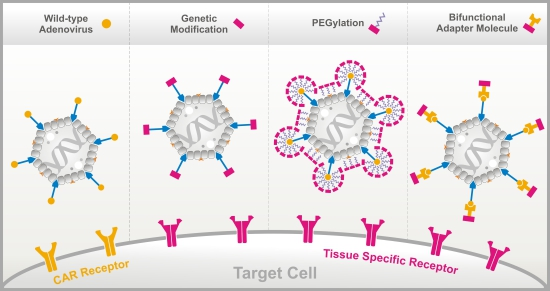
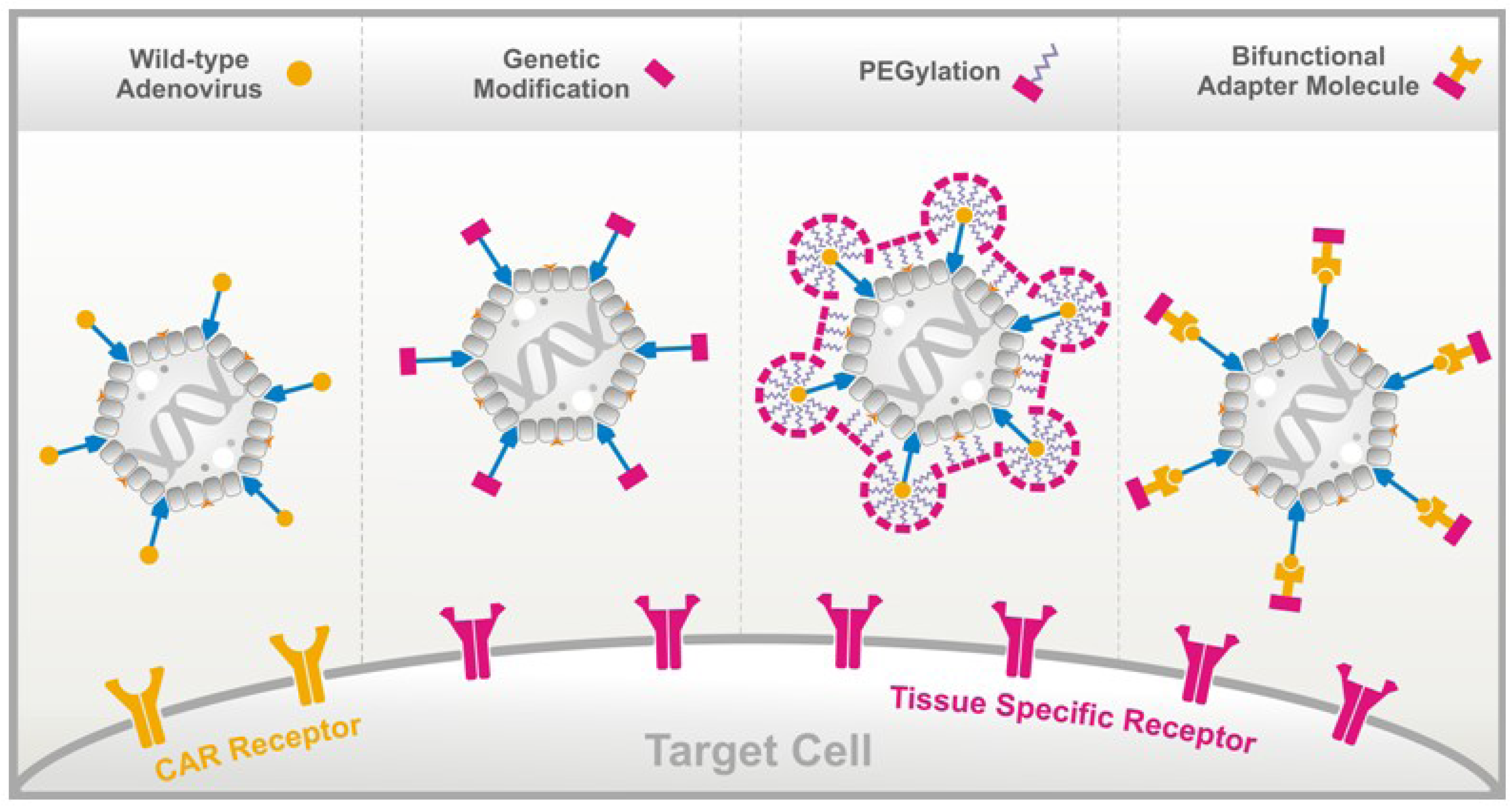
Peptide-Based Technologies to Alter Adenoviral Vector Tropism: Ways and Means for Systemic Treatment of Cancer
Abstract
:1. Introduction
2. Screening for Cell-Specific Ligands
3. Methods to Alter the Natural Ad Vector Tropism
3.1. Genetic Fiber Engineering
3.2. Chemical Peptide Conjugation via PEGylation
3.3. Bifunctional Non-Covalently Linked Adapter Molecules
4. Conclusions
Acknowledgments
Conflicts of Interest
References and Notes
- Morral, N.; O’Neal, W.K.; Rice, K.; Leland, M.M.; Piedra, P.A.; Aguilar-Cordova, E.; Carey, K.D.; Beaudet, A.L.; Langston, C. Lethal toxicity, severe endothelial injury, and a threshold effect with high doses of an adenoviral vector in baboons. Hum. Gene Ther. 2002, 13, 143–154. [Google Scholar] [CrossRef]
- Russell, W.C. Update on adenovirus and its vectors. J. Gen. Virol. 2000, 81, 2573–2604. [Google Scholar]
- Rowe, W.P.; Huebner, R.J.; Gilmore, L.K.; Parrott, R.H.; Ward, T.G. Isolation of a cytopathogenic agent from human adenoids undergoing spontaneous degeneration in tissue culture. Proc. Soc. Exp. Biol. Med. 1953, 84, 570–573. [Google Scholar] [CrossRef]
- Seto, D.; Chodosh, J.; Brister, J.R.; Jones, M.S.; Members of the Adenovirus Research Community. Using the whole-genome sequence to characterize and name human adenoviruses. J. Virol. 2011, 85, 5701–5702. [Google Scholar] [CrossRef] [Green Version]
- Xia, H.; Anderson, B.; Mao, Q.; Davidson, B.L. Recombinant human adenovirus: Targeting to the human transferrin receptor improves gene transfer to brain microcapillary endothelium. J. Virol. 2000, 74, 11359–11366. [Google Scholar] [CrossRef]
- Bergelson, J.M.; Modlin, J.F.; Wieland-Alter, W.; Cunningham, J.A.; Crowell, R.L.; Finberg, R.W. Clinical coxsackievirus B isolates differ from laboratory strains in their interaction with two cell surface receptors. J. Infect. Dis. 1997, 175, 697–700. [Google Scholar] [CrossRef]
- Roelvink, P.W.; Lizonova, A.; Lee, J.G.; Li, Y.; Bergelson, J.M.; Finberg, R.W.; Brough, D.E.; Kovesdi, I.; Wickham, T.J. The coxsackievirus-adenovirus receptor protein can function as a cellular attachment protein for adenovirus serotypes from subgroups A, C, D, E, and F. J. Virol. 1998, 72, 7909–7915. [Google Scholar]
- Wickham, T.J.; Mathias, P.; Cheresh, D.A.; Nemerow, G.R. Integrins alpha v beta 3 and alpha v beta 5 promote adenovirus internalization but not virus attachment. Cell 1993, 73, 309–319. [Google Scholar] [CrossRef]
- Arnberg, N. Adenovirus receptors: Implications for tropism, treatment and targeting. Rev. Med. Virol. 2009, 19, 165–178. [Google Scholar] [CrossRef]
- Chailertvanitkul, V.A.; Pouton, C.W. Adenovirus: A blueprint for non-viral gene delivery. Curr. Opin. Biotechnol. 2010, 21, 627–632. [Google Scholar] [CrossRef]
- Wickham, T.J. Targeting adenovirus. Gene Ther. 2000, 7, 110–114. [Google Scholar] [CrossRef]
- Douglas, J.T. Adenovirus-mediated gene delivery to skeletal muscle. Methods Mol. Biol. 2004, 246, 29–35. [Google Scholar]
- Rauschhuber, C.; Noske, N.; Ehrhardt, A. New insights into stability of recombinant adenovirus vector genomes in mammalian cells. Eur. J. Cell Biol. 2012, 91, 2–9. [Google Scholar] [CrossRef]
- Armendariz-Borunda, J.; Bastidas-Ramirez, B.E.; Sandoval-Rodriguez, A.; Gonzalez-Cuevas, J.; Gomez-Meda, B.; Garcia-Banuelos, J. Production of first generation adenoviral vectors for preclinical protocols: amplification, purification and functional titration. J. Biosci. Bioeng. 2011, 112, 415–421. [Google Scholar]
- Hashimoto, Y.; Kohri, K.; Akita, H.; Mitani, K.; Ikeda, K.; Nakanishi, M. Efficient transfer of genes into senescent cells by adenovirus vectors via highly expressed alpha v beta 5 integrin. Biochem. Biophys. Res. Commun. 1997, 240, 88–92. [Google Scholar] [CrossRef]
- Havenga, M.J.; Lemckert, A.A.; Ophorst, O.J.; van Meijer, M.; Germeraad, W.T.; Grimbergen, J.; van Den Doel, M.A.; Vogels, R.; van Deutekom, J.; Janson, A.A.; et al. Exploiting the natural diversity in adenovirus tropism for therapy and prevention of disease. J. Virol. 2002, 76, 4612–4620. [Google Scholar] [CrossRef]
- Takayama, K.; Ueno, H.; Pei, X.H.; Nakanishi, Y.; Yatsunami, J.; Hara, N. The levels of integrin alpha v beta 5 may predict the susceptibility to adenovirus-mediated gene transfer in human lung cancer cells. Gene Ther. 1998, 5, 361–368. [Google Scholar]
- Zhang, Y.; Bergelson, J.M. Adenovirus receptors. J. Virol. 2005, 79, 12125–12131. [Google Scholar] [CrossRef]
- Sharma, A.; Li, X.; Bangari, D.S.; Mittal, S.K. Adenovirus receptors and their implications in gene delivery. Virus Res. 2009, 143, 184–194. [Google Scholar] [CrossRef]
- Haj-Ahmad, Y.; Graham, F.L. Characterization of an adenovirus type 5 mutant carrying embedded inverted terminal repeats. Virology 1986, 153, 22–34. [Google Scholar] [CrossRef]
- Hedley, S.J.; Auf der Maur, A.; Hohn, S.; Escher, D.; Barberis, A.; Glasgow, J.N.; Douglas, J.T.; Korokhov, N.; Curiel, D.T. An adenovirus vector with a chimeric fiber incorporating stabilized single chain antibody achieves targeted gene delivery. Gene Ther. 2006, 13, 88–94. [Google Scholar] [CrossRef]
- Coughlan, L.; Alba, R.; Parker, A.L.; Bradshaw, A.C.; McNeish, I.A.; Nicklin, S.A.; Baker, A.H. Tropism-modification strategies for targeted gene delivery using adenoviral vectors. Viruses 2010, 2, 2290–2355. [Google Scholar] [CrossRef]
- Shayakhmetov, D.M.; Gaggar, A.; Ni, S.; Li, Z.-Y.; Lieber, A. Adenovirus binding to blood factors results in liver cell infection and hepatotoxicity. J. Virol. 2005, 79, 7478–7491. [Google Scholar] [CrossRef]
