Construction and Testing of orfA +/- FIV Reporter Viruses
Abstract
:1. Introduction
2. Results
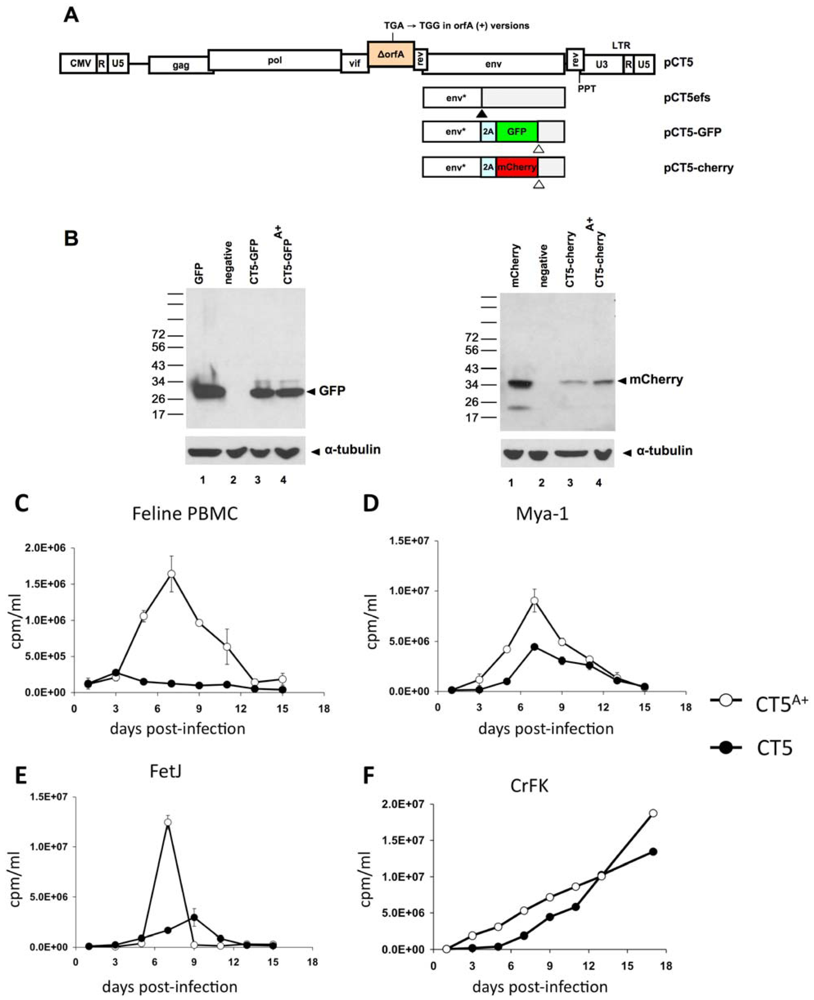
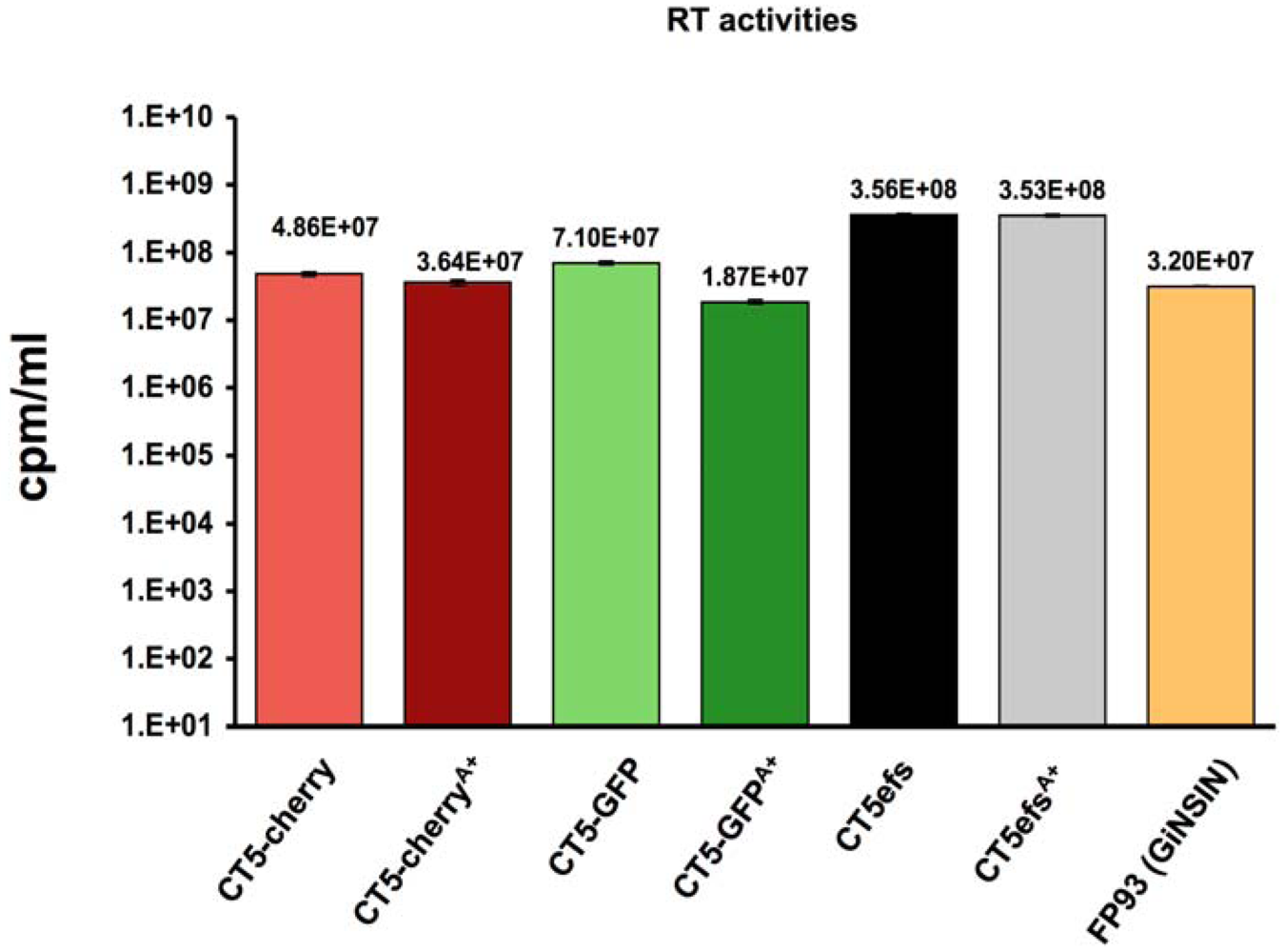
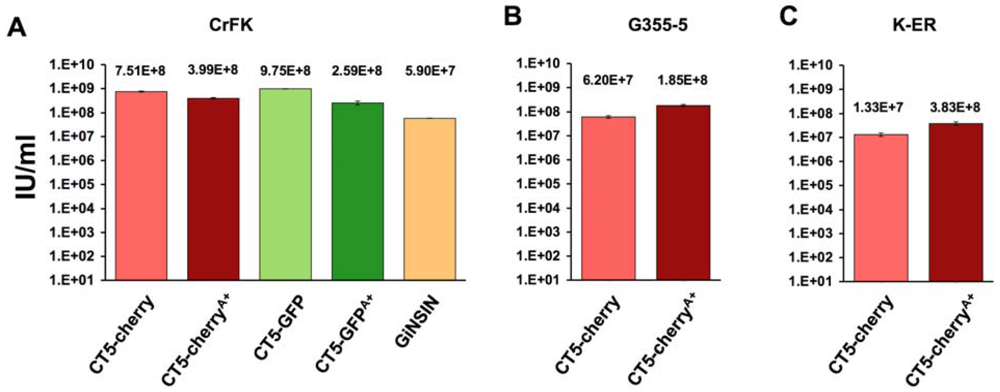
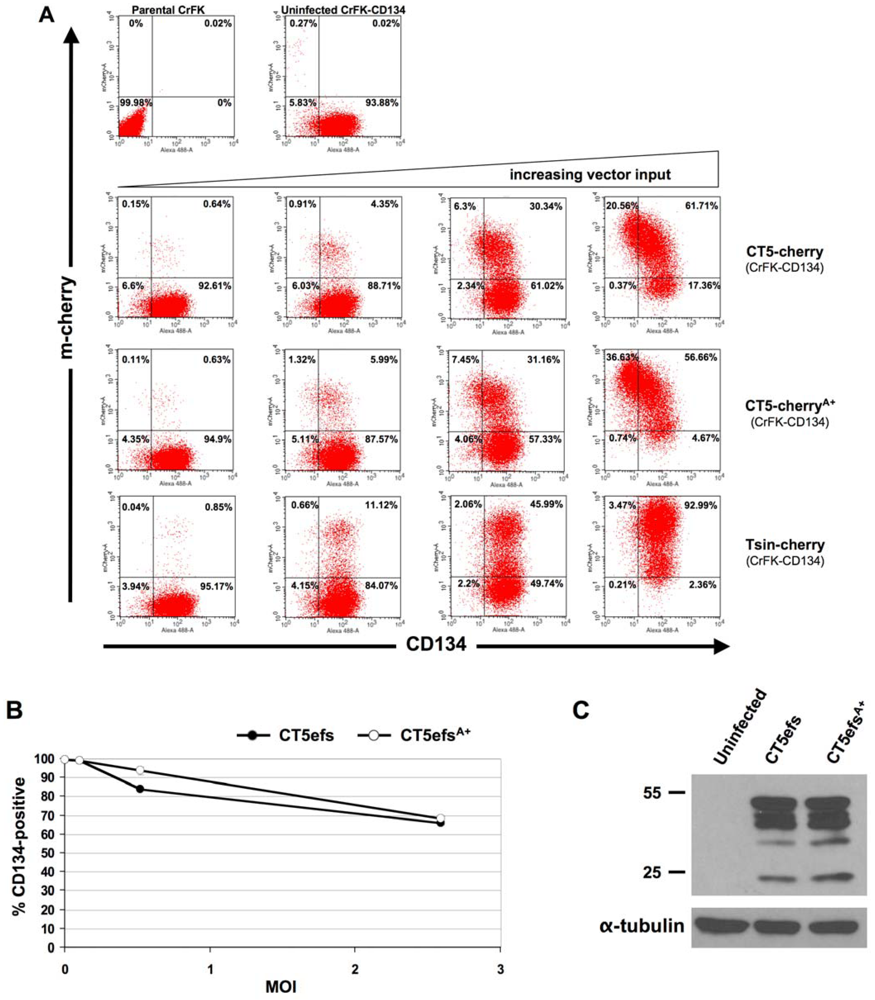
3. Experimental Section
3.1 Cell lines.
3.2 Generation of CrFK-CD134 cells.
3.3 Construction of pCT5 based reporter vectors.
3.4 Single-round FIV reporters and full length replicating FIV production.
3.5 Immunoblotting.
4. Discussion and Conclusions
Acknowledgments
Conflict of Interest
References and Notes
- Naldini, L.; Bloemer, U.; Gallay, P.; Ory, D.; Mulligan, R.; Gage, F.H.; Verma, I.M.; Trono, D. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 1996, 272, 263–267. [Google Scholar]
- Chen, B.K.; Saksela, K.; Andino, R.; Baltimore, D. Distinct modes of human immunodeficiency virus type 1 proviral latency revealed by superinfection of nonproductively infected cell lines with recombinant luciferase-encoding viruses. Journal of Virology 1994, 68, 654–660. [Google Scholar]
- Connor, R.I.; Chen, B.K.; Choe, S.; Landau, N.R. Vpr is required for efficient replication of human immunodeficiency virus type-1 in mononuclear phagocytes. Virology 1995, 206, 935–944. [Google Scholar]
- Planelles, V.; Bachelerie, F.; Jowett, J.B.; Haislip, A.; Xie, Y.; Banooni, P.; Masuda, T.; Chen, I.S. Fate of the human immunodeficiency virus type 1 provirus in infected cells: a role for vpr. Journal of Virology 1995, 69, 5883–5889. [Google Scholar]
- Poeschla, E.; Wong-Staal, F.; Looney, D. Efficient transduction of nondividing cells by feline immunodeficiency virus lentiviral vectors. Nature Medicine 1998, 4, 354–357. [Google Scholar]
- Saenz, D.T.; Teo, W.; Olsen, J.C.; Poeschla, E. Restriction of Feline Immunodeficiency Virus by Ref1, LV1 and Primate TRIM5a Proteins. Journal of Virology 2005, 79, 15175–15188. [Google Scholar]
- Lin, T. Y.; Emerman, M. Cyclophilin A interacts with diverse lentiviral capsids. Retrovirology 2006, 3, 70. [Google Scholar]
