio-Waste Derived Compounds from Potato, Tomato, Olive and Cereals: A Review
Abstract
1. Introduction
2. Valorization of Potato Residues
2.1. Extraction of Phenolic Compounds from Peels
2.2. Liquefaction of Potato Peels
2.3. Aroma Extraction from Liquefied Potato Peels
2.4. Ultrasound Assisted Extraction of Toxic Compounds From Potato Residues
2.4.1. Alkaloids Content in Potatoes
2.4.2. Ultrasounds Assisted Extraction of Alkaloids from Potato Peel
2.5. Production of Food Microorganisms Using Potato Pulp and Process Water
2.6. Protein Extraction from Potato Fruit Juice
2.7. Purification of Potato Pulp
3. Valorization of Tomato Residues
3.1. Fertilizer Production from Cull Tomato and Tomato Processing Residues
3.2. Lycopene Extraction from Tomato Skins
3.3. Cutin Extraction from Tomato Skins
3.4. Further Utilization of Tomato Plant and Tomato Processing Residues
4. Valorization of Cereal Processing Residues
4.1. Cellulose Extraction from Wheat Bran and Oat Husk
4.2. Production and Extraction of Oligosaccharides and Phenolic Acids from Wheat Bran
4.2.1. Extraction of Biomolecules from Bran
4.2.2. Valorization of Ferulic Acid (FA) and Aromatic Compounds as Monomers for Polymer Synthesis
4.2.3. Valorization of Aromatic Compounds as Additives for Polymer Formulation
4.3. Production of Food Microorganisms Using Cereal Processing Residues
5. Valorization of Olive Residues
5.1. Pretreatment of Olive Pomace and Leaves
5.2. Extraction and Purification of Phenolic Compounds from Olive Pomace and Leaves
5.3. Purification of Olive Fibers
5.4. Production of Food Microorganisms Using Olive Mill Wastewaters
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pawelczyk, A. Eu policy and legislation on recycling of organic wastes to agriculture. ISAH 2005, 1, 64–71. [Google Scholar]
- Preparatory Study on Food Waste across EU 27. 2010. Available online: http://ec.europa.eu/environment/eussd/pdf/bio_foodwaste_report.pdf (accessed on 15 August 2017).
- Food and Agriculture Organization of the United Nations. Food Loss and Food Waste. Available online: http://www.fao.org/food-loss-and-food-waste/en/ (accessed on 12 December 2016).
- Mirabella, N.; Castellani, V.; Sala, S. Current options for the valorization of food manufacturing waste: A review. J. Clean. Prod. 2014, 65, 28–41. [Google Scholar] [CrossRef]
- Integrated Product Policy: Building on Environmental Life-Cycle Thinking. 2003. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:52003DC0302 (accessed on 9 March 2017).
- A Resource-Efficient Europe—Flagship Initiative under the Europe 2020 Strategy. 2011. Available online: https://www.eea.europa.eu/policy-documents/a-resource-efficient-europe-flagship (accessed on 15 August 2017).
- European Commission. Innovating for Sustainable Growth: A Bioeconomy for Europe. 2012. Available online: https://www.eea.europa.eu/policy-documents/innovating-for-sustainable-growth-a (accessed on 15 August 2017).
- IndexMundi. Agricultural Production, Supply, and Distribution. Available online: http://www.indexmundi.com/ (accessed on 9 March 2017).
- FAOSTAT Crops. Available online: http://www.fao.org/faostat/en/#home (accessed on 9 March 2017).
- Baiano, A. Recovery of biomolecules from food wastes—A review. Molecules 2014, 19, 14821–14842. [Google Scholar] [CrossRef] [PubMed]
- Guil-Guerrero, J.L.; Ramos, L.; Moreno, C.; Zuniga-Paredes, J.C.; Carlosama-Yepez, M.; Ruales, P. Antimicrobial activity of plant-food by-products: A review focusing on the tropics. Livest. Sci. 2016, 189, 32–49. [Google Scholar] [CrossRef]
- Kalogeropoulos, N.; Chiou, A.; Pyriochou, V.; Peristeraki, A.; Karathanos, V.T. Bioactive phytochemicals in industrial tomatoes and their processing byproducts. LWT Food Sci. Technol. 2012, 49, 213–216. [Google Scholar] [CrossRef]
- Meuser, F.; Walther, G.; Dörfer, J. Design of a New Process for the Extraction of Soluble Beta-Glucan Products from Oat Bran. In Proceedings of the 3rd International Congress’ Flour-Bread 05’and 5th Croatian Congress of Cereal Technologists, Opatija, Croatia, 26–29 October 2005; Faculty of Food Technology, University of Josip Juraj Strossmayer: Osijek, Croatia; pp. 153–161. [Google Scholar]
- Gómez-Caravaca, A.M.; Verardo, V.; Bendini, A.; Toschi, T.G. From wastes to added value by-products: An overview on chemical composition and healthy properties of bioactive compounds of olive oil chain by-products. In Virgin Olive Oil: Production, Composition, Uses and Benefits for Man; De Leonardis, A., Ed.; Nova Science Publishers: New York, NY, USA, 2014; pp. 301–334. [Google Scholar]
- Dordevic, T.; Antov, M. Wheat chaff utilization: Evaluation of antioxidant capacity of waste streams generated by different pretreatments. Ind. Crop Prod. 2016, 94, 649–657. [Google Scholar] [CrossRef]
- Kong, K.W.; Khoo, H.E.; Prasad, K.N.; Ismail, A.; Tan, C.P.; Rajab, N.F. Revealing the power of the natural red pigment lycopene. Molecules 2010, 15, 959–987. [Google Scholar] [CrossRef] [PubMed]
- Lazaridou, A.; Biliaderis, C.G. Molecular aspects of cereal beta-glucan functionality: Physical properties, technological applications and physiological effects. J. Cereal Sci. 2007, 46, 101–118. [Google Scholar] [CrossRef]
- Perretti, G.; Miniati, E.; Montanari, L.; Fantozzi, P. Improving the value of rice by-products by sfe. J Supercrit. Fluid. 2003, 26, 63–71. [Google Scholar] [CrossRef]
- Robles-Ramirez, M.D.; Monterrubio-Lopez, R.; Mora-Escobedo, R.; Beltran-Orozco, M.D. Evaluation of extracts from potato and tomato wastes as natural antioxidant additives. Arch. Latinoam. Nutr. 2016, 66, 66–73. [Google Scholar]
- Cioffi, G.; Pesca, M.S.; De Caprariis, P.; Braca, A.; Severino, L.; De Tommasi, N. Phenolic compounds in olive oil and olive pomace from Cilento (Campania, Italy) and their antioxidant activity. Food Chem. 2010, 121, 105–111. [Google Scholar] [CrossRef]
- Oruna-Concha, M.J.; Duckham, S.C.; Ames, J.H. Comparison of volatile compounds isolated from the skin and flesh of four potato cultivars after baking. J. Agric. Food Chem. 2001, 49, 2414–2421. [Google Scholar] [CrossRef] [PubMed]
- Ties, P.; Barringer, S. Influence of lipid content and lipoxygenase on flavor volatiles in the tomato peel and flesh. J. Food Sci. 2012, 77, C830–C837. [Google Scholar] [CrossRef] [PubMed]
- Oreopoulou, V.; Tzia, C. Utilization of plant by-products for the recovery of proteins, dietary fibers, antioxidants, and colorants. In Utilization of By-Products and Treatment of Waste in the Food Industry; Springer: Berlin, Germany, 2007; pp. 209–232. [Google Scholar]
- Sarkar, A.; Kaul, P. Evaluation of tomato processing by-products: A comparative study in a pilot scale setup. J. Food Process Eng. 2014, 37, 299–307. [Google Scholar] [CrossRef]
- Gupta, P.; Ray, J.; Aggarwal, B.K.; Goyal, P. Good processing residue analysis and its functional components as related to human health: Recent developments. Austin J. Nutr. Food Sci. 2015, 3, 1068. [Google Scholar]
- Nikkilä, M. Cereal Waste Valorization through Development of Functional Key Fibres to Innovate in Fibre Packaging Materials. Available online: http://cordis.europa.eu/project/rcn/110025_en.html (accessed on 15 August 2017).
- Mussatto, S.I.; Teixeira, J.A.; Ballesteros, L.F.; Martins, S. Use of agro-industrial wastes in solid-state fermentation processes. Ind. Waste 2012. [Google Scholar] [CrossRef]
- Eco-Cycle. Waste of Energy—Why Incineration Is Bad for Our Economy, Environment and Community USA. Available online: https://www.ecocycle.org/files/pdfs/WTE_wrong_for_environment_economy_community_by_Eco-Cycle.pdf (accessed on 15 August 2017).
- Ben Sghaier, M.; Skandrani, I.; Nasr, N.; Franca, M.-G.D.; Chekir-Ghedira, L.; Ghedira, K. Flavonoids and sesquiterpenes from tecurium ramosissimum promote antiproliferation of human cancer cells and enhance antioxidant activity: A structure-activity relationship study. Environ. Toxicol. Pharmacol. 2011, 32, 336–348. [Google Scholar] [CrossRef] [PubMed]
- Leitzmann, C.; Müller, C.; Michel, P.; Brehme, U.; Triebel, T.; Hahn, A.; Laube, H. Ernährung in Prävention und Therapie—Ein Lehrbuch; Hippokrates: Stuttgart, Germany, 2009. [Google Scholar]
- Meot-Duros, L.; Magné, C. Antioxidant activity and phenol content of Crithmum maritimum L. Leaves. Plant Physiol. Biochem. 2009, 47, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Watzl, B. Einfluss Sekundärer Pflanzenstoffe auf die Gesundheit; Deutsche Gesellschaft für Ernährung: Bonn, Germany, 2008; pp. 335–379. [Google Scholar]
- Balasundram, N.; Sundram, K.; Samman, S. Phenolic compounds in plants and agri-industrial by-products: Antioxidant activity, occurrence, and potential uses. Food Chem. 2006, 99, 191–203. [Google Scholar] [CrossRef]
- Wijngaard, H.H.; Ballay, M.; Brunton, N. The optimisation of extraction of antioxidants from potato peel by pressurised liquids. Food Chem. 2012, 133, 1123–1130. [Google Scholar] [CrossRef]
- Al-Weshahy, A.; Rao, A.V. Isolation and characterization of functional components from peel samples of six potatoes varieties growing in Ontario. Food Res. Int. 2009, 42, 1062–1066. [Google Scholar] [CrossRef]
- De Sotillo, D.R.; Hadley, M.; Holm, E.T. Potato peel waste—Stability and antioxidant activity of a freeze-dried extract. J. Food Sci. 1994, 59, 1031–1033. [Google Scholar] [CrossRef]
- Tephly, T.R. The toxicity of methanol. Life Sci. 1991, 48, 1031–1041. [Google Scholar] [CrossRef]
- NCBI. Pubchem Compound Database. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/689043 (accessed on 31 January 2017).
- Puertolas, E.; Cregenzan, O.; Luengo, E.; Alvarez, I.; Raso, J. Pulsed-electric-field-assisted extraction of anthocyanins from purple-fleshed potato. Food Chem. 2013, 136, 1330–1336. [Google Scholar] [CrossRef] [PubMed]
- Samarin, A.M.; Poorazarang, H.; Hematyar, N.; Elhamirad, A. Phenolics in potato peels: Extraction and utilization as natural antioxidants. World Appl. Sci. J. 2012, 18, 191–195. [Google Scholar]
- Pereira, R.N.; Rodrigues, R.M.; Genisheva, Z.; Oliveira, H.; de Freitas, V.; Teixeira, J.A.; Vicente, A.A. Effects of ohmic heating on extraction of food-grade phytochemicals from colored potato. LWT Food Sci. Technol. 2016, 74, 493–503. [Google Scholar] [CrossRef]
- Singh, P.P.; Saldana, M.D.A. Subcritical water extraction of phenolic compounds from potato peel. Food Res. Int. 2011, 44, 2452–2458. [Google Scholar] [CrossRef]
- Habeebullah, S.F.K.; Nielsen, N.S.; Jacobsen, C. Potato peel extracts: A potential antioxidant for omega-3-enriched products. INFORM 2012, 23, 58–59. [Google Scholar]
- De Sotillo, D.R.; Hadley, M.; Wolf-Hall, C. Potato peel extract a nonmutagenic antioxidant with potential antimicrobial activity. J. Food Sci. 1998, 63, 907–910. [Google Scholar] [CrossRef]
- Viscidi, K.A.; Dougherty, M.P.; Briggs, J.; Camire, M.E. Complex phenolic compounds reduce lipid oxidation in extruded oat cereals. LWT Food Sci. Technol. 2004, 37, 789–796. [Google Scholar] [CrossRef]
- Amado, I.R.; Franco, D.; Sanchez, M.; Zapata, C.; Vazquez, J.A. Optimisation of antioxidant extraction from solanum tuberosum potato peel waste by surface response methodology. Food Chem. 2014, 165, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Sabally, K.; Kubow, S.; Donnelly, D.J.; Gariepy, Y.; Orsat, V.; Raghavan, G.S.V. Microwave-assisted extraction of phenolic antioxidants from potato peels. Molecules 2011, 16, 2218–2232. [Google Scholar] [CrossRef] [PubMed]
- Mohdaly, A.A.A.; Hassanien, M.F.R.; Mahmoud, A.; Sarhan, M.A.; Smetanska, I. Phenolics extracted from potato, sugar beet, and sesame processing by-products. Int. J. Food Prop. 2013, 16, 1148–1168. [Google Scholar] [CrossRef]
- Arapoglou, D.; Varzakas, T.; Vlyssides, A.; Israilides, C. Ethanol production from potato peel waste (PPW). Waste Manag. 2010, 30, 1898–1902. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Shinomiya, N.; Ohba, K.; Sekikawa, M.; Oda, Y. Enzymatic hydrolysis and ethanol fermentation of by-products from potato processing plants. Food Sci. Technol. Res. 2009, 15, 653–658. [Google Scholar] [CrossRef]
- Ventura, P.; Bordado, J.C.M.; Mateus, M.M.; Galhano dos Santos, R. Upcycling potato peel waste—Data of the pre-screening of the acid-catalyzed liquefaction. Data Brief 2016, 7, 1455–1457. [Google Scholar] [CrossRef] [PubMed]
- Mateus, M.M.; Ventura, P.; Rego, A.; Mota, C.; Castanheira, I.; Bordado, J.; dos Santos, R.G. Acid liquefaction of potato (Solanum tuberosum) and sweet potato (Ipomoea batatas) cultivars peels–pre-screening of antioxidant activity/total phenolic and sugar contents. BioResources 2017, 12, 1463–1478. [Google Scholar] [CrossRef]
- Israilides, C.; Vlyssides, A.; Arapoglou, D.; Varzakas, T.; Marchant, R.; Vlysides, A. Integrated management of potato starch wastes. In Waste 2008: Waste and Resource Management—A Shared Responsibility; Marie, T., Ed.; Waste Conference Ltd.: Stratford, UK, 2008. [Google Scholar]
- Sánchez, Ó.J.; Cardona, C.A. Trends in biotechnological production of fuel ethanol from different feedstocks. Bioresour. Technol. 2008, 99, 5270–5295. [Google Scholar] [CrossRef] [PubMed]
- Briones, R.; Serrano, L.; Labidi, J. Valorization of some lignocellulosic agro-industrial residues to obtain biopolyols. J. Chem. Technol. Biotechnol. 2012, 87, 244–249. [Google Scholar] [CrossRef]
- Abdelmoez, W.; Nage, S.M.; Bastawess, A.; Ihab, A.; Yoshida, H. Subcritical water technology for wheat straw hydrolysis to produce value added products. J. Clean. Prod. 2014, 70, 68–77. [Google Scholar] [CrossRef]
- Karagöz, S.; Bhaskar, T.; Muto, A.; Sakata, Y.; Uddin, M.A. Low-temperature hydrothermal treatment of biomass: Effect of reaction parameters on products and boiling point distributions. Energy Fuels 2004, 18, 234–241. [Google Scholar] [CrossRef]
- Nagamori, M.; Funazukuri, T. Glucose production by hydrolysis of starch under hydrothermal conditions. J. Chem. Technol. Biotechnol. 2004, 79, 229–233. [Google Scholar] [CrossRef]
- Yin, S.; Tan, Z. Hydrothermal liquefaction of cellulose to bio-oil under acidic, neutral and alkaline conditions. Appl. Energy 2012, 92, 234–239. [Google Scholar] [CrossRef]
- Li, F.; Liu, L.; An, Y.; He, W.; Themelis, N.J.; Li, G. Hydrothermal liquefaction of three kinds of starches into reducing sugars. J. Clean. Prod. 2016, 112, 1049–1054. [Google Scholar] [CrossRef]
- Teranishi, R.; Kint, S. Bioactive Volatile Compounds from Plants: An Overview. Available online: http://agris.fao.org/agris-search/search.do?recordID=US9437272 (accessed on 15 August 2017).