- Stone, D.; Liu, Y.; Shayakhmetov, D.; Li, Z.-Y.; Ni, S.; Lieber, A. Adenovirus-platelet interaction in blood causes virus sequestration to the reticuloendothelial system of the liver. J. Virol. 2007, 81, 8466–8471. [Google Scholar]
- Doronin, K.; Flatt, J.W.; di Paolo, N.C.; Khare, R.; Kalyuzhniy, O.; Acchione, M.; Sumida, J.P.; Ohto, U.; Shimizu, T.; Akashi-Takamura, S.; et al. Coagulation factor X activates innate immunity to human species C adenovirus. Science 2012, 338, 795–798. [Google Scholar] [CrossRef]
- Huard, J.; Lochmuller, H.; Acsadi, G.; Jani, A.; Massie, B.; Karpati, G. The route of administration is a major determinant of the transduction efficiency of rat tissues by adenoviral recombinants. Gene Ther. 1995, 2, 107–115. [Google Scholar]
- Khare, R.; Chen, C.Y.; Weaver, E.A.; Barry, M.A. Advances and future challenges in adenoviral vector pharmacology and targeting. Curr. Gene Ther. 2011, 11, 241–258. [Google Scholar] [CrossRef]
- Reynolds, P.N.; Nicklin, S.A.; Kaliberova, L.; Boatman, B.G.; Grizzle, W.E.; Balyasnikova, I.V.; Baker, A.H.; Danilov, S.M.; Curiel, D.T. Combined transductional and transcriptional targeting improves the specificity of transgene expression in vivo. Nat. Biotechnol. 2001, 19, 838–842. [Google Scholar]
- Shayakhmetov, D.M.; Li, Z.Y.; Ni, S.; Lieber, A. Analysis of adenovirus sequestration in the liver, transduction of hepatic cells, and innate toxicity after injection of fiber-modified vectors. J. Virol. 2004, 78, 5368–5381. [Google Scholar] [CrossRef]
- Xu, Z.; Qiu, Q.; Tian, J.; Smith, J.S.; Conenello, G.M.; Morita, T.; Byrnes, A.P. Coagulation factor X shields adenovirus type 5 from attack by natural antibodies and complement. Nat. Med. 2013, 19, 452–457. [Google Scholar] [CrossRef]
- Duffy, M.R.; Parker, A.L.; Kalkman, E.R.; White, K.; Kovalskyy, D.; Kelly, S.M.; Baker, A.H. Identification of novel small molecule inhibitors of adenovirus gene transfer using a high throughput screening approach. J. Contr. Release 2013, 170, 132–140. [Google Scholar] [CrossRef]
- Thacker, E.E.; Nakayama, M.; Smith, B.F.; Bird, R.C.; Muminova, Z.; Strong, T.V.; Timares, L.; Korokhov, N.; O’Neill, A.M.; de Gruijl, T.D.; et al. A genetically engineered adenovirus vector targeted to CD40 mediates transduction of canine dendritic cells and promotes antigen-specific immune responses in vivo. Vaccine 2009, 27, 7116–7124. [Google Scholar] [CrossRef]
- Coughlan, L.; Vallath, S.; Gros, A.; Gimenez-Alejandre, M.; van Rooijen, N.; Thomas, G.J.; Baker, A.H.; Cascallo, M.; Alemany, R.; Hart, I.R. Combined fiber modifications both to target alpha(v)beta(6) and detarget the coxsackievirus-adenovirus receptor improve virus toxicity profiles in vivo but fail to improve antitumoral efficacy relative to adenovirus serotype 5. Hum. Gene Ther. 2012, 23, 960–979. [Google Scholar] [CrossRef]
- McConnell, M.J.; Imperiale, M.J. Biology of adenovirus and its use as a vector for gene therapy. Hum. Gene Ther. 2004, 15, 1022–1033. [Google Scholar] [CrossRef]
- Haisma, H.J.; Boesjes, M.; Beerens, A.M.; van der Strate, B.W.; Curiel, D.T.; Pluddemann, A.; Gordon, S.; Bellu, A.R. Scavenger receptor A: A new route for adenovirus 5. Mol. Pharm. 2009, 6, 366–374. [Google Scholar] [CrossRef]
- Beatty, M.S.; Curiel, D.T. Chapter two–Adenovirus strategies for tissue-specific targeting. Adv. Cancer Res. 2012, 115, 39–67. [Google Scholar]
- Dorer, D.E.; Nettelbeck, D.M. Targeting cancer by transcriptional control in cancer gene therapy and viral oncolysis. Adv. Drug Deliv. Rev. 2009, 61, 554–571. [Google Scholar] [CrossRef]
- Haisma, H.J.; Kamps, G.K.; Bouma, A.; Geel, T.M.; Rots, M.G.; Kariath, A.; Bellu, A.R. Selective targeting of adenovirus to alphavbeta3 integrins, VEGFR2 and Tie2 endothelial receptors by angio-adenobodies. Int. J. Pharm. 2010, 391, 155–161. [Google Scholar] [CrossRef]
- Schmidt, A.; Eipel, C.; Furst, K.; Sommer, N.; Pahnke, J.; Putzer, B.M. Evaluation of systemic targeting of RET oncogene-based MTC with tumor-selective peptide-tagged Ad vectors in clinical mouse models. Gene Ther. 2011, 18, 418–423. [Google Scholar] [CrossRef]
- Schmidt, A.; Haas, S.J.; Hildebrandt, S.; Scheibe, J.; Eckhoff, B.; Racek, T.; Kempermann, G.; Wree, A.; Putzer, B.M. Selective targeting of adenoviral vectors to neural precursor cells in the hippocampus of adult mice: new prospects for in situ gene therapy. Stem Cells 2007, 25, 2910–2918. [Google Scholar]
- Reetz, J.; Genz, B.; Meier, C.; Kowtharapu, B.S.; Timm, F.; Vollmar, B.; Herchenroder, O.; Abshagen, K.; Putzer, B.M. Development of adenoviral delivery systems to target hepatic stellate cells in vivo. PLoS One 2013, 8, e67091. [Google Scholar] [CrossRef]
- Vives, E.; Schmidt, J.; Pelegrin, A. Cell-penetrating and cell-targeting peptides in drug delivery. Biochim. Biophys. Acta 2008, 1786, 126–138. [Google Scholar]
- Scott, J.K.; Smith, G.P. Searching for peptide ligands with an epitope library. Science 1990, 249, 386–390. [Google Scholar]
- Arap, W.; Pasqualini, R. The human vascular mapping project. Selection and utilization of molecules for tumor endothelial targeting. Haemostasis 2001, 31, 30–31. [Google Scholar]
- Arap, W.; Pasqualini, R.; Ruoslahti, E. Cancer treatment by targeted drug delivery to tumor vasculature in a mouse model. Science 1998, 279, 377–380. [Google Scholar] [CrossRef]
- Chen, C.Y.; May, S.M.; Barry, M.A. Targeting adenoviruses with factor x-single-chain antibody fusion proteins. Hum. Gene Ther. 2010, 21, 739–749. [Google Scholar] [CrossRef]