- Diaz-Griffero, F.; Kar, A.; Lee, M.; Stremlau, M.; Poeschla, E.; Sodroski, J. Comparative requirements for the restriction of retrovirus infection by TRIM5alpha and TRIMCyp. Virology 2007, 369, 400–410. [Google Scholar]
- Wilson, S.J.; Webb, B.L.; Ylinen, L.M.; Verschoor, E.; Heeney, J.L.; Towers, G.J. Independent evolution of an antiviral TRIMCyp in rhesus macaques. Proc Natl Acad Sci U S A 2008, 105, 3557–3562. [Google Scholar]
- Münk, C.; Zielonka, J.; Constabel, H.; Kloke, B.P.; Rengstl, B.; Battenberg, M.; Bonci, F.; Pistello, M.; Lochelt, M.; Cichutek, K. Multiple restrictions of human immunodeficiency virus type 1 in feline cells. J Virol 2007, 81, 7048–7060. [Google Scholar]
- Münk, C.; Beck, T.; Zielonka, J.; Hotz-Wagenblatt, A.; Chareza, S.; Battenberg, M.; Thielebein, J.; Cichutek, K.; Bravo, I.G.; O'Brien, S.J.; Lochelt, M.; Yuhki, N. Functions, structure, and read-through alternative splicing of feline APOBEC3 genes. Genome Biol 2008, 9, R48. [Google Scholar] [PubMed]
- Schaller, T.; Ylinen, L.M.; Webb, B.L.; Singh, S.; Towers, G.J. Fusion of cyclophilin A to Fv1 enables cyclosporine-sensitive restriction of human and feline immunodeficiency viruses. J Virol 2007, 81, 10055–10063. [Google Scholar]
- Virgen, C.A.; Kratovac, Z.; Bieniasz, P.D.; Hatziioannou, T. Independent genesis of chimeric TRIM5-cyclophilin proteins in two primate species. Proc Natl Acad Sci U S A 2008, 105, 3563–3568. [Google Scholar]
- McEwan, W.A.; Schaller, T.; Ylinen, L.M.; Hosie, M.J.; Towers, G.J.; Willett, B.J. Truncation of TRIM5 in Feliformia explains the absence of retroviral restriction in cells of the domestic cat. J Virol 2009, 16, 8270–8275. [Google Scholar]
- Stern, M.A.; Hu, C.; Saenz, D.T.; Fadel, H.J.; Sims, O.; Peretz, M.; Poeschla, E.M. Productive replication of Vif-chimeric HIV-1 in feline cells. J Virol 2010, 84, 7378–7395. [Google Scholar]