- Galego, L.R.; Da Silva, J.P.; Almeida, V.R.; Bronze, M.R.; Boas, L.V. Preparation of novel distinct highly aromatic liquors using fruit distillates. Int. J. Food Sci. Technol. 2011, 46, 67–73. [Google Scholar] [CrossRef]
- Gounaris, Y. Biotechnology for the production of essential oils, flavours and volatile isolates. A review. Flavour Fragr. J. 2010, 25, 367–386. [Google Scholar] [CrossRef]
- Franz, S.; Willersinn, D.; Kroschel, K. Evaluating sensor effects on perception performance. In Proceedings of the IEEE Intelligent Vehicles Symposium, San Diego, CA, USA, 21–24 June 2010; pp. 634–639. [Google Scholar]
- Flavourtech. Spinning Cone Column. Available online: http://flavourtech.com/products/spinning-cone-column/ (accessed on 30 January 2017).
- Buttery, R.G.; Guadagni, D.G.; Ling, L.C. Volatile components of baked potatoes. J. Sci. Food Agric. 1973, 24, 1125–1131. [Google Scholar] [CrossRef]
- Center, I.P. Potato Facts and Figures. Available online: http://cipotato.org/potato/facts/ (accessed on 8 February 2017).
- Schieber, A.; Saldaña, M.D.A. Potato peels: A source of nutritionally and pharmacologically interesting compounds—A review. Food 2009, 3, 23–29. [Google Scholar]
- Austin, S.; Lojkowska, E.; Ehlenfeldt, K.; Kelman, A.; Helgeson, J.P. Fertile interspecific somatic hybrids of solanum: A novel source of resistance to erwinia soft rot. Phytopathology 1988, 78, 1216–1220. [Google Scholar] [CrossRef]
- Fewell, A.M.; Roddick, J.G. Interactive antifungal activity of the glycoalkaloids alpha-solanine and alpha-chaconine. Phytochemistry 1993, 33, 323–328. [Google Scholar] [CrossRef]
- Sanford, L.L.; Kobayashi, R.S.; Deahl, K.L.; Sinden, S.L. Diploid and tetraploid solanum chacoense genotypes that synthesize leptine glycoalkaloids and deter feeding by colorado potato beetle. Am. Potato J. 1997, 74, 15–21. [Google Scholar] [CrossRef]
- Morris, S.C.; Lee, T.H. The toxicity and teratogenicity of Solanaceae glycoalkaloids, particularly those of the potato (Solanum-tuberosum)—A review. Food Technol. Aust. 1984, 36, 118–124. [Google Scholar]
- Slanina, P. Solanine (glycoalkaloids) in potatoes—Toxicological evaluation. Food Chem. Toxicol. 1990, 28, 759–761. [Google Scholar] [CrossRef]
- Friedman, M.; McDonald, G.M. Postharvest changes in glycoalkaloid content of potatoes. In Impact of Processing on Food Safety; Jackson, L.S., Knize, M.G., Morgan, J.N., Eds.; Springer: Boston, MA, USA, 1999; pp. 121–143. [Google Scholar]
- Ripperger, H. Chapter two—Solanum steroid alkaloids—An update. In Alkaloids: Chemical and Biological Perspectives; Pelletier, S.W., Ed.; John Wiley and Sons: New York, NY, USA, 1998; Volume 12, pp. 103–185. [Google Scholar]
- Friedman, M. Potato glycoalkaloids and metabolites: Roles in the plant and in the diet. J. Agric. Food Chem. 2006, 54, 8655–8681. [Google Scholar] [CrossRef] [PubMed]
- Weissenberg, M.; Levy, A.; Svoboda, J.A.; Ishaaya, I. The effect of some solanum steroidal alkaloids and glycoalkaloids on larvae of the red flour beetle, tribolium castaneum, and the tobacco hornworm, manduca sexta. Phytochemistry 1998, 47, 203–209. [Google Scholar] [CrossRef]
- McGehee, D.S.; Krasowski, M.D.; Fung, D.L.; Wilson, B.; Gronert, G.A.; Moss, J. Cholinesterase inhibition by potato glycoalkaloids slows mivacurium metabolism. ASA 2000, 93, 510–519. [Google Scholar] [CrossRef]
- Michalska, L.; Nagel, G.; Swiniarski, E.; Żydowo, M.M. The effect of α-solanine on the active calcium transport in rat intestine. Gen. Pharmacol. Vasc. Syst. 1985, 16, 69–70. [Google Scholar] [CrossRef]
- Blankemeyer, J.T.; Atherton, R.; Friedman, M. Effect of potato glycoalkaloids Alpha-chaconine and Alpha-solanine on sodium active transport in frog skin. J. Agric. Food Chem. 1995, 43, 636–639. [Google Scholar] [CrossRef]
- Toyoda, M.; Rausch, W.D.; Inoue, K.; Ohno, Y.; Fujiyama, Y.; Takagi, K.; Saito, Y. Comparison of solanaceous glycoalkaloids-evoked Ca2+ influx in different types of cultured cells. Toxicol. In Vitro 1991, 5, 347–351. [Google Scholar] [CrossRef]
- Romanucci, V.; Pisanti, A.; Di Fabio, G.; Davinelli, S.; Scapagnini, G.; Guaragna, A.; Zarrelli, A. Toxin levels in different variety of potatoes: Alarming contents of α-chaconine. Phytochem. Lett. 2016, 16, 103–107. [Google Scholar] [CrossRef]
- Phillips, B.J.; Hughes, J.A.; Phillips, J.C.; Walters, D.G.; Anderson, D.; Tahourdin, C.S.M. A study of the toxic hazard that might be associated with the consumption of green potato tops. Food Chem. Toxicol. 1996, 34, 439–448. [Google Scholar] [CrossRef]
- Hossain, M.B.; Rai, D.K.; Brunton, N.P. Optimisation and validation of ultra-high performance liquid chromatographic-tandem mass spectrometry method for qualitative and quantitative analysis of potato steroidal alkaloids. J. Chrom. B 2015, 997, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Chang, K. Polyphenol Antioxidants from Potato Peels: Extraction Optimization and Application to Stabilizing Lipid Oxidation in Foods. In Proceedings of the National Conference on Undergraduate Research (NCUR), New York, NY, USA, 30 March–2 April 2011. [Google Scholar]
- Glisic, S.B.; Ristic, M.; Skala, D.U. The combined extraction of sage (Salvia officinalis L.): Ultrasound followed by supercritical CO2 extraction. Ultrason. Sonochem. 2011, 18, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Vinatoru, M. An overview of the ultrasonically assisted extraction of bioactive principles from herbs. Ultrason. Sonochem. 2001, 8, 303–313. [Google Scholar] [CrossRef]
- Wijngaard, H.; Hossain, M.B.; Rai, D.K.; Brunton, N. Techniques to extract bioactive compounds from food by-products of plant origin. Food Res. Int. 2012, 46, 505–513. [Google Scholar] [CrossRef]
- Ma, Y.; Ye, X.; Hao, Y.; Xu, G.; Xu, G.; Liu, D. Ultrasound-assisted extraction of hesperidin from penggan (Citrus reticulata) peel. Ultrason. Sonochem. 2008, 15, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Veličković, D.T.; Milenović, D.M.; Ristić, M.S.; Veljković, V.B. Ultrasonic extraction of waste solid residues from the Salvia sp. essential oil hydrodistillation. Biochem. Eng. J. 2008, 42, 97–104. [Google Scholar] [CrossRef]
- Virot, M.; Tomao, V.; Le Bourvellec, C.; Renard, C.M.C.G.; Chemat, F. Towards the industrial production of antioxidants from food processing by-products with ultrasound-assisted extraction. Ultrason. Sonochem. 2010, 17, 1066–1074. [Google Scholar] [CrossRef] [PubMed]
- Chemat, F.; Rombaut, N.; Sicaire, A.G.; Meullemiestre, A.; Fabiano-Tixier, A.S.; Abert-Vian, M. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar] [CrossRef] [PubMed]
- Mason, T.J. Sonochemistry and sonoprocessing: The link, the trends and (probably) the future. Ultrason. Sonochem. 2003, 10, 175–179. [Google Scholar] [CrossRef]
- Toma, M.; Vinatoru, M.; Paniwnyk, L.; Mason, T.J. Investigation of the effects of ultrasound on vegetal tissues during solvent extraction. Ultrason. Sonochem. 2001, 8, 137–142. [Google Scholar] [CrossRef]
- Tang, D.-M.; Zhu, C.-F.; Zhong, S.-A.; Zhou, M.-D. Extraction of naringin from pomelo peels as dihydrochalcone’s precursor. J. Separ. Sci. 2011, 34, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Vilkhu, K.; Mawson, R.; Simons, L.; Bates, D. Applications and opportunities for ultrasound assisted extraction in the food industry—A review. Innov. Food Sci. Emerg. Technol. 2008, 9, 161–169. [Google Scholar] [CrossRef]
- Pingret, D.; Fabiano-Tixier, A.S.; Chemat, F. Ultrasound-assisted extraction. In Natural Product Extraction: Principles and Applications; Rostagno, M.A., Prado, J.M., Eds.; Royal Society of Chemistry: London, UK, 2013; pp. 89–112. [Google Scholar]
- Vinatoru, M. Ultrasonically assisted extraction (UAE) of natural products some guidelines for good practice and reporting. Ultrason. Sonochem. 2015, 25, 94–95. [Google Scholar] [CrossRef] [PubMed]
- Gallego-Juarez, J.A. High-power ultrasonic processing: Recent developments and prospective advances. Phys. Procedia 2010, 3, 35–47. [Google Scholar] [CrossRef]
- Qiyun, S.; Liang, Q. Study on the production of SCP feed from potato mash residue. Cereal Feed Ind. 2004, 9, 32–33. [Google Scholar]
- Jianmin, Yun. Study on production of SCP feed from potato residue. J. Microbiol. 1998, 19, 23–27. [Google Scholar]
- Gharsallah, N. Production of single cell protein from olive mill waste water by yeasts. Environ. Technol. 1993, 14, 391–395. [Google Scholar] [CrossRef]
- Schultz, N.; Chang, L.; Hauck, A.; Reuss, M.; Syldatk, C. Microbial production of single-cell protein from deproteinized whey concentrates. Appl. Microbiol. Biotechnol. 2006, 69, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Moon, N.J.; Hammond, E.G.; Glatz, B.A. Conversion of cheese whey and whey permeate to oil and single-cell protein1. J. Dairy Sci. 1978, 61, 1537–1547. [Google Scholar] [CrossRef]
- Nigam, P.; Vogel, M. Bioconversion of sugar industry by-products—Molasses and sugar beet pulp for single cell protein production by yeasts. Biomass Bioenergy 1991, 1, 339–345. [Google Scholar] [CrossRef]
- Tanaka, M.; Matsuno, R. Conversion of lignocellulosic materials to single-cell protein (SCP): Recent developments and problems. Enzym. Microb. Technol. 1985, 7, 197–206. [Google Scholar] [CrossRef]
- Kavšček, M.; Stražar, M.; Curk, T.; Natter, K.; Petrovič, U. Yeast as a cell factory: Current state and perspectives. Microb. Cell Fact. 2015, 14, 1. [Google Scholar] [CrossRef] [PubMed]
- Rumbold, K.; van Buijsen, H.J.J.; Overkamp, K.M.; van Groenestijn, J.W.; Punt, P.J.; van der Werf, M.J. Microbial production host selection for converting second-generation feedstocks into bioproducts. Microb. Cell Fact. 2009. [Google Scholar] [CrossRef] [PubMed]
- Ostergaard, S.; Olsson, L.; Nielsen, J. Metabolic engineering of Saccharomyces cerevisiae. Microbiol. Mol. Biol. Rev. 2000, 64, 34–50. [Google Scholar] [CrossRef] [PubMed]
- Altıntaş, M.M.; Ülgen, K.Ö.; Kırdar, B.; Önsan, Z.İ.; Oliver, S.G. Optimal substrate feeding policy for fed-batch cultures of S. cerevisiae expressing bifunctional fusion protein displaying amylolytic activities. Enzym. Microb. Technol. 2003, 33, 262–269. [Google Scholar] [CrossRef]
- Bacha, U.; Nasir, M.; Khalique, A.; Anjum, A.A.; Jabbar, M.A. Comparative assessment of various agro-industrial wastes for saccharomyces cerevisiae biomass production and its quality evaluation as single cell protein. J. Anim. Plant Sci. 2011, 21, 844–849. [Google Scholar]
- Chang, M.; Zhou, S.G.; Lu, N.; Ni, J.R. Starch processing wastewater as a new medium for production of bacillus thuringiensis. World J. Microbiol. Biotechnol. 2008, 24, 441–447. [Google Scholar] [CrossRef]
- Srisamai, S.; Srikhampa, P.; Pathom-aree, W. Production of probiotic streptomyces biomass from starchy wastewater. Chiang Mai J. Sci. 2013, 40, 294–298. [Google Scholar]
- Calleja, G.B.; Yaguchi, M.; Levy-Rick, S.; Seguin, J.R.H.; Roy, C.; Lusena, C.V. Single-cell protein production from potato starch by the yeast schwanniomyces alluvius. J. Ferment. Technol. 1986, 64, 71–75. [Google Scholar] [CrossRef]
- Kleekayai, T.; Suntornsuk, W. Production and characterization of chitosan obtained from rhizopus oryzae grown on potato chip processing waste. World J. Microbiol. Biotechnol. 2011, 27, 1145–1154. [Google Scholar] [CrossRef]
- Izmirlioglu, G.; Demirci, A. Strain selection and medium optimization for glucoamylase production from industrial potato waste by aspergillus Niger. J. Sci. Food Agric. 2016, 96, 2788–2795. [Google Scholar] [CrossRef] [PubMed]
- Pagana, I.; Morawicki, R.; Hager, T.J. Lactic acid production using waste generated from sweet potato processing. Int. J. Food Sci. Technol. 2014, 49, 641–649. [Google Scholar] [CrossRef]