- Dias-Neto, E.; Nunes, D.N.; Giordano, R.J.; Sun, J.; Botz, G.H.; Yang, K.; Setubal, J.C.; Pasqualini, R.; Arap, W. Next-generation phage display: integrating and comparing available molecular tools to enable cost-effective high-throughput analysis. PLoS One 2009, 4, e8338. [Google Scholar] [CrossRef]
- Essler, M.; Ruoslahti, E. Molecular specialization of breast vasculature: A breast-homing phage-displayed peptide binds to aminopeptidase P in breast vasculature. Proc. Natl. Acad. Sci. USA 2002, 99, 2252–2257. [Google Scholar] [CrossRef]
- Kugler, J.; Zantow, J.; Meyer, T.; Hust, M. Oligopeptide m13 phage display in pathogen research. Viruses 2013, 5, 2531–2545. [Google Scholar] [CrossRef]
- Nicklin, S.A.; White, S.J.; Watkins, S.J.; Hawkins, R.E.; Baker, A.H. Selective targeting of gene transfer to vascular endothelial cells by use of peptides isolated by phage display. Circulation 2000, 102, 231–237. [Google Scholar] [CrossRef]
- Bignone, P.A.; Krupa, R.A.; Sternberg, H.; Funk, W.D.; Snyder, E.Y.; West, M.D.; Larocca, D. Identification of human embryonic progenitor cell targeting peptides using phage display. PLoS One 2013, 8, e58200. [Google Scholar]
- Pasqualini, R.; Koivunen, E.; Kain, R.; Lahdenranta, J.; Sakamoto, M.; Stryhn, A.; Ashmun, R.A.; Shapiro, L.H.; Arap, W.; Ruoslahti, E. Aminopeptidase N is a receptor for tumor-homing peptides and a target for inhibiting angiogenesis. Cancer Res. 2000, 60, 722–727. [Google Scholar]
- Pasqualini, R.; Ruoslahti, E. Organ targeting in vivo using phage display peptide libraries. Nature 1996, 380, 364–366. [Google Scholar] [CrossRef]
- White, S.J.; Nicklin, S.A.; Sawamura, T.; Baker, A.H. Identification of peptides that target the endothelial cell-specific LOX-1 receptor. Hypertension 2001, 37, 449–455. [Google Scholar] [CrossRef]
- Bewley, M.C.; Springer, K.; Zhang, Y.B.; Freimuth, P.; Flanagan, J.M. Structural analysis of the mechanism of adenovirus binding to its human cellular receptor, CAR. Science 1999, 286, 1579–1583. [Google Scholar] [CrossRef]
- Liu, Z.; Wu, K. Peptides homing to tumor vasculature: Imaging and therapeutics for cancer. Recent Pat. Anticancer Drug Discov. 2008, 3, 202–208. [Google Scholar] [CrossRef]
- Laakkonen, P.; Porkka, K.; Hoffman, J.A.; Ruoslahti, E. A tumor-homing peptide with a targeting specificity related to lymphatic vessels. Nat. Med. 2002, 8, 751–755. [Google Scholar]
- Odermatt, A.; Audige, A.; Frick, C.; Vogt, B.; Frey, B.M.; Frey, F.J.; Mazzucchelli, L. Identification of receptor ligands by screening phage-display peptide libraries ex vivo on microdissected kidney tubules. J. Am. Soc. Nephrol. 2001, 12, 308–316. [Google Scholar]
- Barry, M.A.; Dower, W.J.; Johnston, S.A. Toward cell-targeting gene therapy vectors: Selection of cell-binding peptides from random peptide-presenting phage libraries. Nat. Med. 1996, 2, 299–305. [Google Scholar] [CrossRef]
- Mazzucchelli, L.; Burritt, J.B.; Jesaitis, A.J.; Nusrat, A.; Liang, T.W.; Gewirtz, A.T.; Schnell, F.J.; Parkos, C.A. Cell-specific peptide binding by human neutrophils. Blood 1999, 93, 1738–1748. [Google Scholar]
- Ravera, M.W.; Carcamo, J.; Brissette, R.; Alam-Moghe, A.; Dedova, O.; Cheng, W.; Hsiao, K.C.; Klebanov, D.; Shen, H.; Tang, P.; et al. Identification of an allosteric binding site on the transcription factor p53 using a phage-displayed peptide library. Oncogene 1998, 16, 1993–1999. [Google Scholar]
- Ivanenkov, V.V.; Felici, F.; Menon, A.G. Targeted delivery of multivalent phage display vectors into mammalian cells. Biochim. Biophys. Acta 1999, 1448, 463–472. [Google Scholar] [CrossRef]
- Sclavons, C.; Burtea, C.; Boutry, S.; Laurent, S.; vander Elst, L.; Muller, R.N. Phage display screening for tumor necrosis factor- alpha -binding peptides: Detection of inflammation in a mouse model of hepatitis. Int. J. Pept. 2013, 2013, 348409. [Google Scholar]
- Cheung, C.S.; Lui, J.C.; Baron, J. Identification of chondrocyte-binding peptides by phage display. J. Orthop. Res. 2013, 31, 1053–1058. [Google Scholar] [CrossRef]
- Nishimoto, T.; Yamamoto, Y.; Yoshida, K.; Goto, N.; Ohnami, S.; Aoki, K. Development of peritoneal tumor-targeting vector by in vivo screening with a random peptide-displaying adenovirus library. PLoS One 2012, 7, e45550. [Google Scholar]
- Ozturk, N.; Erin, N.; Tuzuner, S. Changes in tissue substance P levels in patients with carpal tunnel syndrome. Neurosurgery 2010, 67, 1655–1660; discussion 1660–1661. [Google Scholar]
- Adewuya, A.O.; Owoeye, O.A.; Erinfolami, A.R. Psychopathology and subjective burden amongst primary caregivers of people with mental illness in South-Western Nigeria. Soc. Psychiatry Psychiatr. Epidemiol. 2011, 46, 1251–1256. [Google Scholar] [CrossRef]
- Nnodu, O.; Erinosho, L.; Jamda, M.; Olaniyi, O.; Adelaiye, R.; Lawson, L.; Odedina, F.; Shuaibu, F.; Odumuh, T.; Isu, N.; et al. Knowledge and attitudes towards cervical cancer and human papillomavirus: A Nigerian pilot study. Afr. J. Reprod. Health 2010, 14, 95–108. [Google Scholar]
- Erinjeri, J.P.; Clark, T.W. Cryoablation: Mechanism of action and devices. J. Vasc. Interv. Radiol: JVIR 2010, 21, S187–S191. [Google Scholar]
- Bockmann, M.; Drosten, M.; Putzer, B.M. Discovery of targeting peptides for selective therapy of medullary thyroid carcinoma. J. Gene Med. 2005, 7, 179–188. [Google Scholar] [CrossRef]
- Bockmann, M.; Hilken, G.; Schmidt, A.; Cranston, A.N.; Tannapfel, A.; Drosten, M.; Frilling, A.; Ponder, B.A.; Putzer, B.M. Novel SRESPHP peptide mediates specific binding to primary medullary thyroid carcinoma after systemic injection. Hum. Gene Ther. 2005, 16, 1267–1275. [Google Scholar] [CrossRef]