- Zielonka, J.; Marino, D.; Hofmann, H.; Yuhki, N.; Lochelt, M.; Munk, C. Vif of feline immunodeficiency virus from domestic cats protects against APOBEC3 restriction factors from many felids. J Virol 2010, 84, 7312–7324. [Google Scholar]
- LaRue, R.S.; Lengyel, J.; Jonsson, S.R.; Andresdottir, V.; Harris, R.S. Lentiviral Vif degrades the APOBEC3Z3/APOBEC3H protein of its mammalian host and is capable of cross-species activity. J Virol 2010, 84, 8193–8201. [Google Scholar]
- Ikeda, T.; Ohsugi, T.; Kimura, T.; Matsushita, S.; Maeda, Y.; Harada, S.; Koito, A. The antiretroviral potency of APOBEC1 deaminase from small animal species. Nucleic Acids Res 2008, 36, 6859–6871. [Google Scholar]
- Neagu, M.R.; Ziegler, P.; Pertel, T.; Strambio-De-Castillia, C.; Grutter, C.; Martinetti, G.; Mazzucchelli, L.; Grutter, M.; Manz, M.G.; Luban, J. Potent inhibition of HIV-1 by TRIM5-cyclophilin fusion proteins engineered from human components. J Clin Invest 2009, 119, 3035–3047. [Google Scholar] [Green Version]
- Dietrich, I.; Macintyre, A.; McMonagle, E.; Price, A.J.; James, L.C.; McEwan, W.A.; Hosie, M. J.; Willett, B.J. Potent lentiviral restriction by a synthetic feline TRIM5 cyclophilin A fusion. J Virol 2010, 84, 8980–8985. [Google Scholar]
- Javanbakht, H.; Diaz-Griffero, F.; Yuan, W.; Yeung, D.F.; Li, X.; Song, B.; Sodroski, J. The ability of multimerized cyclophilin A to restrict retrovirus infection. Virology 2007, 367, 19–29. [Google Scholar]
- Poss, M.; Ross, H.A.; Painter, S.L.; Holley, D.C.; Terwee, J.A.; Vandewoude, S.; Rodrigo, A. Feline lentivirus evolution in cross-species infection reveals extensive G-to-A mutation and selection on key residues in the viral polymerase. J Virol 2006, 80, 2728–2737. [Google Scholar]
- Fletcher, A.J.; Hue, S.; Schaller, T.; Pillay, D.; Towers, G.J. Hare TRIM5alpha Restricts Divergent Retroviruses and Exhibits Significant Sequence Variation from Closely Related Lagomorpha TRIM5 Genes. J Virol 2010, 84, 12463–12468. [Google Scholar]