- Fox, S.L.; Bala, G.A. Production of surfactant from bacillus subtilis atcc 21332 using potato substrates. Bioresour. Technol. 2000, 75, 235–240. [Google Scholar] [CrossRef]
- Wojnowska, I.; Poznanski, S.; Bednarski, W. Processing of potato protein-concentrates and their properties. J. Food Sci. 1982, 47, 167–172. [Google Scholar] [CrossRef]
- Waglay, A.; Karboune, S. Enzymatic generation of peptides from potato proteins by selected proteases and characterization of their structural properties. Biotechnol. Progr. 2016, 32, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Zwijnenberg, H.J.; Kemperman, A.J.B.; Boerrigter, M.E.; Lotz, M.; Dijksterhuis, J.F.; Poulsen, P.E.; Koops, G.-H. Native protein recovery from potato fruit juice by ultrafiltration. Desalination 2002, 144, 331–334. [Google Scholar] [CrossRef]
- Knorr, D.; Kohler, G.; Betschart, A. Potato protein concentrates: The influence of various methods of recovery upon yield, compositional and functional characteristics. J. Food Process. Preserv. 1977, 1, 235–247. [Google Scholar] [CrossRef]
- Graf, A.M.; Steinhof, R.; Lotz, M.; Tippkötter, N.; Kasper, C.; Beutel, S.; Ulber, R. Downstream-processing mit membranadsorbern zur isolierung nativer proteinfraktionen aus kartoffelfruchtwasser. Chem. Ing. Tech. 2009, 81, 267–274. [Google Scholar] [CrossRef]
- Alt, V.; Steinhof, R.; Lotz, M.; Ulber, R.; Kasper, C.; Scheper, T. Optimization of glycoalkaloid analysis for use in industrial potato fruit juice downstreaming. Eng. Life Sci. 2005, 5, 562–567. [Google Scholar] [CrossRef]
- Van Koningsveld, G.A.; Walstra, P.; Voragen, A.G.J.; Kuijpers, I.J.; Van Boekel, M.A.J.S.; Gruppen, H. Effects of protein composition and enzymatic activity on formation and properties of potato protein stabilized emulsions. J. Agric. Food Chem. 2006, 54, 6419–6427. [Google Scholar] [CrossRef] [PubMed]
- Bartova, V.; Barta, J.; Brabcova, A.; Zdrahal, Z.; Horackova, V. Amino acid composition and nutritional value of four cultivated south american potato species. J. Food Compos. Anal. 2015, 40, 78–85. [Google Scholar] [CrossRef]
- Bártová, V.; Bárta, J. Chemical composition and nutritional value of protein concentrates isolated from potato (Solanum tuberosum L.) fruit juice by precipitation with ethanol or ferric chloride. J. Agric. Food Chem. 2009, 57, 9028–9034. [Google Scholar] [CrossRef] [PubMed]
- Creusot, N.; Wierenga, P.A.; Laus, M.C.; Giuseppin, M.L.; Gruppen, H. Rheological properties of patatin gels compared with β-lactoglobulin, ovalbumin, and glycinin. J. Agric. Food Chem. 2011, 91, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Kudo, K.; Onodera, S.; Takeda, Y.; Benkeblia, N.; Shiomi, N. Antioxidative activities of some peptides isolated from hydrolyzed potato protein extract. J. Funct. Foods 2009, 1, 170–176. [Google Scholar] [CrossRef]
- Bergthaller, W.; Witt, W.; Goldau, H.P. Potato starch technology. Starch Stärke 1999, 51, 235–242. [Google Scholar] [CrossRef]
- Kinsella, J.E.; Melachouris, N. Functional properties of proteins in foods: A survey. CRC Crit. Rev. Food Sci. Nutr. 1976, 7, 219–280. [Google Scholar] [CrossRef]
- Knorr, D. Protein quality of potato and potato protein-concentrates. Lebensm.Wiss. Technol. 1978, 11, 109–115. [Google Scholar]
- Boruch, M.; Makowski, J.; Wachowicz, M.; Dubla, W. Rückgewinnung der stickstoffverbindungen aus kartoffelfruchtwasser. Food Nahrung 1989, 33, 67–76. [Google Scholar] [CrossRef]
- Edens, L.; Van der Lee, J.A.B.; Plijter, J.J. Novel Food Compositions. Patent EP0912105 A1, 12 May 1997. [Google Scholar]
- Van Koningsveld, G.A.; Gruppen, H.; de Jongh, H.H.J.; Wijngaards, G.; van Boekel, M.A.J.S.; Walstral, P.; Voragen, A.G.J. The solubility of potato proteins from industrial potato fruit juice as influenced by pH and various additives. J. Sci. Food Agric. 2002, 82, 134–142. [Google Scholar] [CrossRef]
- Gonzalez, J.M.; Lindamood, J.B.; Desai, N. Recovery of protein from potato plant waste effluents by complexation with carboxymethylcellulose. Food Hydrocoll. 1991, 4, 355–363. [Google Scholar] [CrossRef]
- Lindner, P.; Keren, R.; Bengera, I. Precipitation of proteins from potato juice with bentonite. J. Sci. Food Agric. 1981, 32, 1177–1182. [Google Scholar] [CrossRef] [PubMed]
- Knorr, D. Effect of recovery methods on yield, quality and functional-properties of potato protein-concentrates. J. Food Sci. 1980, 45, 1183–1186. [Google Scholar] [CrossRef]
- Knorr, D. Effect of recovery methods on the functionality of vegetable proteins. J. Am. Oil Chem. Soc. 1982, 59, A302. [Google Scholar]
- Meister, E.; Thompson, N.R. Physical-chemical methods for recovery of protein from waste effluent of potato-chip processing. J. Agric. Food Chem. 1976, 24, 919–923. [Google Scholar] [CrossRef]
- Kamnerdpetch, C.; Weiss, M.; Kasper, C.; Scheper, T. An improvement of potato pulp protein hydrolyzation process by the combination of protease enzyme systems. Enzym. Microb. Technol. 2007, 40, 508–514. [Google Scholar] [CrossRef]
- Cheng, Y.; Xiong, Y.L.L.; Chen, J. Fractionation, separation, and identification of antioxidative peptides in potato protein hydrolysate that enhance oxidative stability of soybean oil emulsions. J. Food Sci. 2010, 75, C760–C765. [Google Scholar] [CrossRef] [PubMed]
- Miedzianka, J.; Peksa, A.; Pokora, M.; Rytel, E.; Tajner-Czopek, A.; Kita, A. Improving the properties of fodder potato protein concentrate by enzymatic hydrolysis. Food Chem. 2014, 159, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Dabestani, S.; Arcot, J.; Chen, V. Protein recovery from potato processing water: Pre-treatment and membrane fouling minimization. J. Food Eng. 2017, 195, 85–96. [Google Scholar] [CrossRef]
- Talburt, W.F.; Smith, O. Potato Processing; AVI Publishing Company: Westport, CT, USA, 1975. [Google Scholar]
- Reboul, E.; Borel, P.; Mikail, C.; Abou, L.; Charbonnier, M.; Caris-Veyrat, C.; Goupy, P.; Portugal, H.; Lairon, D.; Amiot, M.J. Enrichment of tomato paste with 6% tomato peel increases lycopene and β-carotene bioavailability in men. J. Nutr. 2005, 135, 790–794. [Google Scholar] [PubMed]
- Mayer, F. Potato pulp: Properties, physical modification and applications. Polym. Degrad. Stab. 1998, 59, 231–235. [Google Scholar] [CrossRef]
- Kempf, W. Beitrag zur mechanischen entwässerung von kartoffelpülpe. Starch-Stärke 1980, 32, 14–20. [Google Scholar] [CrossRef]
- Hillebrandt, J.-O. Kartoffelpülpe, ein Landwirtschaftliches Abfallprodukt und ein Nachwachsender Rohstoff: Versuche zu Ihrer Mikrobiologischen Charakterisierung, Ihrer Konservierung und Ihrem Abbau; Cuvillier: Göttingen, Germany, 1994. [Google Scholar]
- Lisinska, G.; Leszczynski, W. Potato Science and Technology; Springer Science & Business Media: Berlin, Germany, 1989. [Google Scholar]
- Tegge, G. Stärke und Stärkederivate; Behr’s Verlag: Hamburg, Germany, 2004. [Google Scholar]
- Slomińska, L.; Starogardzka, G. Application of a multi-enzyme complex in the utilization of potato pulp. Starch-Stärke 1987, 39, 121–125. [Google Scholar] [CrossRef]
- Waglay, A.; Karboune, S. A novel enzymatic approach based on the use of multi-enzymatic systems for the recovery of enriched protein extracts from potato pulp. Food Chem. 2017, 220, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Abousteit, O.; Kempf, W. Pektingewinnung aus kartoffelpülpe und ihre mögliche bedeutung für die kartoffelstärkeindustrie. Starch-Stärke 1974, 26, 417–421. [Google Scholar] [CrossRef]
- Turquois, T.; Rinaudo, M.; Taravel, F.R.; Heyraud, A. Extraction of highly gelling pectic substances from sugar beet pulp and potato pulp: Influence of extrinsic parameters on their gelling properties. Food Hydrocoll. 1999, 13, 255–262. [Google Scholar] [CrossRef]
- Klingspohn, U.; Bader, J.; Kruse, B.; Vijai Kishore, P.; Schügerl, K.; Kracke-Helm, H.A.; Likidis, Z. Utilization of potato pulp from potato starch processing. Process Biochem. 1993, 28, 91–98. [Google Scholar] [CrossRef]
- Khodaei, N.; Karboune, S.; Orsat, V. Microwave-assisted alkaline extraction of galactan-rich rhamnogalacturonan i from potato cell wall by-product. Food Chem. 2016, 190, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Batista, J.E.R.; Demorais, M.P.; Caliari, M.; Soares, M.S.J. Physical, microbiological and sensory quality of gluten-free biscuits prepared from rice flour and potato pulp. J. Food Nutr. Res. 2016, 55, 101–107. [Google Scholar]
- Obidziński, S. Analysis of usability of potato pulp as solid fuel. Fuel Process. Technol. 2012, 94, 67–74. [Google Scholar] [CrossRef]
- Ercolano, M.R.; Gomez, L.D.; Andolfi, A.; Simister, R.; Troise, C.; Angelino, G.; Borrelli, C.; McQueen-Mason, S.J.; Evidente, A.; Frusciante, L.; et al. Residual biomass saccharification in processing tomato is affected by cultivar and nitrogen fertilization. Biomass Bioenerg 2015, 72, 242–250. [Google Scholar] [CrossRef]
- Shrestha, N.; Fogg, A.; Wilder, J.; Franco, D.; Komisar, S.; Gadhamshetty, V. Electricity generation from defective tomatoes. Bioelectrochemistry 2016, 112, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Ventura, M.R.; Pieltain, M.C.; Castanon, J.I.R. Evaluation of tomato crop by-products as feed for goats. Anim. Feed Sci. Technol. 2009, 154, 271–275. [Google Scholar] [CrossRef]
- Lenucci, M.S.; Leucci, M.R.; Piro, G.; Dalessandro, G. Variability in the content of soluble sugars and cell wall polysaccharides in red-ripe cherry and high-pigment tomato cultivars. J. Sci. Food Agric. 2008, 88, 1837–1844. [Google Scholar] [CrossRef]
- Riggi, E.; Avola, G. Fresh tomato packinghouses waste as high added-value biosource. Resour. Conserv. Recyl. 2008, 53, 96–106. [Google Scholar] [CrossRef]
- Bacenetti, J.; Duca, D.; Negri, M.; Fusi, A.; Fiala, M. Mitigation strategies in the agro-food sector: The anaerobic digestion of tomato puree by-products. An Italian case study. Sci. Total Environ. 2015, 526, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Riggi, E.; Avola, G. Quantification of the waste stream from fresh tomato packinghouses and its fluctuations: Implications for waste management planning. Resour. Conserv. Recyl. 2010, 54, 436–441. [Google Scholar] [CrossRef]
- Ribaudo, M.O.; Gollehon, N.R.; Agapoff, J. Land application of manure by animal feeding operations: Is more land needed? J. Soil Water Conserv. 2003, 58, 30–38. [Google Scholar]
- Ayalon, O.; Avnimelech, Y.; Shechter, M. Solid waste treatment as a high-priority and low-cost alternative for greenhouse gas mitigation. Environ. Manag. 2001, 27, 697–704. [Google Scholar]
- Ravindran, R.; Jaiswal, A.K. Exploitation of food industry waste for high-value products. Trends Biotechnol. 2016, 34, 58–69. [Google Scholar] [CrossRef] [PubMed]
- Jurado, M.M.; Suarez-Estrella, F.; Lopez, M.J.; Vargas-Garcia, M.C.; Lopez-Gonzalez, J.A.; Moreno, J. Enhanced turnover of organic matter fractions by microbial stimulation during lignocellulosic waste composting. Bioresour. Technol. 2015, 186, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Gonzalez, J.A.; Lopez, M.J.; Vargas-Garcia, M.C.; Suarez-Estrella, F.; Jurado, M.; Moreno, J. Tracking organic matter and microbiota dynamics during the stages of lignocellulosic waste composting. Bioresour. Technol. 2013, 146, 574–584. [Google Scholar] [CrossRef] [PubMed]
- Sheets, J.P.; Yang, L.; Ge, X.; Wang, Z.; Li, Y. Beyond land application: Emerging technologies for the treatment and reuse of anaerobically digested agricultural and food waste. Waste Manag. 2015, 44, 94–115. [Google Scholar] [CrossRef] [PubMed]
- Sweeten, J.M. Composting Manure and Sludge. Available online: http://hdl.handle.net/1969.1/87650 (accessed on 15 August 2017).