- Arap, W.; Kolonin, M.G.; Trepel, M.; Lahdenranta, J.; Cardo-Vila, M.; Giordano, R.J.; Mintz, P.J.; Ardelt, P.U.; Yao, V.J.; Vidal, C.I.; et al. Steps toward mapping the human vasculature by phage display. Nat. Med. 2002, 8, 121–127. [Google Scholar] [CrossRef]
- Chang, D.K.; Chiu, C.Y.; Kuo, S.Y.; Lin, W.C.; Lo, A.; Wang, Y.P.; Li, P.C.; Wu, H.C. Antiangiogenic targeting liposomes increase therapeutic efficacy for solid tumors. J. Biol. Chem. 2009, 284, 12905–12916. [Google Scholar]
- Seung-Min, L.; Gil-Suk, Y.; Eun-Sang, Y.; Tae-Gyun, K.; In-San, K.; Byung-Heon, L. Application of phage display to discovery of tumor-specific homing peptides: Developing strategies for therapy and molecular imaging of cancer. Methods Mol. Biol. 2009, 512, 355–363. [Google Scholar] [CrossRef]
- Rangel, R.; Dobroff, A.S.; Guzman-Rojas, L.; Salmeron, C.C.; Gelovani, J.G.; Sidman, R.L.; Pasqualini, R.; Arap, W. Targeting mammalian organelles with internalizing phage (iPhage) libraries. Nat. Protoc. 2013, 8, 1916–1939. [Google Scholar] [CrossRef]
- Rangel, R.; Guzman-Rojas, L.; le Roux, L.G.; Staquicini, F.I.; Hosoya, H.; Barbu, E.M.; Ozawa, M.G.; Nie, J.; Jr, K.D.; Langley, R.R.; et al. Combinatorial targeting and discovery of ligand-receptors in organelles of mammalian cells. Nat. Commun. 2012, 3, 788. [Google Scholar] [CrossRef]
- Jedlicka, P.; Hrdy, I.; Kuldova, J.; Wimmer, Z. The systemic effects of juvenoids on the red firebug Pyrrhocoris apterus and on the pea aphid Acyrthosiphon pisum with data on life table response. Pest Manag. Sci. 2007, 63, 1026–1035. [Google Scholar] [CrossRef]
- Wimmer, Z.; Jurcek, O.; Jedlicka, P.; Hanus, R.; Kuldova, J.; Hrdy, I.; Bennettova, B.; Saman, D. Insect pest management agents: Hormonogen esters (juvenogens). J. Agric. Food Chem. 2007, 55, 7387–7393. [Google Scholar]
- Song, K.M.; Lee, S.; Ban, C. Aptamers and their biological applications. Sensors (Basel) 2012, 12, 612–631. [Google Scholar] [CrossRef]
- Sundaram, P.; Kurniawan, H.; Byrne, M.E.; Wower, J. Therapeutic RNA aptamers in clinical trials. Eur. J. Pharm. Sci. 2013, 48, 259–271. [Google Scholar] [CrossRef]
- Zimbres, F.M.; Tarnok, A.; Ulrich, H.; Wrenger, C. Aptamers: Novel molecules as diagnostic markers in bacterial and viral infections? Biomed. Res. Int. 2013, 2013, 731516. [Google Scholar]
- Tan, W.; Donovan, M.J.; Jiang, J. Aptamers from cell-based selection for bioanalytical applications. Chem. Rev. 2013, 113, 2842–2862. [Google Scholar] [CrossRef]
- Hu, M.; Zhang, K. The application of aptamers in cancer research: An up-to-date review. Fut. Oncol. 2013, 9, 369–376. [Google Scholar] [CrossRef]
- Zhu, G.; Ye, M.; Donovan, M.J.; Song, E.; Zhao, Z.; Tan, W. Nucleic acid aptamers: An emerging frontier in cancer therapy. Chem. Commun. (Camb.) 2012, 48, 10472–10480. [Google Scholar]
- Wickham, T.J.; Tzeng, E.; Shears, L.L., 2nd; Roelvink, P.W.; Li, Y.; Lee, G.M.; Brough, D.E.; Lizonova, A.; Kovesdi, I. Increased in vitro and in vivo gene transfer by adenovirus vectors containing chimeric fiber proteins. J. Virol. 1997, 71, 8221–8229. [Google Scholar]
- Krasnykh, V.; Dmitriev, I.; Mikheeva, G.; Miller, C.R.; Belousova, N.; Curiel, D.T. Characterization of an adenovirus vector containing a heterologous peptide epitope in the HI loop of the fiber knob. J. Virol. 1998, 72, 1844–1852. [Google Scholar]
- Einfeld, D.A.; Schroeder, R.; Roelvink, P.W.; Lizonova, A.; King, C.R.; Kovesdi, I.; Wickham, T.J. Reducing the native tropism of adenovirus vectors requires removal of both CAR and integrin interactions. J. Virol. 2001, 75, 11284–11291. [Google Scholar] [CrossRef]
- Pereboev, A.V.; Nagle, J.M.; Shakhmatov, M.A.; Triozzi, P.L.; Matthews, Q.L.; Kawakami, Y.; Curiel, D.T.; Blackwell, J.L. Enhanced gene transfer to mouse dendritic cells using adenoviral vectors coated with a novel adapter molecule. Mol. Ther. 2004, 9, 712–720. [Google Scholar] [CrossRef]
- Krenek, K.; Kuldova, M.; Hulikova, K.; Stibor, I.; Lhotak, P.; Dudic, M.; Budka, J.; Pelantova, H.; Bezouska, K.; Fiserova, A.; et al. N-acetyl-D-glucosamine substituted calix[4]arenes as stimulators of NK cell-mediated antitumor immune response. Carbohydr. Res. 2007, 342, 1781–1792. [Google Scholar] [CrossRef]
- Dmitriev, I.; Krasnykh, V.; Miller, C.R.; Wang, M.; Kashentseva, E.; Mikheeva, G.; Belousova, N.; Curiel, D.T. An adenovirus vector with genetically modified fibers demonstrates expanded tropism via utilization of a coxsackievirus and adenovirus receptor-independent cell entry mechanism. J. Virol. 1998, 72, 9706–9713. [Google Scholar]
- Davison, A.J.; Benko, M.; Harrach, B. Genetic content and evolution of adenoviruses. J. Gen. Virol. 2003, 84, 2895–2908. [Google Scholar] [CrossRef]
- Koizumi, N.; Mizuguchi, H.; Sakurai, F.; Yamaguchi, T.; Watanabe, Y.; Hayakawa, T. Reduction of natural adenovirus tropism to mouse liver by fiber-shaft exchange in combination with both CAR- and alphav integrin-binding ablation. J. Virol. 2003, 77, 13062–13072. [Google Scholar] [CrossRef]
- Nettelbeck, D.M.; Rivera, A.A.; Kupsch, J.; Dieckmann, D.; Douglas, J.T.; Kontermann, R.E.; Alemany, R.; Curiel, D.T. Retargeting of adenoviral infection to melanoma: combining genetic ablation of native tropism with a recombinant bispecific single-chain diabody (scDb) adapter that binds to fiber knob and HMWMAA. Int. J. Cancer 2004, 108, 136–145. [Google Scholar] [CrossRef]
- Glasgow, J.N.; Kremer, E.J.; Hemminki, A.; Siegal, G.P.; Douglas, J.T.; Curiel, D.T. An adenovirus vector with a chimeric fiber derived from canine adenovirus type 2 displays novel tropism. Virology 2004, 324, 103–116. [Google Scholar] [CrossRef]