- Ylinen, L.M.; Price, A.J.; Rasaiyaah, J.; Hue, S.; Rose, N.J.; Marzetta, F.; James, L.C.; Towers, G.J. Conformational adaptation of Asian macaque TRIMCyp directs lineage specific antiviral activity. PLoS Pathog 2010, 6, 8. [Google Scholar]
- Llano, M.; Saenz, D.T.; Meehan, A.; Wongthida, P.; Peretz, M.; Walker, W.H.; Teo, W.; Poeschla, E.M. An Essential Role for LEDGF/p75 in HIV Integration. Science 2006, 314, 461–464. [Google Scholar]
- Luttge, B.G.; Shehu-Xhilaga, M.; Demirov, D.G.; Adamson, C.S.; Soheilian, F.; Nagashima, K.; Stephen, A.G.; Fisher, R.J.; Freed, E.O. Molecular characterization of feline immunodeficiency virus budding. J Virol 2008, 82, 2106–2119. [Google Scholar]
- Luttge, B.G.; Freed, E.O. FIV Gag: Virus assembly and host-cell interactions. Vet Immunol Immunopathol 2009. [Google Scholar]
- Krishnan, L.; Matreyek, K.A.; Oztop, I.; Lee, K.; Tipper, C.H.; Li, X.; Dar, M.J.; Kewalramani, V.N.; Engelman, A. The requirement for cellular transportin 3 (TNPO3 or TRN-SR2) during infection maps to human immunodeficiency virus type 1 capsid and not integrase. J Virol 2009, 84, 397–406. [Google Scholar]
- Kemler, I.; Meehan, A.; Poeschla, E.M. Live-cell coimaging of the genomic RNAs and Gag proteins of two lentiviruses. J Virol 2010, 84, 6352–6366. [Google Scholar]
- Lee, K.; Ambrose, Z.; Martin, T.D.; Oztop, I.; Mulky, A.; Julias, J.G.; Vandegraaff, N.; Baumann, J.G.; Wang, R.; Yuen, W.; Takemura, T.; Shelton, K.; Taniuchi, I.; Li, Y.; Sodroski, J.; Littman, D.R.; Coffin, J.M.; Hughes, S.H.; Unutmaz, D.; Engelman, A.; KewalRamani, V.N. Flexible use of nuclear import pathways by HIV-1. Cell Host Microbe 2010, 7, 221–233. [Google Scholar]
- Lin, Y.C.; Torbett, B.E.; Elder, J.H. Generation of infectious feline immunodeficiency virus (FIV) encoding FIV/human immunodeficiency virus chimeric protease. J Virol 2010, 84, 6799–6809. [Google Scholar]
- Poeschla, E.; Looney, D. CXCR4 is required by a non-primate lentivirus: heterologous expression of feline immunodeficiency virus in human, rodent and feline cells. Journal of Virology 1998, 72, 6858–6866. [Google Scholar]