- Kalamdhad, A.S.; Singh, Y.K.; Ali, M.; Khwairakpam, M.; Kazmi, A.A. Rotary drum composting of vegetable waste and tree leaves. Bioresour. Technol. 2009, 100, 6442–6450. [Google Scholar] [CrossRef] [PubMed]
- Suluk, K.; Tosun, I.; Ekinci, K. Co-composting of two-phase olive-mill pomace and poultry manure with tomato harvest stalks. Environ. Technol. 2017, 38, 923–932. [Google Scholar] [CrossRef] [PubMed]
- Sevik, F.; Tosun, I.; Ekinci, K. Composting of olive processing wastes and tomato stalks together with sewage sludge or dairy manure. Int. J. Environ. Sci. Technol. 2016, 13, 1207–1218. [Google Scholar] [CrossRef]
- Vargas-Garcia, M.C.; Suarez-Estrella, F.; Lopez, M.J.; Moreno, J. Microbial population dynamics and enzyme activities in composting processes with different starting materials. Waste Manag. 2010, 30, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Gonzalez, J.A.; Suarez-Estrella, F.; Vargas-Garcia, M.C.; Lopez, M.J.; Jurado, M.M.; Moreno, J. Dynamics of bacterial microbiota during lignocellulosic waste composting: Studies upon its structure, functionality and biodiversity. Bioresour. Technol. 2015, 175, 406–416. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wang, S.T.; Zhang, J.; Xu, T. Biochar influences the microbial community structure during tomato stalk composting with chicken manure. Bioresour. Technol. 2014, 154, 148–154. [Google Scholar]
- Kulcu, R. Composting of greenhouse tomato plant residues, wheat straw, and separated dairy manure, and the effect of free air space on the process. Pol. J. Environ. Stud. 2014, 23, 1341–1346. [Google Scholar]
- Gavilanes-Teran, I.; Jara-Samaniego, J.; Idrovo-Novillo, J.; Bustamante, M.A.; Moral, R.; Paredes, C. Windrow composting as horticultural waste management strategy—A case study in ecuador. Waste Manag. 2016, 48, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Mendoza-Hernandez, D.; Fornes, F.; Belda, R.M. Compost and vermicompost of horticultural waste as substrates for cutting rooting and growth of rosemary. Sci. Hortic. 2014, 178, 192–202. [Google Scholar] [CrossRef]
- Pane, C.; Celano, G.; Piccolo, A.; Villecco, D.; Spaccini, R.; Palese, A.M.; Zaccardelli, M. Effects of on-farm composted tomato residues on soil biological activity and yields in a tomato cropping system. Chem. Biol. Technol. Agric. 2015, 2, 4. [Google Scholar] [CrossRef]
- Tits, M.; Elsen, A.; Bries, J.; Vandendriessche, H. Short-term and long-term effects of vegetable, fruit and garden waste compost applications in an arable crop rotation in flanders. Plant Soil 2014, 376, 43–59. [Google Scholar] [CrossRef]
- Zaller, J.G. Vermicompost in seedling potting media can affect germination, biomass allocation, yields and fruit quality of three tomato varieties. Eur. J. Soil Biol. 2007, 43, S332–S336. [Google Scholar] [CrossRef]
- Gutierrez-Miceli, F.A.; Santiago-Borraz, J.; Molina, J.A.M.; Nafate, C.C.; Abud-Archila, M.; Llaven, M.A.O.; Rincon-Rosales, R.; Dendooven, L. Vermicompost as a soil supplement to improve growth, yield and fruit quality of tomato (Lycopersicum esculentum). Bioresour. Technol. 2007, 98, 2781–2786. [Google Scholar] [CrossRef] [PubMed]
- Arancon, N.Q.; Edwards, C.A.; Bierman, P.; Metzger, J.D.; Lee, S.; Welch, C. Effects of vermicomposts on growth and marketable fruits of field-grown tomatoes, peppers and strawberries. Pedobiologia 2003, 47, 731–735. [Google Scholar] [CrossRef]
- Fernandez-Gomez, M.J.; Diaz-Ravina, M.; Romero, E.; Nogales, R. Recycling of environmentally problematic plant wastes generated from greenhouse tomato crops through vermicomposting. Int. J. Environ. Sci. Technol. 2013, 10, 697–708. [Google Scholar] [CrossRef]
- Ertani, A.; Pizzeghello, D.; Baglieri, A.; Cadili, V.; Tambone, F.; Gennari, M.; Nardi, S. Humic-like substances from agro-industrial residues affect growth and nitrogen assimilation in maize (Zea mays L.) plantlets. J. Geochem. Explor. 2013, 129, 103–111. [Google Scholar] [CrossRef]
- Nardi, S.; Carletti, P.; Pizzeghello, D. Biological activities of humic substances. In Biophysico-Chemical Processes Involving Natural Nonliving Organic Matter in Environmental Systems; Senesi, N., Xing, B., Huang, P., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2009; Volume 2, pp. 305–339. [Google Scholar]
- Lamar, R.T.; Olk, D.C.; Mayhew, L.; Bloom, P.R. A new standardized method for quantification of humic and fulvic acids in humic ores and commercial products. J. AOAC Int. 2014, 97, 721–730. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Magen, H.; Riov, J. Humic substances originating from rapidly decomposing organic matter: Properties and effects on plant growth. In Humic Substances in the Global Environment and Implications on Human Health; Senesi, N., Miano, T.M., Eds.; Elsevier: New York, NY, USA, 1994; pp. 427–443. [Google Scholar]
- Sortino, O.; Dipasquale, M.; Montoneri, E.; Tomasso, L.; Avetta, P.; Bianco Prevot, A. 90% yield increase of red pepper with unexpectedly low doses of compost soluble substances. Agron. Sustain. Dev. 2013, 33, 433–441. [Google Scholar] [CrossRef]
- Valdrighi, M.M.; Pera, A.; Agnolucci, M.; Frassinetti, S.; Lunardi, D.; Vallini, G. Effects of compost-derived humic acids on vegetable biomass production and microbial growth within a plant (Cichorium intybus)-soil system: A comparative study. Agric. Ecosyst. Environ. 1996, 58, 133–144. [Google Scholar] [CrossRef]
- Eyheraguibel, B.; Silvestre, J.; Morard, P. Effects of humic substances derived from organic waste enhancement on the growth and mineral nutrition of maize. Bioresour. Technol. 2008, 99, 4206–4212. [Google Scholar] [CrossRef] [PubMed]
- Rose, M.T.; Patti, A.F.; Little, K.R.; Jackson, W.R.; Cavagnaro, T.R. A meta-analysis and review of plant-growth response to humic substances: Practical implications for agriculture. In Advances in Agronomy; Sparks, D.L., Ed.; Academic Press: Cambridge, MA, USA, 2014; Volume 124, pp. 37–89. [Google Scholar]
- Moreno Casco, J.; Suárez Estrella, F.; López López, M.J.; Vargas García, M.C. Procedimiento de Obtención de Abono Líquido Orgánico Rico en Sustancias Húmicas a Partir de Compost de Origen Vegetal. Available online: http://hdl.handle.net/10835/4367 (accessed on 15 August 2017).
- Jurado, M.M.; Suarez-Estrella, F.; Vargas-Garcia, M.C.; Lopez, M.J.; Lopez-Gonzalez, J.A.; Moreno, J. Evolution of enzymatic activities and carbon fractions throughout composting of plant waste. J. Environ. Manag. 2014, 133, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Garcia, M.D.; Suarez-Estrella, F.F.; Lopez, M.J.; Moreno, J. Influence of microbial inoculation and co-composting material on the evolution of humic-like substances during composting of horticultural wastes. Process Biochem. 2006, 41, 1438–1443. [Google Scholar] [CrossRef]
- Bashan, Y.; de-Bashan, L.E.; Prabhu, S.R.; Hernandez, J.P. Advances in plant growth-promoting bacterial inoculant technology: Formulations and practical perspectives (1998–2013). Plant Soil 2014, 378, 1–33. [Google Scholar] [CrossRef]
- Lugtenberg, B.; Kamilova, F. Plant-growth-promoting rhizobacteria. Annu. Rev. Microbiol. 2009, 63, 541–556. [Google Scholar] [CrossRef] [PubMed]
- Borriss, R. Use of plant-associated bacillus strains as biofertilizers and biocontrol agents in agriculture. In Bacteria in Agrobiology: Plant Growth Responses; Maheshwari, D.K., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 41–76. [Google Scholar]
- Olivares, F.L.; Aguiar, N.O.; Rosa, R.C.C.; Canellas, L.P. Substrate biofortification in combination with foliar sprays of plant growth promoting bacteria and humic substances boosts production of organic tomatoes. Sci. Hortic. 2015, 183, 100–108. [Google Scholar] [CrossRef]
- Vessey, J.K. Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 2003, 255, 571–586. [Google Scholar] [CrossRef]
- Suarez-Estrella, F.; Jurado, M.M.; Vargas-Garcia, M.C.; Lopez, M.J.; Moreno, J. Isolation of bio-protective microbial agents from eco-composts. Biol. Control. 2013, 67, 66–74. [Google Scholar] [CrossRef]
- Canellas, L.P.; Olivares, F.L. Physiological responses to humic substances as plant growth promoter. Chem. Biol. Technol. Agric. 2014, 1, 3. [Google Scholar] [CrossRef]
- Martinez-Balmori, D.; Olivares, F.L.; Spaccini, R.; Aguiar, K.P.; Araujo, M.F.; Aguiar, N.O.; Guridi, F.; Canellas, L.P. Molecular characteristics of vermicompost and their relationship to preservation of inoculated nitrogen-fixing bacteria. J. Anal. Appl. Pyrol. 2013, 104, 540–550. [Google Scholar] [CrossRef]
- Clinton, S.K. Lycopene: Chemistry, biology, and implications for human health and disease. Nutr. Rev. 1998, 56, 35–51. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K.; LeMaguer, M. Lycopene in tomatoes and tomato pulp fraction. Ital. J. Food Sci. 1996, 8, 107–113. [Google Scholar]
- Heber, D.; Lu, Q.Y. Overview of mechanisms of action of lycopene. Exp. Biol. Med. 2002, 227, 920–923. [Google Scholar] [CrossRef]
- Giovannucci, E. Tomato products, lycopene, and prostate cancer: A review of the epidemiological literature. J. Nutr. 2005, 135, 2030–2031. [Google Scholar]
- Rao, A.V.; Rao, L.G. Carotenoids and human health. Pharmacol. Res. 2007, 55, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Naviglio, D.; Caruso, T.; Iannece, P.; Aragòn, A.; Santini, A. Characterization of high purity lycopene from tomato wastes using a new pressurized extraction approach. J. Agric. Food Chem. 2008, 56, 6227–6231. [Google Scholar] [CrossRef] [PubMed]
- Strati, I.F.; Oreopoulou, V. Recovery of carotenoids from tomato processing by-products—A review. Food Res. Int. 2014, 65, 311–321. [Google Scholar] [CrossRef]
- Cadoni, E.; De Giorgi, M.R.; Medda, E.; Poma, G. Supercritical CO2 extraction of lycopene and β-carotene from ripe tomatoes. Dyes Pigments 1999, 44, 27–32. [Google Scholar] [CrossRef]
- Park, J.W. Coloring technology for surimi seafood. ACS Symp. Ser. 2008, 983, 254–266. [Google Scholar]
- Metkar, S.; Saptarshi, S.; Kadam, A. Studies on extraction, isolation and applications of lycopene. IAJPR 2014, 4, 5017–5028. [Google Scholar]
- Stajčić, S.; Ćetković, G.; Čanadanović-Brunet, J.; Djilas, S.; Mandić, A.; Četojević-Simin, D. Tomato waste: Carotenoids content, antioxidant and cell growth activities. Food Chem. 2014, 172, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Hyde, K.D.; Bahkali, A.H.; Moslem, M.A. Fungi—An unusual source for cosmetics. Fungal Divers. 2010, 43, 1–9. [Google Scholar] [CrossRef]
- Kanno, E. Functional Cosmetic Composition. Patent JP2003212709A, 22 January 2002. [Google Scholar]
- Ishida, B.K.; Chapman, M.H. Carotenoid extraction from plants using a novel, environmentally friendly solvent. J. Agric. Food Chem. 2009, 57, 1051–1059. [Google Scholar] [CrossRef] [PubMed]
- Kaur, D.; Wani, A.A.; Oberoi, D.P.S.; Sogi, D.S. Effect of extraction conditions on lycopene extractions from tomato processing waste skin using response surface methodology. Food Chem. 2008, 108, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Periago, M.J.; Rincón, F.; Agüera, M.D.; Ros, G. Mixture approach for optimizing lycopene extraction from tomato and tomato products. J. Agric. Food Chem. 2004, 52, 5796–5802. [Google Scholar] [CrossRef] [PubMed]
- Vertzoni, M.; Kartezini, T.; Reppas, C.; Archontaki, H.; Valsami, G. Solubilization and quantification of lycopene in aqueous media in the form of cyclodextrin binary systems. Int. J. Pharm. 2006, 309, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.S.M.; Silva, V.M.T.M.; Rodrigues, A.E. Ethyl lactate as a solvent: Properties, applications and production processes—A review. Green Chem. 2011, 13, 2658–2671. [Google Scholar] [CrossRef]
- Clark, J.H.; Tavener, S.J. Alternative solvents: Shades of green. Org. Process Res. Dev. 2007, 11, 149–155. [Google Scholar] [CrossRef]
- Strati, I.F.; Oreopoulou, V. Effect of extraction parameters on the carotenoid recovery from tomato waste. Int. J. Food Sci. Technol. 2011, 46, 23–29. [Google Scholar] [CrossRef]
- Chemat-Djenni, Z.; Ferhat, M.A.; Tomao, V.; Chemat, F. Carotenoid extraction from tomato using a green solvent resulting from orange processing waste. J. Essent. Oil Bear. Plants 2010, 13, 139–147. [Google Scholar] [CrossRef]