- Coughlan, L.; Vallath, S.; Saha, A.; Flak, M.; McNeish, I.A.; Vassaux, G.; Marshall, J.F.; Hart, I.R.; Thomas, G.J. In vivo retargeting of adenovirus type 5 to alphavbeta6 integrin results in reduced hepatotoxicity and improved tumor uptake following systemic delivery. J. Virol. 2009, 83, 6416–6428. [Google Scholar] [CrossRef]
- Wang, D.; Liu, S.; Mao, Q.; Zhao, J.; Xia, H. A novel vector for a rapid generation of fiber-mutant adenovirus based on one step ligation and quick screening of positive clones. J. Biotechnol. 2011, 152, 72–76. [Google Scholar] [CrossRef]
- Belousova, N.; Krendelchtchikova, V.; Curiel, D.T.; Krasnykh, V. Modulation of adenovirus vector tropism via incorporation of polypeptide ligands into the fiber protein. J. Virol. 2002, 76, 8621–8631. [Google Scholar] [CrossRef]
- Nicklin, S.A.; Dishart, K.L.; Buening, H.; Reynolds, P.N.; Hallek, M.; Nemerow, G.R.; von Seggern, D.J.; Baker, A.H. Transductional and transcriptional targeting of cancer cells using genetically engineered viral vectors. Cancer Lett. 2003, 201, 165–173. [Google Scholar] [CrossRef]
- Work, L.M.; Nicklin, S.A.; Brain, N.J.; Dishart, K.L.; Von Seggern, D.J.; Hallek, M.; Buning, H.; Baker, A.H. Development of efficient viral vectors selective for vascular smooth muscle cells. Mol. Ther. 2004, 9, 198–208. [Google Scholar]
- Cao, C.; Dong, X.; Wu, X.; Wen, B.; Ji, G.; Cheng, L.; Liu, H. Conserved fiber-penton base interaction revealed by nearly atomic resolution cryo-electron microscopy of the structure of adenovirus provides insight into receptor interaction. J. Virol. 2012, 86, 12322–12329. [Google Scholar] [CrossRef]
- Tanaka, T.; Kuroki, M.; Hamada, H.; Kato, K.; Kinugasa, T.; Shibaguchi, H.; Zhao, J. Cancer-targeting gene therapy using tropism-modified adenovirus. Anticancer Res. 2007, 27, 3679–3684. [Google Scholar]
- Kurachi, S.; Koizumi, N.; Sakurai, F.; Kawabata, K.; Sakurai, H.; Nakagawa, S.; Hayakawa, T.; Mizuguchi, H. Characterization of capsid-modified adenovirus vectors containing heterologous peptides in the fiber knob, protein IX, or hexon. Gene Ther. 2007, 14, 266–274. [Google Scholar] [CrossRef]
- Terao, S.; Acharya, B.; Suzuki, T.; Aoi, T.; Naoe, M.; Hamada, K.; Mizuguchi, H.; Gotoh, A. Improved gene transfer into renal carcinoma cells using adenovirus vector containing RGD motif. Anticancer Res. 2009, 29, 2997–3001. [Google Scholar]
- Katayama, K.; Furuki, R.; Yokoyama, H.; Kaneko, M.; Tachibana, M.; Yoshida, I.; Nagase, H.; Tanaka, K.; Sakurai, F.; Mizuguchi, H.; et al. Enhanced in vivo gene transfer into the placenta using RGD fiber-mutant adenovirus vector. Biomaterials 2011, 32, 4185–4193. [Google Scholar] [CrossRef]
- Bayo-Puxan, N.; Gimenez-Alejandre, M.; Lavilla-Alonso, S.; Gros, A.; Cascallo, M.; Hemminki, A.; Alemany, R. Replacement of adenovirus type 5 fiber shaft heparan sulfate proteoglycan-binding domain with RGD for improved tumor infectivity and targeting. Hum. Gene Ther. 2009, 20, 1214–1221. [Google Scholar] [CrossRef]
- Rojas, J.J.; Gimenez-Alejandre, M.; Gil-Hoyos, R.; Cascallo, M.; Alemany, R. Improved systemic antitumor therapy with oncolytic adenoviruses by replacing the fiber shaft HSG-binding domain with RGD. Gene Ther. 2012, 19, 453–457. [Google Scholar] [CrossRef]
- Roelvink, P.W.; Mi Lee, G.; Einfeld, D.A.; Kovesdi, I.; Wickham, T.J. Identification of a conserved receptor-binding site on the fiber proteins of CAR-recognizing adenoviridae. Science 1999, 286, 1568–1571. [Google Scholar] [CrossRef]
- Leissner, P.; Legrand, V.; Schlesinger, Y.; Hadji, D.A.; van Raaij, M.; Cusack, S.; Pavirani, A.; Mehtali, M. Influence of adenoviral fiber mutations on viral encapsidation, infectivity and in vivo tropism. Gene Ther. 2001, 8, 49–57. [Google Scholar] [CrossRef]
- Mizuguchi, H.; Koizumi, N.; Hosono, T.; Ishii-Watabe, A.; Uchida, E.; Utoguchi, N.; Watanabe, Y.; Hayakawa, T. CAR- or alphav integrin-binding ablated adenovirus vectors, but not fiber-modified vectors containing RGD peptide, do not change the systemic gene transfer properties in mice. Gene Ther. 2002, 9, 769–776. [Google Scholar] [CrossRef]
- Alemany, R.; Curiel, D.T. CAR-binding ablation does not change biodistribution and toxicity of adenoviral vectors. Gene Ther. 2001, 8, 1347–1353. [Google Scholar] [CrossRef]
- Nakamura, T.; Sato, K.; Hamada, H. Reduction of natural adenovirus tropism to the liver by both ablation of fiber-coxsackievirus and adenovirus receptor interaction and use of replaceable short fiber. J. Virol. 2003, 77, 2512–2521. [Google Scholar] [CrossRef]
- Smith, T.A.; Idamakanti, N.; Rollence, M.L.; Marshall-Neff, J.; Kim, J.; Mulgrew, K.; Nemerow, G.R.; Kaleko, M.; Stevenson, S.C. Adenovirus serotype 5 fiber shaft influences in vivo gene transfer in mice. Hum. Gene Ther. 2003, 14, 777–787. [Google Scholar] [CrossRef]
- Smith, T.A.; Idamakanti, N.; Marshall-Neff, J.; Rollence, M.L.; Wright, P.; Kaloss, M.; King, L.; Mech, C.; Dinges, L.; Iverson, W.O.; et al. Receptor interactions involved in adenoviral-mediated gene delivery after systemic administration in non-human primates. Hum. Gene Ther. 2003, 14, 1595–1604. [Google Scholar] [CrossRef]
- Magnusson, M.K.; Kraaij, R.; Leadley, R.M.; de Ridder, C.M.; van Weerden, W.M.; van Schie, K.A.; van der Kroeg, M.; Hoeben, R.C.; Maitland, N.J.; Lindholm, L. A transductionally retargeted adenoviral vector for virotherapy of Her2/neu-expressing prostate cancer. Hum. Gene Ther. 2012, 23, 70–82. [Google Scholar] [CrossRef]
- Myhre, S.; Henning, P.; Friedman, M.; Stahl, S.; Lindholm, L.; Magnusson, M.K. Re-targeted adenovirus vectors with dual specificity; binding specificities conferred by two different Affibody molecules in the fiber. Gene Ther. 2009, 16, 252–261. [Google Scholar] [CrossRef]