- Talbott, R.L.; Sparger, E.E.; Lovelace, K.M.; Fitch, W.M.; Pedersen, N.C.; Luciw, P.A.; Elder, J.H. Nucleotide sequence and genomic organization of feline immunodeficiency virus. Proceedings of the National Academy of Sciences of the United States of America 1989, 86, 5743–5747. [Google Scholar]
- de Parseval, A.; Chatterji, U.; Morris, G.; Sun, P.; Olson, A.J.; Elder, J.H. Structural mapping of CD134 residues critical for interaction with feline immunodeficiency virus. Nat Struct Mol Biol 2005, 12, 60–66. [Google Scholar]
- Ryan, M.D.; King, A.M.; Thomas, G.P. Cleavage of foot-and-mouth disease virus polyprotein is mediated by residues located within a 19 amino acid sequence. J Gen Virol 1991, 72, 2727–2732. [Google Scholar]
- Szymczak, A.L.; Workman, C.J.; Wang, Y.; Vignali, K.M.; Dilioglou, S.; Vanin, E.F.; Vignali, D.A. Correction of multi-gene deficiency in vivo using a single 'self-cleaving' 2A peptide-based retroviral vector. Nat Biotechnol 2004, 22, 589–594. [Google Scholar]
- Tomonaga, K.; Miyazawa, T.; Sakuragi, J.; Mori, T.; Adachi, A.; Mikami, T. The feline immunodeficiency virus ORF-A gene facilitates efficient viral replication in established T-cell lines and peripheral blood lymphocytes. J Virol 1993, 67, 5889–5895. [Google Scholar]
- Waters, A.K.; De Parseval, A.P.; Lerner, D.L.; Neil, J.C.; Thompson, F.J.; Elder, J.H. Influence of ORF2 on host cell tropism of feline immunodeficiency virus. Virology 1996, 215, 10–16. [Google Scholar]
- Gemeniano, M.C.; Sawai, E.T.; Leutenegger, C.M.; Sparger, E.E. Feline immunodeficiency virus ORF-Ais required for virus particle formation and virus infectivity. J Virol 2003, 77, 8819–8830. [Google Scholar]
- Saenz, D.T.; Barraza, R.; Loewen, N.; Teo, W.; Poeschla, E. Production and Use of Feline Immunodeficiency Virus (FIV)-based lentiviral vectors. In Gene Transfer: A Cold Spring Harbor Laboratory Manual; Rossi, J., Friedman, T., Eds.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, 2006; pp. 57–74. [Google Scholar]