- Eh, A.L.-S.; Teoh, S.-G. Novel modified ultrasonication technique for the extraction of lycopene from tomatoes. Ultrason. Sonochem. 2012, 19, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Bonrath, W. Chemical reactions under “non-classical conditions”, microwaves and ultrasound in the synthesis of vitamins. Ultrason. Sonochem. 2004, 11, 1–4. [Google Scholar] [CrossRef]
- Ho, K.K.H.Y.; Ferruzzi, M.G.; Liceaga, A.M.; San Martín-González, M.F. Microwave-assisted extraction of lycopene in tomato peels: Effect of extraction conditions on all-trans and cis-isomer yields. LWT Food Sci. Technol. 2015, 62, 160–168. [Google Scholar] [CrossRef]
- Kusuma, H.S.; Mahfud, M. Preliminary study: Kinetics of oil extraction from sandalwood by microwave-assisted hydrodistillation. In Proceedings of the IOP Conference Series: Materials Science and Engineering, Dresden, Germany, 19–20 September 2017. [Google Scholar]
- Lianfu, Z.; Zelong, L. Optimization and comparison of ultrasound/microwave assisted extraction (UMAE) and ultrasonic assisted extraction (UAE) of lycopene from tomatoes. Ultrason. Sonochem. 2008, 15, 731–737. [Google Scholar] [CrossRef] [PubMed]
- Baiano, A.; Bevilacqua, L.; Terracone, C.; Contò, F.; Del Nobile, M.A. Single and interactive effects of process variables on microwave-assisted and conventional extractions of antioxidants from vegetable solid wastes. J. Food Eng. 2014, 120, 135–145. [Google Scholar] [CrossRef]
- Choudhari, S.M.; Ananthanarayan, L. Enzyme aided extraction of lycopene from tomato tissues. Food Chem. 2007, 102, 77–81. [Google Scholar] [CrossRef]
- Lavecchia, R.; Zuorro, A. Improved lycopene extraction from tomato peels using cell-wall degrading enzymes. Eur. Food Res. Technol. 2008, 228, 153. [Google Scholar] [CrossRef]
- Papaioannou, E.H.; Karabelas, A.J. Lycopene recovery from tomato peel under mild conditions assisted by enzymatic pre-treatment and non-ionic surfactants. Acta Biochim. Pol. 2012, 59, 71–74. [Google Scholar] [PubMed]
- Ranveer, R.C.; Patil, S.N.; Sahoo, A.K. Effect of different parameters on enzyme-assisted extraction of lycopene from tomato processing waste. Food Bioprod. Process. 2013, 91, 370–375. [Google Scholar] [CrossRef]
- Cuccolini, S.; Aldini, A.; Visai, L.; Daglia, M.; Ferrari, D. Environmentally friendly lycopene purification from tomato peel waste: Enzymatic assisted aqueous extraction. J. Agric. Food Chem. 2013, 61, 1646–1651. [Google Scholar] [CrossRef] [PubMed]
- Puri, M.; Sharma, D.; Barrow, C.J. Enzyme-assisted extraction of bioactives from plants. Trends Biotechnol. 2012, 30, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Baysal, T.; Ersus, S.; Starmans, D.A.J. Supercritical CO2 extraction of β-carotene and lycopene from tomato paste waste. J. Agi. Food Chem. 2000, 48, 5507–5511. [Google Scholar] [CrossRef]
- Rozzi, N.L.; Singh, R.K.; Vierling, R.A.; Watkins, B.A. Supercritical fluid extraction of lycopene from tomato processing byproducts. J. Agric. Food Chem. 2002, 50, 2638–2643. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Weller, C.L. Recent advances in extraction of nutraceuticals from plants. Trends Food Sci. Technol. 2006, 17, 300–312. [Google Scholar] [CrossRef]
- Kolattukudy, P. Biosynthetic Pathways of Cutin and Waxes, and their Sensitivity to Environmental Stresses. Plant Cuticles: An Integrated Functional Approach; BIOS Scientific Publishers Ltd.: Oxford, UK, 1996; pp. 83–108. [Google Scholar]
- Kolattukudy, P.E. Cutin from plants. Biopolymers 2005. [Google Scholar] [CrossRef]
- Benı́tez, J.J.; Garcı́a-Segura, R.; Heredia, A. Plant biopolyester cutin: A tough way to its chemical synthesis. Biochim. Biophys. Acta 2004, 1674, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Heredia, A. Biophysical and biochemical characteristics of cutin, a plant barrier biopolymer. Biochim. Biophys. Acta 2003, 1620, 1–7. [Google Scholar] [CrossRef]
- Li, Y.H.; Beisson, F.; Koo, A.J.K.; Molina, I.; Pollard, M.; Ohlrogge, J. Identification of acyltransferases required for cutin biosynthesis and production of cutin with suberin-like monomers. Proc. Natl. Acad. Sci. USA 2007, 104, 18339–18344. [Google Scholar] [CrossRef] [PubMed]
- Tsatsaronis, G.C.; Boskou, D.G. Amino-acid and mineral salt content of tomato seed and skin waste. J. Sci. Food Agric. 1975, 26, 421–423. [Google Scholar] [CrossRef] [PubMed]
- Kallio, H.; Nieminen, R.; Tuomasjukka, S.; Hakala, M. Cutin composition of five Finnish berries. J. Sci. Food Agric. 2006, 54, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Järvinen, R.; Silvestre, A.J.D.; Gil, A.M.; Kallio, H. Solid state 13C CP-MAS NMR and FT-IR spectroscopic analysis of cuticular fractions of berries and suberized membranes of potato. J. Food Compos. Anal. 2011, 24, 334–345. [Google Scholar] [CrossRef]
- Koskimies, S.; Hulkko, J.; Pitkänen, P.; Heiskanen, N.; Yli-Kauhaluoma, J.; Wähälä, K. Method for the Manufacture of Oligo- and Polyesters from a Mixture of Carboxylic Acid Obtained from Suberin and/or Cutin and Use Thereof. U.S. Patent US20110319504 A1, 8 September 2011. [Google Scholar]
- Osman, S.F.; Irwin, P.; Fett, W.F.; O’Conno, J.V.; Parris, N. Preparation, isolation, and characterization of cutin monomers and oligomers from tomato peels. J. Sci. Food Agric. 1999, 47, 799–802. [Google Scholar] [CrossRef]
- Luque, P.; Bruque, S.; Heredia, A. Water permeability of isolated cuticular membranes: A structural analysis. Arch. Biochem. Biophys. 1995, 317, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Chefetz, B. Decomposition and sorption characterization of plant cuticles in soil. Plant Soil 2007, 298, 21–30. [Google Scholar] [CrossRef]
- Shechter, M.; Xing, B.; Kopinke, F.-D.; Chefetz, B. Competitive sorption−desorption behavior of triazine herbicides with plant cuticular fractions. J. Sci. Food Agric. 2006, 54, 7761–7768. [Google Scholar] [CrossRef] [PubMed]
- Hauff, S.; Chefetz, B.; Shechter, M.; Vetter, W. Determination of hydroxylated fatty acids from the biopolymer of tomato cutin and their fate during incubation in soil. Phytochem Anal. 2010, 21, 582–589. [Google Scholar] [CrossRef] [PubMed]
- Cigognini, I.; Montanari, A.; De la Torre Carreras, R.; Cardoso, G. Extraction Method of a Polyester Polymer or Cutin from the Wasted Tomato Peels and Polyester Polimer so Extracted. Patent WO2015028299 (A1), 5 March 2015. [Google Scholar]
- Montanari, A.; Bolzoni, L.; Cigognini, I.M.; Carreras, R.D. Tomato bio-based lacquer for sustainable metal packaging. Agro Food Ind. Hi Tec 2014, 25, 50–54. [Google Scholar] [CrossRef]
- Encinar, J.M.; González, J.F.; Martínez, G. Energetic use of the tomato plant waste. Fuel Process. Technol. 2008, 89, 1193–1200. [Google Scholar] [CrossRef]
- Zhang, R.Y. What to Do with Tomato Plant Wastes after Ketchup? Available online: http://www.biofuelnet.ca/author/ryan-yongsheng-zhang/ (accessed on 3 May 2015).
- Tabasso, S.; Montoneri, E.; Carnaroglio, D.; Caporaso, M.; Cravotto, G. Microwave-assisted flash conversion of non-edible polysaccharides and post-harvest tomato plant waste to levulinic acid. Green Chem. 2014, 16, 73–76. [Google Scholar] [CrossRef]
- Buttery, R.G.; Ling, L.C.; Light, D.M. Tomato leaf volatile aroma components. J. Agric. Food Chem. 1987, 35, 1039–1042. [Google Scholar] [CrossRef]
- Baldwin, E.A.; Scott, J.W.; Shewmaker, C.K.; Schuch, W. Flavor trivia and tomato aroma: Biochemistry and possible mechanisms for control of important aroma components. Hortscience 2000, 35, 1013–1022. [Google Scholar]
- Tabart, J.; Kevers, C.; Sipel, A.; Pincemail, J.; Defraigne, J.O.; Dommes, J. Optimisation of extraction of phenolics and antioxidants from black currant leaves and buds and of stability during storage. Food Chem. 2007, 105, 1268–1275. [Google Scholar] [CrossRef]
- Sun, T.; Ho, C.T. Antioxidant activities of buckwheat extracts. Food Chem. 2005, 90, 743–749. [Google Scholar] [CrossRef]
- Savatović, S.M.; Ćetković, G.S.; Čanadanović-Brunet, J.M.; Đilas, S.M. Utilization of tomato waste as a source of polyphenolic antioxidants. Acta Period. Technol. 2010, 41, 187–194. [Google Scholar] [CrossRef]
- Chandi, G.K.; Singh, S.P.; Gill, B.S.; Sogi, D.S.; Singh, P. Optimization of carotenoids by rhodotorula glutinis. Food Sci. Biotechnol. 2010, 19, 881–887. [Google Scholar] [CrossRef]
- Moayedi, A.; Hashemi, M.; Safari, M. Valorization of tomato waste proteins through production of antioxidant and antibacterial hydrolysates by proteolytic bacillus subtilis: Optimization of fermentation conditions. J. Food Sci. Technol. 2016, 53, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Di Gioia, D.; Fava, F.; Luziatelli, F.; Ruzzi, M. Vanillin production from agro-industrial wastes. In Comprehensive Biotechnology, 2nd ed.; Murray, M.-Y., Ed.; Academic Press: Burlington, VT, USA, 2011. [Google Scholar]
- FAO. Fao Cereal Supply and Demand Brief. Available online: www.fao.org/worldfoodsituation/csdb/en (accessed on 11 November 2016).
- FAOSTAT Crops. Available online: http://faostat3.fao.org/browse/Q/QC/E (accessed on 11 November 2016).
- Reisinger, M.; Tirpanalan, O.; Pruckler, M.; Huber, F.; Kneifel, W.; Novalin, S. Wheat bran biorefinery—A detailed investigation on hydrothermal and enzymatic treatment. Bioresour. Technol. 2013, 144, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Pruckler, M.; Siebenhandl-Ehn, S.; Apprich, S.; Holtinger, S.; Haas, C.; Schmid, E.; Kneifel, W. Wheat bran-based biorefinery 1: Composition of wheat bran and strategies of functionalization. LWT Food Sci. Technol. 2014, 56, 211–221. [Google Scholar] [CrossRef]
- Soukoulis, C.; Aprea, E. Cereal bran fractionation: Processing techniques for the recovery of functional components and their applications to the food industry. Recent Patent. Food Nutr. Agric. 2012, 4, 61–77. [Google Scholar]
- Akil, H.M.; Omar, M.F.; Mazuki, A.A.M.; Safiee, S.; Ishak, Z.A.M.; Abu Bakar, A. Kenaf fiber reinforced composites: A review. Mater. Des. 2011, 32, 4107–4121. [Google Scholar] [CrossRef]
- Kozlowski, R.; Mieleniak, B.; Helwig, M.; Przepiera, A. Flame resistant lignocellulosic-mineral composite particleboards. Polym. Degrad Stabil. 1999, 64, 523–528. [Google Scholar] [CrossRef]
- Kim, J.; Yun, S.; Ounaies, Z. Discovery of cellulose as a smart material. Macromolecules 2006, 39, 4202–4206. [Google Scholar] [CrossRef]
- Hunter, I.R. Bamboo resources, uses and trade: The future? J. Bamboo Rattan 2003, 2, 319–326. [Google Scholar] [CrossRef]
- Parameswaran, N.; Liese, W. On the fine structure of bamboo fibres. Wood Sci. Technol. 1976, 10, 231–246. [Google Scholar]
- Mohanty, A.K.; Misra, M.; Hinrichsen, G. Biofibres, biodegradable polymers and biocomposites: An overview. Macromol. Mater. Eng. 2000, 276, 1–24. [Google Scholar] [CrossRef]
- Henriksson, M.; Henriksson, G.; Berglund, L.A.; Lindstrom, T. An environmentally friendly method for enzyme-assisted preparation of microfibrillated cellulose (MFC) nanofibers. Eur. Polym. J. 2007, 43, 3434–3441. [Google Scholar] [CrossRef]
- Isogai, A.; Saito, T.; Fukuzumi, H. Tempo-oxidized cellulose nanofibers. Nanoscale 2011, 3, 71–85. [Google Scholar] [CrossRef] [PubMed]
- Paakko, M.; Ankerfors, M.; Kosonen, H.; Nykanen, A.; Ahola, S.; Osterberg, M.; Ruokolainen, J.; Laine, J.; Larsson, P.T.; Ikkala, O.; et al. Enzymatic hydrolysis combined with mechanical shearing and high-pressure homogenization for nanoscale cellulose fibrils and strong gels. Biomacromolecules 2007, 8, 1934–1941. [Google Scholar] [CrossRef] [PubMed]
- Moon, R.J.; Martini, A.; Nairn, J.; Simonsen, J.; Youngblood, J. Cellulose nanomaterials review: Structure, properties and nanocomposites. Chem. Soc. Rev. 2011, 40, 3941–3994. [Google Scholar] [CrossRef] [PubMed]
- Jonoobi, M.; Oladi, R.; Davoudpour, Y.; Oksman, K.; Dufresne, A.; Hamzeh, Y.; Davoodi, R. Different preparation methods and properties of nanostructured cellulose from various natural resources and residues: A review. Cellulose 2015, 22, 935–969. [Google Scholar] [CrossRef]