- Wu, H.; Seki, T.; Dmitriev, I.; Uil, T.; Kashentseva, E.; Han, T.; Curiel, D.T. Double modification of adenovirus fiber with RGD and polylysine motifs improves coxsackievirus-adenovirus receptor-independent gene transfer efficiency. Hum. Gene Ther. 2002, 13, 1647–1653. [Google Scholar] [CrossRef]
- Belousova, N.; Korokhov, N.; Krendelshchikova, V.; Simonenko, V.; Mikheeva, G.; Triozzi, P.L.; Aldrich, W.A.; Banerjee, P.T.; Gillies, S.D.; Curiel, D.T.; et al. Genetically targeted adenovirus vector directed to CD40-expressing cells. J. Virol. 2003, 77, 11367–11377. [Google Scholar] [CrossRef]
- O'Neill, A.M.; Smith, A.N.; Spangler, E.A.; Whitley, E.M.; Schleis, S.E.; Bird, R.C.; Curiel, D.T.; Thacker, E.E.; Smith, B.F. Resistance of canine lymphoma cells to adenoviral infection due to reduced cell surface RGD binding integrins. Cancer Biol. Ther. 2011, 11, 651–658. [Google Scholar]
- Yu, D.; Jin, C.; Leja, J.; Majdalani, N.; Nilsson, B.; Eriksson, F.; Essand, M. Adenovirus with hexon Tat-protein transduction domain modification exhibits increased therapeutic effect in experimental neuroblastoma and neuroendocrine tumors. J. Virol. 2011, 85, 13114–13123. [Google Scholar] [CrossRef]
- Yu, D.; Jin, C.; Ramachandran, M.; Xu, J.; Nilsson, B.; Korsgren, O.; le Blanc, K.; Uhrbom, L.; Forsberg-Nilsson, K.; Westermark, B.; et al. Adenovirus serotype 5 vectors with Tat-PTD modified hexon and serotype 35 fiber show greatly enhanced transduction capacity of primary cell cultures. PLoS One 2013, 8, e54952. [Google Scholar]
- Brouwer, E.; Havenga, M.J.; Ophorst, O.; de Leeuw, B.; Gijsbers, L.; Gillissen, G.; Hoeben, R.C.; ter Horst, M.; Nanda, D.; Dirven, C.; et al. Human adenovirus type 35 vector for gene therapy of brain cancer: improved transduction and bypass of pre-existing anti-vector immunity in cancer patients. Cancer Gene Ther. 2007, 14, 211–219. [Google Scholar] [CrossRef]
- Sova, P.; Ren, X.W.; Ni, S.; Bernt, K.M.; Mi, J.; Kiviat, N.; Lieber, A. A tumor-targeted and conditionally replicating oncolytic adenovirus vector expressing TRAIL for treatment of liver metastases. Mol. Ther. 2004, 9, 496–509. [Google Scholar] [CrossRef]
- Hesse, A.; Kosmides, D.; Kontermann, R.E.; Nettelbeck, D.M. Tropism modification of adenovirus vectors by peptide ligand insertion into various positions of the adenovirus serotype 41 short-fiber knob domain. J. Virol. 2007, 81, 2688–2699. [Google Scholar] [CrossRef]
- Matsui, H.; Sakurai, F.; Katayama, K.; Kurachi, S.; Tashiro, K.; Sugio, K.; Kawabata, K.; Mizuguchi, H. Enhanced transduction efficiency of fiber-substituted adenovirus vectors by the incorporation of RGD peptides in two distinct regions of the adenovirus serotype 35 fiber knob. Virus Res. 2011, 155, 48–54. [Google Scholar] [CrossRef]
- Magnusson, M.K.; Hong, S.S.; Henning, P.; Boulanger, P.; Lindholm, L. Genetic retargeting of adenovirus vectors: Functionality of targeting ligands and their influence on virus viability. J. Gene Med. 2002, 4, 356–370. [Google Scholar] [CrossRef]
- Mizuguchi, H.; Hayakawa, T. Targeted adenovirus vectors. Hum. Gene Ther. 2004, 15, 1034–1044. [Google Scholar] [CrossRef]
- Kreppel, F.; Kochanek, S. Modification of adenovirus gene transfer vectors with synthetic polymers: A scientific review and technical guide. Mol. Ther. 2008, 16, 16–29. [Google Scholar] [CrossRef]
- Elkon, K.B.; Liu, C.C.; Gall, J.G.; Trevejo, J.; Marino, M.W.; Abrahamsen, K.A.; Song, X.; Zhou, J.L.; Old, L.J.; Crystal, R.G.; et al. Tumor necrosis factor alpha plays a central role in immune-mediated clearance of adenoviral vectors. Proc. Natl. Acad. Sci. USA 1997, 94, 9814–9819. [Google Scholar] [CrossRef]
- Zaiss, A.K.; Liu, Q.; Bowen, G.P.; Wong, N.C.; Bartlett, J.S.; Muruve, D.A. Differential activation of innate immune responses by adenovirus and adeno-associated virus vectors. J. Virol. 2002, 76, 4580–4590. [Google Scholar] [CrossRef]
- Croyle, M.A.; Callahan, S.M.; Auricchio, A.; Schumer, G.; Linse, K.D.; Wilson, J.M.; Brunner, L.J.; Kobinger, G.P. PEGylation of a vesicular stomatitis virus G pseudotyped lentivirus vector prevents inactivation in serum. J. Virol. 2004, 78, 912–921. [Google Scholar] [CrossRef]
- Mok, H.; Palmer, D.J.; Ng, P.; Barry, M.A. Evaluation of polyethylene glycol modification of first-generation and helper-dependent adenoviral vectors to reduce innate immune responses. Mol. Ther. 2005, 11, 66–79. [Google Scholar]
- Gao, J.Q.; Eto, Y.; Yoshioka, Y.; Sekiguchi, F.; Kurachi, S.; Morishige, T.; Yao, X.; Watanabe, H.; Asavatanabodee, R.; Sakurai, F.; et al. Effective tumor targeted gene transfer using PEGylated adenovirus vector via systemic administration. J. Contr. Release 2007, 122, 102–110. [Google Scholar] [CrossRef]
- Alemany, R.; Suzuki, K.; Curiel, D.T. Blood clearance rates of adenovirus type 5 in mice. J. Gen. Virol. 2000, 81, 2605–2609. [Google Scholar]
- O'Riordan, C.R.; Lachapelle, A.; Delgado, C.; Parkes, V.; Wadsworth, S.C.; Smith, A.E.; Francis, G.E. PEGylation of adenovirus with retention of infectivity and protection from neutralizing antibody in vitro and in vivo. Hum. Gene Ther. 1999, 10, 1349–1358. [Google Scholar] [CrossRef]
- Romanczuk, H.; Galer, C.E.; Zabner, J.; Barsomian, G.; Wadsworth, S.C.; O'Riordan, C.R. Modification of an adenoviral vector with biologically selected peptides: A novel strategy for gene delivery to cells of choice. Hum. Gene Ther. 1999, 10, 2615–2626. [Google Scholar] [CrossRef]