- Barraza, R.; Poeschla, E. Human gene therapy vectors derived from feline lentiviruses. Veterinary Immunology and Immunopathology 2008, 123, 23–31. [Google Scholar]
- Llano, M.; Gaznick, N.; Poeschla, E.M. Rapid, controlled and intensive lentiviral vector-based RNAi. In HIV Protocols, second edition; Kalpana, G., Prasad, V., Eds.; Humana Press: Totowa, NJ, 2008. [Google Scholar]
- Barraza, R.A.; Rasmussen, C.A.; Loewen, N.; Cameron, J.D.; Gabelt, B.T.; Teo, W.L.; Kaufman, P.L.; Poeschla, E.M. Prolonged transgene expression with lentiviral vectors in the aqueous humor outflow pathway of nonhuman primates. Hum Gene Ther 2009, 20, 191–200. [Google Scholar]
- Sparger, E.E.; Shacklett, B.L.; Renshaw-Gegg, L.; Barry, P.A.; Pedersen, N.C.; Elder, J.H.; Luciw, P.A. Regulation of gene expression directed by the long terminal repeat of the feline immunodeficiency virus. Virology 1992, 187, 165–177. [Google Scholar]
- de Parseval, A.; Elder, J.H. Demonstration that orf2 encodes the feline immunodeficiency virus transactivating (Tat) protein and characterization of a unique gene product with partial rev activity. J Virol 1999, 73, 608–617. [Google Scholar]
- Hong, Y.; Fink, E.; Hu, Q.Y.; Kiosses, W.B.; Elder, J.H. OrfA downregulates feline immunodeficiency virus primary receptor CD134 on the host cell surface and is important in viral infection. J Virol 2010, 84, 7225–7232. [Google Scholar]
- Gemeniano, M.C.; Sawai, E.T.; Sparger, E.E. Feline immunodeficiency virus Orf-A localizes to the nucleus and induces cell cycle arrest. Virology 2004, 325, 167–174. [Google Scholar]
- Chatterji, U.; de Parseval, A.; Elder, J.H. Feline immunodeficiency virus OrfA is distinct from other lentivirus transactivators. J Virol 2002, 76, 9624–9634. [Google Scholar]
- Sundstrom, M.; Chatterji, U.; Schaffer, L.; de Rozieres, S.; Elder, J.H. Feline immunodeficiency virus OrfA alters gene expression of splicing factors and proteasome-ubiquitination proteins. Virology 2008, 371, 394–404. [Google Scholar]