- Alemdar, A.; Sain, M. Isolation and characterization of nanofibers from agricultural residues - wheat straw and soy hulls. Bioresour. Technol. 2008, 99, 1664–1671. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, A.; Singh, M. Isolation and characterization of cellulose nanofibrils from wheat straw using steam explosion coupled with high shear homogenization. Carbohydr. Res. 2011, 346, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Jonoobi, M.; Khazaeian, A.; Tahir, P.M.; Azry, S.S.; Oksman, K. Characteristics of cellulose nanofibers isolated from rubberwood and empty fruit bunches of oil palm using chemo-mechanical process. Cellulose 2011, 18, 1085–1095. [Google Scholar] [CrossRef]
- Dufresne, A.; Cavaille, J.Y.; Vignon, M.R. Mechanical behavior of sheets prepared from sugar beet cellulose microfibrils. J. Appl. Polym. Sci. 1997, 64, 1185–1194. [Google Scholar] [CrossRef]
- Dufresne, A.; Vignon, M.R. Improvement of starch film performances using cellulose microfibrils. Macromolecules 1998, 31, 2693–2696. [Google Scholar] [CrossRef]
- Bruce, D.M.; Hobson, R.N.; Farrent, J.W.; Hepworth, D.G. High-performance composites from low-cost plant primary cell walls. Compos. Appl. Sci. Manuf. 2005, 36, 1486–1493. [Google Scholar] [CrossRef]
- Bhattacharya, D.; Germinario, L.T.; Winter, W.T. Isolation, preparation and characterization of cellulose microfibers obtained from bagasse. Carbohydr. Polym. 2008, 73, 371–377. [Google Scholar] [CrossRef]
- Hassan, M.L.; Mathew, A.P.; Hassan, E.A.; El-Wakil, N.A.; Oksman, K. Nanofibers from bagasse and rice straw: Process optimization and properties. Wood Sci. Technol. 2012, 46, 193–205. [Google Scholar] [CrossRef]
- Zuluaga, R.; Putaux, J.L.; Cruz, J.; Velez, J.; Mondragon, I.; Ganan, P. Cellulose microfibrils from banana rachis: Effect of alkaline treatments on structural and morphological features. Carbohydr. Polym. 2009, 76, 51–59. [Google Scholar] [CrossRef]
- Tibolla, H.; Pelissari, F.M.; Menegalli, F.C. Cellulose nanofibers produced from banana peel by chemical and enzymatic treatment. LWT Food Sci. Technol. 2014, 59, 1311–1318. [Google Scholar] [CrossRef]
- Spence, K.L.; Venditti, R.A.; Rojas, O.J.; Habibi, Y.; Pawlak, J.J. A comparative study of energy consumption and physical properties of microfibrillated cellulose produced by different processing methods. Cellulose 2011, 18, 1097–1111. [Google Scholar] [CrossRef]
- Purkait, B.S.; Ray, D.; Sengupta, S.; Kar, T.; Mohanty, A.; Misra, M. Isolation of cellulose nanoparticles from sesame husk. Ind. Eng. Chem. Res. 2011, 50, 871–876. [Google Scholar] [CrossRef]
- Apprich, S.; Tirpanalan, O.; Hell, J.; Reisinger, M.; Bohmdorfer, S.; Siebenhandl-Ehn, S.; Novalin, S.; Kneifel, W. Wheat bran-based biorefinery 2: Valorization of products. LWT Food Sci. Technol. 2014, 56, 222–231. [Google Scholar] [CrossRef]
- Luthria, D.L.; Lu, Y.J.; John, K.M.M. Bioactive phytochemicals in wheat: Extraction, analysis, processing, and functional properties. J. Funct. Foods 2015, 18, 910–925. [Google Scholar] [CrossRef]
- Kroon, P.A.; Faulds, C.B.; Ryden, P.; Robertson, J.A.; Williamson, G. Release of covalently bound ferulic acid from fiber in the human colon. J. Agric. Food Chem. 1997, 45, 661–667. [Google Scholar] [CrossRef]
- Liyana-Pathirana, C.M.; Shahidi, F. Importance of insoluble-bound phenolics to antioxidant properties of wheat. J. Agric. Food Chem. 2006, 54, 1256–1264. [Google Scholar] [CrossRef] [PubMed]
- Ou, S.; Kwok, K.-C. Ferulic acid: Pharmaceutical functions, preparation and applications in foods. J. Sci. Food Agric. 2004, 84, 1261–1269. [Google Scholar] [CrossRef]
- Fava, F.; Zanaroli, G.; Vannini, L.; Guerzoni, E.; Bordoni, A.; Viaggi, D.; Robertson, J.; Waldron, K.; Bald, C.; Esturo, A.; et al. New advances in the integrated management of food processing by-products in Europe: Sustainable exploitation of fruit and cereal processing by-products with the production of new food products (NAMASTE EU). New Biotechnol. 2013, 30, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Tirpanalan, O.; Reisinger, M.; Huber, F.; Kneifel, W.; Novalin, S. Wheat bran biorefinery: An investigation on the starch derived glucose extraction accompanied by pre- and post-treatment steps. Bioresour. Technol. 2014, 163, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Wood, I.P.; Cook, N.M.; Wilson, D.R.; Ryden, P.; Robertson, J.A.; Waldron, K.W. Ethanol from a biorefinery waste stream: Saccharification of amylase, protease and xylanase treated wheat bran. Food Chem. 2016, 198, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Brewer, L.R.; Kubola, J.; Siriamornpun, S.; Herald, T.J.; Shi, Y.C. Wheat bran particle size influence on phytochemical extractability and antioxidant properties. Food Chem. 2014, 152, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Hell, J.; Donaldson, L.; Michlmayr, H.; Kraler, M.; Kneifel, W.; Potthast, A.; Rosenau, T.; Bohmdorfer, S. Effect of pretreatment on arabinoxylan distribution in wheat bran. Carbohydr. Polym. 2015, 121, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.Y.; Zhao, M.L.; Liu, X.X.; Zhong, K.; Tong, L.T.; Zhou, X.R.; Zhou, S.M. Effect of steam explosion-assisted extraction on phenolic acid profiles and antioxidant properties of wheat bran. J. Sci. Food Agric. 2016, 96, 3484–3491. [Google Scholar] [CrossRef] [PubMed]
- Merali, Z.; Collins, S.R.A.; Elliston, A.; Wilson, D.R.; Kasper, A.; Waldron, K.W. Characterization of cell wall components of wheat bran following hydrothermal pretreatment and fractionation. Biotechnol. Biofuels 2015. [Google Scholar] [CrossRef] [PubMed]
- Reisinger, M.; Tirpanalan, O.; Huber, F.; Kneifel, W.; Novalin, S. Investigations on a wheat bran biorefinery involving organosolv fractionation and enzymatic treatment. Bioresour. Technol. 2014, 170, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Hromadkova, Z.; Kost’alova, Z.; Ebringerova, A. Comparison of conventional and ultrasound-assisted extraction of phenolics-rich heteroxylans from wheat bran. Ultrason. Sonochem. 2008, 15, 1062–1068. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.J.; Luthria, D. Influence of gelatinization on the extraction of phenolic acids from wheat fractions. Food Chem. 2016, 194, 1138–1142. [Google Scholar] [CrossRef] [PubMed]
- Lasrado, L.D.; Gudipati, M. Antioxidant property of synbiotic combination of lactobacillus sp and wheat bran xylo-oligosaccharides. J. Food Sci. Technol. 2015, 52, 4551–4557. [Google Scholar] [CrossRef] [PubMed]
- Hromadkova, Z.; Paulsen, B.S.; Polovka, M.; Kost’alova, Z.; Ebringerova, A. Structural features of two heteroxylan polysaccharide fractions from wheat bran with anti-complementary and antioxidant activities. Carbohydr. Polym. 2013, 93, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Llevot, A.; Grau, E.; Carlotti, S.; Grelier, S.; Cramail, H. From lignin-derived aromatic compounds to novel biobased polymers. Macromol. Rapid Comm. 2016, 37, 9–28. [Google Scholar] [CrossRef] [PubMed]
- Bock, L.H.; Anderson, J.K. Linear polyesters derived from vanillic acid. J. Polym. Sci. 1955, 17, 553–558. [Google Scholar] [CrossRef]
- Era, V.; Hannula, J. Polyesters from vanillin-synthesis and characterization. Paperi ja Puu 1974, 56, 489. [Google Scholar]
- Ishikawa, K. Nippon Kagaku Zasshi. Journal of the Chemical Society of Japan: Pure Chemistry Section. Available online: http://www.worldcat.org/title/journal-of-the-chemical-society-of-japan-pure-chemistry-section-nippon-kagaku-zasshi/oclc/476489674 (accessed on 15 August 2017).
- Lange, W.; Kordsachia, O. Preparation and properties of polyesters from vanillin and syringaaldehyde a contribution to possible utilization of hardwood lignins. Holz als Roh-und Werkstoff 1981, 39, 107–112. [Google Scholar] [CrossRef]
- Mialon, L.; Pemba, A.G.; Miller, S.A. Biorenewable polyethylene terephthalate mimics derived from lignin and acetic acid. Green Chem. 2010, 12, 1704–1706. [Google Scholar] [CrossRef]
- Mialon, L.; Vanderhenst, R.; Pemba, A.G.; Miller, S.A. Polyalkylenehydroxybenzoates (PAHBs): Biorenewable aromatic/aliphatic polyesters from lignin. Macromol. Rapid Commun. 2011, 32, 1386–1392. [Google Scholar] [CrossRef] [PubMed]
- Gioia, C.; Banella, M.B.; Marchese, P.; Vannini, M.; Colonna, M.; Celli, A. Advances in the synthesis of bio-based aromatic polyesters: Novel copolymers derived from vanillic acid and epsilon-caprolactone. Polym. Chem. UK 2016, 7, 5396–5406. [Google Scholar] [CrossRef]
- Kreye, O.; Oelmann, S.; Meier, M.A. Renewable aromatic–aliphatic copolyesters derived from rapeseed. Macromolecul. Chem. Phys. 2013, 214, 1452–1464. [Google Scholar] [CrossRef]
- Firdaus, M.; Meier, M.A.R. Renewable co-polymers derived from vanillin and fatty acid derivatives. Eur. Polym. J. 2013, 49, 156–166. [Google Scholar] [CrossRef]
- Pang, C.C.; Zhang, J.; Wu, G.L.; Wang, Y.N.; Gao, H.; Ma, J.B. Renewable polyesters derived from 10-undecenoic acid and vanillic acid with versatile properties. Polym. Chem. UK 2014, 5, 2843–2853. [Google Scholar] [CrossRef]
- Pion, F.; Reano, A.F.; Ducrot, P.H.; Allais, F. Chemo-enzymatic preparation of new bio-based bis-and trisphenols: New versatile building blocks for polymer chemistry. RSC Adv. 2013, 3, 8988–8997. [Google Scholar] [CrossRef]
- Llevot, A.; Grau, E.; Carlotti, S.; Grelier, S.; Cramail, H. Renewable (SEMI) aromatic polyesters from symmetrical vanillin-based dimers. Polym. Chem. UK 2015, 6, 6058–6066. [Google Scholar] [CrossRef]
- Goto, T.; Ishii, D.; Enomoto-Rogers, Y.; Takemura, A.; Iwata, T. Synthesis and characterization of poly [(ferulic acid)-alt-(glycine)]. Polymer 2017, 112, 385–392. [Google Scholar] [CrossRef]
- Graf, E. Antioxidant potential of ferulic acid. Free Radic. Biol. Med. 1992, 13, 435–448. [Google Scholar] [CrossRef]
- Mori, H.; Kawabata, K.; Yoshimi, N.; Tanaka, T.; Murakami, T.; Okada, T.; Murai, H. Chemopreventive effects of ferulic acid on oral and rice germ on large bowel carcinogenesis. Anticancer Res. 1999, 19, 3775–3778. [Google Scholar] [PubMed]
- Ooishi, N.; Shibata, T.; Ikeda, K. Stabilized oral preparations containing ulcer inhibitors. Jpn. Kokai Tokkyo Koho JP 1993, 5, 194–225. [Google Scholar]
- Ouimet, M.A.; Griffin, J.; Carbone-Howell, A.L.; Wu, W.H.; Stebbins, N.D.; Di, R.; Uhrich, K.E. Biodegradable ferulic acid-containing poly(anhydride-ester): Degradation products with controlled release and sustained antioxidant activity. Biomacromolecules 2013, 14, 854–861. [Google Scholar] [CrossRef] [PubMed]
- Ouimet, M.A.; Faig, J.J.; Yu, W.; Uhrich, K.E. Ferulic acid-based polymers with glycol functionality as a versatile platform for topical applications. Biomacromolecules 2015, 16, 2911–2919. [Google Scholar] [CrossRef] [PubMed]
- Coelho, C.; Hennous, M.; Verney, V.; Leroux, F. Functionalisation of polybutylene succinate nanocomposites: From structure to reinforcement of uv-absorbing and mechanical properties. RSC Adv. 2012, 2, 5430–5438. [Google Scholar] [CrossRef]
- Reano, A.F.; Cherubin, J.; Peru, A.M.M.; Wang, Q.; Clement, T.; Domenek, S.; Allais, F. Structure-activity relationships and structural design optimization of a series of p-hydroxycinnamic acids-based bis- and trisphenols as novel sustainable antiradical/antioxidant additives. ACS. Sustain. Chem. Eng. 2015, 3, 3486–3496. [Google Scholar] [CrossRef]
- Reano, A.F.; Domenek, S.; Pernes, M.; Beaugrand, J.; Allais, F. Ferulic acid-based bis/trisphenols as renewable antioxidants for polypropylene and poly (butylene succinate). ACS. Sustain. Chem. Eng. 2016, 4, 6562–6571. [Google Scholar] [CrossRef]
- Cerruti, P.; Malinconico, M.; Rychly, J.; Matisova-Rychla, L.; Carfagna, C. Effect of natural antioxidants on the stability of polypropylene films. Polym. Degrad Stabil. 2009, 94, 2095–2100. [Google Scholar] [CrossRef]
- Parisi, O.I.; Puoci, F.; Iemma, F.; De Luca, G.; Curcio, M.; Cirillo, G.; Spizzirri, U.G.; Picci, N. Antioxidant and spectroscopic studies of crosslinked polymers synthesized by grafting polymerization of ferulic acid. Polym. Advan. Technol. 2010, 21, 774–779. [Google Scholar] [CrossRef]
- Iemma, F.; Puoci, F.; Curcio, M.; Parisi, O.I.; Cirillo, G.; Spizzirri, U.G.; Picci, N. Ferulic acid as a comonomer in the synthesis of a novel polymeric chain with biological properties. J. Appl. Polym. Sci. 2010, 115, 784–789. [Google Scholar] [CrossRef]
- Du, J.; Fang, Y.Y.; Zheng, Y.B. Synthesis and characterization of poly (l-lactic acid) reinforced by biomesogenic units. Polym. Degrad Stabil. 2008, 93, 838–845. [Google Scholar] [CrossRef]