- Eto, Y.; Yoshioka, Y.; Ishida, T.; Yao, X.; Morishige, T.; Narimatsu, S.; Mizuguchi, H.; Mukai, Y.; Okada, N.; Kiwada, H.; et al. Optimized PEGylated adenovirus vector reduces the anti-vector humoral immune response against adenovirus and induces a therapeutic effect against metastatic lung cancer. Biol. Pharm. Bull. 2010, 33, 1540–1544. [Google Scholar] [CrossRef]
- Lee, G.K.; Maheshri, N.; Kaspar, B.; Schaffer, D.V. PEG conjugation moderately protects adeno-associated viral vectors against antibody neutralization. Biotechnol. Bioeng. 2005, 92, 24–34. [Google Scholar] [CrossRef]
- Hofherr, S.E.; Shashkova, E.V.; Weaver, E.A.; Khare, R.; Barry, M.A. Modification of adenoviral vectors with polyethylene glycol modulates in vivo tissue tropism and gene expression. Mol. Ther. 2008, 16, 1276–1282. [Google Scholar] [CrossRef]
- Wonganan, P.; Croyle, M.A. PEGylated adenoviruses: From mice to monkeys. Viruses 2010, 2, 468–502. [Google Scholar] [CrossRef]
- Lanciotti, J.; Song, A.; Doukas, J.; Sosnowski, B.; Pierce, G.; Gregory, R.; Wadsworth, S.; O'Riordan, C. Targeting adenoviral vectors using heterofunctional polyethylene glycol FGF2 conjugates. Mol. Ther. 2003, 8, 99–107. [Google Scholar] [CrossRef]
- Eto, Y.; Gao, J.Q.; Sekiguchi, F.; Kurachi, S.; Katayama, K.; Mizuguchi, H.; Hayakawa, T.; Tsutsumi, Y.; Mayumi, T.; Nakagawa, S. Neutralizing antibody evasion ability of adenovirus vector induced by the bioconjugation of methoxypolyethylene glycol succinimidyl propionate (MPEG-SPA). Biol. Pharm. Bull. 2004, 27, 936–938. [Google Scholar] [CrossRef]
- Ogawara, K.; Rots, M.G.; Kok, R.J.; Moorlag, H.E.; van Loenen, A.M.; Meijer, D.K.; Haisma, H.J.; Molema, G. A novel strategy to modify adenovirus tropism and enhance transgene delivery to activated vascular endothelial cells in vitro and in vivo. Hum. Gene Ther. 2004, 15, 433–443. [Google Scholar] [CrossRef]
- Eto, Y.; Gao, J.Q.; Sekiguchi, F.; Kurachi, S.; Katayama, K.; Maeda, M.; Kawasaki, K.; Mizuguchi, H.; Hayakawa, T.; Tsutsumi, Y.; et al. PEGylated adenovirus vectors containing RGD peptides on the tip of PEG show high transduction efficiency and antibody evasion ability. J. Gene Med. 2005, 7, 604–612. [Google Scholar] [CrossRef]
- Xiong, Z.; Cheng, Z.; Zhang, X.; Patel, M.; Wu, J.C.; Gambhir, S.S.; Chen, X. Imaging chemically modified adenovirus for targeting tumors expressing integrin alphavbeta3 in living mice with mutant herpes simplex virus type 1 thymidine kinase PET reporter gene. J. Nucl. Med. 2006, 47, 130–139. [Google Scholar]
- Kuldo, J.M.; Asgeirsdottir, S.A.; Zwiers, P.J.; Bellu, A.R.; Rots, M.G.; Schalk, J.A.; Ogawara, K.I.; Trautwein, C.; Banas, B.; Haisma, H.J.; et al. Targeted adenovirus mediated inhibition of NF-kappaB-dependent inflammatory gene expression in endothelial cells in vitro and in vivo. J. Contr. Release 2013, 166, 57–65. [Google Scholar] [CrossRef]
- Kim, P.H.; Sohn, J.H.; Choi, J.W.; Jung, Y.; Kim, S.W.; Haam, S.; Yun, C.O. Active targeting and safety profile of PEG-modified adenovirus conjugated with herceptin. Biomaterials 2011, 32, 2314–2326. [Google Scholar] [CrossRef]
- Yao, X.; Yoshioka, Y.; Morishige, T.; Eto, Y.; Watanabe, H.; Okada, Y.; Mizuguchi, H.; Mukai, Y.; Okada, N.; Nakagawa, S. Systemic administration of a PEGylated adenovirus vector with a cancer-specific promoter is effective in a mouse model of metastasis. Gene Ther. 2009, 16, 1395–1404. [Google Scholar] [CrossRef]
- Kaneda, Y.; Tsutsumi, Y.; Yoshioka, Y.; Kamada, H.; Yamamoto, Y.; Kodaira, H.; Tsunoda, S.; Okamoto, T.; Mukai, Y.; Shibata, H.; et al. The use of PVP as a polymeric carrier to improve the plasma half-life of drugs. Biomaterials 2004, 25, 3259–3266. [Google Scholar] [CrossRef]
- Yao, X.; Yoshioka, Y.; Morishige, T.; Eto, Y.; Narimatsu, S.; Kawai, Y.; Mizuguchi, H.; Gao, J.Q.; Mukai, Y.; Okada, N.; et al. Tumor vascular targeted delivery of polymer-conjugated adenovirus vector for cancer gene therapy. Mol. Ther. 2011, 19, 1619–1625. [Google Scholar] [CrossRef]
- Yao, X.L.; Yoshioka, Y.; Ruan, G.X.; Chen, Y.Z.; Mizuguchi, H.; Mukai, Y.; Okada, N.; Gao, J.Q.; Nakagawa, S. Optimization and internalization mechanisms of PEGylated adenovirus vector with targeting peptide for cancer gene therapy. Biomacromolecules 2012, 13, 2402–2409. [Google Scholar] [CrossRef]
- Jiang, Z.K.; Koh, S.B.; Sato, M.; Atanasov, I.C.; Johnson, M.; Zhou, Z.H.; Deming, T.J.; Wu, L. Engineering polypeptide coatings to augment gene transduction and in vivo stability of adenoviruses. J. Contr. Release 2013, 166, 75–85. [Google Scholar] [CrossRef]
- Deming, T.J. Facile synthesis of block copolypeptides of defined architecture. Nature 1997, 390, 386–389. [Google Scholar] [CrossRef]
- Curiel, D.T. Strategies to adapt adenoviral vectors for targeted delivery. Ann. N. Y. Acad. Sci. 1999, 886, 158–171. [Google Scholar] [CrossRef]
- Glasgow, J.N.; Everts, M.; Curiel, D.T. Transductional targeting of adenovirus vectors for gene therapy. Cancer Gene Ther. 2006, 13, 830–844. [Google Scholar] [CrossRef]
- Everts, M.; Curiel, D.T. Transductional targeting of adenoviral cancer gene therapy. Curr. Gene Ther. 2004, 4, 337–346. [Google Scholar] [CrossRef]
- Douglas, J.T.; Miller, C.R.; Kim, M.; Dmitriev, I.; Mikheeva, G.; Krasnykh, V.; Curiel, D.T. A system for the propagation of adenoviral vectors with genetically modified receptor specificities. Nat. Biotechnol. 1999, 17, 470–475. [Google Scholar] [CrossRef]
- Rancourt, C.; Robertson, M.W., 3rd; Wang, M.; Goldman, C.K.; Kelly, J.F.; Alvarez, R.D.; Siegal, G.P.; Curiel, D.T. Endothelial cell vehicles for delivery of cytotoxic genes as a gene therapy approach for carcinoma of the ovary. Clin. Cancer Res. 1998, 4, 265–270. [Google Scholar]