- Remington, K.M.; Chesebro, B.; Wehrly, K.; Pedersen, N.C.; North, T.W. Mutants of feline immunodeficiency virus resistant to 3'-azido-3'-deoxythymidine. Journal of Virology 1991, 65, 308–312. [Google Scholar]
- Willett, B.J.; Cannon, C.A.; Hosie, M.J. Upregulation of surface feline CXCR4 expression following ectopic expression of CCR5: implications for studies of the cell tropism of feline immunodeficiency virus. J Virol 2002, 76, 9242–9252. [Google Scholar]
- Soneoka, Y.; Cannon, P.M.; Ramsdale, E.E.; Griffiths, J.C.; Romano, G.; Kingsman, S.M.; Kingsman, A.J. A transient three-plasmid expression system for the production of high titer retroviral vectors. Nucleic Acids Research 1995, 23, 628–633. [Google Scholar]
- Kemler, I.; Barraza, R.; Poeschla, E.M. Mapping of the encapsidation determinants of feline immunodeficiency virus. J Virol 2002, 76, 11889–11903. [Google Scholar]
- Malim, M.H.; Emerman, M. HIV-1 accessory proteins--ensuring viral survival in a hostile environment. Cell Host Microbe 2008, 3, 388–398. [Google Scholar]
- Willey, R.L.; Maldarelli, F.; Martin, M.A.; Strebel, K. Human immunodeficiency virus type 1 Vpu protein induces rapid degradation of CD4. J Virol 1992, 66, 7193–7200. [Google Scholar]
- Neil, S.J.; Zang, T.; Bieniasz, P.D. Tetherin inhibits retrovirus release and is antagonized by HIV-1 Vpu. Nature 2008, 451, 425–430. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fadel, H.J.; Saenz, D.T.; Poeschla, E.M. Construction and Testing of orfA +/- FIV Reporter Viruses. Viruses 2012, 4, 184-199. https://doi.org/10.3390/v4010184
Fadel HJ, Saenz DT, Poeschla EM. Construction and Testing of orfA +/- FIV Reporter Viruses. Viruses. 2012; 4(1):184-199. https://doi.org/10.3390/v4010184
Chicago/Turabian StyleFadel, Hind J., Dyana T. Saenz, and Eric M. Poeschla. 2012. "Construction and Testing of orfA +/- FIV Reporter Viruses" Viruses 4, no. 1: 184-199. https://doi.org/10.3390/v4010184