- Cerruti, P.; Santagata, G.; d’Ayala, G.G.; Ambrogi, V.; Carfagna, C.; Malinconico, M.; Persico, P. Effect of a natural polyphenolic extract on the properties of a biodegradable starch-based polymer. Polym. Degrad Stabil. 2011, 96, 839–846. [Google Scholar] [CrossRef]
- Kim, D.J.; Kim, W.S.; Lee, D.H.; Min, K.E.; Park, L.S.; Kang, I.K.; Jeon, I.R.; Seo, K.H. Modification of poly(Butylene succinate) with peroxide: Crosslinking, physical and thermal properties, and biodegradation. J. Appl. Polym. Sci. 2001, 81, 1115–1124. [Google Scholar] [CrossRef]
- Rizzarelli, P.; Carroccio, S. Thermo-oxidative processes in biodegradable poly (Butylene succinate). Polym. Degrad Stabil. 2009, 94, 1825–1838. [Google Scholar] [CrossRef]
- Poopathi, S.; Archana, B. Production of bacillus thuringiensis subsp israelensis from agro-based product (corncob). Curr. Sci. 2011, 101, 1009–1010. [Google Scholar]
- Velasquez, A.; Arias, R. Effect of substrate on the in vitro protein digestibility of extracts generated by saccharomyces cerevisiae. Ciencia E Investigación Agraria 2013, 40, 503–511. [Google Scholar] [CrossRef]
- Yunus, F.U.N.; Nadeem, M.; Rashid, F. Single-cell protein production through microbial conversion of lignocellulosic residue (wheat bran) for animal feed. J. I. Brewing 2015, 121, 553–557. [Google Scholar] [CrossRef]
- Tejeda-Ricardez, J.; Vaca-Garcia, C.; Borredon, M.E. Design of a batch solvolytic liquefaction reactor for the valorization of residues from the agricultural foodstuff. Chem. Eng. Res. Des. 2003, 81, 1066–1070. [Google Scholar] [CrossRef]
- Demirbas, A.; Caglar, A.; Akdeniz, F.; Gullu, D. Conversion of olive husk to liquid fuel by pyrolysis and catalytic liquefaction. Energy Sources 2000, 22, 631–639. [Google Scholar]
- Çağlar, A.; Demirbaş, A. Hydrogen rich gas mixture from olive husk via pyrolysis. Energy Convers. Manag. 2002, 43, 109–117. [Google Scholar] [CrossRef]
- Rao, T.R.; Sharma, A. Pyrolysis rates of biomass materials. Energy 1998, 23, 973–978. [Google Scholar] [CrossRef]
- Georgieva, T.I.; Ahring, B.K. Potential of agroindustrial waste from olive oil industry for fuel ethanol production. Biotechnol. J. 2007, 2, 1547–1555. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Bolanos, J.; Rodriguez, G.; Rodriguez, R.; Guillen, R.; Jimenez, A. Extraction of interesting organic compounds from olive oil waste. Grasas Aceites 2006, 57, 95–106. [Google Scholar] [CrossRef]
- Reboredo-Rodríguez, P.; González-Barreiro, C.; Cancho-Grande, B.; Simal-Gándara, J. Dynamic headspace/gc–ms to control the aroma fingerprint of extra-virgin olive oil from the same and different olive varieties. Food Control. 2012, 25, 684–695. [Google Scholar] [CrossRef]
- Reboredo-Rodriguez, P.; Gonzalez-Barreiro, C.; Cancho-Grande, B.; Simal-Gandara, J. Aroma biogenesis and distribution between olive pulps and seeds with identification of aroma trends among cultivars. Food Chem. 2013, 141, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Mason, W. Apparatus for and Explosion Fibration of Lignocellulose Material. U.S. Patent 1655618A, 10 January 1928. [Google Scholar]
- Biswas, A.K.; Umeki, K.; Yang, W.; Blasiak, W. Change of pyrolysis characteristics and structure of woody biomass due to steam explosion pretreatment. Fuel Process. Technol. 2011, 92, 1849–1854. [Google Scholar] [CrossRef]
- Kataria, R.; Mol, A.; Schulten, E.; Happel, A.; Mussatto, S.I. Bench scale steam explosion pretreatment of acid impregnated elephant grass biomass and its impacts on biomass composition, structure and hydrolysis. Ind. Crop. Prod. 2016, 106, 48–58. [Google Scholar] [CrossRef]
- Duque, A.; Manzanares, P.; Ballesteros, I.; Ballesteros, M. Biomass Fractionation Technologies for a Lignocellulosic Feedstock Based Biorefinery, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 349–368. [Google Scholar]
- Cara, C.; Ruiz, E.; Ballesteros, I.; Negro, M.J.; Castro, E. Enhanced enzymatic hydrolysis of olive tree wood by steam explosion and alkaline peroxide delignification. Process Biochem. 2006, 41, 423–429. [Google Scholar] [CrossRef]
- Ballesteros, I.; Oliva, J.M.; Saez, F.; Ballesteros, M. Ethanol production from lignocellulosic byproducts of olive oil extraction. Appl. Biochem. Biotechnol. 2001, 91, 237–252. [Google Scholar] [CrossRef]
- Spinelli, R.; Picchi, G. Industrial harvesting of olive tree pruning residue for energy biomass. Bioresour. Technol. 2010, 101, 730–735. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros, I.; Ballesteros, M.; Cara, C.; Sáez, F.; Castro, E.; Manzanares, P.; Negro, M.J.; Oliva, J.M. Effect of water extraction on sugars recovery from steam exploded olive tree pruning. Bioresour. Technol. 2011, 102, 6611–6616. [Google Scholar] [CrossRef] [PubMed]
- Conde, E.; Cara, C.; Moure, A.; Ruiz, E.; Castro, E.; Domínguez, H. Antioxidant activity of the phenolic compounds released by hydrothermal treatments of olive tree pruning. Food Chem. 2009, 114, 806–812. [Google Scholar] [CrossRef]
- Dermeche, S.; Nadour, M.; Larroche, C.; Moulti-Mati, F.; Michaud, P. Olive mill wastes: Biochemical characterizations and valorization strategies. Process Biochem. 2013, 48, 1532–1552. [Google Scholar] [CrossRef]
- Fernandez-Bolanos, J.; Felizon, B.; Heredia, A.; Guillen, R.; Jimenez, A. Characterization of the lignin obtained by alkaline delignification and of the cellulose residue from steam-exploded olive stones. Bioresour. Technol. 1999, 68, 121–132. [Google Scholar] [CrossRef]
- Felizón, B.; Fernández-Bolaños, J.; Heredia, A.; Guillén, R. Steam-explosion pretreatment of olive cake. J. Am. Oil Chem. Soc. 2000, 77, 15–22. [Google Scholar] [CrossRef]
- Fernández-Bolaños, J.; Felizón, B.; Brenes, M.; Guillén, R.; Heredia, A. Hydroxytyrosol and tyrosol as the main compounds found in the phenolic fraction of steam-exploded olive stones. J. Am. Oil Chem. Soc. 1998, 75, 1643–1649. [Google Scholar] [CrossRef]
- Berthet, M.A.; Angellier-Coussy, H.; Machado, D.; Hilliou, L.; Staebler, A.; Vicente, A.; Gontard, N. Exploring the potentialities of using lignocellulosic fibres derived from three food by-products as constituents of biocomposites for food packaging. Ind. Crop. Prod. 2015, 69, 110–122. [Google Scholar] [CrossRef]
- Bouknana, D.; Hammouti, B.; Salghi, R.; Jodeh, S.; Zarrouk, A.; Warad, I.; Aouniti, A.; Sbaa, M. Physicochemical characterization of olive oil mill wastewaters in the eastern region of morocco. J. Mater. Environ. Sci. 2014, 5, 1039–1058. [Google Scholar]
- Meftah, H.; Latrache, H.; Hamadi, F.; Hanine, H.; Zahir, H.; El louali, M. Comparaison des caractéristiques physicochimiques des huiles d’olives issus de différentes zones de la région tadla azilal (maroc)[comparison of the physico-chemical characteristics of the olive oil coming from different zones in tadla azilal area (morocco)]. J. Mater. Environ. Sci. 2014, 5, 641–646. [Google Scholar]
- Taamalli, A.; Arráez-Román, D.; Barrajón-Catalán, E.; Ruiz-Torres, V.; Pérez-Sánchez, A.; Herrero, M.; Ibañez, E.; Micol, V.; Zarrouk, M.; Segura-Carretero, A.; et al. Use of advanced techniques for the extraction of phenolic compounds from Tunisian olive leaves: Phenolic composition and cytotoxicity against human breast cancer cells. Food Chem. Toxicol. 2012, 50, 1817–1825. [Google Scholar] [CrossRef] [PubMed]
- Zahari, A.; Tazi, A.; Azzi, M. Optimisation des conditions de traitement des margines par un superoxydant k3fexmnyo8 [optimization of treatment conditions of olive oil mill wastewater by superoxidant k3fexmnyo8]. J. Mater. Environ. Sci. 2014, 5, 2484–2489. [Google Scholar]
- Bucić-Kojić, A.; Planinić, M.; Tomas, S.; Jakobek, L.; Šeruga, M. Influence of solvent and temperature on extraction of phenolic compounds from grape seed, antioxidant activity and colour of extract. Int. J. Food Sci. Technol. 2009, 44, 2394–2401. [Google Scholar] [CrossRef]
- Spigno, G.; Tramelli, L.; De Faveri, D.M. Effects of extraction time, temperature and solvent on concentration and antioxidant activity of grape marc phenolics. J. Food Eng. 2007, 81, 200–208. [Google Scholar] [CrossRef]
- Paini, M.; Casazza, A.A.; Aliakbarian, B.; Perego, P.; Binello, A.; Cravotto, G. Influence of ethanol/water ratio in ultrasound and high-pressure/high-temperature phenolic compound extraction from agri-food waste. Int. J. Food Sci. Technol. 2016, 51, 349–358. [Google Scholar] [CrossRef]
- Winkelhausen, E.; Pospiech, R.; Laufenberg, G. Antifungal activity of phenolic compounds extracted from dried olive pomace. Bull. Chem. Technol. Macedonia 2005, 24, 41–46. [Google Scholar]
- Vergani, L.; Vecchione, G.; Baldini, F.; Voci, A.; Ferrari, P.F.; Aliakbarian, B.; Casazza, A.A.; Perego, P. Antioxidant and hepatoprotective potentials of phenolic compounds from olive pomace. Chem. Eng. Trans. 2016, 49, 475–480. [Google Scholar]
- Cardoso, S.M.; Guyot, S.; Marnet, N.; Lopes-da-Silva, J.A.; Renard, C.M.G.C.; Coimbra, M.A. Characterisation of phenolic extracts from olive pulp and olive pomace by electrospray mass spectrometry. J. Sci. Food Agric. 2005, 85, 21–32. [Google Scholar] [CrossRef]
- Li, C.; Zheng, Y.Y.; Wang, X.F.; Feng, S.L.; Di, D.L. Simultaneous separation and purification of flavonoids and oleuropein from Olea europaea L. (olive) leaves using macroporous resin. J. Sci. Food Agric. 2011, 91, 2826–2834. [Google Scholar] [CrossRef] [PubMed]
- Japón-Luján, R.; de Castro, M.D.L. Superheated liquid extraction of oleuropein and related biophenols from olive leaves. J. Chromatogr. A 2006, 1136, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Khemakhem, I.; Ahmad-Qasem, M.H.; Catalán, E.B.; Micol, V.; García-Pérez, J.V.; Ayadi, M.A.; Bouaziz, M. Kinetic improvement of olive leaves’ bioactive compounds extraction by using power ultrasound in a wide temperature range. Ultrason. Sonochem. 2017, 34, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.; Kazemipour, M.; Fathi, S. Development of a simple green extraction procedure and hplc method for determination of oleuropein in olive leaf extract applied to a multi-source comparative study. J. Iran. Chem. Soc. 2011, 8, 38–47. [Google Scholar] [CrossRef]
- Heemken, O.P.; Theobald, N.; Wenclawiak, B.W. Comparison of ASE and SFE with soxhlet, sonication, and methanolic saponification extractions for the determination of organic micropollutants in marine particulate matter. Anal. Chem. 1997, 69, 2171–2180. [Google Scholar] [CrossRef] [PubMed]
- Procopio, A.; Alcaro, S.; Nardi, M.; Oliverio, M.; Ortuso, F.; Sacchetta, P.; Pieragostino, D.; Sindona, G. Synthesis, biological evaluation, and molecular modeling of oleuropein and its semisynthetic derivatives as cyclooxygenase inhibitors. J. Agric. Food Chem. 2009, 57, 11161–11167. [Google Scholar] [CrossRef] [PubMed]
- Sifaoui, I.; Chammem, N.; Abderrabba, M.; Mejri, M. Optimization of phenolic compounds extraction from olive leaves using experimental design methodology. J. Mater. Environ. Sci. 2016, 7, 1119–1127. [Google Scholar]
- Mkaouar, S.; Bahloul, N.; Gelicus, A.; Allaf, K.; Kechaou, N. Instant controlled pressure drop texturing for intensifying ethanol solvent extraction of olive (Olea europaea) leaf polyphenols. Separ. Purif. Tech. 2015, 145, 139–146. [Google Scholar] [CrossRef]
- Tsakona, S.; Galanakis, C.M.; Gekas, V. Hydro-ethanolic mixtures for the recovery of phenols from mediterranean plant materials. Food Bioprocess Technol. 2012, 5, 1384–1393. [Google Scholar] [CrossRef]
- Hayes, J.E.; Allen, P.; Brunton, N.; O’Grady, M.N.; Kerry, J.P. Phenolic composition and in vitro antioxidant capacity of four commercial phytochemical products: Olive leaf extract (Olea europaea L.), lutein, sesamol and ellagic acid. Food Chem. 2011, 126, 948–955. [Google Scholar] [CrossRef]
- Niaounakis, M.; Halvadakis, C.P. Olive Processing Waste Management: Literature Review and Patent Survey, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2006; Volume 5. [Google Scholar]
- Rafiee, Z.; Jafari, S.M.; Alami, M.; Khomeiri, M. Antioxidant effect of microwave-assisted extracts of olive leaves on sunflower oil. J. Agric. Sci. Technol. 2012, 14, 1497–1509. [Google Scholar]
- Components as related to human health: Recent developments. Austin. J. Nutr. Food Sci. 2015, 3, 3.