- Bauerschmitz, G.J.; Barker, S.D.; Hemminki, A. Adenoviral gene therapy for cancer: From vectors to targeted and replication competent agents (review). Int. J. Oncol. 2002, 21, 1161–1174. [Google Scholar]
- Reynolds, P.N.; Zinn, K.R.; Gavrilyuk, V.D.; Balyasnikova, I.V.; Rogers, B.E.; Buchsbaum, D.J.; Wang, M.H.; Miletich, D.J.; Grizzle, W.E.; Douglas, J.T.; et al. A targetable, injectable adenoviral vector for selective gene delivery to pulmonary endothelium in vivo. Mol. Ther. 2000, 2, 562–578. [Google Scholar] [CrossRef]
- Griffioen, A.W.; Molema, G. Angiogenesis: Potentials for pharmacologic intervention in the treatment of cancer, cardiovascular diseases, and chronic inflammation. Pharmacol. Rev. 2000, 52, 237–268. [Google Scholar]
- Nettelbeck, D.M.; Miller, D.W.; Jerome, V.; Zuzarte, M.; Watkins, S.J.; Hawkins, R.E.; Muller, R.; Kontermann, R.E. Targeting of adenovirus to endothelial cells by a bispecific single-chain diabody directed against the adenovirus fiber knob domain and human endoglin (CD105). Mol. Ther. 2001, 3, 882–891. [Google Scholar] [CrossRef]
- Van Beusechem, V.W.; van Rijswijk, A.L.; van Es, H.H.; Haisma, H.J.; Pinedo, H.M.; Gerritsen, W.R. Recombinant adenovirus vectors with knobless fibers for targeted gene transfer. Gene Ther. 2000, 7, 1940–1946. [Google Scholar] [CrossRef]
- Haisma, H.J.; Grill, J.; Curiel, D.T.; Hoogeland, S.; van Beusechem, V.W.; Pinedo, H.M.; Gerritsen, W.R. Targeting of adenoviral vectors through a bispecific single-chain antibody. Cancer Gene Ther. 2000, 7, 901–904. [Google Scholar]
- van Zeeburg, H.J.; van Beusechem, V.W.; Huizenga, A.; Haisma, H.J.; Korokhov, N.; Gibbs, S.; Leemans, C.R.; Brakenhoff, R.H. Adenovirus retargeting to surface expressed antigens on oral mucosa. J. Gene Med. 2010, 12, 365–376. [Google Scholar] [CrossRef]
- Hemminki, A.; Zinn, K.R.; Liu, B.; Chaudhuri, T.R.; Desmond, R.A.; Rogers, B.E.; Barnes, M.N.; Alvarez, R.D.; Curiel, D.T. In vivo molecular chemotherapy and noninvasive imaging with an infectivity-enhanced adenovirus. J. Natl. Cancer Inst. 2002, 94, 741–749. [Google Scholar] [CrossRef]
- Dmitriev, I.; Kashentseva, E.; Rogers, B.E.; Krasnykh, V.; Curiel, D.T. Ectodomain of coxsackievirus and adenovirus receptor genetically fused to epidermal growth factor mediates adenovirus targeting to epidermal growth factor receptor-positive cells. J. Virol. 2000, 74, 6875–6884. [Google Scholar] [CrossRef]
- Kashentseva, E.A.; Seki, T.; Curiel, D.T.; Dmitriev, I.P. Adenovirus targeting to c-erbB-2 oncoprotein by single-chain antibody fused to trimeric form of adenovirus receptor ectodomain. Cancer Res. 2002, 62, 609–616. [Google Scholar]
- Hangalapura, B.N.; Timares, L.; Oosterhoff, D.; Scheper, R.J.; Curiel, D.T.; de Gruijl, T.D. CD40-targeted adenoviral cancer vaccines: the long and winding road to the clinic. J. Gene Med. 2012, 14, 416–427. [Google Scholar] [CrossRef]
- Williams, B.J.; Bhatia, S.; Adams, L.K.; Boling, S.; Carroll, J.L.; Li, X.L.; Rogers, D.L.; Korokhov, N.; Kovesdi, I.; Pereboev, A.V.; et al. Dendritic cell based PSMA immunotherapy for prostate cancer using a CD40-targeted adenovirus vector. PLoS One 2012, 7, e46981. [Google Scholar] [CrossRef]
- Echeverria, I.; Pereboev, A.; Silva, L.; Zabaleta, A.; Riezu-Boj, J.I.; Bes, M.; Cubero, M.; Borras-Cuesta, F.; Lasarte, J.J.; Esteban, J.I.; et al. Enhanced T cell responses against hepatitis C virus by ex vivo targeting of adenoviral particles to dendritic cells. Hepatology 2011, 54, 28–37. [Google Scholar] [CrossRef]
- Kim, Y.S.; Kim, Y.J.; Lee, J.M.; Han, S.H.; Ko, H.J.; Park, H.J.; Pereboev, A.; Nguyen, H.H.; Kang, C.Y. CD40-targeted recombinant adenovirus significantly enhances the efficacy of antitumor vaccines based on dendritic cells and B cells. Hum. Gene Ther. 2010, 21, 1697–1706. [Google Scholar] [CrossRef]
- Tillman, B.W.; Hayes, T.L.; DeGruijl, T.D.; Douglas, J.T.; Curiel, D.T. Adenoviral vectors targeted to CD40 enhance the efficacy of dendritic cell-based vaccination against human papillomavirus 16-induced tumor cells in a murine model. Cancer Res. 2000, 60, 5456–5463. [Google Scholar]
- Watkins, S.J.; Mesyanzhinov, V.V.; Kurochkina, L.P.; Hawkins, R.E. The ‘adenobody’ approach to viral targeting: Specific and enhanced adenoviral gene delivery. Gene Ther. 1997, 4, 1004–1012. [Google Scholar]
- Dreier, B.; Honegger, A.; Hess, C.; Nagy-Davidescu, G.; Mittl, P.R.; Grutter, M.G.; Belousova, N.; Mikheeva, G.; Krasnykh, V.; Pluckthun, A. Development of a generic adenovirus delivery system based on structure-guided design of bispecific trimeric DARPin adapters. Proc. Natl. Acad. Sci. USA 2013, 110, E869–E877. [Google Scholar] [CrossRef]
- Dreier, B.; Mikheeva, G.; Belousova, N.; Parizek, P.; Boczek, E.; Jelesarov, I.; Forrer, P.; Pluckthun, A.; Krasnykh, V. Her2-specific multivalent adapters confer designed tropism to adenovirus for gene targeting. J. Mol. Biol. 2011, 405, 410–426. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Reetz, J.; Herchenröder, O.; Pützer, B.M. Peptide-Based Technologies to Alter Adenoviral Vector Tropism: Ways and Means for Systemic Treatment of Cancer. Viruses 2014, 6, 1540-1563. https://doi.org/10.3390/v6041540
Reetz J, Herchenröder O, Pützer BM. Peptide-Based Technologies to Alter Adenoviral Vector Tropism: Ways and Means for Systemic Treatment of Cancer. Viruses. 2014; 6(4):1540-1563. https://doi.org/10.3390/v6041540
Chicago/Turabian StyleReetz, Julia, Ottmar Herchenröder, and Brigitte M. Pützer. 2014. "Peptide-Based Technologies to Alter Adenoviral Vector Tropism: Ways and Means for Systemic Treatment of Cancer" Viruses 6, no. 4: 1540-1563. https://doi.org/10.3390/v6041540