- Skaltsounis, A.L.; Argyropoulou, A.; Aligiannis, N.; Xynos, N. Recovery of high added value compounds from olive tree products and olive processing byproducts. In Olive and Olive Oil Bioactive Constituents, 1st ed.; Dimitrios, B., Ed.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 333–391. [Google Scholar]
- Taamalli, A.; Arráez-Román, D.; Zarrouk, M.; Valverde, J.; Segura-Carretero, A.; Fernández-Gutiérrez, A. The occurrence and bioactivity of polyphenols in Tunisian olive products and by-products: A review. J. Food Sci. 2012, 77, R83–R92. [Google Scholar] [CrossRef] [PubMed]
- Cirico, T.L.; Omaye, S.T. Additive or synergetic effects of phenolic compounds on human low density lipoprotein oxidation. Food Chem. Toxicol. 2006, 44, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Sun, A.Y.; Simonyi, A.; Sun, G.Y. The “french paradox” and beyond: Neuroprotective effects of polyphenols. Free Radic. Biol. Med. 2002, 32, 314–318. [Google Scholar] [CrossRef]
- Brul, S.; Coote, P. Preservative agents in foods: Mode of action and microbial resistance mechanisms. Int. J.Food Microbiol. 1999, 50, 1–17. [Google Scholar] [CrossRef]
- Ejechi, B.O.; Akpomedaye, D.E. Activity of essential oil and phenolic acid extracts of pepperfruit (Dennetia tripetala G. Barker; Anonaceae) against some food-borne microorganisms. Afr. J. Biotechnol. 2005, 4, 258–261. [Google Scholar]
- Naczk, M.; Shahidi, F. Nutritional and Pharmacological Effects of Food Phenolics, 2004 ed.; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar] [CrossRef]
- Bracke, M.E.; Depypere, H.T.; Boterberg, T.; van Marck, V.L.; Vennekens, K.M.; Vanluchene, E.; Nuytinck, M.; Serreyn, R.; Mareel, M.M. Influence of tangeretin on tamoxifen’s therapeutic benefit in mammary cancer. J. Natl Canc Inst. 1999, 91, 354–359. [Google Scholar] [CrossRef]
- DeJong, S.; Lanari, M.C. Extracts of olive polyphenols improve lipid stability in cooked beef and pork: Contribution of individual phenolics to the antioxidant activity of the extract. Food Chem. 2009, 116, 892–897. [Google Scholar] [CrossRef]
- González-Larena, M.; Cilla, A.; García-Llatas, G.; Barberá, R.; Lagarda, M.J. Plant sterols and antioxidant parameters in enriched beverages: Storage stability. J. Agric. Food Chem. 2012, 60, 4725–4734. [Google Scholar] [CrossRef] [PubMed]
- Tarko, T.; Duda-Chodak, A.; Semik, D.; Nycz, M. The use of fruit extracts for production of beverages with high antioxidative activity. Potravinarstvo 2015, 9, 280–283. [Google Scholar] [CrossRef]
- Kranz, P.; Braun, N.; Schulze, N.; Kunz, B. Sensory quality of functional beverages: Bitterness perception and bitter masking of olive leaf extract fortified fruit smoothies. J. Food Sci. 2010, 75, S308–S311. [Google Scholar] [CrossRef] [PubMed]
- Guillén, R.; Heredia, A.; Felizón, B.; Jiménez, A.; Montaño, A.; Fernández-Bolaños, J. Fibre fraction carbohydrates in olea europaea (gordal and manzanilla var.). Food Chem. 1992, 44, 173–178. [Google Scholar] [CrossRef]
- Jimenez, A.; Guillen, R.; Fernandezbolanos, J.; Heredia, A. Cell-wall composition of olives. J. Food Sci. 1994, 59, 1192–1196. [Google Scholar] [CrossRef]
- Galanakis, C.M. Olive fruit dietary fiber: Components, recovery and applications. Trends Food Sci. Technol. 2011, 22, 175–184. [Google Scholar] [CrossRef]
- Coimbra, M.A.; Waldron, K.W.; Selvendran, R.R. Isolation and characterisation of cell wall polymers from the heavily lignified tissues of olive (Olea europaea) seed hull. Carbohydr. Polymer. 1995, 27, 285–294. [Google Scholar] [CrossRef]
- Laufenberg, G. Regulierung Einer Mikroorganismenflora mit Oliventresterextrakten-Nutzung des Fungiziden und Bakteriziden Potentials Natürlicher Polyphenole zur Kontrolle von Pflanzenkrankheiten. 2007. Available online: https://www.ilt.uni-bonn.de/papierkorb/upgrading/regulierung-einer-mikroorganismenflora-mit-oliventresterextrakten (accessed on 14 August 2017).
- Valiente, C.; Arrigoni, E.; Esteban, R.M.; Amado, R. Chemical-composition of olive by-product and modifications through enzymatic treatments. J. Sci. Food Agric. 1995, 69, 27–32. [Google Scholar] [CrossRef]
- Dufresne, A.; Dupeyre, D.; Paillet, M. Lignocellulosic flour-reinforced poly (hydroxybutyrate-co-valerate) composites. J. Appl. Polym. Sci. 2003, 87, 1302–1315. [Google Scholar] [CrossRef]
- Amar, B.; Salem, K.; Hocine, D.; Chadia, I.; Juan, M.J. Study and characterization of composites materials based on polypropylene loaded with olive husk flour. J. Appl. Polym. Sci. 2011, 122, 1382–1394. [Google Scholar] [CrossRef]
- Cardoso, S.M.; Coimbra, M.A.; Lopes da Silva, J.A. Calcium-mediated gelation of an olive pomace pectic extract. Carbohydr. Polymer. 2003, 52, 125–133. [Google Scholar] [CrossRef]
- Galanakis, C.M.; Tornberg, E.; Gekas, V. A study of the recovery of the dietary fibres from olive mill wastewater and the gelling ability of the soluble fibre fraction. LWT Food Sci. Technol. 2010, 43, 1009–1017. [Google Scholar] [CrossRef]
- Cardoso, S.M.; Coimbra, M.A.; da Silva, J.A.L. Temperature dependence of the formation and melting of pectin-Ca 2 + networks: A rheological study. Food Hydrocoll. 2003, 17, 801–807. [Google Scholar] [CrossRef]
- Brozzoli, V.; Crognale, S.; Sampedro, I.; Federici, F.; D’Annibale, A.; Petruccioli, M. Assessment of olive-mill wastewater as a growth medium for lipase production by candida cylindracea in bench-top reactor. Bioresour. Technol. 2009, 100, 3395–3402. [Google Scholar] [CrossRef] [PubMed]
- Sarris, D.; Galiotou-Panayotou, M.; Koutinas, A.A.; Komaitis, M.; Papanikolaou, S. Citric acid, biomass and cellular lipid production by yarrowia lipolytica strains cultivated on olive mill wastewater-based media. J. Chem. Technol. Biotechnol. 2011, 86, 1439–1448. [Google Scholar] [CrossRef]
- Laconi, S.; Molle, G.; Cabiddu, A.; Pompei, R. Bioremediation of olive oil mill wastewater and production of microbial biomass. Biodegradation 2007, 18, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Aouidi, F.; Khelifi, E.; Asses, N.; Ayed, L.; Hamdi, M. Use of cheese whey to enhance geotrichum candidum biomass production in olive mill wastewater. J. Ind. Microbiol. Biotechnol. 2010, 37, 877–882. [Google Scholar] [CrossRef] [PubMed]
- Arous, F.; Azabou, S.; Jaouani, A.; Zouari-Mechichi, H.; Nasri, M.; Mechichi, T. Biosynthesis of single-cell biomass from olive mill wastewater by newly isolated yeasts. Environ. Sci. Pollut. Res. 2016, 23, 6783–6792. [Google Scholar] [CrossRef] [PubMed]
- Papankolaou, S.; Galiotou-Panayotou, M.; Fakas, S.; Komaitis, M.; Aggelis, G. Citric acid production by yarrowia lipolytica cultivated on olive-mill wastewater-based media. Bioresour. Technol. 2008, 99, 2419–2428. [Google Scholar] [CrossRef] [PubMed]
- Haddadin, M.S.; Abdulrahim, S.M.; Al-Khawaldeh, G.Y.; Robinson, R.K. Solid state fermentation of waste pomace from olive processing. J. Chem. Technol. Biotechnol. 1999, 74, 613–618. [Google Scholar] [CrossRef]
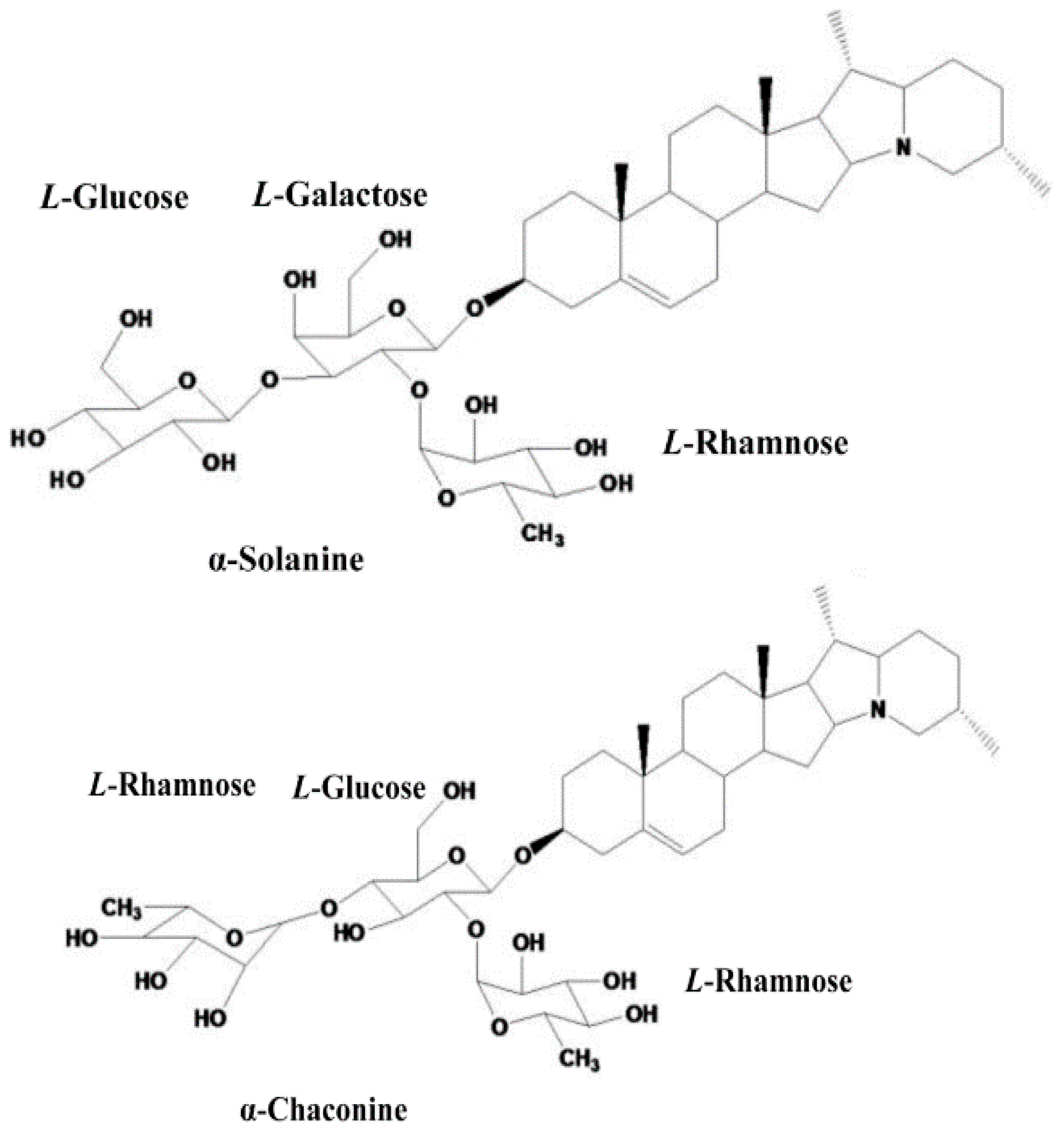

| Cultivar | Extraction Method | Optimal Conditions | Levels Reported | Authors |
|---|---|---|---|---|
| Agria (White) | SLE | 34 min at 89.9 °C and ethanol concentrations of 71.2% | 3.2–10.3 mg/100 g db | Amado, et al. [46] |
| Russett Burbank (brown) | MWAE | MeOH (67.33%), time of 15 min and a MP of 14.67% | 120–390 mg GAE 100/g db | Singh, et al. [47] |
| Lady Claire (cream) | PLE | 70% ethanol, 125 °C | 409 mg/100 g db | Wijngaard, Ballay and Brunton [34] |
| Lady Claire (cream) | SLE | 75% ethanol, 80 °C, 22 min | 431 mg/100 g db | Wijngaard, Ballay and Brunton [34] |
| Diamond (white) | SLE | Methanol | 112–291 mg GAE/100g db | Mohdaly, et al. [48] |
| Red | Subcritical water extraction | 180 °C and extraction time of 30 min | 81.83 mg/100 g wb | Singh and Saldana [42] |
| Vitelotte (Purple) | Ohmic heating assisted | 100 °C for 1 s 200 V/cm, water | 84 mg/100 g product | Pereira, Rodrigues, Genisheva, Oliveira, de Freitas, Teixeira and Vicente [41] |
| Vitelotte (Purple) | PEF aided extraction | 3.4 kV/cm and 105 µs (35 pulses of 3 µs), water | 65.8 mg/100 g fw | Puertolas, Cregenzan, Luengo, Alvarez and Raso [39] |
| Ramus | UAE | sonicated in a continuous air stream ultrasonic bath for 15 min | 593.3 mg/100 g db | Samarin, Poorazarang, Hematyar and Elhamirad [40] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fritsch, C.; Staebler, A.; Happel, A.; Cubero Márquez, M.A.; Aguiló-Aguayo, I.; Abadias, M.; Gallur, M.; Cigognini, I.M.; Montanari, A.; López, M.J.; et al. Processing, Valorization and Application of Bio-Waste Derived Compounds from Potato, Tomato, Olive and Cereals: A Review. Sustainability 2017, 9, 1492. https://doi.org/10.3390/su9081492
Fritsch C, Staebler A, Happel A, Cubero Márquez MA, Aguiló-Aguayo I, Abadias M, Gallur M, Cigognini IM, Montanari A, López MJ, et al. Processing, Valorization and Application of Bio-Waste Derived Compounds from Potato, Tomato, Olive and Cereals: A Review. Sustainability. 2017; 9(8):1492. https://doi.org/10.3390/su9081492
Chicago/Turabian StyleFritsch, Caroline, Andreas Staebler, Anton Happel, Miguel Angel Cubero Márquez, Ingrid Aguiló-Aguayo, Maribel Abadias, Miriam Gallur, Ilaria Maria Cigognini, Angela Montanari, Maria Jose López, and et al. 2017. "Processing, Valorization and Application of Bio-Waste Derived Compounds from Potato, Tomato, Olive and Cereals: A Review" Sustainability 9, no. 8: 1492. https://doi.org/10.3390/su9081492
APA StyleFritsch, C., Staebler, A., Happel, A., Cubero Márquez, M. A., Aguiló-Aguayo, I., Abadias, M., Gallur, M., Cigognini, I. M., Montanari, A., López, M. J., Suárez-Estrella, F., Brunton, N., Luengo, E., Sisti, L., Ferri, M., & Belotti, G. (2017). Processing, Valorization and Application of Bio-Waste Derived Compounds from Potato, Tomato, Olive and Cereals: A Review. Sustainability, 9(8), 1492. https://doi.org/10.3390/su9081492







